Isoliquiritigen Enhances the Antitumour Activity and Decreases the Genotoxic Effect of Cyclophosphamide
Abstract
:1. Introduction
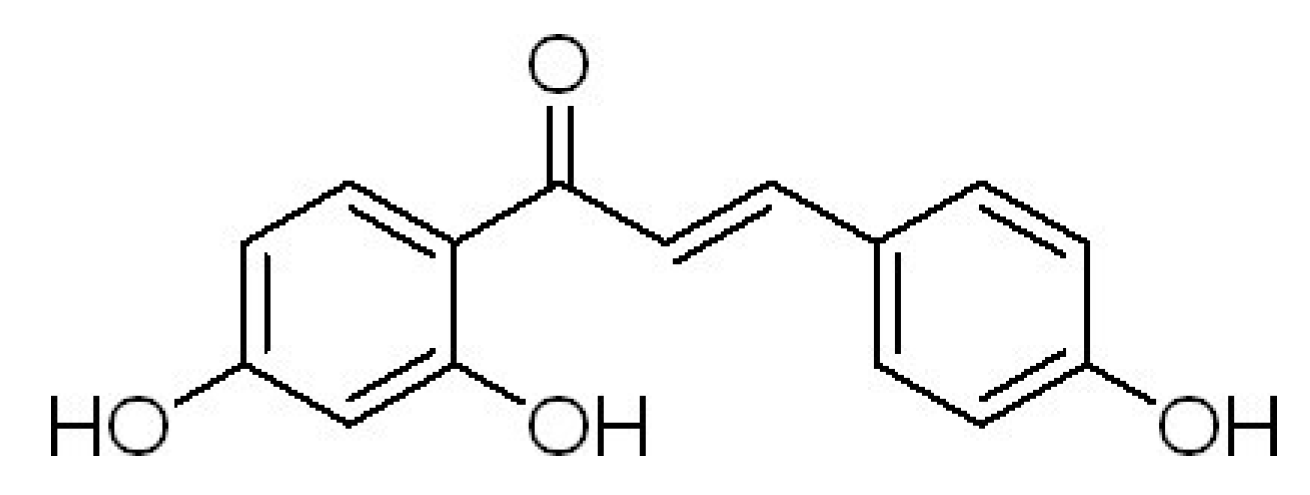
2. Results and Discussion
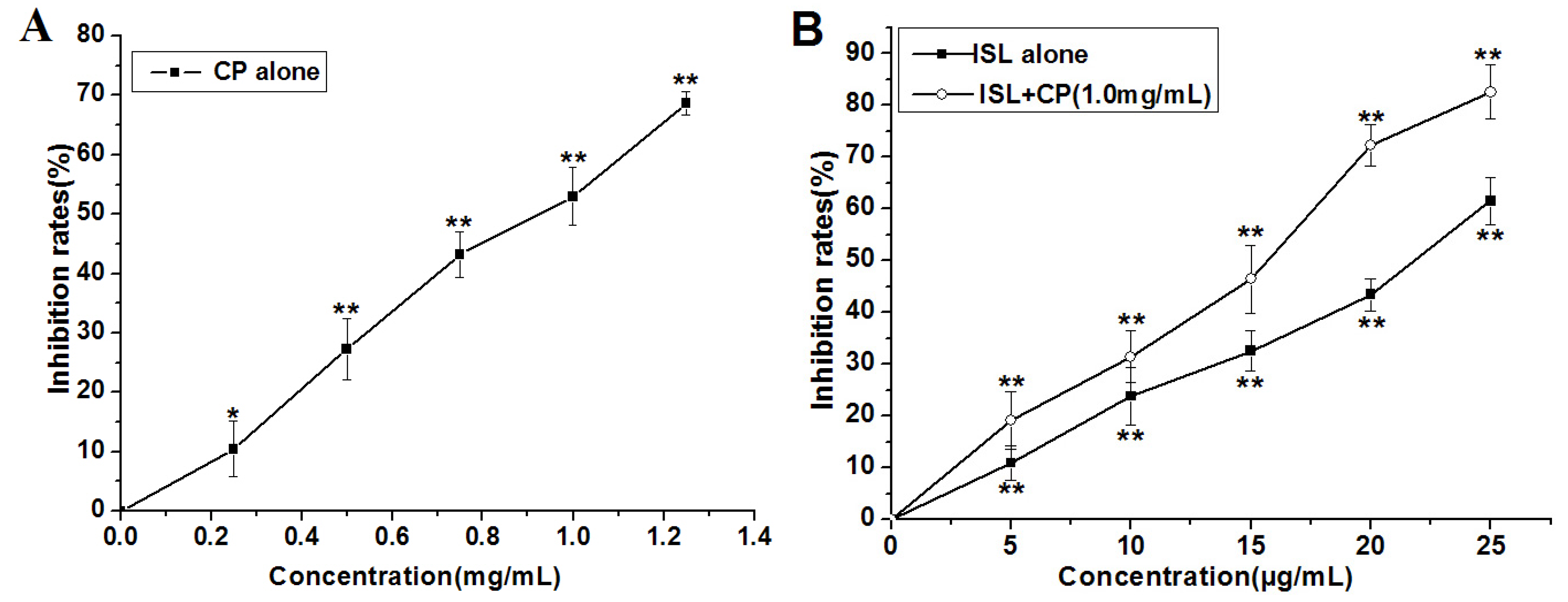
2.1. High Concentration of ISL Significantly Enhances CP-Induced Inhibition of U14 Cells Proliferation in Vitro
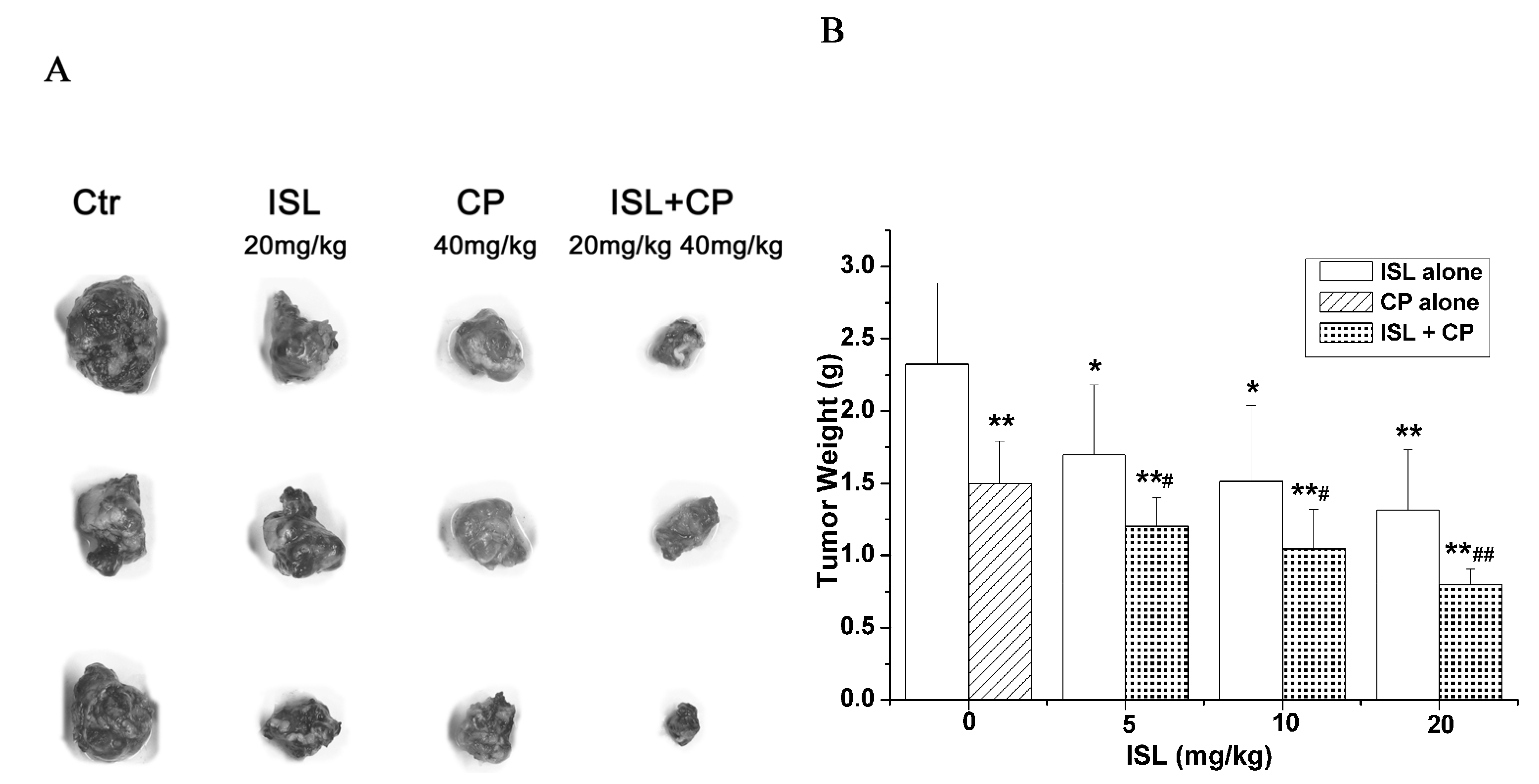
2.2. Co-Treatment with ISL and CP Synergestically Decreases the Tumour Growth in Vivo
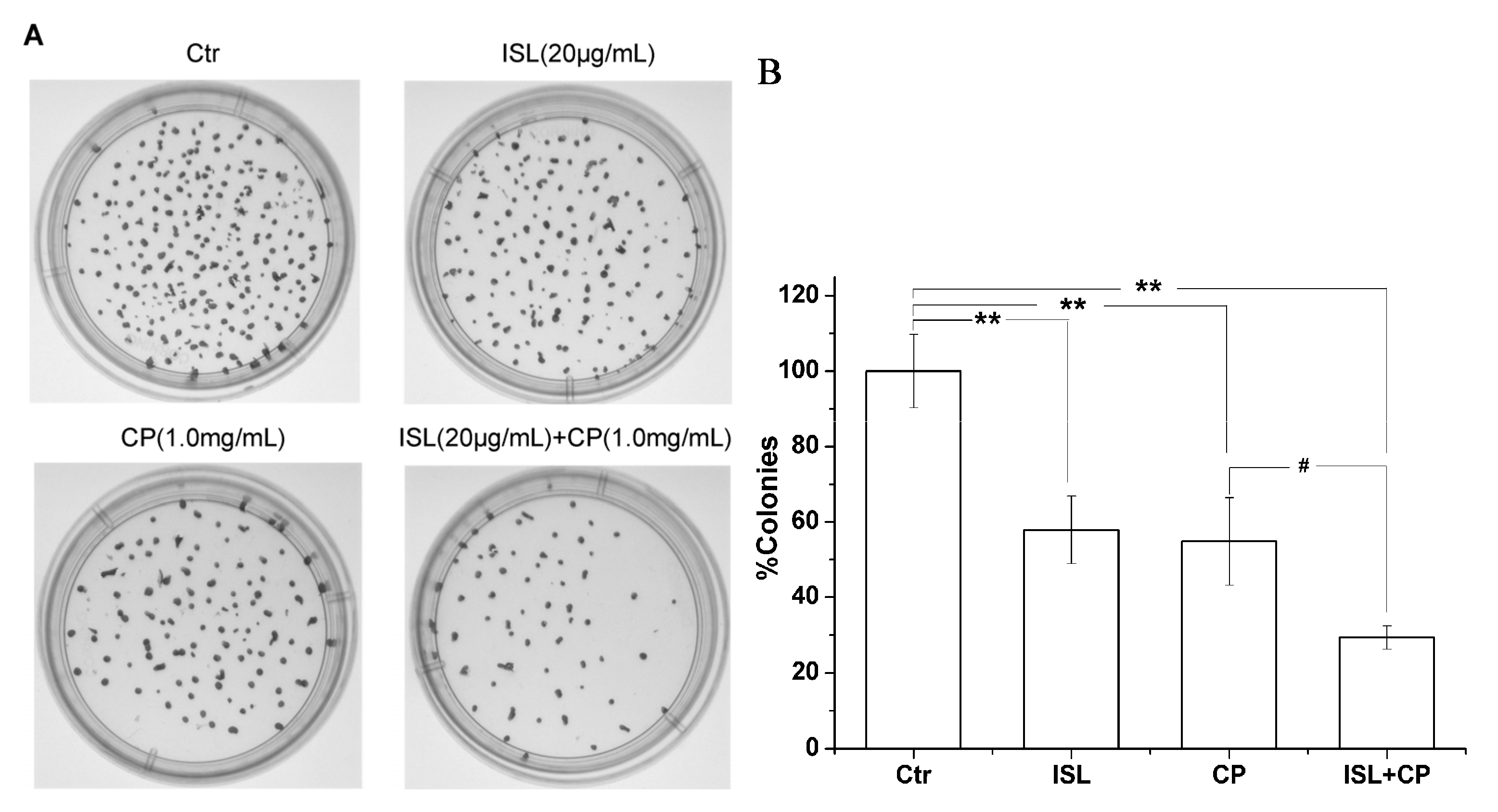
2.3. ISL Inhibits the Micronuclei Yield Induced by CP

2.4. ISL Inhibits the DNA Damage Induced by CP
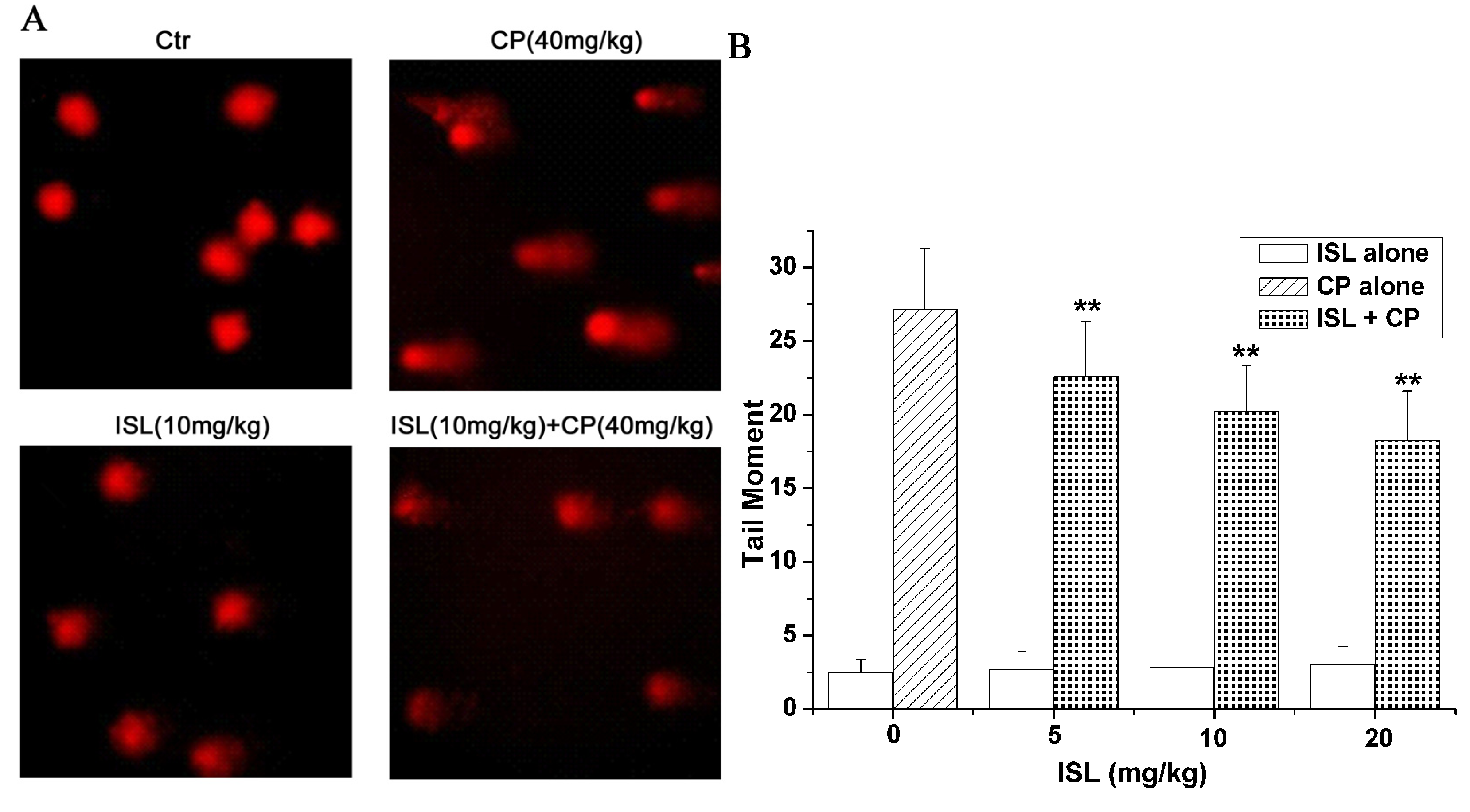
2.5. Discussion
3. Experimental
3.1. Materials
3.2. Animal Preparation
3.3. Cell Culture
3.4. Cell Viability Assay
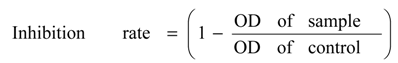
3.5. Colony Formation Assay
3.6. In Vivo Antitumour Activity Assay
3.7. Micronucleus Formation Assay
3.8. Comet Assay
3.9. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Jemal, A.; Bray, F.; Center, M.M.; Ferlay, J.; Ward, E.; Forman, D. Global cancer statistics. CA Cancer J. Clin. 2011, 61, 69–90. [Google Scholar] [CrossRef]
- Attar, E.C.; Ervin, T.; Janicek, M.; Deykin, A.; Godleski, J. Side effects of chemotherapy. Case 3. Acute interstitial pneumonitis related to gemcitabine. J. Clin. Oncol. 2000, 18, 697–698. [Google Scholar]
- Kerbel, R.S.; Kamen, B.A. The anti-angiogenic basis of metronomic chemotherapy. Nat. Rev. Cancer 2004, 4, 423–436. [Google Scholar] [CrossRef]
- Hanafelt, T.D.; Lin, T.; Geyer, S.M.; Zent, C.S.; Leung, N.; Kabat, B.; Bowen, D.; Grever, M.R.; Byrd, J.C.; Kay, N.E. Pentostatin, cyclophosphamide, and rituximab regimen in older patients with chronic lymphocytic leukemia. Cancer 2007, 109, 2291–2298. [Google Scholar] [CrossRef]
- Perroud, H.A.; Rico, M.J.; Alasino, C.M.; Queralt, F.; Mainetti, L.E.; Pezzotto, S.M.; Rozados, V.R.; Scharovsky, O.G. Safety and therapeutic effect of metronomic chemotherapy with cyclophosphamide and celecoxib in advanced breast cancer patients. Future Oncol. 2013, 9, 451–462. [Google Scholar] [CrossRef]
- Lebeau, B.; Chouaid, C.; Baud, M.; Masanes, M.J.; Febvre, M. Oral second- and third-line lomustine-etoposide-cyclophosphamide chemotherapy for small cell lung cancer. Lung Cancer 2010, 67, 188–193. [Google Scholar] [CrossRef]
- Hoffman, M.S.; Roberts, W.S.; Bryson, S.C.; Kavanagh, J.J., Jr.; Cavanagh, D.; Lyman, G.H. Treatment of recurrent and metastatic cervical cancer with cis-platin, doxorubicin, and cyclophosphamide. Gynecol. Oncol. 1988, 29, 32–36. [Google Scholar] [CrossRef]
- Oki, Y.; Ogura, M.; Kato, H.; Kikuchi, A.; Taji, H.; Kagami, Y.; Oshiro, A.; Tsujimura, A.; Yamamoto, K.; Morishima, Y. Phase II study of a salvage regimen using cyclophosphamide, high-dose cytarabine, dexamethasone, etoposide, and rituximab in patients with relapsed or refractory B-cell non-Hodgkin’s lymphoma. Cancer Sci. 2008, 99, 179–184. [Google Scholar]
- Meistrich, M.L.; Wilson, G.; Brown, B.W.; da Cunha, M.F.; Lipshultz, L.I. Impact of cyclophosphamide on long-term reduction in sperm count in men treated with combination chemotherapy for Ewing and soft tissue sarcomas. Cancer 1992, 70, 2703–2712. [Google Scholar] [CrossRef]
- Franke, S.I.; Pra, D.; da Silva, J.; Erdtmann, B.; Henriques, J.A. Possible repair action of vitamin C on DNA damage induced by methyl methanesulfonate, cyclophosphamide, FeSO4 and CuSO4 in mouse blood cells in vivo. Mutat. Res. 2005, 583, 75–84. [Google Scholar] [CrossRef]
- Rehman, M.U.; Tahir, M.; Ali, F.; Qamar, W.; Lateef, A.; Khan, R.; Quaiyoom, A.; Oday, O.H.; Sultana, S. Cyclophosphamide-induced nephrotoxicity, genotoxicity, and damage in kidney genomic DNA of Swiss albino mice: The protective effect of Ellagic acid. Mol. Cell Biochem. 2012, 365, 119–127. [Google Scholar] [CrossRef]
- Zhang, Q.H.; Wu, C.F.; Duan, L.; Yang, J.Y. Protective effects of ginsenoside Rg(3) against cyclophosphamide-induced DNA damage and cell apoptosis in mice. Arch. Toxicol. 2008, 82, 117–123. [Google Scholar] [CrossRef]
- Fintelmann, V. Modern phytotherapy and its uses in gastrointestinal conditions. Planta Med. 1991, 57, S48–S52. [Google Scholar] [CrossRef]
- Kamei, J.; Saitoh, A.; Asano, T.; Nakamura, R.; Ichiki, H.; Iiduka, A.; Kubo, M. Pharmacokinetic and pharmacodynamic profiles of the antitussive principles of Glycyrrhizae radix (licorice), a main component of the Kampo preparation Bakumondo-to (Mai-men-dong-tang). Eur. J. Pharmacol. 2005, 507, 163–168. [Google Scholar] [CrossRef]
- Haggag, E.G.; Abou-Moustafa, M.A.; Boucher, W.; Theoharides, T.C. The effect of a herbal water-extract on histamine release from mast cells and on allergic asthma. J. Herb. Pharmacother. 2003, 3, 41–54. [Google Scholar] [CrossRef]
- Fukai, T.; Satoh, K.; Nomura, T.; Sakagami, H. Preliminary evaluation of antinephritis and radical scavenging activities of glabridin from Glycyrrhiza glabra. Fitoterapia 2003, 74, 624–629. [Google Scholar] [CrossRef]
- Yokota, T.; Nishio, H.; Kubota, Y.; Mizoguchi, M. The inhibitory effect of glabridin from licorice extracts on melanogenesis and inflammation. Pigment Cell Res. 1998, 11, 355–361. [Google Scholar] [CrossRef]
- Inoue, H.; Saito, H.; Koshihara, Y.; Murota, S. Inhibitory effect of glycyrrhetinic acid derivatives on lipoxygenase and prostaglandin synthetase. Chem. Pharm. Bull. (Tokyo) 1986, 34, 897–901. [Google Scholar] [CrossRef]
- Zhou, S.; Koh, H.L.; Gao, Y.; Gong, Z.Y.; Lee, E.J. Herbal bioactivation: The good, the bad and the ugly. Life Sci. 2004, 74, 935–968. [Google Scholar] [CrossRef]
- Haraguchi, H.; Ishikawa, H.; Mizutani, K.; Tamura, Y.; Kinoshita, T. Antioxidative and superoxide scavenging activities of retrochalcones in Glycyrrhiza inflata. Bioorg. Med. Chem. 1998, 6, 339–347. [Google Scholar] [CrossRef]
- Tawata, M.; Aida, K.; Noguchi, T.; Ozaki, Y.; Kume, S.; Sasaki, H.; Chin, M.; Onaya, T. Anti-platelet action of isoliquiritigenin, an aldose reductase inhibitor in licorice. Eur. J. Pharmacol. 1992, 212, 87–92. [Google Scholar] [CrossRef]
- Tamir, S.; Eizenberg, M.; Somjen, D.; Izrael, S.; Vaya, J. Estrogen-like activity of glabrene and other constituents isolated from licorice root. J. Steroid Biochem. Mol. Biol. 2001, 78, 291–298. [Google Scholar] [CrossRef]
- Chowdhury, S.A.; Kishino, K.; Satoh, R.; Hashimoto, K.; Kikuchi, H.; Nishikawa, H.; Shirataki, Y.; Sakagami, H. Tumor-specificity and apoptosis-inducing activity of stilbenes and flavonoids. Anticancer Res. 2005, 25, 2055–2063. [Google Scholar]
- Zhou, G.S.; Song, L.J.; Yang, B. Isoliquiritigenin inhibits proliferation and induces apoptosis of U87 human glioma cells in vitro. Mol. Med. Rep. 2013, 7, 531–536. [Google Scholar]
- Kim, D.C.; Ramachandran, S.; Baek, S.H.; Kwon, S.H.; Kwon, K.Y.; Cha, S.D.; Bae, I.; Cho, C.H. Induction of growth inhibition and apoptosis in human uterine leiomyoma cells by isoliquiritigenin. Reprod. Sci. 2008, 15, 552–558. [Google Scholar] [CrossRef]
- Takahashi, T.; Takasuka, N.; Iigo, M.; Baba, M.; Nishino, H.; Tsuda, H.; Okuyama, T. Isoliquiritigenin, a flavonoid from licorice, reduces prostaglandin E2 and nitric oxide, causes apoptosis, and suppresses aberrant crypt foci development. Cancer Sci. 2004, 95, 448–453. [Google Scholar] [CrossRef]
- Jung, J.I.; Chung, E.; Seon, M.R.; Shin, H.K.; Kim, E.J.; Lim, S.S.; Chung, W.Y.; Park, K.K.; Park, J.H. Isoliquiritigenin (ISL) inhibits ErbB3 signaling in prostate cancer cells. Biofactors 2006, 28, 159–168. [Google Scholar] [CrossRef]
- Hsu, Y.L.; Kuo, P.L.; Lin, L.T.; Lin, C.C. Isoliquiritigenin inhibits cell proliferation and induces apoptosis in human hepatoma cells. Planta Med. 2005, 71, 130–134. [Google Scholar] [CrossRef]
- Wang, K.L.; Hsia, S.M.; Chan, C.J.; Chang, F.Y.; Huang, C.Y.; Bau, D.T.; Wang, P.S. Inhibitory effects of isoliquiritigenin on the migration and invasion of human breast cancer cells. Expert Opin. Ther. Targets 2013, 17, 337–349. [Google Scholar] [CrossRef]
- Yamazaki, S.; Morita, T.; Endo, H.; Hamamoto, T.; Baba, M.; Joichi, Y.; Kaneko, S.; Okada, Y.; Okuyama, T.; Nishino, H.; et al. Isoliquiritigenin suppresses pulmonary metastasis of mouse renal cell carcinoma. Cancer Lett. 2002, 183, 23–30. [Google Scholar] [CrossRef]
- Li, D.; Wang, Z.; Chen, H.; Wang, J.; Zheng, Q.; Shang, J.; Li, J. Isoliquiritigenin induces monocytic differentiation of HL-60 cells. Free Radic. Biol. Med. 2009, 46, 731–736. [Google Scholar] [CrossRef]
- Chen, H.; Zhang, B.; Yao, Y.; Chen, N.; Chen, X.; Tian, H.; Wang, Z.; Zheng, Q. NADPH oxidase-derived reactive oxygen species are involved in the HL-60 cell monocytic differentiation induced by isoliquiritigenin. Molecules 2012, 17, 13424–13438. [Google Scholar] [CrossRef]
- Chen, X.; Zhang, B.; Yuan, X.; Yang, F.; Liu, J.; Zhao, H.; Liu, L.; Wang, Y.; Wang, Z.; Zheng, Q. Isoliquiritigenin-induced differentiation in mouse melanoma B16F0 cell line. Oxid. Med. Cell Longev. 2012, 2012, 534934. [Google Scholar]
- Yuan, X.; Zhang, B.; Chen, N.; Chen, X.Y.; Liu, L.L.; Zheng, Q.S.; Wang, Z.P. Isoliquiritigenin treatment induces apoptosis by increasing intracellular ROS levels in HeLa cells. J. Asian Nat. Prod. Res. 2012, 14, 789–798. [Google Scholar] [CrossRef]
- Yuan, X.; Zhang, B.; Gan, L.; Wang, Z.H.; Yu, B.C.; Liu, L.L.; Zheng, Q.S.; Wang, Z.P. Involvement of the Mitochondrion-dependent and the endoplasmic reticulum stress-signaling pathways in isoliquiritigenin-induced apoptosis of HeLa Cell. Biomed. Environ. Sci. 2013, 26, 268–276. [Google Scholar]
- Yuan, X.; Yu, B.; Wang, Y.; Jiang, J.; Liu, L.; Zhao, H.; Qi, W.; Zheng, Q. Involvement of endoplasmic reticulum stress in isoliquiritigenin-induced SKOV-3 cell apoptosis. Recent Pat. Anticancer Drug Discov. 2013, 8, 191–199. [Google Scholar]
- Yuan, X.; Li, D.; Chen, H.; Sun, C.; Chen, N.; Wang, Z.; Zheng, Q. Isoliquiritigen Enhances Antitumour Activity of Cyclophosphamid. In Proceedings of the 2010 International Conference on Bioinformatics and Biomedical Technology, Chengdu, China, 18–20 June 2010; pp. 170–173.
- Kaldor, J.M.; Day, N.E.; Clarke, E.A.; van Leeuwen, F.E.; Henry-Amar, M.; Fiorentino, M.V.; Bell, J.; Pedersen, D.; Band, P.; Assouline, D.; et al. Leukemia following Hodgkin’s disease. N. Engl. J. Med. 1990, 322, 7–13. [Google Scholar] [CrossRef]
- Levine, E.G.; Bloomfield, C.D. Leukemias and myelodysplastic syndromes secondary to drug, radiation, and environmental exposure. Semin. Oncol. 1992, 19, 47–84. [Google Scholar]
- Yokoyama, Y.; Futagami, M.; Fukushi, Y.; Sakamoto, T.; Higuchi, T.; Fujii, S.; Sato, S.; Takami, H.; Saito, Y. Secondary acute nonlymphocytic leukemia following successful chemotherapy combining cisplatin, doxorubicin, and cyclophosphamide for stage IV epithelial ovarian cancer. Arch. Gynecol. Obstet. 2000, 263, 206–207. [Google Scholar] [CrossRef]
- Agarwala, S.; Hemal, A.K.; Seth, A.; Gupta, A.K.; Bhatnagar, V.; Mitra, D.K. Transitional cell carcinoma of the urinary bladder following exposure to cyclophosphamide in childhood. Eur. J. Pediatr. Surg. 2001, 11, 207–210. [Google Scholar] [CrossRef]
- Heller, A.; Trifonov, V.; Rubtsov, N.; Sauerbrey, A.; Starke, H.; Loncarevic, I.F.; Claussen, U.; Liehr, T. Complex chromosomal rearrangements in a secondary acute myeloblastic leukemia after chemotherapy in TRAPS. Oncol. Rep. 2003, 10, 1789–1792. [Google Scholar]
- Habs, M.R.; Schmahl, D. Prevention of urinary bladder tumors in cyclophosphamide-treated rats by additional medication with the uroprotectors sodium 2-mercaptoethane sulfonate (mesna) and disodium 2,2'-dithio-bis-ethane sulfonate (dimesna). Cancer 1983, 51, 606–609. [Google Scholar] [CrossRef]
- Jenderny, J.; Walk, R.A.; Hackenberg, U.; Rohrborn, G. Chromosomal abnormalities and sister-chromatid exchange in bone marrow cells of mice and Chinese hamsters after inhalation and intraperitoneal administration. II. Cyclophosphamide. Mutat. Res. 1988, 203, 1–10. [Google Scholar] [CrossRef]
- Agrawal, R.C.; Kumar, S. Prevention of cyclophosphamide-induced micronucleus formation in mouse bone marrow by indole-3-carbinol. Food Chem. Toxicol. 1998, 36, 975–977. [Google Scholar] [CrossRef]
- Ozcan, A.; Korkmaz, A.; Oter, S.; Coskun, O. Contribution of flavonoid antioxidants to the preventive effect of mesna in cyclophosphamide-induced cystitis in rats. Arch. Toxicol. 2005, 79, 461–465. [Google Scholar] [CrossRef]
- Lee, C.K.; Son, S.H.; Park, K.K.; Park, J.H.; Lim, S.S.; Chung, W.Y. Isoliquiritigenin inhibits tumor growth and protects the kidney and liver against chemotherapy-induced toxicity in a mouse xenograft model of colon carcinoma. J. Pharmacol. Sci. 2008, 106, 444–451. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Yallapu, M.M.; Maher, D.M.; Sundram, V.; Bell, M.C.; Jaggi, M.; Chauhan, S.C. Curcumin induces chemo/radio-sensitization in ovarian cancer cells and curcumin nanoparticles inhibit ovarian cancer cell growth. J. Ovarian. Res. 2010. [Google Scholar] [CrossRef]
- Tinwell, H.; Ashby, J. Comparison of acridine orange and Giemsa stains in several mouse bone marrow micronucleus assays—Including a triple dose study. Mutagenesis 1989, 4, 476–481. [Google Scholar] [CrossRef]
- Wang, Z.; Zheng, Q.; Liu, K.; Li, G.; Zheng, R. Ginsenoside Rh(2) enhances antitumour activity and decreases genotoxic effect of cyclophosphamide. Basic Clin. Pharmacol. Toxicol. 2006, 98, 411–415. [Google Scholar]
- Konca, K.; Lankoff, A.; Banasik, A.; Lisowska, H.; Kuszewski, T.; Gozdz, S.; Koza, Z.; Wojcik, A. A cross-platform public domain PC image-analysis program for the comet assay. Mutat. Res. 2003, 534, 15–20. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds Isoliquiritigenin and Cyclophosphamide are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhao, H.; Yuan, X.; Li, D.; Chen, H.; Jiang, J.; Wang, Z.; Sun, X.; Zheng, Q. Isoliquiritigen Enhances the Antitumour Activity and Decreases the Genotoxic Effect of Cyclophosphamide. Molecules 2013, 18, 8786-8798. https://doi.org/10.3390/molecules18088786
Zhao H, Yuan X, Li D, Chen H, Jiang J, Wang Z, Sun X, Zheng Q. Isoliquiritigen Enhances the Antitumour Activity and Decreases the Genotoxic Effect of Cyclophosphamide. Molecules. 2013; 18(8):8786-8798. https://doi.org/10.3390/molecules18088786
Chicago/Turabian StyleZhao, Hong, Xuan Yuan, Defang Li, Hongmei Chen, Jiangtao Jiang, Zhiping Wang, Xiling Sun, and Qiusheng Zheng. 2013. "Isoliquiritigen Enhances the Antitumour Activity and Decreases the Genotoxic Effect of Cyclophosphamide" Molecules 18, no. 8: 8786-8798. https://doi.org/10.3390/molecules18088786
APA StyleZhao, H., Yuan, X., Li, D., Chen, H., Jiang, J., Wang, Z., Sun, X., & Zheng, Q. (2013). Isoliquiritigen Enhances the Antitumour Activity and Decreases the Genotoxic Effect of Cyclophosphamide. Molecules, 18(8), 8786-8798. https://doi.org/10.3390/molecules18088786



