Preventive Effects of Eleutherococcus senticosus Bark Extract in OVX-Induced Osteoporosis in Rats
Abstract
:1. Introduction
2. Results and Discussion
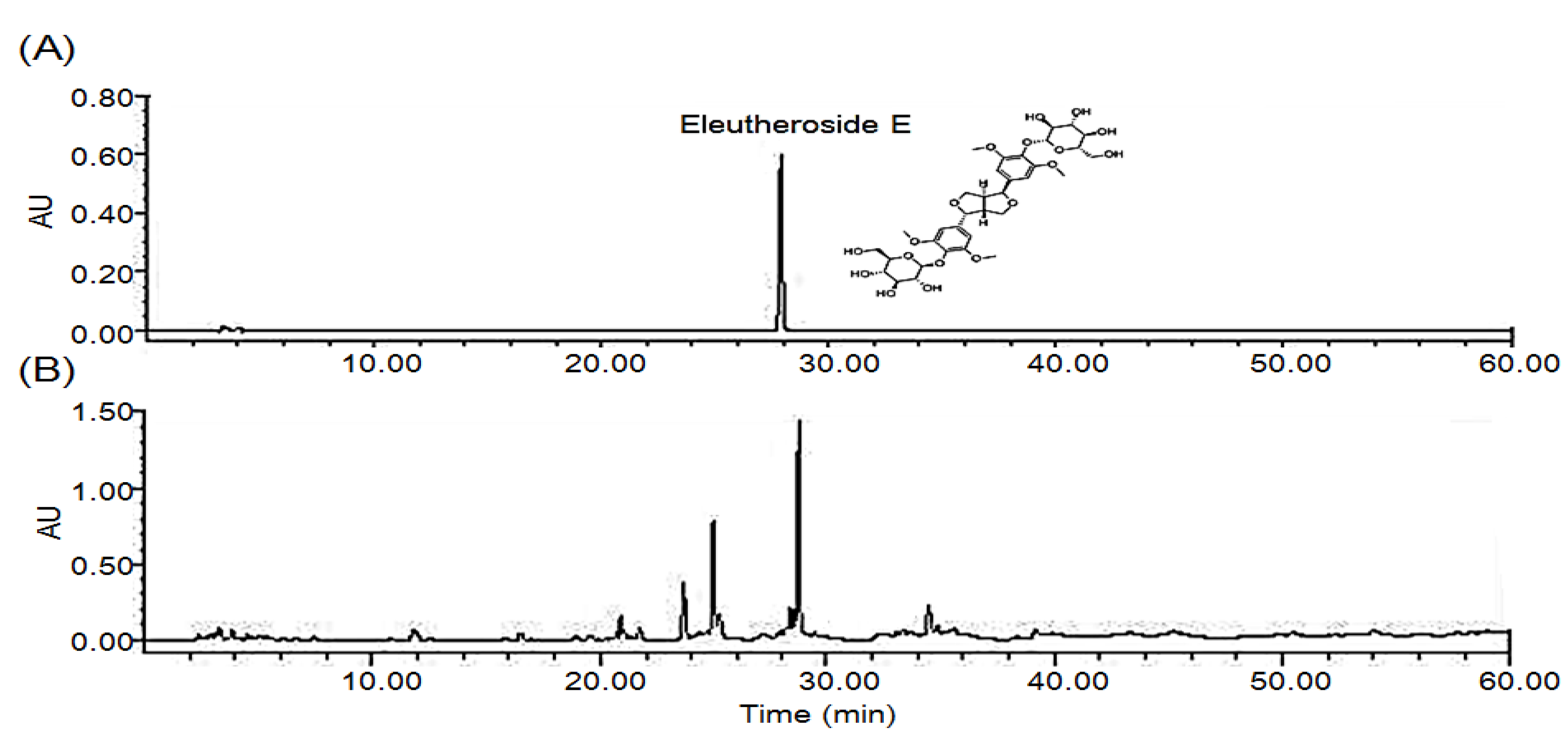
2.1. HPLC Chromatograms for Standardization of DES
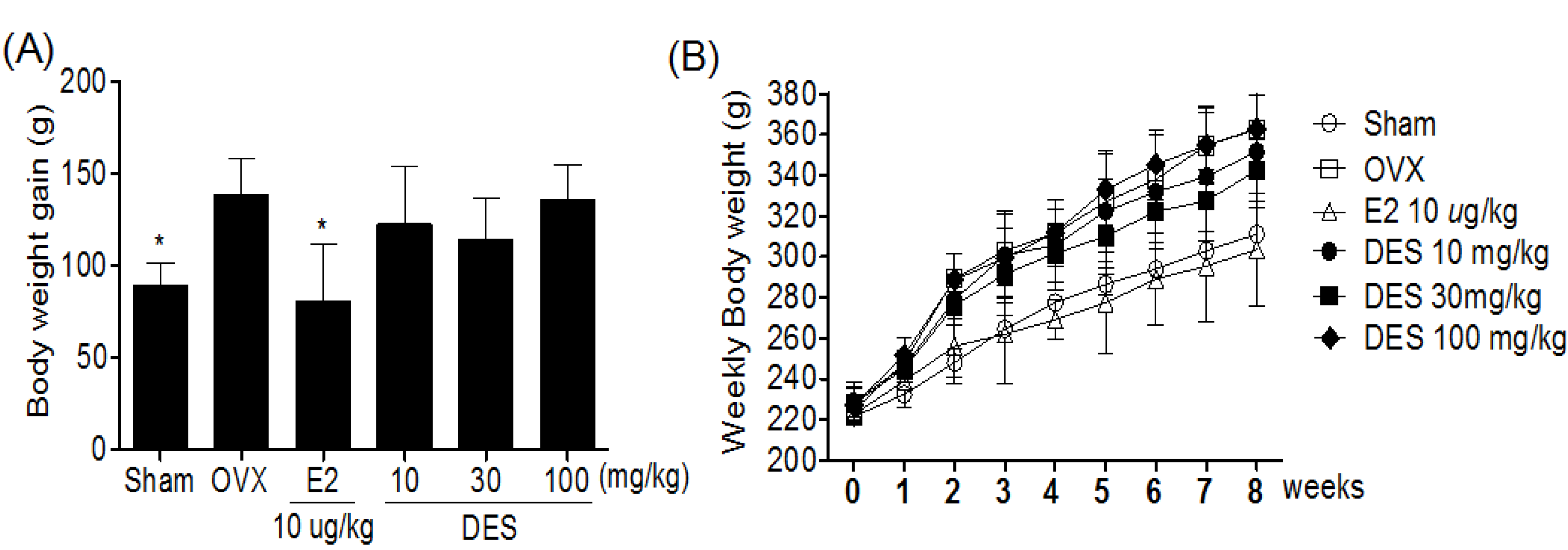
2.2. Weekly Body Weight in Treatments of DES
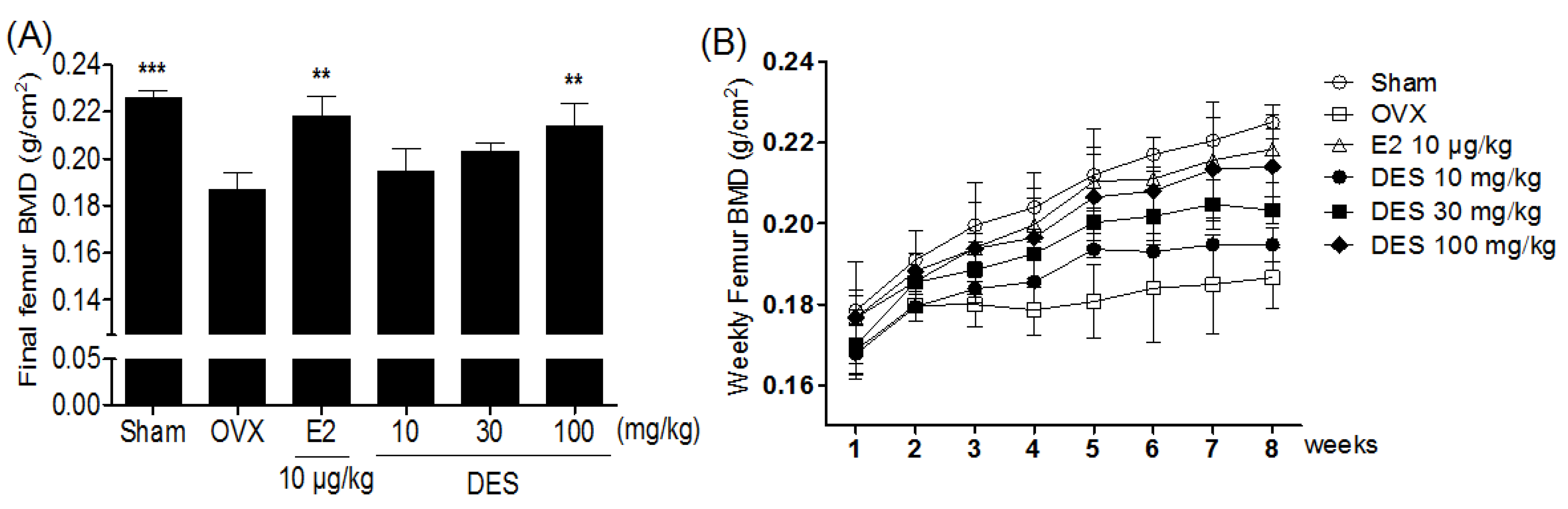
2.3. Bone Mineral Density of the femur in Treatments of DES
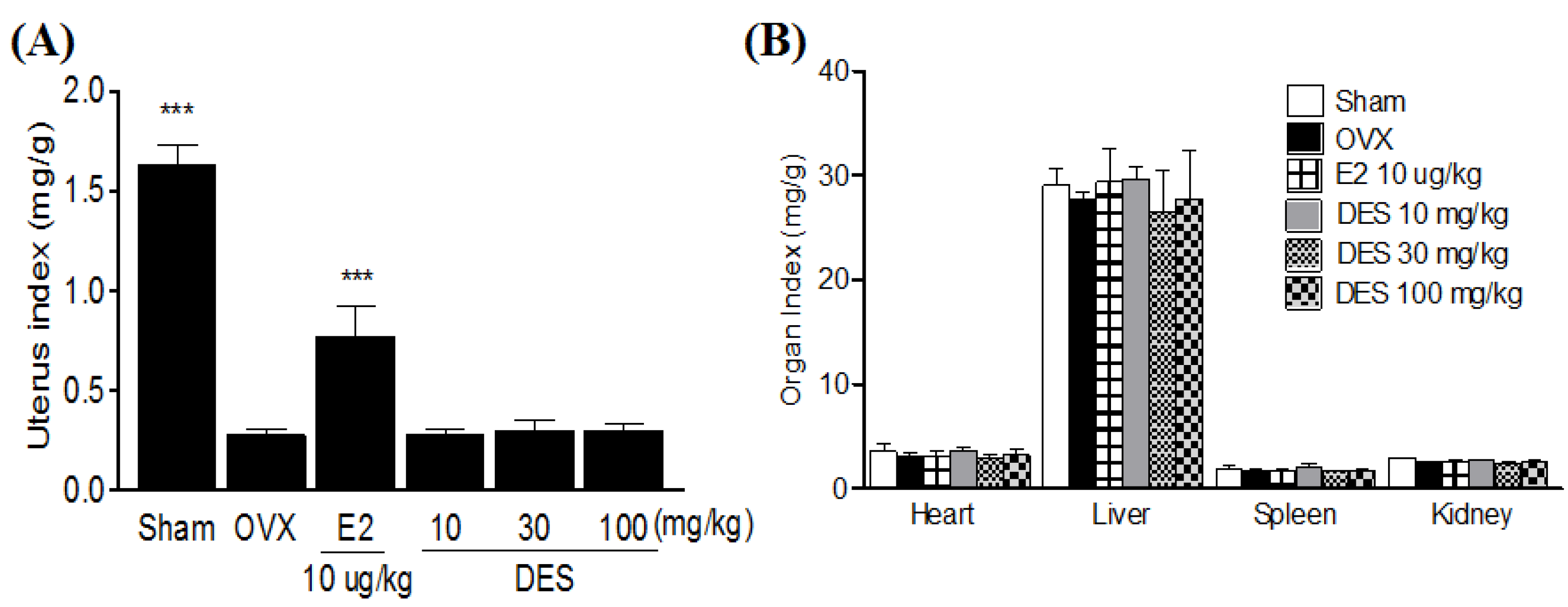
2.4. Uterus and Organ Index in Treatments of DES
2.5. Serum Bone Marker in Treatments of DES
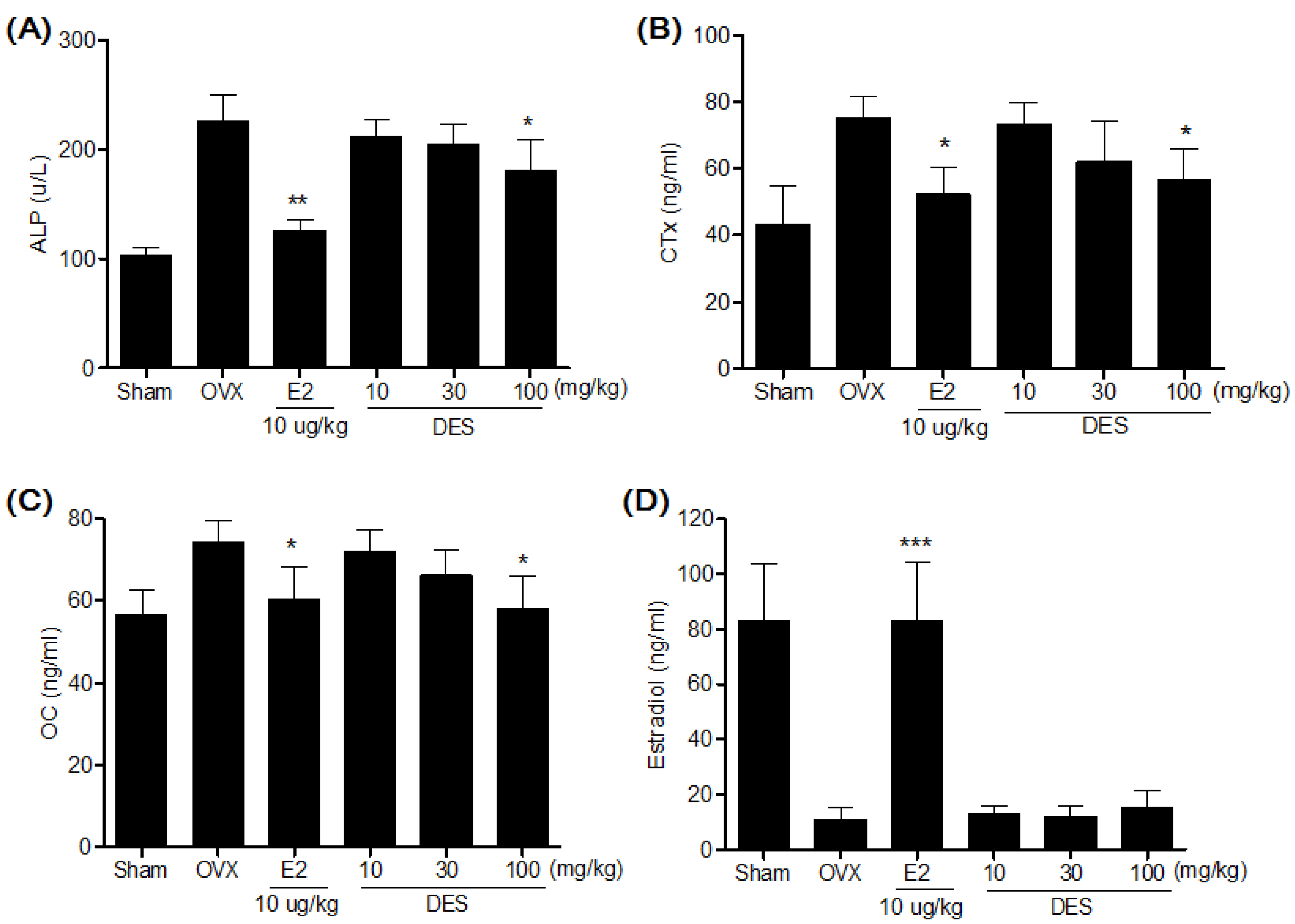
2.6. Discussion
3. Experimental
3.1. Sample Preparation and HPLC Analysis
3.2. Animals and Treatments
3.3. Bone Mineral Density Measurements
3.4. Serum Estradiol and Bone marker Analysis
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Compston, J. Clinical and therapeutic aspects of osteoporosis. Eur. J. Radiol. 2009, 71, 388–391. [Google Scholar] [CrossRef]
- Hansen, M.A.; Overgaard, K.; Riis, B.J.; Christiansen, C. Potential risk factors for development of postmenopausal osteoporosis—Examined over a 12-year period. Osteoporos. Int. 1991, 1, 95–102. [Google Scholar] [CrossRef]
- Miazgowski, T.; Kleerekoper, M.; Felsenberg, D.; Stepan, J.J.; Szulc, P. Secondary osteoporosis: endocrine and metabolic causes of bone mass deterioration. J. Osteoporos. 2012, 2012, 907214. [Google Scholar]
- Nelson, H.D.; Humphrey, L.L.; Nygren, P.; Teutsch, S.M.; Allan, J.D. Postmenopausal hormone replacement therapy: scientific review. JAMA 2002, 288, 872–881. [Google Scholar] [CrossRef]
- Pradhan, A.D.; Manson, J.E.; Rossouw, J.E.; Siscovick, D.S.; Mouton, C.P.; Rifai, N.; Wallace, R.B.; Jackson, R.D.; Pettinger, M.B.; Ridker, P.M. Inflammatory biomarkers, hormone replacement therapy, and incident coronary heart disease: Prospective analysis from the Women’s Health Initiative observational study. JAMA 2002, 288, 980–987. [Google Scholar] [CrossRef]
- Reid, I.R. Bisphosphonate therapy for secondary osteoporosis: Adult perspective. Horm. Res. Paediatr. 2011, 76, 28–32. [Google Scholar] [CrossRef]
- McClung, M.; Harris, S.T.; Miller, P.D.; Bauer, D.C.; Davison, K.S.; Dian, L.; Hanley, D.A.; Kendler, D.L.; Yuen, C.K.; Lewiecki, E.M. Bisphosphonate therapy for osteoporosis: Benefits, risks, and drug holiday. Am. J. Med. 2013, 126, 13–20. [Google Scholar] [CrossRef]
- Banu, J.; Varela, E.; Fernandes, G. Alternative therapies for the prevention and treatment of osteoporosis. Nutr. Rev. 2012, 70, 22–40. [Google Scholar] [CrossRef]
- Tseng, S.H.; Sung, H.C.; Chen, L.G.; Lai, Y.J.; Wang, K.T.; Sung, C.H.; Wang, C.C. Effects of velvet antler with blood on bone in ovariectomized rats. Molecules 2012, 17, 10574–10585. [Google Scholar] [CrossRef]
- Wu, Y.B.; Zheng, C.J.; Qin, L.P.; Sun, L.N.; Han, T.; Jiao, L.; Zhang, Q.Y.; Wu, J.Z. Antiosteoporotic activity of anthraquinones from Morinda officinalis on osteoblasts and osteoclasts. Molecules 2009, 14, 573–583. [Google Scholar] [CrossRef]
- Lim, D.W.; Kim, Y.T. Dried Root of Rehmannia glutinosa Prevents Bone Loss in Ovariectomized Rats. Molecules 2013, 18, 5804–5813. [Google Scholar] [CrossRef]
- Wang, X.; Hai, C.X.; Liang, X.; Yu, S.X.; Zhang, W.; Li, Y.L. The protective effects of Acanthopanax senticosus Harms aqueous extracts against oxidative stress: role of Nrf2 and antioxidant enzymes. J. Ethnopharmacol. 2010, 127, 424–432. [Google Scholar] [CrossRef]
- Jung, H.J.; Park, H.J.; Kim, R.G.; Shin, K.M.; Ha, J.; Choi, J.W.; Kim, H.J.; Lee, Y.S.; Lee, K.T. In vivo anti-inflammatory and antinociceptive effects of liriodendrin isolated from the stem bark of Acanthopanax senticosus. Planta Med. 2003, 69, 610–616. [Google Scholar] [CrossRef]
- Yoon, T.J.; Yoo, Y.C.; Lee, S.W.; Shin, K.S.; Choi, W.H.; Hwang, S.H.; Ha, E.S.; Jo, S.K.; Kim, S.H.; Park, W.M. Anti-metastatic activity of Acanthopanax senticosus extract and its possible immunological mechanism of action. J. Ethnopharmacol. 2004, 93, 247–253. [Google Scholar] [CrossRef]
- Kimura, Y.; Sumiyoshi, M. Effects of various Eleutherococcus senticosus cortex on swimming time, natural killer activity and corticosterone level in forced swimming stressed mice. J. Ethnopharmacol. 2004, 95, 447–453. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, S.G.; Kang, S.K.; Chung, S.H. Acanthopanax senticosus reverses fatty liver disease and hyperglycemia in ob/ob mice. Arch. Pharmacal Res. 2006, 29, 768–776. [Google Scholar] [CrossRef]
- Bu, Y.; Jin, Z.H.; Park, S.Y.; Baek, S.; Rho, S.; Ha, N.; Park, S.K.; Kim, H.; Sun Yeo, K. Siberian ginseng reduces infarct volume in transient focal cerebral ischaemia in Sprague-Dawley rats. PTR 2005, 19, 167–169. [Google Scholar]
- Huang, L.; Zhao, H.; Huang, B.; Zheng, C.; Peng, W.; Qin, L. Acanthopanax senticosus: review of botany, chemistry and pharmacology. Die Pharm. 2011, 66, 83–97. [Google Scholar]
- Manolagas, S.C. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis. Endocr. Rev. 2000, 21, 115–137. [Google Scholar] [CrossRef]
- Evans, D.B.; Bunning, R.A.; Russell, R.G. The effects of recombinant human interleukin-1 beta on cellular proliferation and the production of prostaglandin E2, plasminogen activator, osteocalcin and alkaline phosphatase by osteoblast-like cells derived from human bone. Biochem. Biophys. Res. Commun. 1990, 166, 208–216. [Google Scholar] [CrossRef]
- Stashenko, P.; Dewhirst, F.E.; Peros, W.J.; Kent, R.L.; Ago, J.M. Synergistic interactions between interleukin 1, tumor necrosis factor, and lymphotoxin in bone resorption. J. Immunol. 1987, 138, 1464–1468. [Google Scholar]
- Wilkinson, J.M.; Hamer, A.J.; Rogers, A.; Stockley, I.; Eastell, R. Bone mineral density and biochemical markers of bone turnover in aseptic loosening after total hip arthroplasty. J. Orthop. Res. 2003, 21, 691–696. [Google Scholar] [CrossRef]
- Ryu, J.H.; Ahn, H.; Kim, J.Y.; Kim, Y.K. Inhibitory activity of plant extracts on nitric oxide synthesis in LPS-activated macrophages. PTR 2003, 17, 485–489. [Google Scholar]
- Kropotov, A.V.; Kolodnyak, O.L.; Koldaev, V.M. Effects of Siberian ginseng extract and ipriflavone on the development of glucocorticoid-induced osteoporosis. Bull. Exp. Biol. Med. 2002, 133, 252–254. [Google Scholar] [CrossRef]
- Mazziotti, G.; Angeli, A.; Bilezikian, J.P.; Canalis, E.; Giustina, A. Glucocorticoid-induced osteoporosis: an update. TEM 2006, 17, 144–149. [Google Scholar]
- Hwang, Y.C.; Jeong, I.K.; Ahn, K.J.; Chung, H.Y. The effects of Acanthopanax senticosus extract on bone turnover and bone mineral density in Korean postmenopausal women. J. Bone Miner. Metab. 2009, 27, 584–590. [Google Scholar] [CrossRef]
- Hoegh-Andersen, P.; Tanko, L.B.; Andersen, T.L.; Lundberg, C.V.; Mo, J.A.; Heegaard, A.M.; Delaisse, J.M.; Christgau, S. Ovariectomized rats as a model of postmenopausal osteoarthritis: validation and application. Arthritis Res. Therapy 2004, 6, R169–R180. [Google Scholar] [CrossRef]
- Jee, W.S.; Yao, W. Overview: animal models of osteopenia and osteoporosis. J. Musculoskelet. Neuronal Interact. 2001, 1, 193–207. [Google Scholar]
- Lelovas, P.P.; Xanthos, T.T.; Thoma, S.E.; Lyritis, G.P.; Dontas, I.A. The laboratory rat as an animal model for osteoporosis research. Comp. Med. 2008, 58, 424–430. [Google Scholar]
- Bahlous, A.; Kalai, E.; Hadj Salah, M.; Bouzid, K.; Zerelli, L. Biochemical markers of bone remodeling: recent data of their applications in managing postmenopausal osteoporosis. La Tunis. Med. 2006, 84, 751–757. [Google Scholar]
- Thiede, M.A.; Smock, S.L.; Petersen, D.N.; Grasser, W.A.; Thompson, D.D.; Nishimoto, S.K. Presence of messenger ribonucleic acid encoding osteocalcin, a marker of bone turnover, in bone marrow megakaryocytes and peripheral blood platelets. Endocrinology 1994, 135, 929–937. [Google Scholar] [CrossRef]
- Coleman, R.E. The clinical use of bone resorption markers in patients with malignant bone disease. Cancer 2002, 94, 2521–2533. [Google Scholar] [CrossRef]
- Yin, J.; Tezuka, Y.; Subehan; Shi, L.; Nobukawa, M.; Nobukawa, T.; Kadota, S. In vivo anti-osteoporotic activity of isotaxiresinol, a lignan from wood of Taxus yunnanensis. Phytomedicine 2006, 13, 37–42. [Google Scholar] [CrossRef]
- Park, J.A.; Ha, S.K.; Kang, T.H.; Oh, M.S.; Cho, M.H.; Lee, S.Y.; Park, J.H.; Kim, S.Y. Protective effect of apigenin on ovariectomy-induced bone loss in rats. Life Sci. 2008, 82, 1217–1223. [Google Scholar] [CrossRef]
- Devareddy, L.; Khalil, D.A.; Smith, B.J.; Lucas, E.A.; Soung, D.Y.; Marlow, D.D.; Arjmandi, B.H. Soy moderately improves microstructural properties without affecting bone mass in an ovariectomized rat model of osteoporosis. Bone 2006, 38, 686–693. [Google Scholar] [CrossRef]
- Dang, Z.C.; Van Bezooijen, R.L.; Karperien, M.; Papapoulos, S.E.; Lowik, C.W.G.M. Exposure of KS483 cells to estrogen enhances osteogenesis and inhibits adipogenesis. J. Bone Miner. Res. 2002, 17, 394–405. [Google Scholar] [CrossRef]
- Heine, P.A.; Taylor, J.A.; Iwamoto, G.A.; Lubahn, D.B.; Cooke, P.S. Increased adipose tissue in male and female estrogen receptor-alpha knockout mice. Proc. Natl. Acad. Sci. USA 2000, 97, 12729–12734. [Google Scholar] [CrossRef]
- Joyner, J.M.; Hutley, L.J.; Cameron, D.P. Estrogen receptors in human preadipocytes. Endocrine 2001, 15, 225–230. [Google Scholar] [CrossRef]
- Hewitt, S.C.; Korach, K.S. Oestrogen receptor knockout mice: roles for oestrogen receptors alpha and beta in reproductive tissues. Reproduction 2003, 125, 143–149. [Google Scholar] [CrossRef]
- Umland, E.M.; Cauffield, J.S.; Kirk, J.K.; Thomason, T.E. Phytoestrogens as therapeutic alternatives to traditional hormone replacement in postmenopausal women. Pharmacotherapy 2000, 20, 981–990. [Google Scholar] [CrossRef]
- Gaffney, B.T.; Hugel, H.M.; Rich, P.A. The effects of Eleutherococcus senticosus and Panax ginseng on steroidal hormone indices of stress and lymphocyte subset numbers in endurance athletes. Life Sci. 2001, 70, 431–442. [Google Scholar] [CrossRef]
- Sample Availability: Samples of DES are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lim, D.W.; Kim, J.G.; Lee, Y.; Cha, S.H.; Kim, Y.T. Preventive Effects of Eleutherococcus senticosus Bark Extract in OVX-Induced Osteoporosis in Rats. Molecules 2013, 18, 7998-8008. https://doi.org/10.3390/molecules18077998
Lim DW, Kim JG, Lee Y, Cha SH, Kim YT. Preventive Effects of Eleutherococcus senticosus Bark Extract in OVX-Induced Osteoporosis in Rats. Molecules. 2013; 18(7):7998-8008. https://doi.org/10.3390/molecules18077998
Chicago/Turabian StyleLim, Dong Wook, Jae Goo Kim, Youngseok Lee, Seok Ho Cha, and Yun Tai Kim. 2013. "Preventive Effects of Eleutherococcus senticosus Bark Extract in OVX-Induced Osteoporosis in Rats" Molecules 18, no. 7: 7998-8008. https://doi.org/10.3390/molecules18077998
APA StyleLim, D. W., Kim, J. G., Lee, Y., Cha, S. H., & Kim, Y. T. (2013). Preventive Effects of Eleutherococcus senticosus Bark Extract in OVX-Induced Osteoporosis in Rats. Molecules, 18(7), 7998-8008. https://doi.org/10.3390/molecules18077998




