New Resveratrol Oligomer Derivatives from the Roots of Rheum lhasaense
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structural Elucidation of the New Compounds
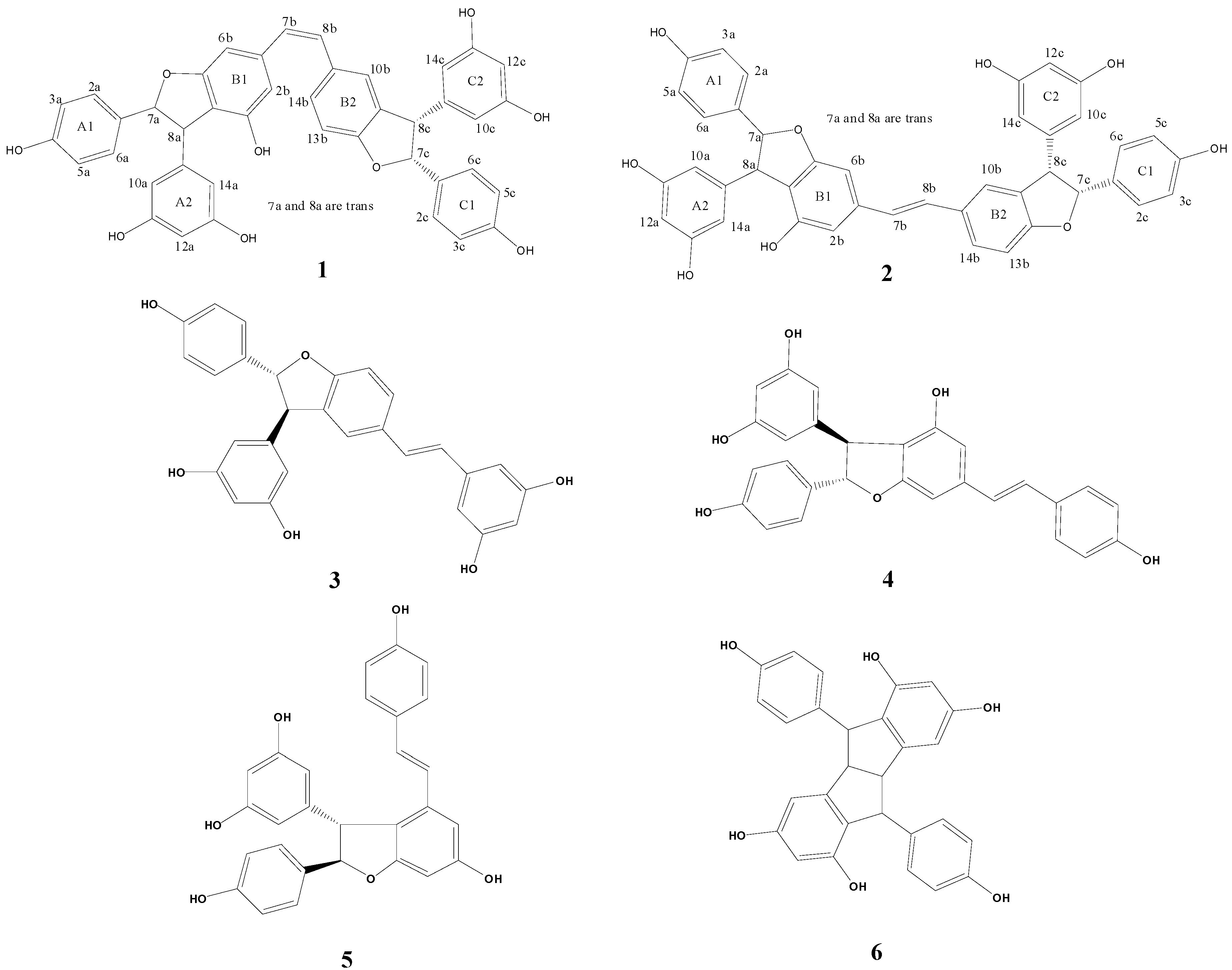
| Positions | 1 | 2 | ||||
|---|---|---|---|---|---|---|
| δH (mult, J in Hz, I) | δC | ROSEY | δH (mult, J in Hz, I) | δC | ROSEY | |
| 1a | 134.2 | 134.3 | ||||
| 2a(6a) | 7.12 (d, 8.5, 2H) | 128.1 | 7a, 8a | 7.13 (d, 8.5, 2H) | 128.1 | 7a, 8a |
| 3a(5a) | 6.77 (d, 8.5, 2H) | 116.3 | 6.76 (d, 8.5, 2H) | 116.3 | ||
| 4a | 158.5 | 158.4 | ||||
| 7a | 5.28 (d, 5.4, 1H) | 94.5 | 5.30 (d, 5.4, 1H) | 94.5 | ||
| 8a | 4.31 (d, 5.4, 1H) | 56.7 | 4.33 (d, 5.4, 1H) | 56.6 | ||
| 9a | 146.5 | 146.6 | ||||
| 10a(14a) | 6.10 (d, 2.0 Hz, 2H) | 107.1 | 7a, 8a | 6.13 (d, 1.7 ,2H) | 107.0 | 7a, 8a |
| 11a(13a) | 159.6 | 159.6 | ||||
| 12a | 6.14 (t, 2.0 Hz, 1H) | 102.0 | 6.16 (t, 1.7, 1H) | 102.0 | ||
| 1b | 141.2 | 141.5 | ||||
| 2b | 6.34 (s, 1H) | 110.1 | 6.50 (s, 1H) | 108.0 | ||
| 3b | 155.5 | 155.6 | ||||
| 4b | 115.1 | 115.4 | ||||
| 5b | 163.0 | 163.3 | ||||
| 6b | 6.32 (s, 1H) | 102.5 | 6.60 (s, 1H) | 99.6 | ||
| 7b | 6.40 (d, 12.2, 1H) | 129.8 | 8b | 6.85 (d, 16.2, 1H) | 127.4 | |
| 8b | 6.46 (d, 12.2, 1H) | 131.0 | 7b | 7.02 (d, 16.2, 1H) | 129.3 | |
| 9b | 131.7 | 132.3 | ||||
| 10b | 6.94 (br s, 1H) | 127.4 | 7.20 (br s, 1H) | 124.1 | ||
| 11b | 131.7 | 132.4 | ||||
| 12b | 160.4 | 160.9 | ||||
| 13b | 6.74 (d, 8.0, 1H) | 109.8 | 6.84 (d, 8.3, 1H) | 110.3 | ||
| 14b | 7.21 (d, 8.0, 1H) | 130.6 | 7.37 (d, 8.3, 1H) | 128.7 | ||
| 1c | 132.9 | 132.8 | ||||
| 2c(6c) | 7.16 (d, 8.5, 2H) | 128.7 | 7c, 8c | 7.17 (d, 8.5, 2H) | 128.7 | 7c, 8c |
| 3c(5c) | 6.75 (d, 8.5, 2H) | 116.3 | 6.79 (d, 8.5, 2H) | 116.3 | ||
| 4c | 158.7 | 158.6 | ||||
| 7c | 5.36 (d, 8.3, 1H) | 94.7 | 8c | 5.39 (d, 8.4, 1H) | 94.9 | 8c |
| 8c | 4.36 (d, 8.3, 1H) | 58.7 | 7c, 10b | 4.40 (d, 8.4, 1H) | 58.7 | 7c, 10b |
| 9c | 145.4 | 145.3 | ||||
| 10c(14c) | 6.10 (d, 2.0, 2H) | 107.7 | 7c, 8c | 6.15 (d, 1.7, 2H) | 107.9 | 7c, 8c |
| 11c(13c) | 159.8 | 159.8 | ||||
| 12c | 6.17 (t, 2.0, 1H) | 102.5 | 6.22 (t, 1.7, 1H) | 102.5 | ||
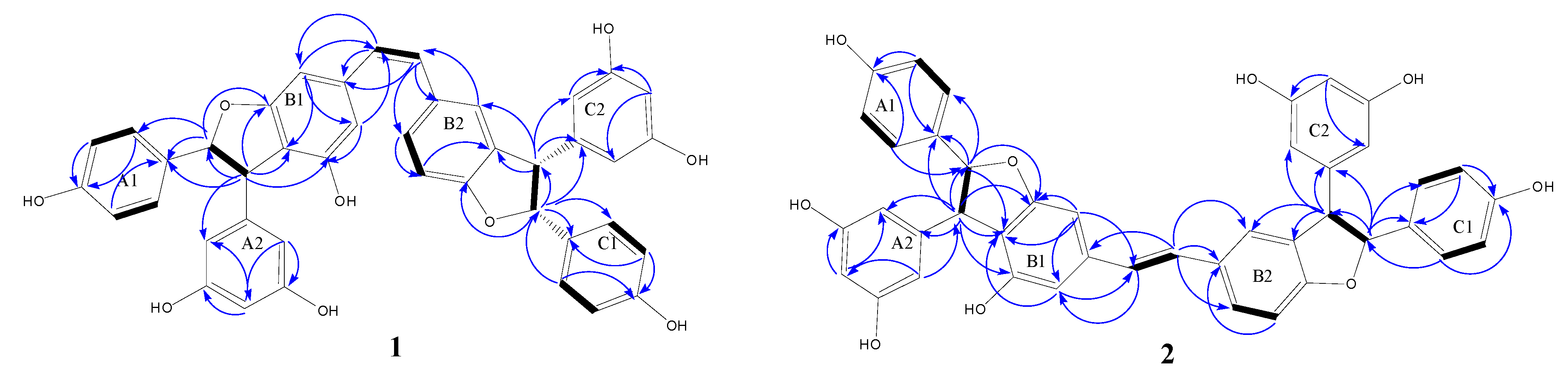
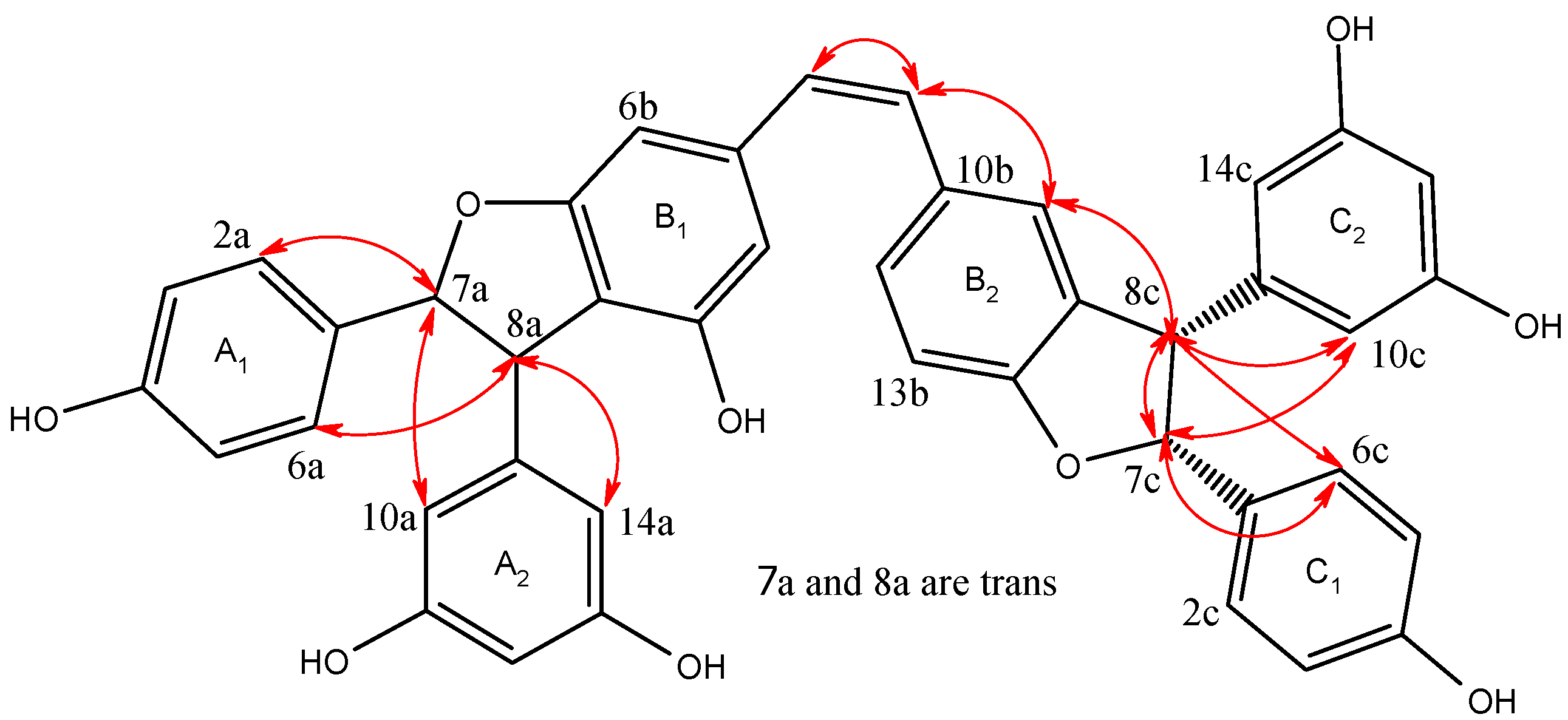
2.2. Antioxidant Activities by DPPH Scavenging Capacities
| Compds. | DPPH radical IC50 (μM) a |
|---|---|
| 1 | 49.7 ± 2.3 |
| 2 | 31.3 ± 1.5 |
| 3 | 28.7 ± 1.0 |
| 4 | 69.8 ± 2.3 |
| 5 | 52.6 ± 1.1 |
| 6 | 190.2 ± 3.8 |
| Vitamin E | 27.9 ± 0.9 |
3. Experimental
3.1. General
3.2. Plant Materials
3.3. Extraction and Isolation of the Compounds
3.4. Spectroscopic Data
3.5. DPPH Assays
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Shen, T.; Wang, X.N.; Lou, H.X. Natural stilbenes: An overview. Nat. Prod. Rep. 2009, 26, 916–935. [Google Scholar] [CrossRef]
- He, S.; Wu, B.; Pan, Y.; Jiang, L. Stilbene oligomers from Parthenocissus laetevirens: Isolation, biomimetic synthesis, absolute configuration, and implication of antioxidative defense system in the plant. J. Org. Chem. 2008, 73, 5233–5241. [Google Scholar] [CrossRef]
- Li, C.; Lu, J.; Xu, X.; Hu, R.; Pan, Y. pH-switched HRP-catalyzed dimerization of resveratrol: A selective biomimetic synthesis. Green Chem. 2012, 14, 3281–3284. [Google Scholar] [CrossRef]
- Xiao, K.; Zhang, H.J.; Xuan, L.J.; Zhang, J.; Xu, Y.M.; Bai, D.L. Stilbenoids: Chemistry and Bioactivities. In Studies in Natural Products Chemistry; Elsevier Science: Amsterdam, The Netherlands, 2008; Volume 34, pp. 453–646. [Google Scholar]
- He, S.; Yan, X. From resveratrol to its derivatives: New sources of natural antioxidant. Curr. Med. Chem. 2013, 20, 1005–1017. [Google Scholar]
- Cichewicz, R.H.; Kouzi, S.A. Resveratrol Oligomers: Structure, Chemistry, and Biological Activity. In Studies in Natural Products Chemistry; Elsevier Science: Amsterdam, The Netherlands, 2002; Volume 26, pp. 507–579. [Google Scholar]
- Rivière, C.; Pawlus, A.D.; Mérillon, J.M. Natural stilbenoids: distribution in the plant kingdom and chemotaxonomic interest in Vitaceae. Nat. Prod. Rep. 2012, 29, 1317–1333. [Google Scholar] [CrossRef]
- Flamini, R.; de Rosso, M.; de Marchi, F.; Dalla Vedova, A.; Panighel, A.; Gardiman, M.; Maoz, I.; Bavaresco, L. An innovative approach to grape metabolomics: stilbene profiling by suspect screening analysis. Metabolomics 2013. [Google Scholar] [CrossRef]
- Sotheeswaran, S.; Pasupathy, V. Distribution of resveratrol oligomers in plants. Phytochemistry 1993, 32, 1083–1092. [Google Scholar] [CrossRef]
- Li, A.; Bao, B.; Alisa, E.G.; Suk-pyo, H.; John, M.; Sergei, L.M.; Hideaki, O.; Chong-wook, P. Polygonaceae. In Flora of China; Science Press & Missouri Botanical Garden: Beijing, China and St. Louis, MO, USA, 2003; Volume 5, pp. 277–350. [Google Scholar]
- Xiao, P.; He, L.; Wang, L. Ethnopharmacologic study of Chinese Rhubarb. J. Ethnopharmacol. 1984, 10, 275–293. [Google Scholar] [CrossRef]
- Xiao, W.; Deng, H.Z.; Ma, Y. Summarization of the clinical and laboratory study on the rhubarb in treating chronic renal failure. Zhongguo Zhongyao Zazhi 2002, 27, 241–242, 262. [Google Scholar]
- Wu, C.Y. Flora of Xizang; Science Press: Beijing, China, 1983; Volume 1, p. 598. [Google Scholar]
- Xiao, P.; Chen, B.Z.; Wang, L.W.; He, L.Y.; Luo, S.R.; Guo, H.Z. A preliminary study of the correlation between phylogeny, chemical constituents and therapeutic effects of Rheum speices. Acta Pharm. Sin. 1980, 15, 33–39. [Google Scholar]
- Kato, E.; Tokunaga, Y.; Sakan, F. Stilbenoids isolated from the seeds of melinjo (Gnetum gnemon L.) and their biological activity. J. Agric. Food Chem. 2009, 57, 2544–2549. [Google Scholar] [CrossRef]
- Iliya, I.; Ali, Z.; Tanaka, T.; Iinuma, M.; Furusawa, M.; Nakaya, K. I.; Murata, J.; Darnaedi, D.; Matsuura, N.; Ubukata, M. Stilbene derivatives from Gnetum gnemon Linn. Phytochemistry 2003, 62, 601–606. [Google Scholar] [CrossRef]
- Iliya, I.; Tanaka, T.; Iinuma, M.; Furusawa, M.; Ali, Z.; Nakaya, K.I.; Murata, J.; Darnaedi, D. Five stilbene glucosides from Gnetum gnemonoides and Gnetum africanum. Helv. Chim. Acta 2002, 85, 2394–2402. [Google Scholar] [CrossRef]
- Shikishima, Y.; Takaishi, Y.; Honda, G.; Shikishima, Y.; Takaishi, Y.; Honda, G.; Ito, M.; Takeda, Y.; Kodzhimatov, O.K.; Ashurmetov, O. Phenylbutanoids and stilbene derivatives of Rheum maximowiczii. Phytochemistry 2001, 56, 377–381. [Google Scholar] [CrossRef]
- Lins, A.P.; Ribeiro, M.N.D.S.; Gottlieb, O.R.; Gottlieb, H.E. Gnetins: Resveratrol oligomers from Gnetum species. J.Nat. Prod. 1982, 45, 754–761. [Google Scholar] [CrossRef]
- Amira-Guebailia, H.; Valls, J.; Richard, T.; Vitrac, X.; Monti, J.P.; Delaunay, J.C.; Mérillon, J.M. Centrifugal partition chromatography followed by HPLC for the isolation of cis-ε-viniferin, a resveratrol dimer newly extracted from a red Algerian wine. Food Chem. 2009, 113, 320–324. [Google Scholar] [CrossRef]
- Ngoc, T.M.; Hung, T.M.; Thuong, P.T.; Na, M.; Kim, H.; Ha, D.T.; Min, B.; Minh, P.T.H.; Bae, K. Inhibition of human low density lipoprotein and high density lipoprotein oxidation by oligostilbenes from rhubarb. Biol. Pharm. Bull. 2008, 31, 1809–1812. [Google Scholar] [CrossRef]
- Snyder, S.A.; Breazzano, S.P.; Ross, A.G.; Lin, Y.; Zografos, A.L. Total synthesis of diverse carbogenic complexity within the resveratrol class from a common building block. J. Am. Chem. Soc. 2009, 131, 1753–1765. [Google Scholar]
- Sample Availability: Not available.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, W.-B.; Hu, L.; Hu, Q.; Chen, N.-N.; Yang, Q.-S.; Wang, F.-F. New Resveratrol Oligomer Derivatives from the Roots of Rheum lhasaense. Molecules 2013, 18, 7093-7102. https://doi.org/10.3390/molecules18067093
Liu W-B, Hu L, Hu Q, Chen N-N, Yang Q-S, Wang F-F. New Resveratrol Oligomer Derivatives from the Roots of Rheum lhasaense. Molecules. 2013; 18(6):7093-7102. https://doi.org/10.3390/molecules18067093
Chicago/Turabian StyleLiu, Wen-Bo, Lin Hu, Qun Hu, Na-Na Chen, Qing-Song Yang, and Fang-Fang Wang. 2013. "New Resveratrol Oligomer Derivatives from the Roots of Rheum lhasaense" Molecules 18, no. 6: 7093-7102. https://doi.org/10.3390/molecules18067093
APA StyleLiu, W.-B., Hu, L., Hu, Q., Chen, N.-N., Yang, Q.-S., & Wang, F.-F. (2013). New Resveratrol Oligomer Derivatives from the Roots of Rheum lhasaense. Molecules, 18(6), 7093-7102. https://doi.org/10.3390/molecules18067093




