Dihydrochalcones with Antiinflammatory Activity from Leaves and Twigs of Cyathostemma argenteum
Abstract
:1. Introduction
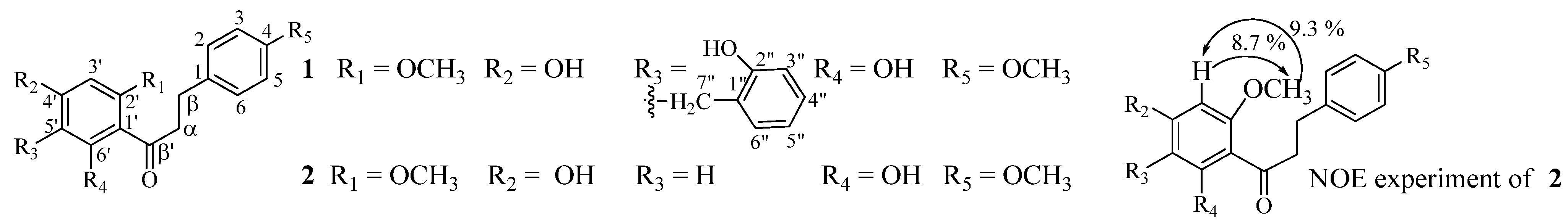
2. Results and Discussion
| Position | 4',6'-Dihydroxy-2',4-dimethoxy-5'-(2''-hydroxybenzyl)dihydrochalcone (1) | Position | 4',6'-Dihydroxy-2',4-dimethoxydihydrochalcone (2) | ||
|---|---|---|---|---|---|
| * δ 1H (J Hz) | δ 13C (DEPT) | * δ 1H (J Hz) | δ 13C (DEPT) | ||
| 1 | - | 133.64 (C) | 1 | - | 133.72 (C) |
| 2 | 7.18 m | 129.30 (CH) | 2 | 7.18 m | 129.37 (CH) |
| 3 | 6.83 obsc. | 113.70 (CH) | 3 | 6.87 dd (2.07, 6.6) | 113.87 (CH) |
| 4(OCH3) | 3.75 s | 158.07 (C) | 4(OCH3) | 3.82s | 157.88 (C) |
| 5 | 6.83 obsc. | 133.70 (CH) | 5 | 6.87 dd (2.07, 6.6) | 113.87 (CH) |
| 6 | 7.18 m | 129.30 (CH) | 6 | 7.18 m | 129.37 (CH) |
| α | 3.28 t (7.7) | 45.89 (CH2) | α | 3.31 t (7.8) | 46.08 (CH2) |
| β | 2.90 t (7.7) | 29.75 (CH2) | β | 2.96 t (7.8) | 29.81 (CH2) |
| β' | - | 205.40 (C) | β' | - | 204.68 (C) |
| 1' | - | 104.87(C) | 1' | - | 105.95 (C) |
| 2′(OCH3) | 3.86s | 161.81 (C) | 2'(OCH3) | 3.87s | 162.31 (C) |
| 3' | 6.13s | 91.01 (CH) | 3' | 5.93 d (2.4) | 90.64 (CH) |
| 4'(OH) | - | 162.26 (C) | 4'(OH) | 5.56 br s | 163.48 (C) |
| 5' | - | 106.93(C) | 5' | 6.02 d (2.4) | 96.60 (CH) |
| 6'(OH) | 14.76s | 164.70 (C) | 6'(OH) | 13.97s | 167.34 (C) |
| 1'' | - | 126.99 (C) | 4(OCH3) | - | 55.27 (CH3) |
| 2''(OH) | - | 154.13 (C) | 2′(OCH3) | - | 55.68 (CH3) |
| 3'' | 6.83 obsc. | 115.05 (CH) | |||
| 4'' | 7.01ddd (1.6, 7.5, 7.7) | 126.95 (CH) | |||
| 5'' | 6.74 ddd (1.6, 7.5, 7.7) | 119.85 (CH) | |||
| 6'' | 7.23 dd (1.6, 7.5) | 130.39 (CH) | |||
| 7'' | 3.89s | 21.87 (CH2) | |||
| 4(OCH3) | 54.52 (CH3) | ||||
| 2'(OCH3) | 55.19 (CH3) | ||||
| Position H | 4',6'-Dihydroxy-2',4-dimethoxy-5'-(2''-hydroxybenzyl)-dihydrochalcone (1) | Position H | 4',6'-dihydroxy-2',4-dimethoxy-dihydrochalcone (2) | ||
|---|---|---|---|---|---|
| HMBC Correlation | COSY Correlation | HMBC Correlation | COSY Correlation | ||
| 1 | - | - | 1 | - | - |
| 2 | C-1, 3, 4, 6, β | H-3 | 2 | C-1, 3, 4, β | H-3 |
| 3 | C-1, 2, 3, 4, 5 | H-2 | 3 | C-1, 2, 4, 5 | H-2 |
| 4(OCH3) | C-4 | - | 4(OCH3) | C-4 | - |
| 5 | C-1, 3, 4, 6 | H-6 | 5 | C-1, 3, 4 | H-6 |
| 6 | C-1, 2, 4, 5, β | H-5 | 6 | C-1, 2, 4, 5, β | H-5 |
| α | C-1, 2, 6, β, β′ | H-α, β | α | C- 1, β, β′ | H-α, β |
| β | C-1, 6, α, β′ | H-α, β | β | C- 1, 2, 6, α, β' | H-α, β |
| β' | - | - | β' | - | - |
| 1' | - | - | 1' | - | - |
| 2'(OCH3) | C-2' | - | 2′(OCH3) | C- 2' | - |
| 3' | C-1', 2', 4', 5', β′ | - | 3' | C- 1', 2', 4', 5' | - |
| 4' | - | - | 4'(OH) | - | - |
| 5' | - | - | 5' | C-1', 3' | - |
| 6'(OH) | C-1', 5', 6', 7'' | - | 6'(OH) | C-1', 5', 6' | - |
| 1'' | - | - | 4(OCH3) | C-4 | - |
| 2'' | - | - | |||
| 3'' | C-1'', 2'', 4'', 5'' | - | |||
| 4'' | C-2'', 3'', 5'', 6'' | H-5'', 6'' | |||
| 5'' | C-1'', 3'', 4'', 6'' | H-4'', 6'' | |||
| 6'' | C-1'', 2'', 4'', 5'', 7'' | H-5'', 4'' | |||
| 7'' | C-4', 5', 6', 1'', 2'', 6'' | H-7'' | |||
| 4(OCH3) | C-4 | - | |||
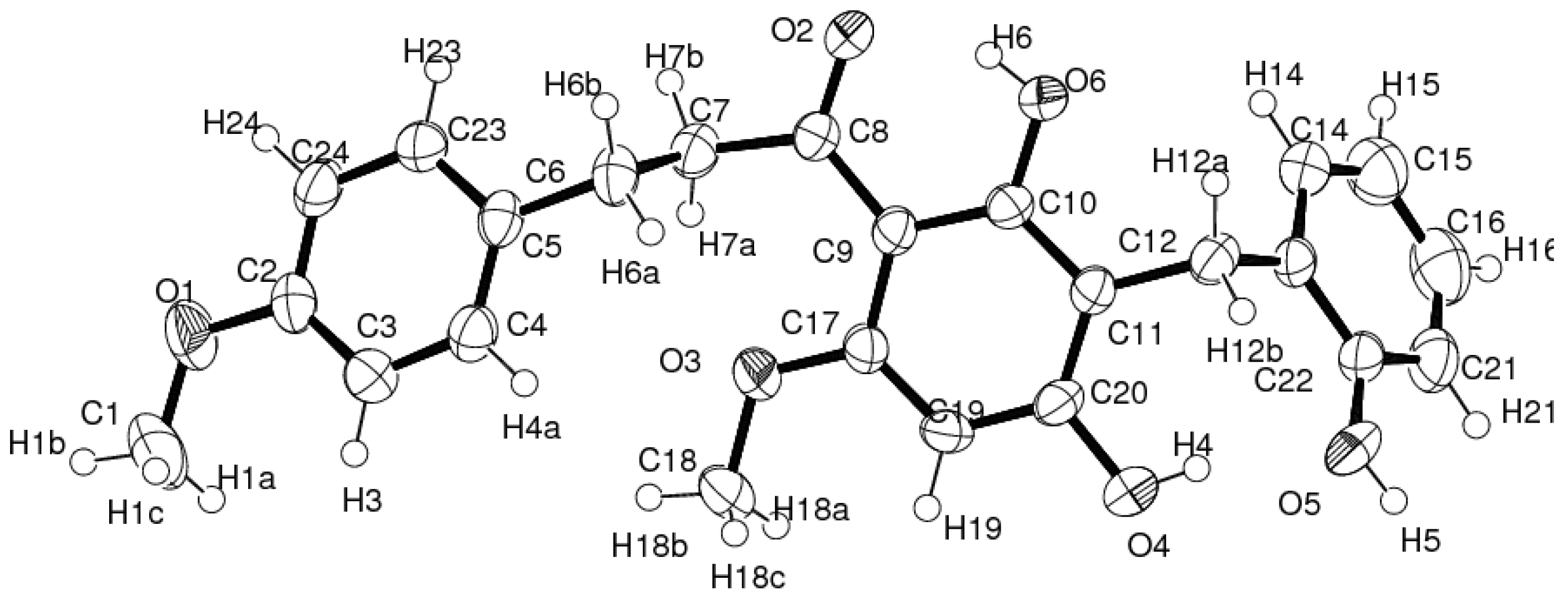
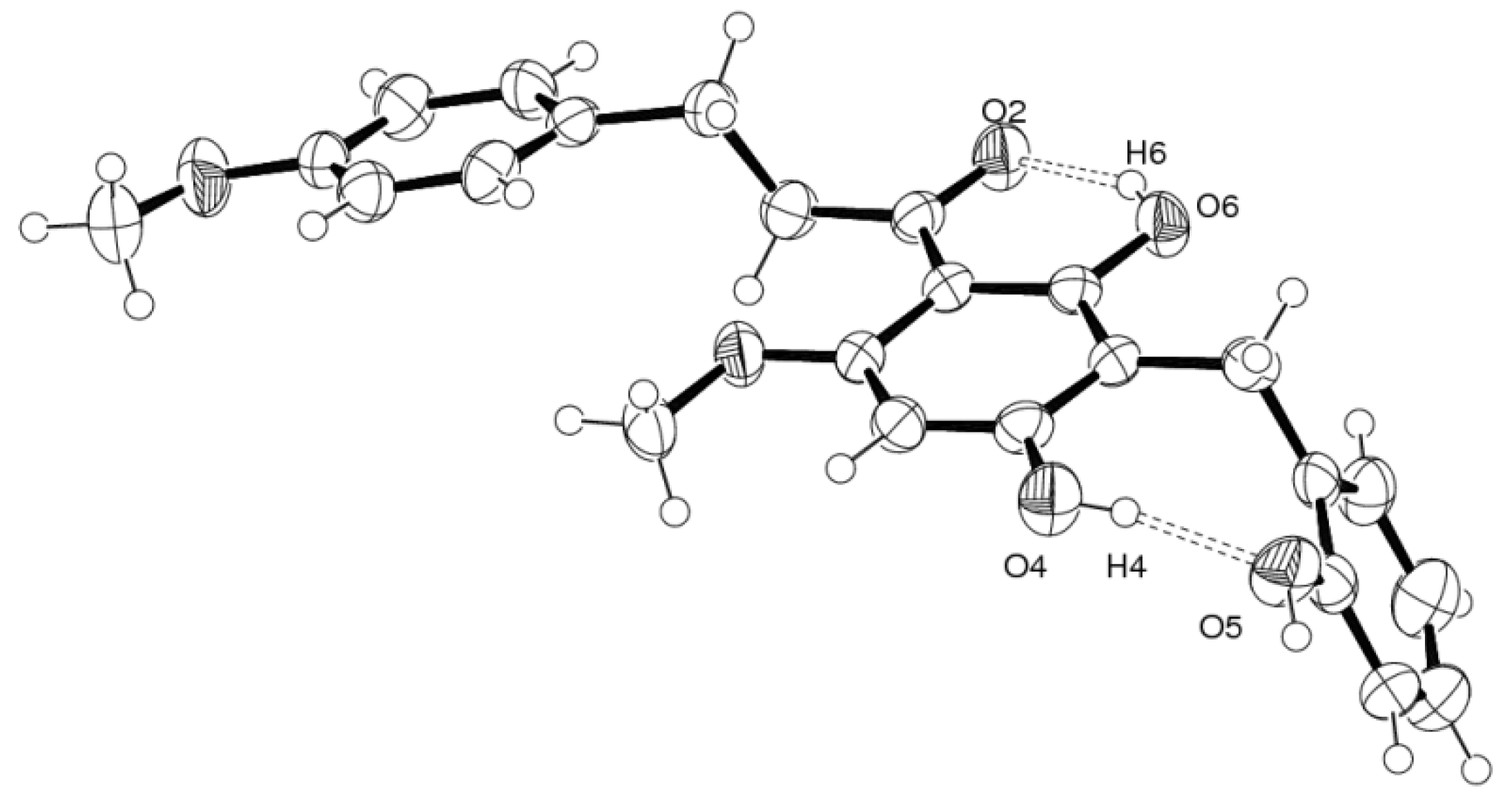
| D–H…A | d[D–H] (Å) | d[H…A] (Å) | d[D…A] (Å) | ∠[D–H…A] (°) |
|---|---|---|---|---|
| O-4–H-4…O-5 | 0.820 | 1.856 | 2.662(3) | 168 |
| O-5–H-5…O-2 i | 0.820 | 1.903 | 2.704(2) | 165 |
| O-6–H-6…O-2 | 0.820 | 1.743 | 2.477(2) | 148 |

| Group | Dose (mg/ear) | Edema thickness ( m) | % edema inhibition | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 15 min | 30 min | 1 h | 2 h | 15 min | 30 min | 1 h | 2 h | ||
| Control (Acetone) | - | 160.00 ± 7.30 | 180.00 ± 5.16 | 203.33 ± 8.03 | 193.33 ± 4.94 | - | - | - | - |
| Phenylbutazone | 1 | 30.00 ± 13.41 * | 43.33 ± 9.54 * | 73.33 ± 12.29 * | 90 ± 4.47 * | 81.25 | 75.93 | 63.93 | 53.45 |
| Compound 1 | 1 | 16.67 ± 2.11 * | 40.00 ± 5.16 * | 76.67 ± 8.03 * | 83.33 ± 3.33 * | 89.58 | 77.78 | 62.30 | 56.90 |
| Compound 2 | 1 | 1.67 ± 1.67 * | 23.33 ± 2.11 * | 63.33 ± 4.22 * | 66.67 ± 8.82 * | 98.96 | 87.04 | 68.85 | 65.52 |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectroscopic Data
3.5. X-ray Crystallographic Analysis of 1
3.6. Antiinflammatory Activity: Ethyl Phenylpropiolate (EPP)-induced Ear Edema in Rats
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Smitinand, T. Thai Plant Name., revised ed.; Prachachon: Bangkok, Thailand, 2001. [Google Scholar]
- Khamis, S.B.; Brown, J.E.; Kirkpatrick, H.E.; Knox, R.; Walker, A.; Wright, C.W. Preliminary investigation of Cyathostemma argenteum, a plant species used in traditional medicine for the treatment of breast cancer. J. Pharm. Pharmacol. 1998, 50, 222. [Google Scholar]
- Khamis, S.; Bibby, M.C.; Brown, J.E.; Cooper, P.A.; Scowen, I.; Wright, C.W. Phytochemistry and preliminary biological evaluation of Cyathostemma argenteum, a Malaysian plant used traditionally for the treatment of breast cancer. Phytpther. Res. 2004, 18, 507–510. [Google Scholar] [CrossRef]
- Mahmood, K.; Fontaine, C.; Pais, M.; Ali, H.M.; Hapipah, M.; Hadi, H.A.; Guittet, E. Cyathostemmine, a new shikimic acid derivative from Cyathostemma viridiflorum Griff. Tetrahedron Lett. 1991, 32, 6117–6120. [Google Scholar]
- Mahmood, K.; Sable, S.; Pais, M.; Ali, H.M.; Hadi, H.A.; Guittet, E. Cyathoviridine, a cytotoxic metabolite from Cyathostemma viridiflorum. Nat. Prod. Lett. 1993, 3, 245–249. [Google Scholar]
- Passador, E.A.P.; Das, M.F.; Silva, G.F.D.; Fo, E.R.; Fernandes, J.B.; Vieira, P.C.; Pirani, J.R. A pyrano chalcone and a flavanone from Neoraputia magnifica. Photochemistry 1997, 45, 1533–1537. [Google Scholar]
- Atta-ur-Rahman. Studies in Natural Products Chemistry., 1st ed.; Elsevier B.V.: London, UK., 2013. [Google Scholar]
- Majumder, P.L.; Lahiri, S.; Mukhoti, N. Chalcone and dihydrochalcone derivatives from the orchid Lusia volucris. Phytochemistry 1995, 40, 271–274. [Google Scholar]
- Ng, S.L.; Razak, I.A.; Fun, H.K.; Boonsri, S.; Chantrapromma, S.; Prawat, U. A second monoclinic polymorph of 1-(2,4-dihydroxy-6-methoxyphenyl)-3-(4-methoxyphenyl)propan-1-one. Acta Crystallogr., Sect. E: Struct. Rep. Online 2005, 61, 3234–3236. [Google Scholar]
- Carlson, R.P.; O’Neill-Davis, L.; Chang, J.; Lewis, A.J. Modulation of mouse ear edema by cyclooxygenase and lipooxygenase inhibitors and other pharmacologic agents. Agents Actions 1985, 17, 197–204. [Google Scholar]
- Brattsand, R.; Thalen, A.; Roempke, K.; Kallstrom, L.; Gruvstad, E. Influence of 16α, 17α-acetal substitution and steroid nucleus fluorinative on the topical to systemic activity ratio of glucocorticoids. J. Steroid. Biochem. 1982, 16, 786–779. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Burnett, M.N.; Johnson, C.K. ORTEP-III: Oak ridge Thermal Ellipsoid Plot Program for Crystal Structure Illustration; Report ORNL-6895; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 1996. [Google Scholar]
- Sample Availability: Samples of the compounds 1–2 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Somsrisa, J.; Meepowpan, P.; Krachodnok, S.; Thaisuchat, H.; Punyanitya, S.; Nantasaen, N.; Pompimon, W. Dihydrochalcones with Antiinflammatory Activity from Leaves and Twigs of Cyathostemma argenteum. Molecules 2013, 18, 6898-6907. https://doi.org/10.3390/molecules18066898
Somsrisa J, Meepowpan P, Krachodnok S, Thaisuchat H, Punyanitya S, Nantasaen N, Pompimon W. Dihydrochalcones with Antiinflammatory Activity from Leaves and Twigs of Cyathostemma argenteum. Molecules. 2013; 18(6):6898-6907. https://doi.org/10.3390/molecules18066898
Chicago/Turabian StyleSomsrisa, Jariya, Puttinan Meepowpan, Samroeng Krachodnok, Haruthai Thaisuchat, Sittiporn Punyanitya, Narong Nantasaen, and Wilart Pompimon. 2013. "Dihydrochalcones with Antiinflammatory Activity from Leaves and Twigs of Cyathostemma argenteum" Molecules 18, no. 6: 6898-6907. https://doi.org/10.3390/molecules18066898
APA StyleSomsrisa, J., Meepowpan, P., Krachodnok, S., Thaisuchat, H., Punyanitya, S., Nantasaen, N., & Pompimon, W. (2013). Dihydrochalcones with Antiinflammatory Activity from Leaves and Twigs of Cyathostemma argenteum. Molecules, 18(6), 6898-6907. https://doi.org/10.3390/molecules18066898




