Chemical Constituents from the Rhizomes of Smilax glabra and Their Antimicrobial Activity
Abstract
:1. Introduction
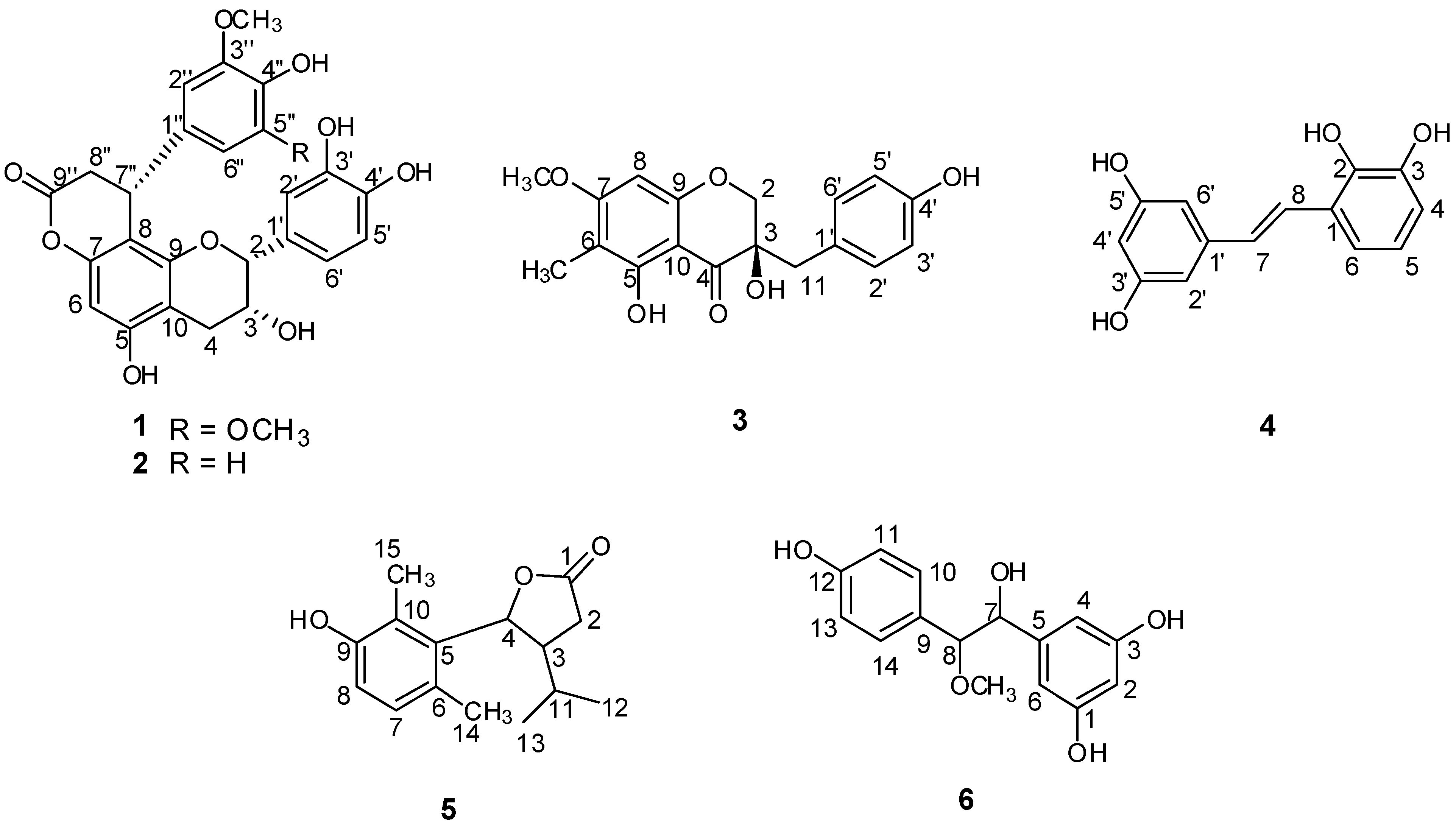
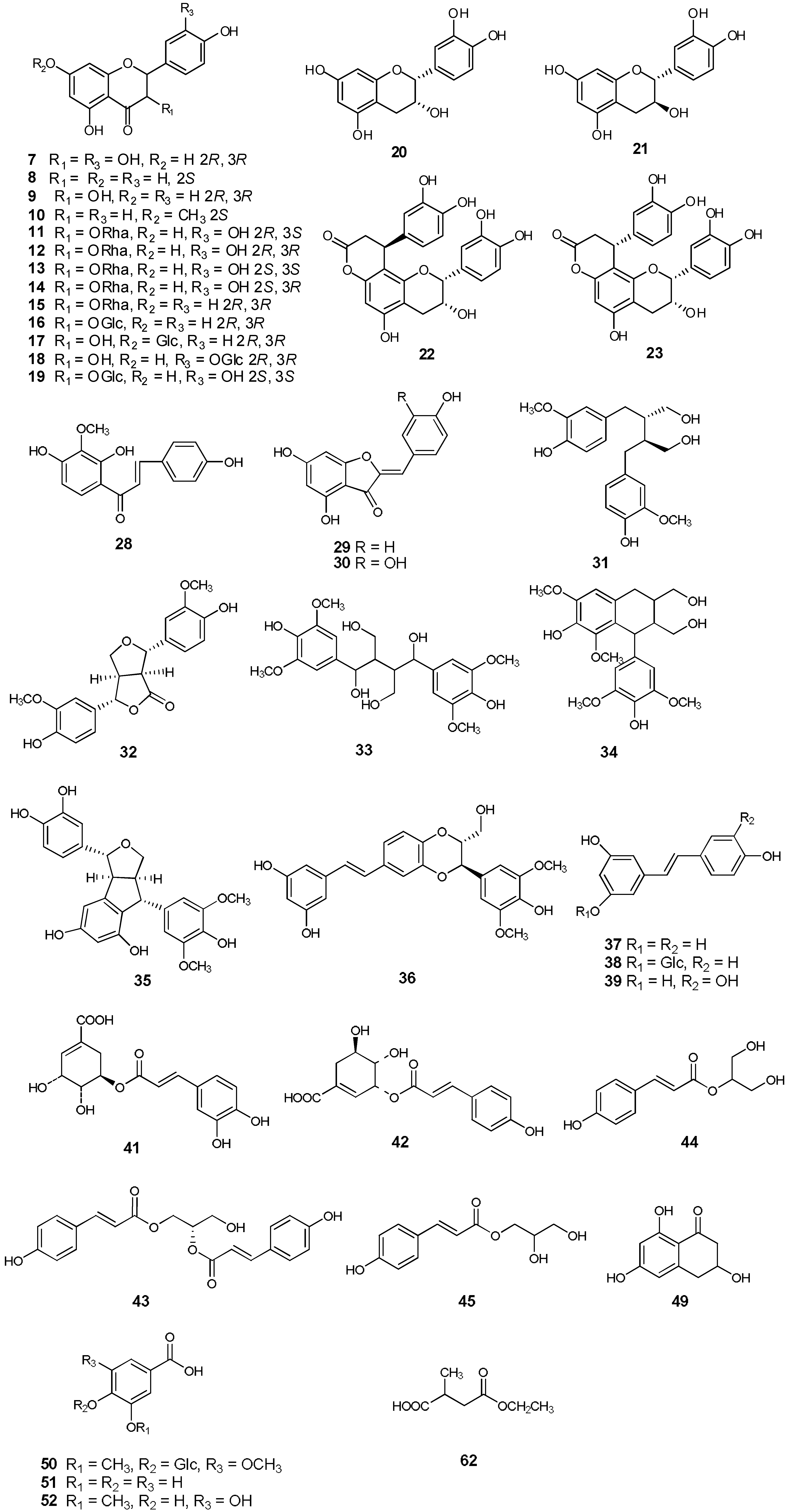
2. Results and Discussion
2.1. Structural Elucidation of the New Compounds
| Position | 1 | 2 | ||
|---|---|---|---|---|
| δH (J in Hz) | δC | δH (J in Hz) | δC | |
| 2 | 4.81 (1H, s) | 79.8 | 4.82 (1H, s) | 79.8 |
| 3 | 4.26 (1H, m) | 66.8 | 4.25 (1H, m) | 66.7 |
| 4 | 2.85 (1H, dd, J = 17.2, 1.7) | 29.8 | 2.85 (1H, dd, J = 17.2, 2.0) | 29.7 |
| 2.92 (1H, dd, J = 17.2, 4.5) | 2.92 (1H, dd, J = 7.2, 4.4) | |||
| 5 | – | 157.4 | – | 157.4 |
| 6 | 6.20 (1H, s) | 96.3 | 6.19 (1H, s) | 96.3 |
| 7 | – | 151.6 | – | 151.7 |
| 8 | – | 106.2 | – | 106.2 |
| 9 | – | 153.6 | – | 153.5 |
| 10 | – | 105.4 | – | 105.3 |
| 1′ | – | 132.0 | – | 132.0 |
| 2′ | 7.06 (1H, d, J = 1.9) | 115.1 | 7.03 (1H, d, J = 1.8) | 115.1 |
| 3′ | – | 146.2 | – | 146.1 |
| 4′ | – | 145.9 | – | 145.8 |
| 5′ | 6.79 (1H, d, J = 8.1) | 116.1 | 6.781 (1H, d, J = 8.2) | 116.0 |
| 6′ | 6.84 (1H, dd, J = 8.1, 1.9) | 119.1 | 6.83 (1H, dd, J = 8.2, 1.8) | 119.1 |
| 1′′ | – | 135.2 | – | 135.8 |
| 2′′ | 6.43 (1H, s) | 105.0 | 6.776 (1H, d, J = 2.0) | 112.2 |
| 3′′ | – | 149.2 | – | 148.8 |
| 4′′ | – | 135.1 | – | 146.3 |
| 5′′ | – | 149.2 | 6.62 (1H, d, J = 8.2) | 116.2 |
| 6′′ | 6.43 (1H, s) | 105.0 | 6.50 (1H, dd, J = 8.2, 2.0) | 119.4 |
| 7′′ | 4.60 (1H, br. d, J = 6.3) | 36.2 | 4.60 (1H, br. d, J = 6.3) | 35.7 |
| 8′′ | 2.91 (1H, dd, J = 15.9, 1.5) | 37.5 | 2.90 (1H, dd, J = 15.9, 1.5) | 37.8 |
| 3.12 (1H, dd, J = 15.9, 7.5) | 3.10 (1H, dd, J = 15.9, 7.4) | |||
| 9′′ | – | 171.0 | – | 170.9 |
| 3′′-OCH3 | – | – | 3.56 (3H, s) | 56.0 |
| 3′′, 5′′-OCH3 | 3.59 (6H, s) | 56.4 | – | – |
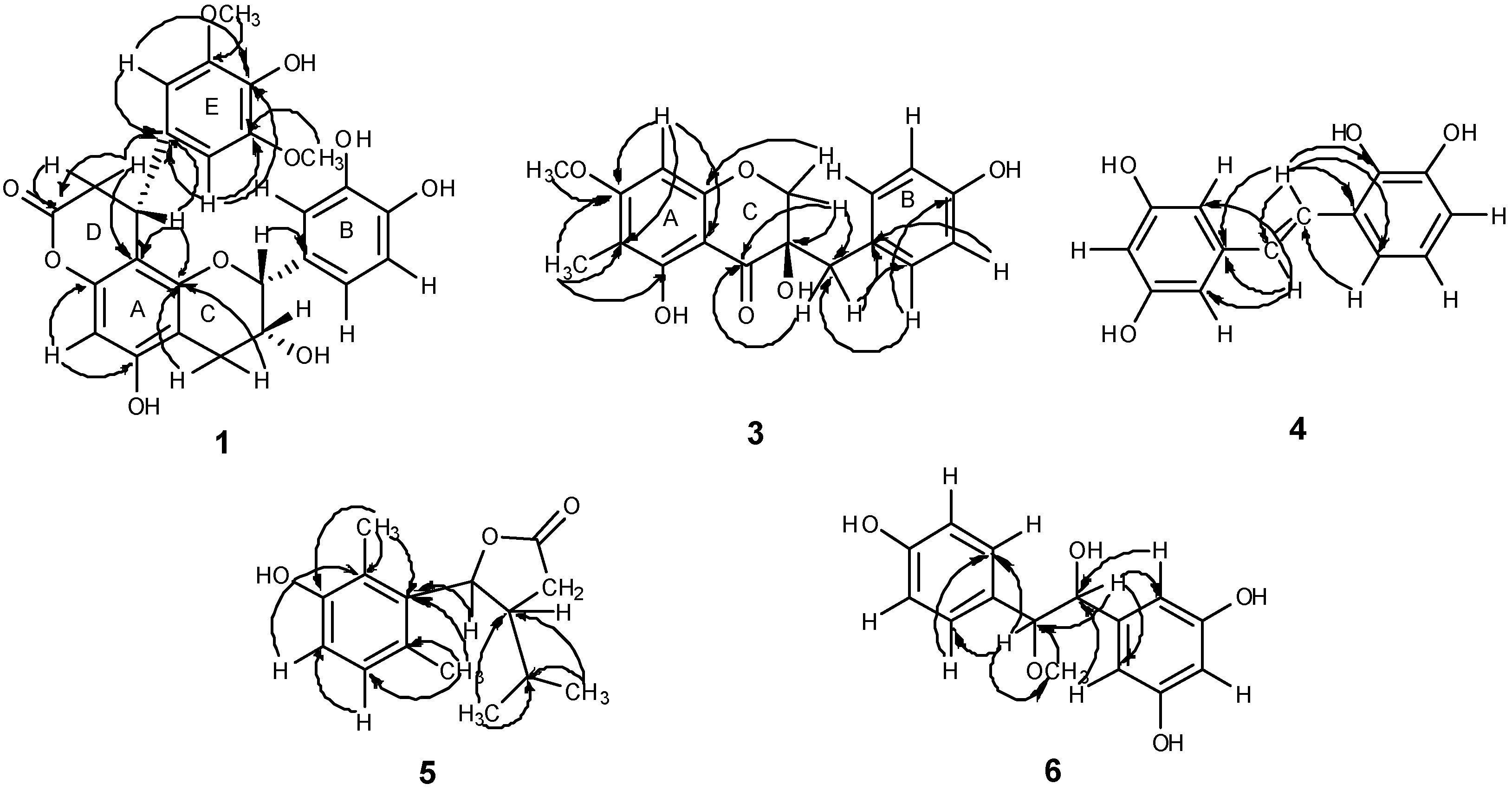
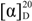 +178.72), indicating the R configuration at C-3 [15]. Consequently, the structure of compound 3 was identified as (3R)-3,5-dihydroxy-7-methoxy-6-methyl-3-(4-hydroxybenzyl)chroman-4-one, and trivially named smilachromanone.
+178.72), indicating the R configuration at C-3 [15]. Consequently, the structure of compound 3 was identified as (3R)-3,5-dihydroxy-7-methoxy-6-methyl-3-(4-hydroxybenzyl)chroman-4-one, and trivially named smilachromanone.| Position | δH (J in Hz) | δC |
|---|---|---|
| 2 | 4.06 (1H, d, J = 11.0) | 72.9 |
| 3.96 (1H, d, J = 11.0) | – | |
| 3 | – | 73.6 |
| 4 | – | 200.6 |
| 5 | – | 161.6 |
| 6 | – | 106.7 |
| 7 | – | 167.5 |
| 8 | 6.14 (1H, s) | 91.8 |
| 9 | – | 162.7 |
| 10 | – | 101.8 |
| 11 | 2.90 (1H, d, J = 14.5) | 40.8 |
| 2.86 (1H, d, J = 14.5) | – | |
| 1′ | – | 126.8 |
| 2′, 6′ | 7.05 (2H, d, J = 8.5) | 132.8 |
| 3′, 5′ | 6.70 (2H, d, J = 8.5) | 115.9 |
| 4′ | – | 157.5 |
| 7-OCH3 | 3.86 (3H, s) | 56.5 |
| 6-CH3 | 1.95 (3H, s) | 7.0 |
| Position | δH (J in Hz) | δC |
|---|---|---|
| 1 | – | 126.0 |
| 2 | – | 144.6 |
| 3 | – | 146.5 |
| 4 | 6.69 (1H, dd, J = 7.8, 1.8) | 114.9 |
| 5 | 6.66 (1H, t, J = 7.8) | 120.5 |
| 6 | 7.02 (1H, dd, J = 7.8, 1.8) | 118.3 |
| 7 | 6.96 (1H, d, J = 16.8) | 129.5 |
| 8 | 7.37 (1H, d, J = 16.8) | 124.7 |
| 1′ | – | 141.6 |
| 2′, 6′ | 6.48 (2H, d, J = 1.8) | 105.9 |
| 3′, 5′ | – | 159.7 |
| 4′ | 6.17 (1H, t, J = 1.8) | 102.8 |
| Position | δH (J in Hz) | δC |
|---|---|---|
| 1 | – | 179.6 |
| 2 | 2.65 (2H, m) | 33.8 |
| 3 | 2.75 (1H, m) | 49.5 |
| 4 | 5.72 (1H, d, J = 9.0) | 85.1 |
| 5 | – | 136.3 |
| 6 | – | 128.9 |
| 7 | 6.85 (1H, d, J = 8.4) | 130.7 |
| 8 | 6.69 (1H, d, J = 8.4) | 116.2 |
| 9 | – | 156.2 |
| 10 | – | 125.2 |
| 11 | 1.76 (1H, m) | 31.3 |
| 12 | 0.76 (3H, d, J = 6.6) | 21.9 |
| 13 | 0.98 (3H, d, J = 6.6) | 19.9 |
| 14 | 2.30 (3H, s) | 20.8 |
| 15 | 2.22 (3H, s) | 13.0 |
| Position | δH (J in Hz) | δC |
|---|---|---|
| 1, 3 | – | 159.7 |
| 2 | 6.88 (1H, t, J = 2.4) | 102.9 |
| 4, 6 | 6.97 (2H, d, J = 2.4) | 107.1 |
| 5 | – | 145.4 |
| 7 | 5.13 (1H, d, J = 7.8) | 78.7 |
| 8 | 4.56 (1H, d, J = 7.8) | 89.5 |
| 9 | – | 130.0 |
| 10, 14 | 7.30 (2H, d, J = 8.4) | 129.9 |
| 11, 13 | 7.06 (2H, d, J = 8.4) | 115.7 |
| 12 | – | 158.4 |
| 1, 3-OH | 11.24 (2H, br. s) | – |
| 7-OH | 3.59 (1H, br. s) | – |
| 8-OCH3 | 3.28 (3H, s) | 56.7 |
| 12-OH | 11.38 (1H, br. s) | – |
2.2. Structural Elucidation of the Known Isolates
2.3. Antimicrobial Activity
| Compound | EC | PA | KP | MRSA | SA | EF | CA |
|---|---|---|---|---|---|---|---|
| 1 | 0.604 | 0.604 | 0.604 | 0.302 | 0.302 | 0.302 | 0.146 |
| 3 | >1.21 | >1.21 | >1.21 | 0.605 | 0.303 | 0.605 | 0.605 |
| 4 | 1.64 | 1.64 | 1.64 | 0.409 | 0.205 | 0.819 | 0.819 |
| 9 | >2.10 | >2.10 | >2.10 | 2.10 | 2.10 | >2.10 | >2.10 |
| 10 | >2.10 | >2.10 | >2.10 | 0.524 | 0.524 | 1.05 | >2.10 |
| 11 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 |
| 12 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 | 0.666 |
| 13 | >1.33 | >1.33 | >1.33 | 1.33 | >1.33 | >1.33 | >1.33 |
| 14 | 1.33 | 1.33 | 1.33 | 1.33 | 0.666 | 1.33 | 1.33 |
| 15 | >1.38 | >1.38 | >1.38 | >1.38 | >1.38 | >1.38 | >1.38 |
| 16 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 | >1.33 |
| 17 | >1.33 | >1.33 | >1.33 | 1.33 | 1.33 | 1.33 | >1.33 |
| 20 | >2.07 | >2.07 | >2.07 | 1.03 | 0.517 | 1.03 | >2.07 |
| 22 | >1.33 | 0.663 | >1.33 | 0.0801 | 0.0801 | 0.160 | 0.160 |
| 33 | >0.880 | >0.880 | >0.880 | >0.880 | >0.880 | >0.880 | >0.880 |
| 36 | 0.663 | 0.663 | 0.663 | 1.33 | 0.663 | 1.33 | 0.332 |
| 37 | >2.63 | >2.63 | >2.63 | 0.159 | 0.0794 | 0.159 | 0.657 |
| 38 | >1.54 | >1.54 | >1.54 | 0.768 | 0.768 | 1.54 | >1.54 |
| 39 | 1.64 | 1.64 | 1.64 | 0.409 | 0.205 | >1.64 | 0.819 |
| 44 | 2.52 | 2.52 | 2.52 | 0.630 | 0.630 | 1.26 | 0.630 |
| 49 | 3.09 | 3.09 | 3.09 | 3.09 | 1.55 | 3.09 | 3.09 |
| 51 | >2.60 | >2.60 | >2.60 | >2.60 | >2.60 | >2.60 | >2.60 |
| Cipro | 0.00302 | 0.00302 | 0.00604 | ||||
| Van | 0.000690 | 0.000690 | 0.00138 | ||||
| Keto | 0.0000301 |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectroscopic Data
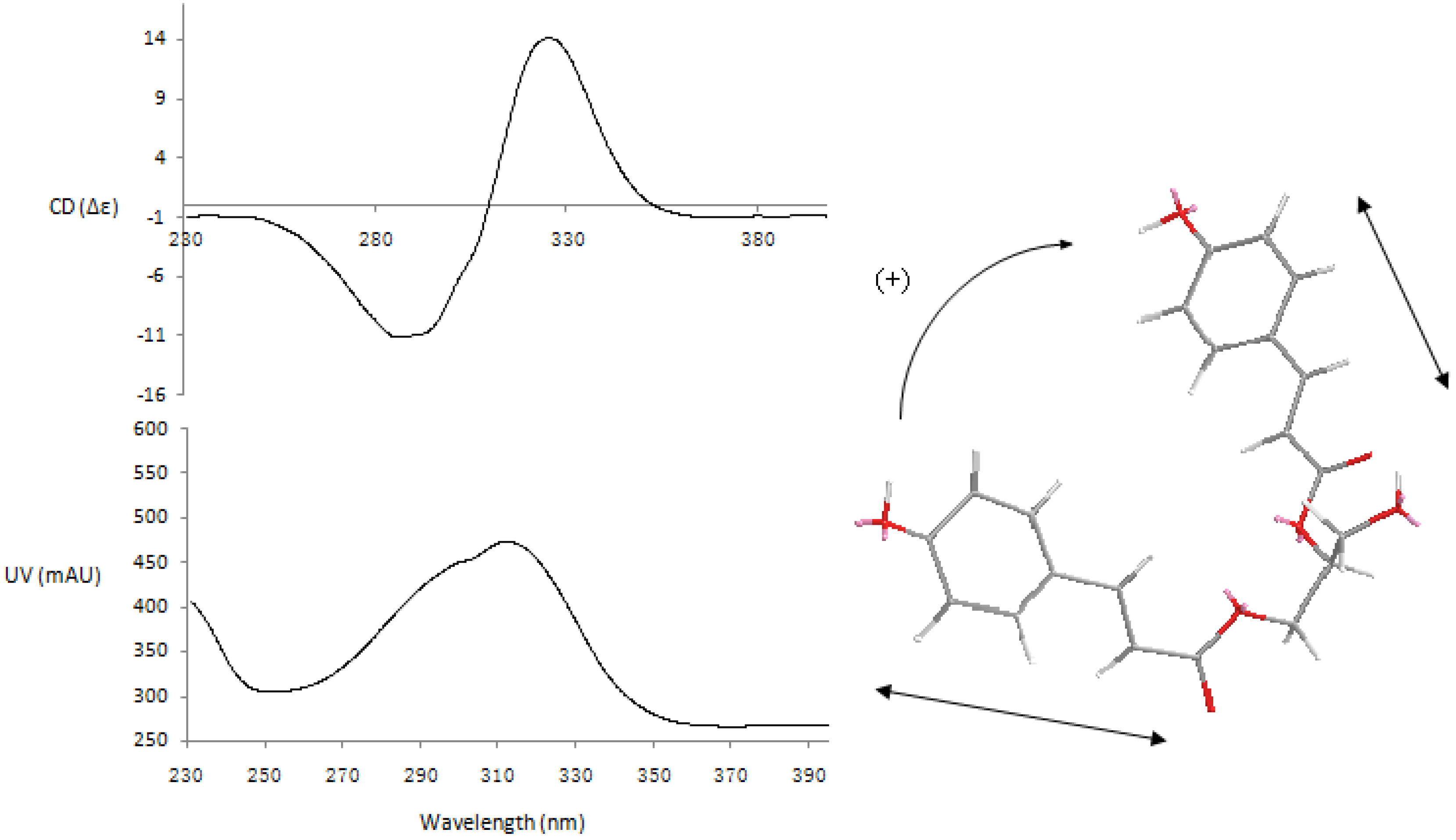
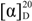 63.6 (c 0.44, MeOH); CD (2.72 × 10−4 MeOH): Δε (nm) 0.49 (214), −14.46 (235), 0.41 (259), −4.77 (284); UV λmax (MeOH): 202, 230, 280 nm; IR (KBr) νmax (cm−1): 3,452, 2,920, 2,850, 1,751, 1,676, 1,620, 1,520, 1,466, 1,368, 1,278, 1,202, 1,161, 1,112, 1,064, 997, 834, 724; HR-ESI-MS m/z: 495.1299 [M−H]− (calcd. for C26H23O10 495.1291); 1H- and 13C-NMR spectroscopic data see Table 1.
63.6 (c 0.44, MeOH); CD (2.72 × 10−4 MeOH): Δε (nm) 0.49 (214), −14.46 (235), 0.41 (259), −4.77 (284); UV λmax (MeOH): 202, 230, 280 nm; IR (KBr) νmax (cm−1): 3,452, 2,920, 2,850, 1,751, 1,676, 1,620, 1,520, 1,466, 1,368, 1,278, 1,202, 1,161, 1,112, 1,064, 997, 834, 724; HR-ESI-MS m/z: 495.1299 [M−H]− (calcd. for C26H23O10 495.1291); 1H- and 13C-NMR spectroscopic data see Table 1.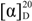 −80.0 (c 0.20, MeOH); CD (4.60 × 10−4 MeOH): Δε (nm) 2.02 (214),−18.04 (234),1.29 (256),−4.84 (284); UV λmax (MeOH): 202, 230, 280 nm; IR (KBr) νmax (cm−1): 3,453, 2,921, 2,853, 1,745, 1,629, 1,542, 1,458, 1,261, 1,165, 1,112, 894, 802; HR-ESI-MS m/z: 465.1189 [M−H]− (calcd. for C25H21O9 465.1186), 467.1327 [M+H]+ (calcd. for C25H23O9 467.1342); 1H- and 13C-NMR spectroscopic data see Table 1.
−80.0 (c 0.20, MeOH); CD (4.60 × 10−4 MeOH): Δε (nm) 2.02 (214),−18.04 (234),1.29 (256),−4.84 (284); UV λmax (MeOH): 202, 230, 280 nm; IR (KBr) νmax (cm−1): 3,453, 2,921, 2,853, 1,745, 1,629, 1,542, 1,458, 1,261, 1,165, 1,112, 894, 802; HR-ESI-MS m/z: 465.1189 [M−H]− (calcd. for C25H21O9 465.1186), 467.1327 [M+H]+ (calcd. for C25H23O9 467.1342); 1H- and 13C-NMR spectroscopic data see Table 1.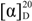 +178.72 (c 0.47, MeOH); CD (1.94 × 10−4 MeOH): Δε (nm) +13.43 (260), +6.22 (312), −29.23 (290); UV λmax (MeOH): 216, 294 nm; IR (KBr) νmax (cm−1): 3,450, 2,921, 1,633, 1,436, 1,376, 1,314, 1,243, 1,165, 1,058, 897, 795; HR-ESI-MS m/z: 329.1033 [M-H]− (calcd. for C18H17O6 329.1025); 1H- and 13C-NMR spectroscopic data see Table 2.
+178.72 (c 0.47, MeOH); CD (1.94 × 10−4 MeOH): Δε (nm) +13.43 (260), +6.22 (312), −29.23 (290); UV λmax (MeOH): 216, 294 nm; IR (KBr) νmax (cm−1): 3,450, 2,921, 1,633, 1,436, 1,376, 1,314, 1,243, 1,165, 1,058, 897, 795; HR-ESI-MS m/z: 329.1033 [M-H]− (calcd. for C18H17O6 329.1025); 1H- and 13C-NMR spectroscopic data see Table 2.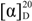 +120 (c 0.1, MeOH); CD (8.06 × 10−4 MeOH): Δε (nm) +34.11 (214),-10.21 (298); UV λmax (MeOH): 201, 289 nm; IR (KBr) νmax (cm−1): 3,357, 1,747, 1,592, 1,489, 1,466, 1,373, 1,321, 1,279, 1,219, 1,160, 1,081, 1,049, 983, 817, 702; HR-EI-MS m/z: 248.1415 [M]+ (calcd. for C15H20O3 248.1412); 1H- and 13C-NMR spectroscopic data see Table 4.
+120 (c 0.1, MeOH); CD (8.06 × 10−4 MeOH): Δε (nm) +34.11 (214),-10.21 (298); UV λmax (MeOH): 201, 289 nm; IR (KBr) νmax (cm−1): 3,357, 1,747, 1,592, 1,489, 1,466, 1,373, 1,321, 1,279, 1,219, 1,160, 1,081, 1,049, 983, 817, 702; HR-EI-MS m/z: 248.1415 [M]+ (calcd. for C15H20O3 248.1412); 1H- and 13C-NMR spectroscopic data see Table 4.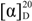 −156.5(c 0.08, MeOH); UV λmax (MeOH): 206, 225, 278nm; IR (KBr) νmax (cm−1): 3,360, 2,927, 1,678, 1,611, 1,515, 1,455, 1,378, 1,341, 1,303, 1,245, 1,158, 1,092, 1,058, 1,007, 923, 838, 718, 695; HR-ESI-MS m/z: 311.06917 [M+Cl]−, 338.08813 [M+NO3]−, 587.16893 [2M+Cl]− (calcd. for C15H16O5Cl 311.06921, C15H16O5NO3 338.08835, C30H32O10Cl 587.16829, respectively); 1H- and 13C-NMR spectroscopic data see Table 5.
−156.5(c 0.08, MeOH); UV λmax (MeOH): 206, 225, 278nm; IR (KBr) νmax (cm−1): 3,360, 2,927, 1,678, 1,611, 1,515, 1,455, 1,378, 1,341, 1,303, 1,245, 1,158, 1,092, 1,058, 1,007, 923, 838, 718, 695; HR-ESI-MS m/z: 311.06917 [M+Cl]−, 338.08813 [M+NO3]−, 587.16893 [2M+Cl]− (calcd. for C15H16O5Cl 311.06921, C15H16O5NO3 338.08835, C30H32O10Cl 587.16829, respectively); 1H- and 13C-NMR spectroscopic data see Table 5.3.5. Antimicrobial Assay
4. Conclusions
Supplementary Materials
Acknowledgments
Conflicts of Interest
References
- Wang, F.Z.; Tang, J; Chen, X.Q.; Zhang, Z.Y.; Dai, L.K.; Liang, S.J.; Tang, Y.C.; Liu, L.; Lang, K.Y. Flora Reipublicae Popularis Sinicae, 1st ed; Science Press: Beijing, China, 1978; Volume 15, pp. 212–214. [Google Scholar]
- Song, L.; Wu, Y.; Hu, L.; Zhang, G.; Xu, G.; Xiao, P.; Ling, Y.; Ding, X.; Cao, C.; Li, Y. Zhong Hua Ben Cao; Shanghai Science and Technology Publisher: Shanghai, China, 1999; Volume 22, pp. 161–164. [Google Scholar]
- Pharmacopoeia Commission of PRC, Pharmacopoeia of the People’s Republic of China; Chinese Medicine Science and Technology Press: Beijing, China, 2010; Volume I, p. 17.
- Gao, Y.J.; Su, Y.H.; Qu, L.K.; Xu, S.; Meng, L.; Cai, S.Q.; Shou, C.C. Mitochondrial apoptosis contributes to the anti-cancer effect of Smilax glabra Roxb. Toxicol. Lett. 2011, 207, 112–120. [Google Scholar] [CrossRef]
- Zhang, Q.F.; Zhang, Z.R.; Cheung, H.Y. Antioxidant activity of Rhizoma Smilacis Glabrae extracts and its key constituent-astilbin. Food Chem. 2009, 115, 297–303. [Google Scholar] [CrossRef]
- Ji, L.L.; Fan, Y.M. Antibacterial activity of extracts from Smilax glabra. Life Sci. Res. 2002, 6, 84–87. [Google Scholar]
- Kuspradini, H.; Mitsunaga, T.; Ohashi, H. Antimicrobial activity against Streptococcus sobrinus and glucosyltransferase inhibitory activity of taxifolin and some flavanonol rhamnosides from kempas (Koompassia malaccensis) extracts. J. Wood Sci. 2009, 55, 308–313. [Google Scholar] [CrossRef]
- Chen, L.; Yin, Y.; Yi, H.W.; Xu, Q.; Chen, T. Simultaneous quantification of five major bioactive flavonoids in Rhizoma Smilacis Glabrae by high-performance liquid chromatography. J. Pharmaceut. Biomed. 2007, 43, 1715–1720. [Google Scholar] [CrossRef]
- You, J.G. Summary on clinical application of the rhizomes of Smilax glabra Roxb. Fujian J. Tradit. Chin. Med. 2002, 33, 50–51. [Google Scholar]
- Ao, C.W.; Higa, T.; Khanh, T.D.; Upadhyay, A.; Tawata, S. Antioxidant phenolic compounds from Smilax sebeana Miq. Food Sci. Technol. 2011, 44, 1681–1686. [Google Scholar]
- Ming, D.S.; López, A.; Hillhouse, B.J.; French, C.J.; Hudson, J.B.; Towers, G.H.N. Bioactive constituents from Iryanthera megistophylla. J. Nat. Prod. 2002, 65, 1412–1416. [Google Scholar] [CrossRef]
- Foo, L.Y. Phenylpropanoid derivatives of catechin, epicatechin and phylloflavan from Phyllocladus trichomanoides. Phytochemistry 1987, 26, 2825–2830. [Google Scholar] [CrossRef]
- Nonaka, G.I.; Nishioka, I. Tannins and related compounds VII phenylpropanoid-substituted epicatechin, cinchonains from Cinchona succirubra. Chem. Pharm. Bull. 1982, 30, 4268–4276. [Google Scholar] [CrossRef]
- Watanabe, Y.; Sanada, S.; Ida, Y.; Shoji, J. Comparative studies on the constituents of Ophiopogonis tuber and its congenes. IV. Studies on the homoisoflavonoids of the subterranean part of Ophiopogon ohwii OKUYAMA and O. jaburan (KUNTH) LODD. Chem. Pharm. Bull. 1985, 33, 5358–5363. [Google Scholar] [CrossRef]
- Tang, Y.P.; Yu, B.; Hu, J.; Wu, T.; Hui, H.Z. Three new homoisoflavanone glycosides from the bulbs of Ornithogalum caudatum. J. Nat. Prod. 2002, 65, 218–220. [Google Scholar] [CrossRef]
- Spassov, S.L. Nuclear magnetic resonance spectra, configuration and conformation of diastereomers: 3-substituted 2,3-diphenylpropanoic acids and their methyl esters. Tetrahedron 1969, 25, 3631–3638. [Google Scholar] [CrossRef]
- Yuan, Z.; Li, X. NMR methods for determining the configuration of 8-O-4′ neolignans. Chin. J. Mag. Reson. 2003, 20, 307–314. [Google Scholar]
- Koshino, H.; Terada, S.; Yoshihara, T.; Sakamura, S.; Shimanuki, T.; Sato, T.; Tajimi, A. Three phenolic acid derivatives from stromata of Epichloe typhina on Phleum pretense. Phytochemistry 1988, 27, 1333–1338. [Google Scholar] [CrossRef]
- Berova, N.; Nakanishi, K. Circular Dichroism: Principles and Applications, 2nd ed; Wiley-VCH: New York, NY, USA, 2000; pp. 337–382. [Google Scholar]
- Adam, W.; Humpf, H.U.; Korb, M.N.; Schreier, P. The configurational assignment of the optically active 5-(1-hydro-peroxyethyl)-3-ethoxycarbonyl-2-methylfuran and its alcohol by exciton-coupled circular dichroism (ECCD). Tetrahedron Asymmetr. 1997, 8, 3555–3558. [Google Scholar] [CrossRef]
- Wungsintaweekul, B.; Umehara, K.; Miyase, T.; Noguchi, H. Estrogenic and anti-estrogenic compounds from the Thai medicinal plant, Smilax corbularia (Smilacaceae). Phytochemistry 2011, 72, 495–502. [Google Scholar] [CrossRef]
- Zhang, X.F.; Hung, T.M.; Phuong, P.T.; Ngoc, T.M.; Min, B.S.; Song, K.S.; Seong, Y.H.; Bae, K. Anti-inflammatory activity of flavonoids from Populus davidiana. Arch. Pharm. Res. 2006, 29, 1102–1108. [Google Scholar] [CrossRef]
- Kasai, R.; Hirono, S.; Chou, W.H.; Tanaka, O.; Chen, F.H. Sweet dihydroflavonol rhamnoside from leaves of Engelhardtia chrysolepis, a Chinese folk medicine, Hung-qi. Chem. Pharm. Bull. 1988, 36, 4167–4170. [Google Scholar] [CrossRef]
- Kang, J; Xie, C.H.; Li, Z.M.; Nagarajan, S; Schauss, A.G.; Wu, T.; Wu, X.L. Flavonoids from acai (Euterpe oleracea Mart.) pulp and their antioxidant and anti-inflammatory activities. Food Chem. 2011, 128, 152–157. [Google Scholar] [CrossRef]
- Si, C.L.; Wu, L.; Zhu, Z.Y. Phenolic glycosides from Populus davidiana bark. Biochem. Syst. Ecol. 2009, 37, 221–224. [Google Scholar] [CrossRef]
- Dübeler, A.; Voltmer, G.; Gora, V.; Lunderstädt, J.; Zeeck, A. Phenols from Fagus sylvatica and their role in defense against Cryptococcus fagisuga. Phytochemistry 1997, 45, 51–57. [Google Scholar]
- Dominguez, X.A.; Garcia, S.; Williams, H.J.; Ortiz, C.; Scott, A.N.; Beibenspies, J.H. Kukulkanins A and B, new chalcones from Mimosa tenuefolia. J. Nat. Prod. 1989, 52, 864–867. [Google Scholar] [CrossRef]
- Haudecoeur, R.; Ahmed-Belkacem, A.; Yi, W.; Fortune, A.; Brillet, R.; Belle, C.; Nicolle, E.; Pallier, C.; Pawlotsky, J.M.; Boumendjel, A. Discovery of naturally occur ring aurones that are potent allosteric inhibitors of hepatitis C virus RNA-dependent RNA polymerase. J. Med. Chem. 2011, 54, 5395–5402. [Google Scholar] [CrossRef]
- Detsi, A.; Majdalani, M.; Kontogiorgis, C.A.; Hadjipavlou-Litina, D.; Kefalas, P. Natural and synthetic 2'-hydroxy-chalcones and aurones: Synthesis, characterization and evaluation of the antioxidant and soybean lipoxygenase inhibitory activity. Bioorgan. Med. Chem. 2009, 17, 8073–8085. [Google Scholar] [CrossRef]
- Achenbach, H.; Waibel, R.; Addae-mansah, I. Lignans and other constituents from Carissa edulis. Phytochemistry 1983, 22, 749–753. [Google Scholar] [CrossRef]
- Otsuka, H.; Takeuchi, M.; Inoshiri, S.; Sato, T.; Yamasaki, K. Phenolic compounds from Coix lachryma-jobi var. Ma-Yuen. Phytochemistry 1989, 28, 883–886. [Google Scholar]
- Freudenberg, K.; Schraube, H. Sinapyl alcohol and the synthesis of syringaresinol. Chemische. Berichte. 1955, 88, 16–23. [Google Scholar] [CrossRef]
- Li, L.Y.; Seeram, N.P. Maple syrup phytochemicals include lignans, coumarins, a stilbene, and other previously unreported antioxidant phenolic compounds. J. Agric. Food Chem. 2010, 58, 11673–11679. [Google Scholar] [CrossRef]
- Kobayashi, M.; Mahmud, T.; Yoshioka, N.; Hori, K.; Shibuya, H.; Kitagawa, I. Indonesian medicinal plants. XVIII. Kompasinol A, a new stibeno-phenylpropanoid from the bark of Koompassia malaccensis (Fabaceae). Chem. Pharm. Bull. 1996, 44, 2249–2253. [Google Scholar] [CrossRef]
- Lee, D.; Cuendet, M.; Vigo, J.S.; Graham, J.G.; Cabieses, F.; Fong, H.H.S.; Pezzuto, J.M.; Kinghorn, A.D. A novel cyclooxygenase-inhibitory stilbenolignan from the seeds of Aiphanes aculeata. Org. Lett. 2001, 3, 2169–2171. [Google Scholar] [CrossRef]
- Ha, D.T.; Chen, Q.C.; Hung, T.M.; Ui, J.Y.; Ngoc, T.M.; Thuong, P.T.; Kim, H.J.; Seong, Y.H.; Min, B.S.; Bae, K.H. Stilbenes and oligostilbenes from leaf and stem of Vitis amurensis and their cytotoxic activity. Arch. Pharm. Res. 2009, 32, 177–183. [Google Scholar] [CrossRef]
- Adam, K.P. Phenolic constituents of the fern Phegopteris connectilis. Phytochemistry 1999, 52, 929–934. [Google Scholar] [CrossRef]
- Jin, D.Z.; Min, Z.D.; Chiou, G.C.Y.; Linuma, M.; Tanaka, T. Two p-coumaroyl glycerides from Juncus Effusus. Phytochemistry 1996, 41, 545–547. [Google Scholar] [CrossRef]
- Luo, J.G.; Li, L.; Kong, L.Y. Preparative separation of phenylpropenoid glycerides from the bulbs of Lilium lancifolium by high-speed counter-current chromatography and evaluation of their antioxidant activities. Food Chem. 2012, 131, 1056–1062. [Google Scholar] [CrossRef]
- Fabrice, V.; Michel, G. Enantiomeric purity of scytalone from different fungai sources. Tetrahedron 1990, 46, 2827–2834. [Google Scholar] [CrossRef]
- Limasset, B.; Doucen, C.; Dore, J.C.; Ojasoo, T.; Damon, M.; de Paulet, A.C. Effects of flavonoids on the release of reactive oxygen species by stimulated human neutrophils. Multivariate analysis of structure-activity relationships (SAR). Biochem. Pharmacol. 1993, 46, 1257–1271. [Google Scholar] [CrossRef]
- Satomi, Y. Inhibitory effects of 3′-methyl-3-hydroxy-rchalcone on proliferation of human malignant tumor cells and on skin carcinogenesis. Int. J. Cancer 1993, 55, 506–514. [Google Scholar] [CrossRef]
- Lam, S.H.; Chen, J.M.; Kang, C.J.; Chen, C.H.; Lee, S.S. α-Glucosidase inhibitors from the seeds of Syagrus romanzoffiana. Phytochemistry 2008, 69, 1173–1178. [Google Scholar] [CrossRef]
- Zhou, J.; Qu, J.; Shou, G.X.; Lü, S.H. Determination of astilbin and engeletin in Smilax glabra Roxb. by RP-HPLC. Drug Stand. Chin. 2009, 10, 372–373. [Google Scholar]
- Yuan, J.Z.; Dou, D.Q.; Chen, Y.J.; Li, W.; Kazuo, K.; Tamotsu, N.; Yao, X.S. Studies on dihydroflavonol glycosides from rhizome of Smilax glabra. Chin. J. Chin. Mater. Med. 2004, 29, 867–870. [Google Scholar]
- Yi, Y.J.; Cao, Z.Z.; Yang, D.L.; Cao, Y.; Wu, Y.P.; Zhao, S.X. Studies on the chemical constituents of Smilax glabra. Acta Pharm. Sin. 1998, 33, 873–875. [Google Scholar]
- Chen, G.Y.; Shen, L.S.; Jiang, P.F. Studies on flavanonol glucosides of Smilax glabra Roxb. Chin. J. Chin. Mater. Med. 1996, 21, 355–357. [Google Scholar]
- Wu, B.; Ma, Y.P.; Yuan, J.Z.; Sun, Q.S. Isolation and identification of chemical constituents from rhizomes of Smilax glabra Roxb. J. Shenyang Pharm. Univ. 2010, 27, 116–119. [Google Scholar]
- Zhang, M.; Li, H.T.; Li, Y. Studies on the chemical constituents of Smilax glabra. Chin. Med. Mat. 1995, 18, 191–193. [Google Scholar]
- Yim, N.H.; Ha, D.T.; Trung, T.N.; Kim, J.P.; Lee, S.M.; Na, M.K.; Jung, H.J.; Kim, H.S.; Kim, Y.H.; Bae, K.H. The antimicrobial activity of compounds from the leaf and stem of Vitis amurensis against two oral pathogens. Bioorg. Med. Chem. Lett. 2010, 20, 1165–1168. [Google Scholar] [CrossRef]
- National Committee for Clinical Laboratory Standard, Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. In Approved standard. NCCLS Document M7-A6, 6th ed; NCCLS: Wayne, USA, 2003; Volume 23, pp. 1–50.
- National Committee for Clinical Laboratory Standards, Reference method for broth dilution antifungal susceptibility testing of yeasts. In Approved Standard. NCCLS Document M27-A2, 2nd ed; NCCLS: Pennsylvania, USA, 2002; Volume 22, pp. 1–29.
- Sample Availability: Samples of the compounds 1–63 are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Xu, S.; Shang, M.-Y.; Liu, G.-X.; Xu, F.; Wang, X.; Shou, C.-C.; Cai, S.-Q. Chemical Constituents from the Rhizomes of Smilax glabra and Their Antimicrobial Activity. Molecules 2013, 18, 5265-5287. https://doi.org/10.3390/molecules18055265
Xu S, Shang M-Y, Liu G-X, Xu F, Wang X, Shou C-C, Cai S-Q. Chemical Constituents from the Rhizomes of Smilax glabra and Their Antimicrobial Activity. Molecules. 2013; 18(5):5265-5287. https://doi.org/10.3390/molecules18055265
Chicago/Turabian StyleXu, Shuo, Ming-Ying Shang, Guang-Xue Liu, Feng Xu, Xuan Wang, Cheng-Chao Shou, and Shao-Qing Cai. 2013. "Chemical Constituents from the Rhizomes of Smilax glabra and Their Antimicrobial Activity" Molecules 18, no. 5: 5265-5287. https://doi.org/10.3390/molecules18055265
APA StyleXu, S., Shang, M.-Y., Liu, G.-X., Xu, F., Wang, X., Shou, C.-C., & Cai, S.-Q. (2013). Chemical Constituents from the Rhizomes of Smilax glabra and Their Antimicrobial Activity. Molecules, 18(5), 5265-5287. https://doi.org/10.3390/molecules18055265




