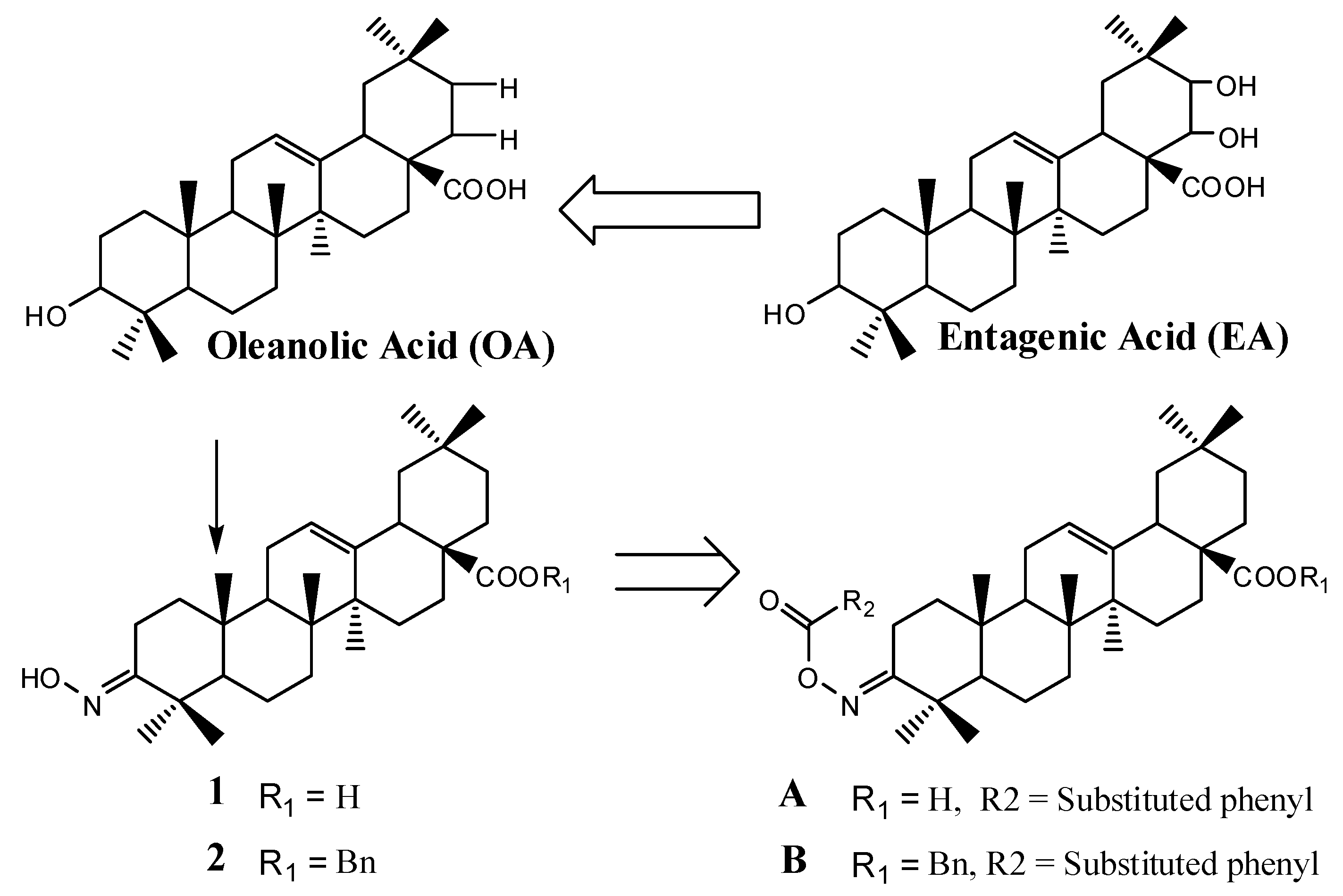
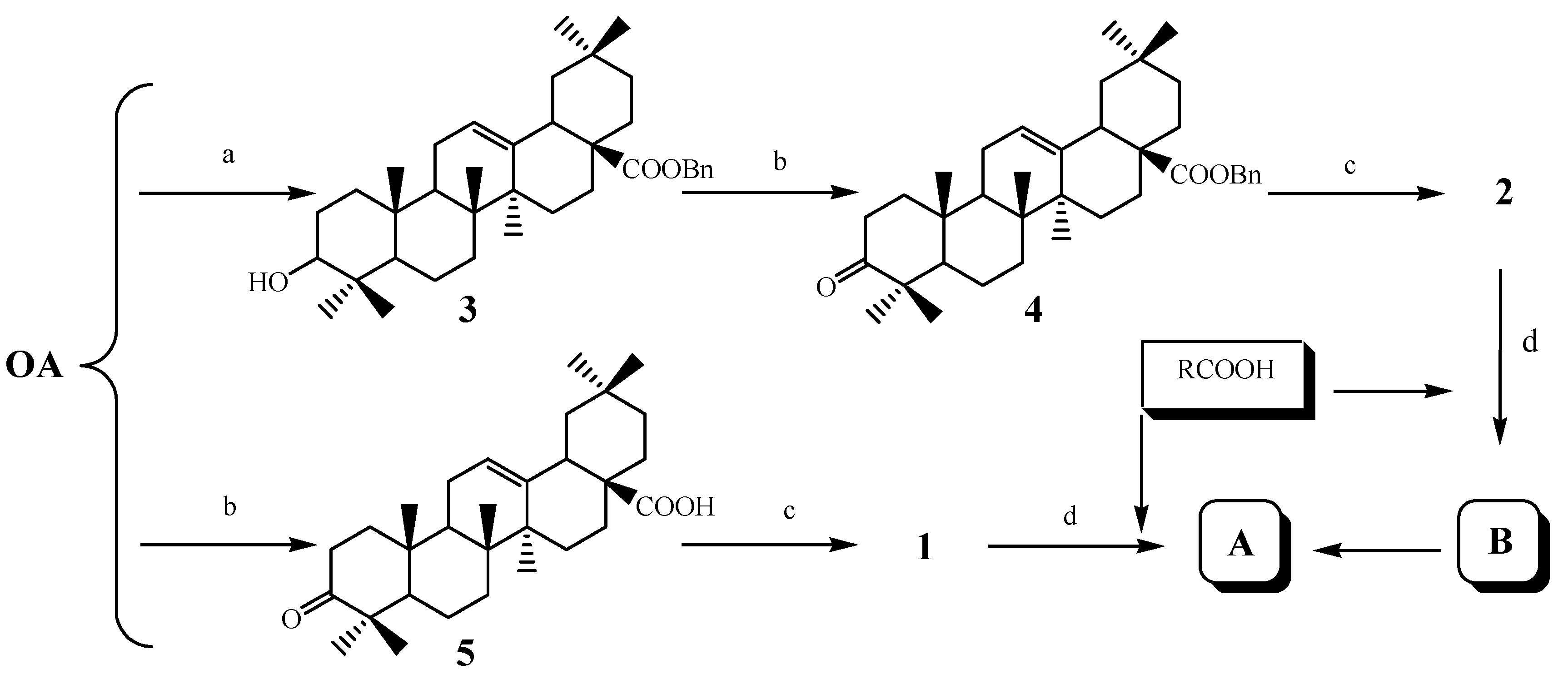
3.2. Chemical Synthesis
Oleanolic acid 3-oxime ester (
A-01). 4-Chlorobenzoic acid (0.66 g, 4.2 mmol) and
N,N′-dicyclohexylcarbodiimide (DCC, 1 g, 5 mmol) were successively added to a soln. of oleanate 3-oxime
1 (1.68 g, 3.5 mmol) which was prepared according to the reported method [
12] in CH
2Cl
2 (50 mL), Then the reaction mixture was refluxed for 8–14 h at the end of which time TLC (4:1 petroleum ether/EtOAc) indicated that the reaction was complete. The reaction mixture was filtered, the soln. was concentrated, and the residue was subjected to column chromatography (6:1 petroleum ether/EtOAc) to give the desired product
A-01 (1.98 g, 93%) as a white foamy solid.
1H-NMR (CDCl
3):
δ 8.00–7.42 (m, 4H, Ar-H), 5.28 (br s, 1 H, H-12), 3.05–3.01 (m, 1H), 2.85–2.80 (m, 1H), 2.48-2.41 (m, 1H), 1.34, 1.19, 1.13, 1.06, 0.93, 0.90, 0.80 (s, 7 × 3H, CH
3);
13C-NMR (CDCl
3): 184.0 (COOH), 176.4 (
COONC), 163.4 (COON
C), 143.8 (C-13), 139.4, 130.9, 130.9, 128.8, 128.8, 128.1 (aromatic carbons), 122.2 (C-12), 55.8, 47.1, 46.6, 45.8, 41.7, 41.0, 39.3, 38.7, 37.1, 33.8, 33.0, 32.4, 32.3, 30.6, 29.7, 27.6, 27.2, 25.8, 23.5, 23.4, 23.2, 22.9, 19.9, 18.9, 17.0, 15.1 (7 ×
CH
3); Anal. Calcd for C
37H
50ClNO
4: C, 73.06; H, 8.29; N, 2.30. found: C, 73.27; H, 8.05; N, 2.51; HRMS calcd for C
37H
50ClNO
4 (M+H)
+: 608.35011, found: 608.34985.
Oleanolic acid 3-oxime ester (A-02). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-02 was obtained in 90% yield. 1H-NMR (CDCl3): δ 8.00–7.42 (m, 4H, Ar-H), 5.28 (br s, 1 H, H-12), 3.05–3.01 (m, 1H), 2.85–2.80 (m, 1H), 2.48–2.41 (m, 1H), 1.34, 1.19, 1.13, 1.06, 0.93, 0.90, 0.80 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 184.3 (COOH), 176.5 (COONC), 163.1 (COONC), 143.7 (C-13), 138.2, 134.3, 132.4, 130.8, 128.4, 127.0 (aromatic carbons), 122.2 (C-12), 55.8, 47.1, 46.5, 45.8, 41.6, 40.9, 39.3, 38.7, 37.0, 33.7, 33.0, 32.4, 32.2, 30.6, 29.6, 27.6, 27.0, 25.8, 23.5, 23.4, 23.0, 22.8, 20.2, 18.9, 17.0, 15.1 (7 × CH3); Anal. Calcd for C37H49Cl2NO4: C, 69.15; H, 7.68; N, 2.18. found: C, 69.35; H, 7.47; N, 2.33; HRMS calcd for C37H50ClNO4 (M+H)+: 642.31114, found: 642.31079.
Oleanolic acid 3-oxime ester (A-03). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-03 was obtained in 91% yield. 1H-NMR (CDCl3): δ 8.01–7.37 (m, 3H, Ar-H), 5.28 (br s, 1 H, H-12), 3.06–3.01 (m, 1H), 2.86–2.80 (m, 1H), 2.47–2.45 (m, 1H), 1.34, 1.19, 1.13, 1.07, 0.93, 0.90, 0.80 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 184.3 (COOH), 176.4 (COONC), 163.0 (COONC), 143.7 (C-13), 134.5, 133.0, 131.4, 129.7, 129.4, 127.6 (aromatic carbons), 122.2 (C-12), 55.8, 47.1, 46.5, 45.8, 41.6, 40.9, 39.3, 38.6, 37.0, 33.7, 33.0, 32.4, 32.2, 30.6, 29.6, 27.6, 27.0, 25.8, 23.5, 23.4, 23.0, 22.8, 20.2, 18.9, 17.0, 15.1 (7 × CH3); Anal. Calcd for C37H50ClNO4: C, 73.06; H, 8.29; N, 2.30. found: C, 73.21; H, 8.43; N, 2.49; HRMS calcd for C37H50ClNO4 (M+H)+: 608.35011, found: 608.34937.
Oleanolic acid 3-oxime ester (A-04). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-04 was obtained in 84% yield. 1H-NMR (CDCl3): δ 8.33–8.20 (m, 4H, Ar-H), 5.33 (br s, 1 H, H-12), 3.07–3.01 (m, 1H), 2.88–2.82 (m, 1H), 2.49–2.43 (m, 1H), 1.36, 1.19, 1.15, 1.07, 0.94, 0.92, 0.83 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.0 (COOH), 172.8 (COONC), 162.3 (COONC), 150.5, 143.3 (C-13), 135.1, 130.5, 130.5, 123.6, 123.6 (aromatic carbons), 122.6 (C-12), 55.8, 48.3, 47.1, 45.6, 41.8, 41.7, 39.4, 38.7, 37.0, 33.6, 32.9, 32.3, 31.9, 30.6, 29.6, 27.4, 27.1, 25.7, 23.5, 23.2, 22.9, 22.6, 20.0, 18.9, 17.1, 15.1 (7 × CH3); Anal. Calcd for C37H50N2O6: C, 71.82; H, 8.14; N, 4.53. found: C, 71.97; H, 8.01; N, 4.69; HRMS calcd for C37H50ClNO4 (M+H)+: 619.37416, found: 619.37708.
Oleanolic acid 3-oxime ester (A-05). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-05 was obtained in 86% yield. 1H-NMR (CDCl3): δ 8.05–7.41(m, 7H, Ar-H), 5.27 (br s, 1 H, H-12), 4.20–4.18 (m, 2H, CH2), 2.84–2.78 (m, 1H), 2.58–2.53 (m, 1H), 1.25, 1.19, 1.09, 1.04, 0.93, 0.90, 0.74 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 184.2 (COOH), 175.5 (COONC), 169.3 (COONC), 143.6 (C-13), 133.7, 132.0, 130.2, 128.6, 127.9, 126.2, 125.7, 125.6, 125.3, 123.8 (aromatic carbons), 122.2 (C-12), 65.4, 55.7, 47.0, 46.5, 45.7, 41.6, 41.3, 40.9, 39.2, 38.2, 36.8, 33.7, 33.0, 32.3, 32.2, 30.6, 29.6, 27.5, 26.9, 25.8, 23.5, 23.3, 22.8, 19.2, 18.7, 16.9, 14.9 (7 × CH3); Anal. Calcd for C42H55NO4: C, 79.08; H, 8.69; N, 2.20. found: C, 79.24; H, 8.37; N, 2.42; HRMS calcd for C37H50ClNO4 (M+H)+: 638.42039, found: 638.42004.
Oleanolic acid 3-oxime ester (A-06). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-06 was obtained in 87% yield. 1H-NMR (CDCl3): δ 7.26–6.81 (m, 4H, Ar-H), 5.28 (br s, 1 H, H-12), 4.82 (s, 1H), 4.72(s, 1H), 2.87–2.82 (m, 1H), 2.30–2.21 (m, 1H), 1.26, 1.24, 1.11, 1.01, 0.93, 0.91, 0.78 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 183.9 (COOH), 175.8 (COONC), 167.8 (COONC), 156.4, 153.1, 143.6 (C-13), 129.3, 126.6, 122.1 (C-12), 116.0, 115.8 (aromatic carbons), 65.4, 55.7, 47.0, 46.4, 45.7, 41.6, 40.9, 39.2, 38.5, 36.9, 33.7, 32.9, 32.5, 32.3, 30.5, 29.6, 27.5, 27.0, 25.7, 23.4, 23.3, 22.9, 22.8, 19.4, 18.8, 16.9, 15.0 (7 × CH3); Anal. Calcd for C38H52ClNO5: C, 71.51; H, 8.21; N, 2.19. found: C, 71.35; H, 8.39; N, 2.35; HRMS calcd for C37H50ClNO4 (M+H)+: 638.36068, found: 638.35919.
Oleanolic acid 3-oxime ester (A-07). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-07 was obtained in 80% yield. 1H-NMR (CDCl3): δ 8.05–7.11(m, 4H, Ar-H), 5.29 (br s, 1 H, H-12), 3.12–3.07 (m, 1H), 2.85–2.82 (m, 1H), 2.49–2.38 (m, 1H), 1.34, 1.19, 1.13, 1.06, 0.93, 0.90, 0.80 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 184.5 (COOH), 176.1 (COONC), 163.1 (COONC), 143.6 (C-13), 134.3, 132.2, 124.0, 122.2 (C-12), 118.0, 116.9, 116.6 (aromatic carbons), 55.7, 47.0, 46.5, 45.7, 41.4, 40.9, 39.2, 38.6, 36.9, 33.7, 32.9, 32.3, 32.2, 30.5, 29.6, 27.5, 27.1, 25.7, 23.4, 23.3, 23.0, 22.7, 19.9, 18.8, 16.9, 15.0 (7 × CH3); Anal. Calcd for C37H50FNO4: C, 75.09; H, 8.52; N, 2.37. found: C, 75.29; H, 8.38; N, 2.16; HRMS calcd for C37H50ClNO4 (M+H)+: 592.37966, found: 592.37909.
Oleanolic acid 3-oxime ester (A-08). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-08 was obtained in 90% yield. 1H-NMR (CDCl3): δ 7.95–7.58 (m, 4H, Ar-H), 5.29 (br s, 1 H, H-12), 3.05–3.00 (m, 1H), 2.86–2.81 (m, 1H), 2.44–2.42 (m, 1H), 1.34, 1.19, 1.13, 1.06, 0.90, 0.88, 0.80 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 184.3 (COOH), 176.4 (COONC), 163.5 (COONC), 143.8 (C-13), 131.9, 131.8, 131.0, 131.0, 128.6, 128.1 (aromatic carbons), 122.3 (C-12), 55.8, 47.2, 45.8, 41.7, 41.6, 41.0, 39.4, 38.7, 37.1, 33.8, 33.0, 32.4, 31.9, 30.7, 29.7, 27.6, 27.2, 25.9, 23.6, 23.5, 23.2, 22.7, 19.9, 19.0, 17.0, 15.1 (7 × CH3); Anal. Calcd for C37H50BrNO4: C, 68.09; H, 7.72; N, 2.15. found: C, 68.39; H, 7.46; N, 2.35; HRMS calcd for C37H50ClNO4 (M+H)+: 652.29960, found: 652.30011.
Oleanolic acid 3-oxime ester (A-09). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-09 was obtained in 91% yield. 1H-NMR (CDCl3): δ 9.25–7.44 (m, 4H, Ar-H), 5.30 (br s, 1 H, H-12), 3.08–3.03 (m, 1H), 2.90–2.84 (m, 1H), 2.48–2.41 (m, 1H), 1.35, 1.21, 1.14, 1.07, 0.94, 0.91, 0.82 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 183.1 (COOH), 176.7 (COONC), 162.7 (COONC), 153.0, 150.1, 143.9 (C-13), 137.3, 125.9, 123.6 (aromatic carbons), 122.0 (C-12), 55.7, 47.1, 46.4, 45.8, 41.6, 41.0, 39.2, 38.6, 37.0, 33.6, 33.0, 32.4, 32.2, 30.6, 29.6, 27.6, 27.1, 25.8, 23.5, 23.4, 23.1, 22.9, 19.9, 18.9, 16.9, 15.1 (7 × CH3); Anal. Calcd for C36H50N2O4: C, 75.22; H, 8.77; N, 4.87. found: C, 75.07; H, 8.64; N, 4.63; HRMS calcd for C37H50ClNO4 (M+H)+: 575.38433, found: 575.38373.
Oleanolic acid 3-oxime ester (A-10). The reaction was run similarly to that used to synthesize A-01. A white foamy solid A-10 was obtained in 86% yield. 1H-NMR (CDCl3): δ 7.63–6.47 (m, 3H, Ar-H), 5.28 (br s, 1 H, H-12), 3.07–3.02 (m, 1H), 2.87–2.81 (m, 1H), 2.48–2.42 (m, 1H), 1.33, 1.17, 1.13, 1.05, 0.93, 0.90, 0.80 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 183.8 (COOH), 176.2 (COONC), 156.6 (COONC), 146.4, 143.8, 143.7 (C-13), 122.1(C-12), 118.0, 111.7 (aromatic carbons), 55.7, 47.1, 46.5, 45.8, 41.5, 40.9, 39.3, 38.6, 37.0, 33.7, 33.0, 32.5, 32.3, 30.6, 30.6, 27.6, 27.1, 25.5, 23.5, 23.4, 23.1, 22.8, 19.7, 18.9, 17.0, 15.0 (7 × CH3); Anal. Calcd for C35H49NO5: C, 74.57; H, 8.76; N, 2.48. found: C, 74.42; H, 8.91; N, 2.25; HRMS calcd for C37H50ClNO4 (M+H)+: 564.36835, found: 564.36804.
Benzyl oleanolic acid 3-oxime ester (
B-01). 4-Chlorobenzoic acid (0.66 g, 4.2 mmol) and DCC (1 g, 5 mmol) were successively added to a soln. of benzyl oleanate 3-oxime
2 (2.00 g, 3.5 mmol) which was prepared according to the reported method [
13] in CH
2Cl
2 (50 mL), Then the reaction mixture was refluxed for 8–14 h at the end of which time TLC (6:1 petroleum ether-EtOAc) indicated that the reaction was complete. The reaction mixture was filtered, the soln was concentrated, and the residue was subjected to column chromatography (8:1 petroleum ether-EtOAc) to give the desired product
B-01 (1.98 g, 80%) as a white foamy solid.
1H-NMR (CDCl
3):
δ 8.01–7.31 (m, 9H, Ar-H), 5.29 (br s, 1 H, H-12), 5.08 (dd, 2H,
J = 12.5, 17.4 Hz, Ar-C
H2), 3.04–2.89 (m, 2H), 2.47–2.33 (m, 1H), 1.34, 1.19, 1.12, 1.03, 0.92, 0.89, 0.64 (s, 7 × 3H, CH
3);
13C-NMR (CDCl
3): 177.3 (
COOBn), 176.4 (
COONC), 163.3 (COON
C), 143.8 (C-13), 139.4, 134.4, 130.8, 130.8, 128.8, 128.8, 128.4, 128.4, 128.4, 128.1, 127.9, 127.9 (aromatic carbons), 122.1 (C-12), 65.9, 55.7, 47.1, 46.7, 45.8, 41.7, 41.5, 41.4, 39.3, 38.7, 36.9, 33.8, 33.0, 32.3, 32.3, 30.6, 27.5, 27.2, 25.7, 23.6, 23.4, 23.2, 23.0, 19.8, 18.9, 16.8, 15.1 (7 ×
CH
3); Anal. Calcd for C
44H
56ClNO
4: C, 75.67; H, 8.08; N, 2.01. found: C, 75.52; H, 8.33; N, 2.17; HRMS calcd for C
37H
50ClNO
4 (M+H)
+: 698.39706, found: 698.39526.
Benzyl oleanolic acid 3-oxime ester (B-02). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-02 was obtained in 83% yield. 1H-NMR (CDCl3): δ 7.80–7.29 (m, 8H, Ar-H), 5.28 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.04–2.89 (m, 2H), 2.48–2.33 (m, 1H), 1.32, 1.18, 1.12, 1.01, 0.92, 0.89, 0.64 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.4 (COOBn), 176.6 (COONC), 163.1 (COONC), 143.9 (C-13), 138.2, 136.4, 134.3, 132.4, 130.8, 128.5, 128.4, 128.4, 128.0, 128.0, 127.9, 127.1 (aromatic carbons), 122.1 (C-12), 65.9, 55.9, 47.1, 46.7, 45.8, 41.8, 41.7, 41.4, 39.3, 38.8, 37.0, 33.9, 33.1, 32.4, 32.3, 30.7, 27.6, 27.1, 25.8, 23.6, 23.5, 23.1, 23.0, 20.2, 19.0, 16.8, 15.1 (7 × CH3); Anal. Calcd for C44H55Cl2NO4: C, 72.11; H, 7.56; N, 1.91. found: C, 72.31; H, 7.49; N, 1.77; HRMS calcd for C37H50ClNO4 (M+H)+: 732.35809, found: 732.35529.
Benzyl oleanolic acid 3-oxime ester (B-03). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-03 was obtained in 79% yield. 1H-NMR (CDCl3): δ 8.01–7.32 (m, 9H, Ar-H), 5.30 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.04–2.88 (m, 2H), 2.48–2.33 (m, 1H), 1.34, 1.19, 1.12, 1.02, 0.92, 0.89, 0.65 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.3 (COOBn), 176.6 (COONC), 163.0 (COONC), 143.8 (C-13), 136.3, 134.5, 133.0, 131.4, 129.8, 129.4, 128.4, 128.4, 127.9, 127.9, 127.9, 127.6 (aromatic carbons), 122.1 (C-12), 65.9, 55.7, 47.1, 46.7, 45.8, 41.7, 41.6, 41.4, 39.3, 38.7, 36.9, 33.8, 33.0, 32.3, 32.3, 30.6, 27.5, 27.1, 25.7, 23.6, 23.4, 23.2, 23.0, 19.9, 18.9, 16.8, 15.1 (7 × CH3); Anal. Calcd for C44H56ClNO4: C, 75.67; H, 8.08; N, 2.01. found: C, 75.52; H, 8.27; N, 2.27; HRMS calcd for C37H50ClNO4 (M+H)+: 698.39706, found: 698.39697.
Benzyl oleanolic acid 3-oxime ester (B-04). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-04 was obtained in 85% yield. 1H-NMR (CDCl3): δ 8.87–7.29 (m, 9H, Ar-H), 5.31 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.06–2.90 (m, 2H), 2.50–2.48 (m, 1H), 1.35, 1.21, 1.12, 1.04, 0.92, 0.90, 0.65 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.2 (COOBn), 177.1 (COONC), 162.1 (COONC), 148.2, 143.8 (C-13), 136.3, 135.1, 131.5, 129.7, 128.3, 128.3, 127.9, 127.9, 127.8, 127.3, 124.2 (aromatic carbons), 122.1 (C-12), 65.8, 55.7, 47.1, 46.7, 45.8, 41.7, 41.6, 41.4, 39.3, 38.6, 36.9, 33.8, 33.0, 32.3, 32.3, 30.6, 27.5, 27.2, 25.7, 23.5, 23.4, 23.2, 23.0, 20.0, 18.9, 16.8, 15.0 (7 × CH3); Anal. Calcd for C44H56N2O6: C, 74.55; H, 7.96; N, 3.95. found: C, 74.40; H, 7.79; N, 3.65; HRMS calcd for C37H50ClNO4 (M+H)+: 709.42111, found: 709.42096.
Benzyl oleanolic acid 3-oxime ester (B-05). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-05 was obtained in 75% yield. 1H-NMR (CDCl3): δ 8.04–7.23 (m, 12H, Ar-H), 5.28 (br s, 1 H, H-12), 5.06 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 4.19 (s, 2 H, CH2C=O), 2.93–2.87 (m, 1H), 2.57–2.50 (m, 1H), 1.19, 1.08, 1.04, 0.92, 0.89, 0.89, 0.60 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.3 (COOBn), 175.6 (COONC), 169.3 (COONC), 143.7 (C-13), 136.3, 133.7, 132.0, 130.2, 128.6, 128.5, 128.3, 127.9, 127.9, 127.8, 127.8, 126.2, 125.7, 125.6, 125.3, 123.8 (aromatic carbons), 122.1 (C-12), 65.8, 55.7, 47.0, 46.6, 45.7, 41.7, 41.3, 41.3, 39.2, 38.5, 38.2, 36.8, 36.8, 33.8, 33.0, 32.2, 30.6, 27.5, 26.9, 25.7, 23.6, 23.3, 22.9, 22.9, 19.3, 18.8, 16.7, 15.0 (7 × CH3); Anal. Calcd for C49H61NO4: C, 80.84; H, 8.45; N, 1.92. found: C, 80.69; H, 8.68; N, 1.69; HRMS calcd for C37H50ClNO4 (M+H)+: 728.46734, found: 728.46869.
Benzyl oleanolic acid 3-oxime ester (B-06). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-06 was obtained in 72% yield. 1H-NMR (CDCl3): δ 7.36–6.85 (m, 9H, Ar-H), 5.29 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 4.82 (s, 2 H, CH2C=O), 2.94–2.82 (m, 2H), 2.32–2.26 (m, 1H), 1.23, 1.12, 1.11, 0.98, 0.92, 0.90, 0.63 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.3 (COOBn), 176.0 (COONC), 167.9 (COONC), 156.5, 143.7 (C-13), 136.3, 129.4, 129.4, 128.4, 128.4, 127.9, 127.9, 127.9, 126.6, 122.1 (C-12), 116.1, 116.1 (aromatic carbons), 65.9, 65.4, 55.8, 47.1, 46.7, 45.7, 41.7, 41.5, 41.3, 39.2, 38.6, 36.9, 33.8, 33.0, 32.3, 30.6, 30.6, 27.5, 27.0, 25.7, 23.6, 23.4, 23.0, 23.0, 19.4, 18.9, 16.8, 15.0 (7 × CH3); Anal. Calcd for C45H58ClNO5: C, 74.20; H, 8.03; N, 1.92. found: C, 74.06; H, 8.29; N, 1.72; HRMS calcd for C37H50ClNO4 (M+H)+: 728.40763, found: 728.40668.
Benzyl oleanolic acid 3-oxime ester (B-07). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-07 was obtained in 75% yield. 1H-NMR (CDCl3): δ 8.80–7.27 (m, 9H, Ar-H), 5.30 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.04–2.89 (m, 2H), 2.47–2.33 (m, 1H), 1.33, 1.18, 1.11, 1.01, 0.92, 0.89, 0.64 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.0 (COOBn), 176.0 (COONC), 162.2 (COONC), 143.5 (C-13), 136.2, 132.6, 132.1, 128.2, 128.2, 128.2, 127.7, 127.7, 127.7, 123.9, 121.9 (C-12), 116.8, 116.5 (aromatic carbons), 65.6, 55.6, 46.9, 46.4, 45.5, 41.5, 41.2, 41.2, 39.0, 38.5, 36.7, 33.6, 32.9, 32.1, 32.1, 30.4, 27.3, 26.9, 25.5, 23.4, 23.2, 22.9, 22.8, 19.8, 18.7, 16.6, 14.8 (7 × CH3); Anal. Calcd for C44H56FNO4: C, 77.50; H, 8.28; N, 2.05. found: C, 77.41; H, 8.41; N, 2.25; HRMS calcd for C37H50ClNO4 (M+H)+: 682.42661, found: 682.42645.
Benzyl oleanolic acid 3-oxime ester (B-08). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-08 was obtained in 78% yield. 1H-NMR (CDCl3): δ 7.93–7.26 (m, 9H, Ar-H), 5.29 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.04–2.89 (m, 2H), 2.44–2.33 (m, 1H), 1.33, 1.19, 1.11, 1.02, 0.91, 0.89, 0.64 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.2 (COOBn), 176.3 (COONC), 163.4 (COONC), 143.8 (C-13), 136.3, 131.7, 131.7, 130.9, 130.9, 128.6, 128.3, 128.3, 128.0, 127.9, 127.9, 127.8 (aromatic carbons), 122.1 (C-12), 65.8, 55.7, 47.0, 46.6, 45.7, 41.7, 41.5, 41.4, 39.3, 38.6, 36.9, 33.7, 33.0, 32.3, 32.3, 30.6, 27.5, 27.2, 25.7, 23.5, 23.4, 23.2, 23.0, 19.8, 18.9, 16.8, 15.0 (7 × CH3); Anal. Calcd for C44H56BrNO4: C, 71.14; H, 7.60; N, 1.89. found: C, 71.35; H, 7.53; N, 1.60; HRMS calcd for C37H50ClNO4 (M+H)+: 742.34655, found: 742.34674.
Benzyl oleanolic acid 3-oxime ester (B-09). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-09 was obtained in 81% yield. 1H-NMR (CDCl3): δ 9.25–7.28 (m, 8H, Ar-H), 5.30 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.06–2.89(m, 2H), 2.48–2.33 (m, 1H), 1.34, 1.20, 1.12, 1.03, 0.92, 0.90, 0.65 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.2 (COOBn), 176.7 (COONC), 162.8 (COONC), 153.3, 150.5, 143.7 (C-13), 136.9, 136.3, 128.3, 128.3, 127.9, 127.9, 127.8, 125.7, 123.4 (aromatic carbons), 122.0 (C-12), 65.8, 55.7, 47.0, 46.6, 45.7, 41.7, 41.6, 41.4, 39.2, 38.6, 36.9, 33.8, 33.0, 32.3, 32.2, 30.6, 27.5, 27.1, 25.7, 23.5, 23.4, 23.1, 23.0, 19.9, 18.9, 16.8, 15.0 (7 × CH3); Anal. Calcd for C43H56N2O4: C, 77.67; H, 8.49; N, 4.21. found: C, 77.73; H, 8.62; N, 4.03; HRMS calcd for C37H50ClNO4 (M+H)+: 665.43128, found: 665.43182.
Benzyl oleanolic acid 3-oxime ester (B-10). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-10 was obtained in 71% yield. 1H-NMR (CDCl3): δ 7.60–6.51(m, 8H, Ar-H), 5.29 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.06–2.88 (m, 2H), 2.43–2.33 (m, 1H), 1.32, 1.25, 1.11, 1.02, 0.92, 0.89, 0.64 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.3 (COOBn), 176.3 (COONC), 156.6 (COONC), 146.4, 143.8, 143.7 (C-13), 136.4, 128.4, 128.4, 128.0, 128.0, 127.9, 122.1 (C-12), 118.0, 111.8 (aromatic carbons), 65.9, 55.7, 47.1, 46.7, 45.8, 41.7, 41.5, 41.4, 39.3, 38.7, 36.9, 33.8, 33.0, 32.3, 32.3, 30.6, 27.5, 27.2, 25.7, 23.6, 23.4, 23.2, 23.0, 19.8, 18.9, 16.8, 15.1 (7 × CH3); Anal. Calcd for C42H55NO5: C, 77.15; H, 8.48; N, 2.14. found: C, 77.01; H, 8.30; N, 2.27; HRMS calcd for C37H50ClNO4 (M+H)+: 654.41530, found: 654.41504.
Benzyl oleanolic acid 3-oxime ester(B-11). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-11 was obtained in 84% yield. 1H-NMR (CDCl3): δ 8.86–7.28 (m, 9H, Ar-H), 5.30 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.06–2.90 (m, 2H), 2.50–2.48 (m, 1H), 1.35, 1.21, 1.12, 1.04, 0.92, 0.90, 0.65 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.2 (COOBn), 177.1 (COONC), 162.1 (COONC), 148.2, 143.7 (C-13), 136.3, 135.1, 131.4, 129.7, 128.3, 128.3, 127.9, 127.9, 127.8, 127.3, 124.2 (aromatic carbons), 122.0 (C-12), 65.8, 55.7, 47.0, 46.6, 45.7, 41.7, 41.6, 41.4, 39.2, 38.6, 36.9, 33.8, 33.0, 32.3, 32.2, 30.6, 27.5, 27.2, 25.7, 23.4, 23.4, 23.2, 22.9, 20.0, 19.0, 16.7, 15.0 (7 × CH3); Anal. Calcd for C44H56N2O6: C, 74.55; H, 7.96; N, 3.95. found: C, 74.30; H, 7.88; N, 3.68; HRMS calcd for C37H50ClNO4 (M+H)+: 709.42111, found: 709.42267.
Benzyl oleanolic acid 3-oxime ester (B-12). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-12 was obtained in 90% yield. 1H-NMR (CDCl3): δ 9.24–7.28 (m, 8H, Ar-H), 5.30 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.04–2.88 (m, 2H), 2.54–2.51 (m, 1H), 1.35, 1.22, 1.13, 1.05, 0.92, 0.90, 0.65 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 178.0 (COOBn), 177.3 (COONC), 160.3 (COONC), 148.7, 143.8 (C-13), 136.3, 133.4, 129.2, 129.2, 128.4, 128.4, 128.0, 128.0, 127.9, 127.9, 122.3 (aromatic carbons), 122.0 (C-12), 65.9, 55.7, 47.1, 46.7, 45.8, 41.9, 41.7, 41.4, 39.3, 38.6, 37.0, 33.8, 33.0, 32.3, 32.3, 30.6, 27.5, 27.1, 25.7, 23.6, 23.4, 23.2, 23.0, 20.2, 18.9, 16.8, 15.1 (7 × CH3); Anal. Calcd for C44H55N3O8: C, 70.10; H, 7.35; N, 5.57. found: C, 70.43; H, 7.31; N, 5.85; HRMS calcd for C37H50ClNO4 (M+H)+: 754.40619, found: 754.40375.
Benzyl oleanolic acid 3-oxime ester (B-13). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-13 was obtained in 76% yield. 1H-NMR (CDCl3): δ 7.82–7.30 (m, 9H, Ar-H), 5.29 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.07–2.88 (m, 2H), 2.42–2.32 (m, 1H), 1.32, 1.18, 1.12, 1.01, 0.92, 0.89, 0.64 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.3 (COOBn), 176.4 (COONC), 163.9 (COONC), 143.8 (C-13), 136.3, 133.1, 132.3, 131.3, 130.8, 130.2, 128.4, 128.4, 127.9, 127.9, 127.9, 126.6 (aromatic carbons), 122.1 (C-12), 65.9, 55.8, 47.1, 46.7, 45.7, 41.7, 41.6, 41.4, 39.2, 38.8, 36.9, 33.8, 33.0, 32.3, 32.3, 30.6, 27.5, 27.0, 25.7, 23.6, 23.4, 23.1, 23.0, 20.1, 18.9, 16.8, 15.1 (7 × CH3); Anal. Calcd for C44H56ClNO4: C, 75.67; H, 8.08; N, 2.01. found: C, 75.55; H, 8.12; N, 2.32; HRMS calcd for C37H50ClNO4 (M+H)+: 698.39706, found: 698.39709.
Benzyl oleanolic acid 3-oxime ester (B-14). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-14 was obtained in 79% yield. 1H-NMR (CDCl3): δ 8.80–7.27 (m, 9H, Ar-H), 5.30 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.04–2.89 (m, 2H), 2.47–2.33 (m, 1H), 1.34, 1.20, 1.12, 1.03, 0.92, 0.90, 0.64 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.1 (COOBn), 176.9 (COONC), 162.5 (COONC), 150.4, 150.4, 143.7 (C-13), 136.8, 136.2, 128.2, 128.2, 128.2, 127.8, 127.8, 127.7, 122.6 (aromatic carbons), 121.9 (C-12), 65.7, 55.6, 46.9, 46.5, 45.6, 41.6, 41.5, 41.2, 39.1, 38.5, 36.8, 33.6, 32.9, 32.1, 32.1, 30.5, 27.4, 27.0, 25.6, 23.4, 23.2, 23.0, 22.8, 19.8, 18.8, 16.6, 14.9 (7 × CH3); Anal. Calcd for C43H56N2O4: C, 77.67; H, 8.49; N, 4.21. found: C, 77.35; H, 8.53; N, 4.02; HRMS calcd for C37H50ClNO4 (M+H)+: 665.43128, found: 665.43146.
Benzyl oleanolic acid 3-oxime ester (B-15). The reaction was run similarly to that used to synthesize B-01. A white foamy solid B-15 was obtained in 70% yield. 1H-NMR (CDCl3): δ 8.07–7.32 (m, 10H, Ar-H), 5.30 (br s, 1 H, H-12), 5.08 (dd, 2H, J = 12.5, 17.4 Hz, Ar-CH2), 3.08–2.88 (m, 2H), 2.47–2.31 (m, 1H), 1.35, 1.19, 1.12, 1.03, 0.92, 0.89, 0.65 (s, 7 × 3H, CH3); 13C-NMR (CDCl3): 177.3 (COOBn), 176.2 (COONC), 164.2 (COONC), 143.8 (C-13), 136.3, 132.9, 129.7, 129.4, 129.4, 128.4, 128.4, 128.4, 128.4, 127.9, 127.9, 127.9 (aromatic carbons), 122.1 (C-12), 65.9, 55.7, 47.1, 46.7, 45.7, 41.7, 41.5, 41.4, 39.3, 38.7, 36.9, 33.8, 33.0, 32.3, 32.3, 30.6, 27.5, 27.1, 25.7, 23.6, 23.4, 23.2, 23.0, 19.8, 18.9, 16.8, 15.1 (7 × CH3); Anal. Calcd for C44H57NO4: C, 79.60; H, 8.65; N, 2.11. found: C, 79.55; H, 8.83; N, 2.38; HRMS calcd for C37H50ClNO4 (M+H)+: 664.43606, found: 664.43500.