Techniques for Analysis of Plant Phenolic Compounds
Abstract
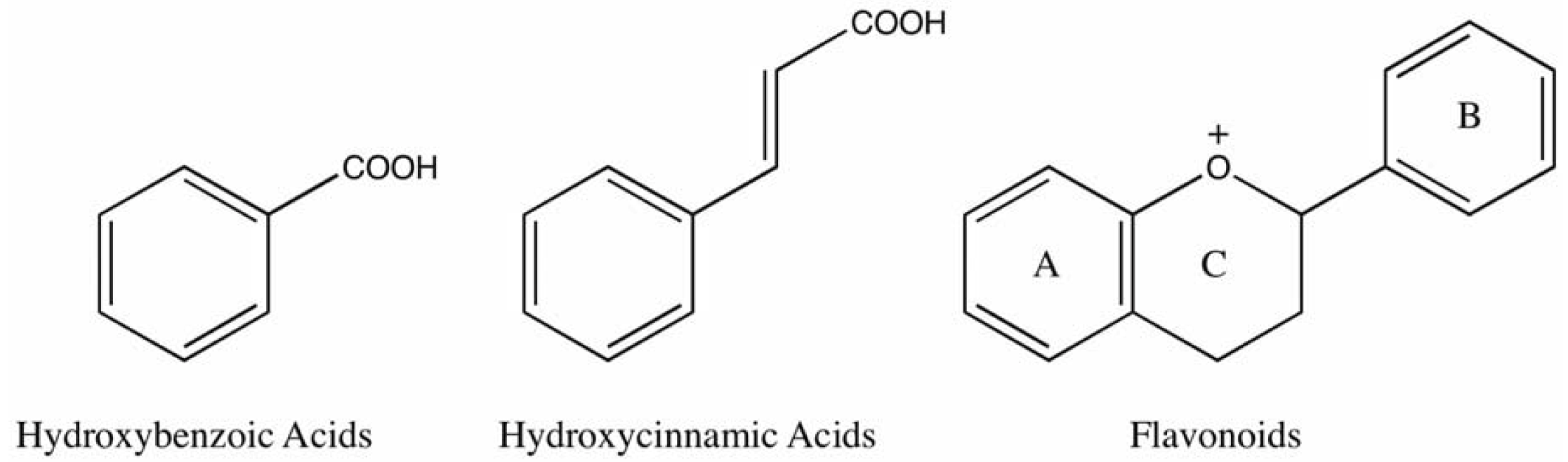
:1. Introduction
2. Sample Preparation
3. Overview of Phenolic Extraction
3.1. Phenolic Acid Extraction
3.2. Flavonoid Extraction
3.3. Anthocyanin/Proanthocyanidin Extraction
4. Modern Extraction Techniques for Phenolics
4.1. Ultrasound-Assisted Extraction (UAE)
| Sample | Solvent | Extraction time (min) | Phenolic class | Yield (mg GAE b/g) | Reference |
|---|---|---|---|---|---|
| Puerariae lobatae radix | Ethanol 80% | 55 | Isoflavones | 128 | [53] |
| Vitis vinifera | Methanol | 60 | TPC a and flavonoid | 55.90 | [36] |
| Galla chinensis | Ethanol 70% | 40 | Tannin | 491.2 | [85] |
| Sunflower meal | Acetone 80% | 30 | TPC a | 30.93 | [37] |
| Orange peel | Ethanol 80% | 30 | TPC a | 2.758 | [86] |
| Satsuma mandarin peel | Methanol 80% | 60 | Hesperidine | 1.446 | [87] |
| Aerial parts of Potentilla atrosanguinea | Ethanol 50% | 60 | TPC a | 27.80 | [35] |
| Soy beans | Ethanol 40–60% | 20 | Isoflavones | 1.353 | [88] |
4.2. Microwave-Assisted Extraction (MAE)
| Solvent | Formula | Boiling point (°C) | Dielectric constant a | Dissipation factor |
|---|---|---|---|---|
| Acetonitrile | C2H3N | 81.60 | 37.50 | 620 |
| Water | H2O | 100 | 78.30 | 1570 |
| Ethanol | C4H8O2 | 78.5 | 24.30 | 2500 |
| Acetone | C3H6O | 56.2 | 20.70 | 5555 |
| Methanol | CH4O | 64.6 | 32.60 | 6400 |
| 2-Propanol | C4H8O | 98 | 19.90 | 6700 |
| Sample | Analyte | Solvent | MAE time (min) | MAE temperature (°C) | MAE power (W) | Solvent/sample (mL/g) | Reference |
|---|---|---|---|---|---|---|---|
| Green Tea | Flavanol | Water | 30 | 80 | 600 | 20 | [101] |
| Tea | Polyphenols | Ethanol 60% | 10 | 80 | 600 | 12 | [102] |
| Ipomoea batatas | TPC a | Ethanol 53% | 2.05 | -- | 302 | 30 | [103] |
| Phaseolus vulgaris | TPC a | Ethanol 50% | 15 | 150 | -- | 49 | [104] |
| Fagopyrum esculentum | TPC a | Ethanol 50% | 15 | 150 | -- | 50 | [105] |
| Visit vinifera | TPC a, Flavonoids | Methanol 100% | 60 | 110 | 60 | 5 | [36] |
| Melilotus officinalis (L.) | Coumarin | Ethanol 50% | 5 | 50 | 100 | 20 | [106] |
| Vanilla beans | Vanillin | Ethanol 70% | 20 | -- | 150 | 25 | [107] |
| Radix angelicae sinensis | Ferulic acid | Ethanol 90% | 9 | -- | 850 | 6 | [108] |
| Saussurea medusa | Flavonoids | Ethanol 80% | 60 | 80 | 1200 | 50 | [109] |
| Sorghum | Phenolic acids | 2 M NaOH | 0.45 | 190 | 1400 | 25 | [110] |
| Spices | Phenolic acids | Ethanol 50% | 18 | 50 | 200 | 20 | [111] |
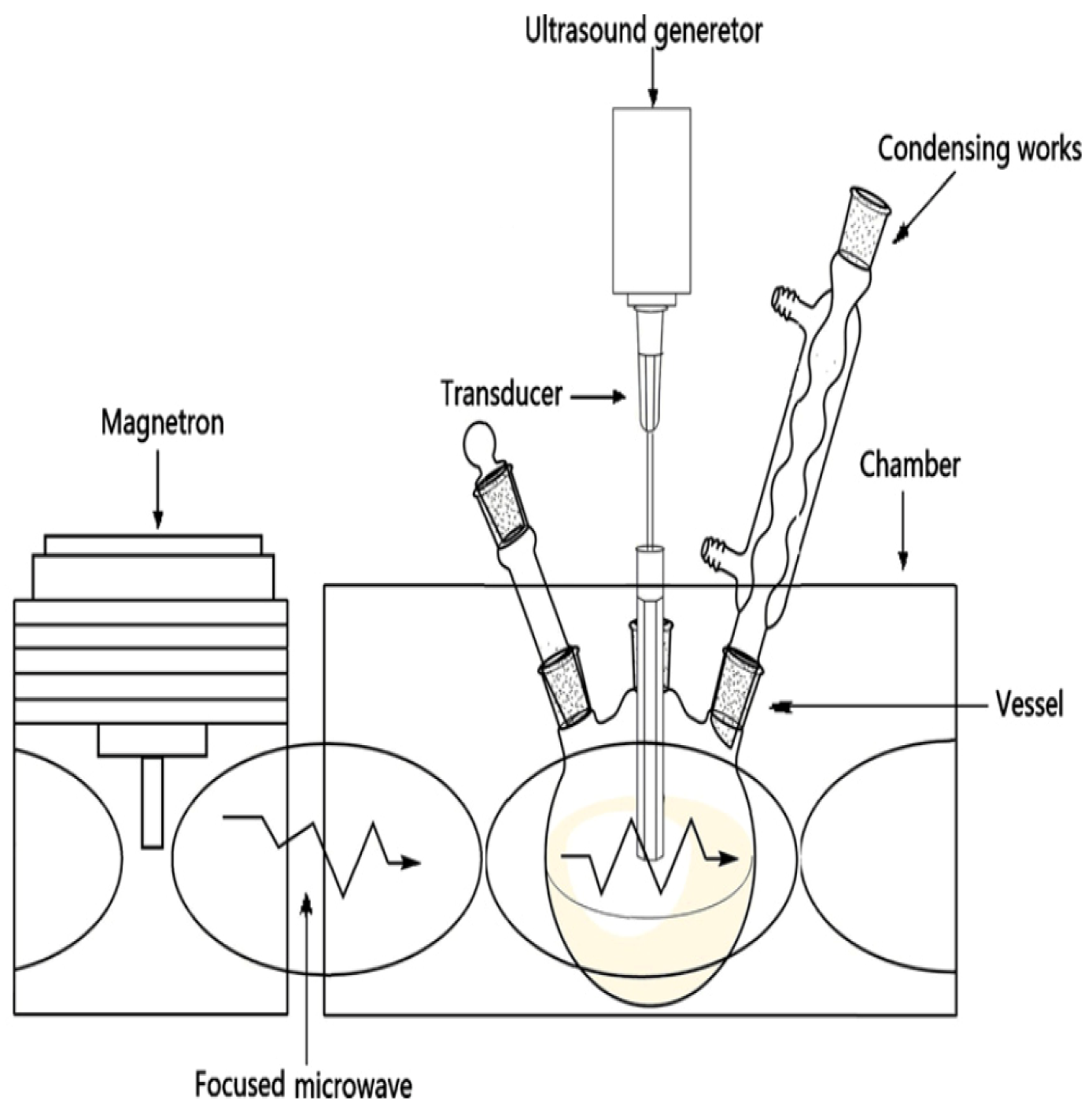
4.3. Ultrasound/Microwave Assisted Extraction (UMAE)
| Sample | Analyte | Solvent | Ultrasound power (W) | Microwave power (W) | UMAE time (s) | UMAE temp (°C) | Solvent/sample (mL/g) | Ref. |
|---|---|---|---|---|---|---|---|---|
| Arctium lappa | Caffeic acid | Ionic solution | 50 | 400 | 30 | -- | 20 | [117] |
| Spatholobus suberectus | Flavonoids | Methanol 70% | 50 | 300 | 450 | 80 | 20 | [115] |
| Tomato | Lycopene | Ethyl acetate | 50 | 98 | 367 | -- | 10.6 | [116] |
| Burdock leaves | Phenoliccompounds | Ethanol 70% | 50 | 500 | 30 | -- | 20 | [114] |
| Anoectochilu roxburghii | Quercetin | Ethanol 50% | 50 | 800 | 900 | 45 | 8 | [118] |
4.4. Supercritical Fluid Extraction (SFE)
| Solvent | Pc (bar) | Tc (°C) | Density (g/mL) |
|---|---|---|---|
| Methane | 46.41 | −82.4 | 0.16 |
| Carbon dioxide | 73.87 | 31.2 | 0.47 |
| Ethane | 48.84 | 32.5 | 0.20 |
| Propane | 42.46 | 97.3 | 0.22 |
| Ammonia | 113.99 | 132.6 | 0.24 |
| Ethanol | 63.83 | 243.6 | 0.28 |
| Benzene | 48.94 | 289.1 | 0.30 |
| Water | 221.19 | 374.3 | 0.32 |
| Sample | Target phenolic class | Temperature (°C) | Time (min) | Pressure (bar) | Modifier | Ref. |
|---|---|---|---|---|---|---|
| Elder berry and grape marc | Phenolic compounds | 40 | -- | 150, 350 | Ethanol | [68] |
| Theobroma cacao hulls | Phenolic compounds | 50 | -- | 100, 200 | Methanol and Acetone | [128] |
| Sweet basil | Phenolic compounds | 35, 50 | 15, 30, 45, 60 | 100, 150, 200, 250, 300 | H2O | [129] |
| Baccharis dracunculifolia leaves | Phenolic compounds | 40, 50, 60 | -- | 200, 300, 400 | -- | [130] |
| Guava seed | Phenolic content | 40, 50, 60 | 120 | 100, 200, 300 | Ethylacetate and Ethanol | [70] |
| Wheat germ | Phenolic content | 40, 60 | 10, 60 | 148, 602 | -- | [125] |
| Pistachio hulls | Phenolic content | 35, 45, 55 | 15, 25, 40 | 100, 200, 350 | Methanol | [131] |
| Bupleurum roots | Phenolic content | 40 | -- | 50, 100, 150, 200 | -- | [132] |
| Bitter melon | Flavonoids | 30, 40, 50 | 40, 50, 60 | 250, 300, 350 | Ethanol | [133] |
| Spearmint leaves | Flavonoids | 40, 50, 60 | 30, 60, 90 | 100, 200, 300 | Ethanol | [134] |
| Pecah Kaca | Flavonoids | 40, 50, 60 | 40,60,80 | 100, 150, 200 | Ethanol | [135] |
| Pueraria lobata | Flavonoids | 40, 50, 60 | 90 | 150, 200, 250 | Ethanol | [136] |
4.5. Subcritical Water Extraction (SCWE)
| Sample | Analyte | Temperature (°C) | Time (min) | Pressure (bar) | Solvent/sample (mL/g) | Ref. |
|---|---|---|---|---|---|---|
| Pomegranate seeds | Phenolic compounds | 80–280 | 15–120 | 60 | 10–50 | [145] |
| Cinnamon bark | Phenolic compounds | 150,200 | 60 | 60 | -- | [146] |
| Potato peel | Phenolic compounds | 100–240 | 30–120 | 60 | -- | [147] |
| Rice bran | Phenolic compounds | 125–200 | 5 | 20 | 2.5 | [148] |
| Terminalia chebula | Phenolic compounds | 120–220 | 10–150 | 40 | -- | [149] |
| Bitter melon | Phenolic compounds | 130–200 | 10–120 | -- | -- | [150] |
| Oregano leaves | Phenolic compounds | 25–200 | 15, 30 | 103.4 | -- | [151] |
| Green tea | Catechin and epicatechin | 140–260 | -- | 38–72 | 20 | [152] |
4.6. High Hydrostatic Pressure Extraction (HHPE)
4.7. Other Extraction Methods
5. Quantification of Phenolics
5.1. Spectrophotometric Assays
5.2. Gas Chromatography
5.3. High Performance Liquid Chromatography
| Sample | Derivatization | Detected phenolics | Detection | Chromatographic assay details | Ref. |
|---|---|---|---|---|---|
| Guarana | Dried phenolic extract derivatized with a mixture of hexamethyldisiloxane and dimethylchlorosilane in pyridine | 3-Hydroxybenzoic acid, benzoic acid, gallic acid, syringic acid, isovanillic acid, protocatechuic acid, catechin, caffeine, epicatechin, quercetin | GC–MS | Zebron ZB-5 ms fused silica capillary column (30 m × 0.25 mm I.D. × 0.25 μm film thickness); Oven temperature: 150 °C held for 5 min, to 295 °C at 3 °C/min, held for 18 min; Injector temperature: 300 °C; Carrier gas: helium flow at 1 mL/min; Ion source temperature: 200 °C; Transfer line temperature: 290 °C | [215] |
| Mirabelle plums | Dried phenolic extractderivatized with N,O-Bis(trimethylsilyl)trifluoro-acetamide | Benzoic acid, p-hydroxybenzaldehyde, p-hydroxybenzoic acid, vanillin, 3,4-dihydroxybenzoic acid, vanillic acid, gallic acid, syringaldehyde, syringic acid, coniferyl aldehyde, 3,5-dimethoxycinnamaldehyde, dehydrodiconiferyl aldehyde, guajacyl-glycerin-coniferyl aldehyde, guajacyl-glycerin-coniferyl aldehyde | GC–MS | HP 5MS capillary column, (30 m × 0.25 mm I.D × 0.25 μm film thickness). Oven temperature: 100–270 °C at 4 °C /min, held for 20 min; Injector temperature: 250 °C; Helium flow at 0.9 mL/s; Ion source temperature: 230 °C; Transfer line temperature: 280 °C | [216] |
| Guava bagasse, Cabernet Sauvignon, Pinot Noir, and Isabella grape marcs wastes | --------------------- | Succinic acid, azelaic acid, syringic acid, p-coumaric acid, gallic acid, ferulic acid, caffeic acid, epicatechin, quercetin, myricetin | GC–MS | RTX 5MS capillary column (30 m × 0.25 mm ID × 0.25 μm film thickness); Oven | [199] |
| Cranberry | Dried phenolic extract derivatized with a mixture of N,O-Bis(trimethylsilyl)-trifluoroacetamide and 1% trimethylchlorosilane in pyridine | Benzoic acid, o-hydroxybenzoic acid, trans-cinnamic acid, m-hydroxybenzoic acid, p-hydroxybenzoic acid, p-hydroxyphenyl acetic acis | GC-MS | Temperature: 80 °C for 1 min, to 250°C, at 20°C/min, held 1 min, to 300°C at 6°C/min, held 5 min, to 310°C at 15 °C/min held 10 min, to 320 °C at 20°C/min, held 10 min; Injector temperature: 280 °C; Transfer line temperature: 280 °C. DB-5 fused-silica capillary column (30 m × 0.32 mm ID × 0.25 μm film thickness) | [217] |
| Saffron corms | Dried phenolic extract derivatized with a mixture of N-methyl-N-(trimethylsilyl) trifluoroacetamide and iodotrimethylsilane | Acetic acid, o-phthalic acid, 2,3-dihydroxy-benzoic acid, vanillic acid, o-hydroxy-cinnamic acid, 2,4-dihydroxy-benzoic acid, p-coumaric acid, ferulic acid, caffeic acid, sinapic acid, epicatechin, catechin. Quercetin, myricetin, m-methylbenzoic acid. catechol, vanillin, salicylic acid, cinnamic acid, p-hydroxybenzoic acid, syringic acid, p-coumaric acid, gallic acid, t-ferulic acid, caffeic acid, gentisic acid | GC-MS | Oven temperature: 80 °C for 1 min, to 220 °C, at 10 °C/min, to 310 °C, at 20 °C/min, held 6 min; Injector temperature: 280 °C; Detector temperature: 305 °C; Transfer line temperature: 280 °C. DB-5 capillary column (30 m × 0.25 mm ID × 0.25 μm film thickness); Oven temperature: 140 °C for 2 min, to 270 °C at 5°C/min | [218] |
| Mangosteen fruit | Dried phenolic extract derivatized with N,O-bis(trimethylsilyl)acetamide | Hydroxybenzoic acid, protocatechuic acid, vanillic acid, caffeic acid, p-coumaric acid, ferulic acid, p-hydroxyphenylacetic acid, 3,4-dihydroxymandelic, cinnamic acid | GC-MS | Held 20 min; Injector temperature: 270 °C; Transfer line temperature: 270 °C. SPB-1 silica-fused capillary column (30 m × 0.25 mm ID × 0.25 μm film thickness); Oven temperature: 120 °C held 2 min, to 260°C at 20 °C /min , held 10 min; Injector temperature: 240 °C; Helium flow at 28 cm3/min; Transfer line temperature: 240 °C. | [208] |
| Green tea | Dried phenolic extract derivatized with trimethyl-sulfonium hydroxide and trimethylsilyl diazomethane | Catechin, epicatechin, epigallocatechin, gallocatechin, kaempferol, quercetin | GC-MS | A ZB-5HT Inferno capillary column (15 m × 0.32 mm ID × 0.10 μm film thickness); Oven temperature: 100°C held for 5 min, to 375°C at 20°C/ min, held for 5 min; Injector temperature: 350°C; Transfer line temperature: 300°C | [200] |
| Various plant extracts | Dried phenolic extract derivatized with a mixture of trimethylchlorosilane and N,O-bis(trimethylsilyl)-trifluoroacetamide with dimethyldichlorosilane in toluene and dimethyldichlorosilane | Gallic acid, p-hydroxybenzoic acid, gentisic acid, p-coumaric acid, vanillic acid, ferulic acid, syringic acid, catechin | GC-MS | CP-Sil 8 capillary column (30 m × 0.32 mm ID × 0.25 μm film thickness) | [195] |
| Propolis | Dried phenolic extract derivatized with N,O-Bis(trimethylsilyl)trifluoro-acetamide | quercetin, apigenin, naringenin, luteolin, caffeic acid, epicatechin, rutin, hydroxytyrosol. Ethyl hydrocinnamate, hydrocinnamic acid, inositol, cinnamic acid, ferulic acid, caffeic acid, pinostrobin | GC-MS | Oven temperature: 70 °C, to 135 °C at 2 °C /min, held for 10 min, to 220 °C at 4°C /min, held for 10 min, to 270 °C at 3.5 °C/min, held for 20 min; Injector temperature: 280 °C; Transfer line temperature: 290 °C. Borosilicate capillary column (20 mm × 0.3 mm ID × 0.1 μm) | [219] |
| Sample | Phenolic class | Column/Detector | Solvent/ Flow rate/ injection volume | Temperature (°C)/Detection time (min) | Ref. |
|---|---|---|---|---|---|
| Mangosteen pericarp | Gallic acid, gentisic acid, protocatechuic acid, gentisic acid, 4-hydroxybenzoic acid, veratric acid, vanillic acid, caffeic acid, syringic acid, p-coumaric acid, sinapic acid, ferulic acid, t-cinnamic acid catechin, epicatechin | Bondapak C18 column (300 mm × 3.9 mm ID × 5 μm)/ PDA b, ESI-MS e | Water : methanol : acetic acid (85:14:1); Flow rate: 1.0 mL/min; Injection volume: 20 μL | Ambient/ 45 | [220] |
| Mulberry fruit | Cyanidin 3-O-rutinoside, cyanidin 3-O-glucoside, pelargonidin 3-O-glucoside, pelargonidin 3-O-rutinoside | RP C18 column (250 mm × 4.6 mm ID, 5 μm)/ PDA b, ESI-MS e | A: water containing 0.1% TFA (trifluoroacetic acid); B: acetonitrile containing 0.1% TFA; Elution profile: 0–2 min, 10% B; 2–35 min, 10–90% B; 35–40 min, 90–100% B; 40–60 min, 100% B/ Flow rate: 1.0 mL/min; NMa | Ambient/ 60 | [221] |
| Fruit juice | Cyanidin, peonidin, delphinidin, petunidin, malvidin, pelargonidin | ODS-3 column (250 mm × 4.6 mm ID × 5 μm)/ PDA b | A: acetonitrile; B: water containing 10% acetic acid and 1% phosphoric acid; Elution profile: 25 min, 2–20% A; 5 min, 20–40%; Flow rate: 1.0 mL/min; Injection volume: 25 μL | NM a/50 | [222] |
| Maytenus aquifolium and Maytenus ilicifolia Leaves | Quercetin, kaempferol derivatives, rutin | Supelcosil C8 and C18 (250 mm × 4.6 mm ID × 5 μm) column/ PDA b | A: water containing 2.0, 2.5 or 3.0% formic acid or 0.3% trifluoroacetic acid; B: acetonitrile or methanol; Various elution profiles; Flow rate 1.0 mL/ min/ Injection volume: 10 μL | 35/ Different detection times | [223] |
| Apple | Gallic acid, chlorogenic acid, catechin, epicatechin, procyanidin, phloridzin, cyanidin 3-galactoside, quercetin 3-rutinoside, quercetin 3-galactoside, quercetin 3-glucoside, quercetin 3-rhamnoside | RP C18 (250 mm × 4.6 mm ID × 4 μm) column/ PDA b | A: water containing 1% TFA, B: ACN containing 1% TFA; Elution profile: 0–10 min, 10% B; 10–45 min, 10–20% B; 45–50 min, 20–50% B; 50–55 min, isocratic 50%; 55–60 min, 50–10% B. Flow rate: 1 mL/min. Injection volume: 10 μL | 40/ 60 | [224] |
| Medicinal plants | Cyanidin glucoside, pelargonidin glucoside, gallocatechin-catechin gallate, afzelechin–catechin dimer, gallocatechin catechin gallate, ferulic acid glucoside, rutin, naringenin-7-O-rutinoside | RP C18 (250 mm × 4.6 mm ID × 5 μm) column/ PDA b, ESI- MS e | A: water containing 1% formic acid, B: acetonitrile; Elution profile: 30 min, 90–75% A; 30–45 min, 75–40% A; Flow rate: 1 mL/min; Injection volume: 20 μL | 25/ 45 | [225] |
| Food samples | Monomeric, dimeric and trimeric procyanidins, catechin, epicatechin | RP 18 (250 mm × 2 mm ID × 5 μm) column/ PDA b, FLD d, ESI-MS/MS | A: water containing 0.1% formic acid; B: acetonitrile containing 0.1% formic acid; Elution profile: 0–10 min, 10% B; 10–30 min, 15% B; 30–65 min, 40% B. Flow rate: 300 μL/min; Injection volume: 20 μL | 25/ 30 | [226] |
| Oregano | Quercetin, fisetin, kaempferol, luteolin, apigenin, eriodictyol, hesperetin, taxifolin, (+)-catechin, (-)-epicatechin | Hypersil C18 ODS (250 mm × 4.6 mm ID × 5 μm) column/ PDA b, ESI-MS-MS | A: water; B: methanol; C: acetonitrile, each containing 0.2% trifluroacetic acid; Elution profile: Initial, 90% A, 6% B, 4% C; 5 min, 85% A, 9% B, 6% C; 5–35 min, 71% A, 17.4% B, 11.6% C; 35–95 min, 0% A, 85% B, 15% C; Flow rate: 1 mL/min; NM a | 30/ NM a | [51] |
| Lotus leaves | Myricetin 3-O-glucoside, quercetin 3-O-arabinopyranosyl, quercetin 3-O-glucuronide, kaempferol 3-O-galactoside, astragalin, isorhamnetin 3-O-glucoside, kaempferol 3-O-glucuronide, quercetin | C18 (150 mm × 4.6 mm ID × 3.5 μm) column/ PDA b, ESI-MS e | A: water containing 0.5% formic acid; B: acetonitrile containing 0.1% formic acid; Elution profile: 0–10 min, 12% B; 10–32 min, 12–20% B; 32–40 min, 20–30% B; 40–48 min, 30–60% B; 48–49 min, 60–12% B; 49–53 min 12% B; Flow rate: 0.6 mL/min; NM a | 30 /53 | [227] |
| Bilberries and Blueberries | Delphinidin-3-O-glucopyranoside, delphinidin-3-O-galactopyranoside, cyanidin-3-O-arabinopyranoside, malvidin-3-O-arabinopyranoside, petunidin-3-O-galactopyranoside | C18 (250 mm × 4.6 mm ID × 3 μm) column/ UV-VISc | A: acetonitrile: water: formic acid (87/3/10); B acetonitrile: water: formic acid (50/40/10); Elution profile: 0–20 min, 2%–14% B; 20–40 min, 14% B; 40–50 min, 15% B; 50–55 min, 19% B; 55–65 min, 20% B/ Flow rate: 0.5 mL/min; Injection volume: 20 μL | Ambient/ 65 | [228] |
| Persian walnut | 3-caffeoylquinic, 3-p-coumaroylquinic, 4-p-coumaroylquinic acid, quercetin 3-galactoside, quercetin 3-arabinoside, quercetin 3-xyloside, quercetin 3-rhamnoside, quercetin 3-pentoside, kaempferol 3-pentoside | LiChroCART RP C 18 (250 mm × 4 mm ID × 5 μm)/ PDA b, MS-MS | A: water containing 0.1% TFA; B: methanol; Elution profile: 30 min, 30–50% B; 30–32 min, 70% B; 32–33 min, 80% B, 33–35 min, 80% B; Flow rate: 1 mL/ min; Injection volume: 5 μL | NM a/35 | [229] |
| Rye grain | Sinapic acid, syringic acid, vanillic acid, ferulic acid, caffeic acid, p-hydroxybenzoic acid, protocatechuic acid, p-coumaric acid, ferulic acid dehydrodimers | Inertsil ODS-3 (150 mm × 4.0 mm ID × 3 μm/ PDA b | A: 50 mM H3PO4 (pH 2.5) B: acetonitrile; Elution profile: 0–5 min, 95% A; 5–17 min, 95–85% A; 17–40 min, 85–80% A; 40–60 min, 80–50% A; 60–65 min 50% A. Flow rate: 0.7 mL/min; Injection volume: 10 μL | 35/ 67 | [230] |
| Pomegranate juices | Delphinidin 3,5-diglucoside, cyanidin 3,5-diglucoside, delphinidin 3-glucoside, pelargonidin 3,5-diglucoside, ellagic acid | RP C18 Nucleosil (125 mm × 5.0 mm ID × 5.0 μm) column/ UV-VIS c | A: water containing 2.5% acetic acid; B: methanol containing 2.5%, acetic acid; Elution profile: 0–5 min, 100% A; 5–15 min, 90%; 15–45 min, 50% A; 45–55 min, 100% A. Flow rate: 1.0 mL/min; Injection volume: 50 μL | NM a/55 | [231] |
| Orange juice | Gallic acid, protocatechuic acid, p-hydroxybenzoic acid, vanillic acid, caffeic acid, chlorogenic acid, p-coumaric acid, ferulic acid, sinapic acid, narirutin, naringin, hesperidin, neohesperidin, didymin | Ultrasphere ODS (250 mm× 4.6 mm ID × 5 μm) column/ UV-VIS c | A: water containing 5% formic acid; B: acetonitrile/solvent A (60:40; v/v); Elution profile: 0-10 min, 0% B; 10–40 min, 0–5% B; 40–58 min, 5–15% B; 48–62 min, 15–25%, 62–93 min, 25–50% B; 93–96 min, 50–100% B; Flow rate: 1.0 mL/min; NM a | 25/ 96 | [232] |
| Quinoa | Apigenin-7-methyl ether, 1-O-galloyl-β-d-glucose, protocatechuic acid 4-O-glucoside, vanillic glucoside, penstebioside, ferulic acid 4-O-glucoside, ethyl-m-digallate, gallocatechin, quercetin, kaempferol, rutin | Kinetex C18 (100 mm × 4.6 mm ID × 2.6 μm) column/ PDA b, ESI-MS e | A: water containing 1% acetic acid; B: acetonitrile/solvent A (40:60; v/v); Elution profile: 0–3.5 min, 2% B; 3.5–4.5 min, 2–6%; 4.5–6 min, 6–10% B; 6–7.5 min, 10–17%; 7.5–13 min, 17–36% B; 13–14 min, 36–38.5% B; 14–19 min, 38.5–60% B; 19–24 min, 60–100% B; 24–30 min, 100% B; 30–32 min, 100–2% B; Flow rate: 0.8 mL/min; Injection volume: 10 μL | 25/ 30 | [233] |
| Pine needle | Catechin, proanthocyanidins | SupelcoSil LC18 (250 mm × 4.6 mm ID × 5 μm) column/ UV c | A: acetonitrile; B: water containing 0.3% phosphoric acid; Elution profile: 0–35 min, 10–20% A; 35–55 min, 20–90% A; Flow rate: 0.7 ml/min; Injection volume: 10 μL | NMa/47 | [234] |
| Apricot fruit | p-aminobenzoic acid, chlorgenic acid, caffeic acid, protocatechuic acid, ferulic acid, rutin, resveratrol, quercetin | Gemini C18 (150 mm × 4.6 mm ID × 3 μm) column/ UV-VIS c | A: citric acid (75 mM); B: ammonium acetate (25 mM); Elution profile: 0–1 min, 5% B; 1–4 min, 5–6% B; 4–20 min, 6–25% B; 20–30 min, 25–100% B; 30–36 min, 100% B; 36–38 min, 100–5% B; 38–45 min, 5% B; Flow rate: 1.0 mL/min; Injection volume: 20 μL | 35/45 | [235] |
| Sage tea | Carnosic acid, epirosmanol, luteolin-rutinoside, salvianolic acid, apigenin-glucuronide, rosmarinic acid, apigenin-rutinoside, luteolin-rutinoside, luteolin-7-O-glucoside, monohydroxy benzoic acid, luteolin-diglucuronide, caffeic acid, caffeoyl-fructosyl-glucose, coumaroyl-hexose, protocatechuic acid | RP C18 (150 mm × 2.1 mm ID × 1.7 μm) column/ PDA b, MS-MS | A: water containing 0.1% formic acid; B: acetonitrile containing 0.1% formic acid; Elution profile: 0–14 min, 4–27% A; 14–28 min, 27–59.7% A; 28–28.2 min, 59.7–100% A; 28.2–30.5 min, 100% A; 30.5–31 min, 100–4% A; 31–34 min, 4% A; Flow rate: 0.4 mL/min; Injection volume: 3 μL | 40/ 28 | [236] |
| Almond skin | Quercetin-3-O-glucoside, isorhamnetin-3-rutinoside, kampferol-3-rutinoside, naringenin-7-O-glucoside, isorhamnetin-3-glucoside, p-hydroxybenzoic acid, naringenin, protocatechuic acid, vanillic acid | RP C18 (50 mm × 2 mm ID, × 2.5 μm) column/ ESI-MS e | A: water containing 0.1% formic acid. B: acetonitrile containing 0.1% formic acid; Elution profile: 0–9.5 min, 1–100% B; Flow rate: 0.5 mL/min; Injection volume: 5 μL | 35/9.5 | [237] |
| Burdock leaves | Quercetin, cynarin, benzoic acid, quercitrin, caffeic acid, luteolin, chlorogenic acid, p-coumaric acid, rutin, arctiin | BEH C18 (150 mm × 2.1 mm ID × 1.7 μm) column/ PDA, ESI-MS-MS | A: water containing 0.1% formic acid; B: acetonitrile/methanol (20/80); Elution profile: 0–10 min, 10–30% B; 10–20 min, 30–50% B; 20–23 min, 50–70% B; 23–25 min, 70–10% B; Flow rate: 0.28 mL/min; NM a | NM a/25 | [114] |
| Grape extract | Malvidin glucoside, delphinidin glucoside, cyanidin glucoside, petunidin glucoside, peonidin glucoside, malvidin acetylglucoside, delphinidin acetylglucoside, cyanidin acetylglucoside, petunidin acetylglucoside, peonidin acetylglucoside, malvidin coumarylglucoside | Zorbax SB-C18 (50 mm × 2.1 mm ID × 1.8 μm) column /PDA b, MS-MS | A: water containing 10%; B: acetonitrile; Elution profile: 0–1.5 min, 10–13% B; 1.5–4.5 min, 13-15% B; 4.5–7.5 min, 15–22% B; 7.5–15 min, 22% B; Flow rate: 0.2 mL/min; Injection volume: 1 μL | NM a/15 | [238] |
| Stem Bark of Acacia confusa | (+)-catechin, (−)-epicatechin, 4β-(2-aminoethylthio) catechin, 4β-(2-aminoethylthio) epicatechin | Hypersil ODS (250 mm × 4.6 mm ID × 2.5 μm) column/ ESI-MS e | A: water containing 0.5 % trifluoroacetic acid; B: acetonitrile containing 0.5% trifluoroacetic acid; Elution profile: 0–5 min, 3% B; 5–15 min, 3%–9% B; 15–45 min, 9%–16% B; 45–60 min, 16%–60% B; Flow rate: 1 mL/min; NM a | Ambient/NM a | [239] |
| Lettuce | Caffeoyltartaric acid, p-coumaroyltartaric acid, caffeoylquinic acid, chlorogenic acid, p-coumaroylquinic acid, caffeoylmalic acid, dicaffeoyltartaric acid, chicoric acid, p -coumaroylcaffeoyltartaric acid, di-p -coumaroyltartaric acid, quercetin-3-O-glucuronide, 3,5-dicaffeoylquinic acid, quercetin malonylglucoside | HSS T3 (100 mm × 2.1 mm ID × 1.8 μm)column/ PDA b, ESI-MS e | A: water:methanol:formic acid (94.9:5.0:0.1); B: methanol:water:formic acid (60.0:39.9:/0.1); Elution profile: 0–30 min, 100–50% A; Flow rate: 0.5 mL/min; Injection volume: 10 μL | 35/NM a | [240] |
| Cocoa and Chocolate products | Catechin, epicatechin | diol-based (250 mm × 4.6 mm ID × 5 μm) /FLD d, MS | A: acetonitrile:acetic acid (98:2); B: methanol:water:acetic acid (95:3:2); 0–35 min, 100–60% A; 35–39 min, 60% A; 39–41 min, 60–0% A; 41–47 min, 0.0% A; 47–51 min, 0–100% A; Flow rate: 1.0 mL/min; Injection volume: 10 μL | 30/ 51 | [241] |
| Wild mushroom | Benzoic acids, p-hydroxybenzoic, protocatechuic, vanillic, cinnamic, p-coumaric acids | Spherisorb RP C18 (150 mm × 4.6 mm ID × 3 μm) column/ PDA b, ESI-MS e | A: water containing 2.5% acetic acid; B: acetic acid 2.5%: acetonitrile (90:10); C: acetonitrile; Elution profile: 10 min, 100% A; 10–20 min, 50% A and 50% B; 20–35 min, 100% B; 35–45 min, 90% B and 10% C; 45–55 min, 70% B and 30% C; 55–60 min, 50% B and 50% C; 60–65 min, 20% B and 80% C; 65–70 min, 100% A; Flow rate: 0.50 mL/min; NM a | 25/NM a | [242] |
| Cocoa, apple | Quercetin, phloridzin, clovamide, p-coumaroylquinic acid, caffeoylquinic acid, quercetin-3-O-galactoside, quercetin-3-O-arabinoside, quercetin-3-O-xyloside, dideoxyclovamide, quercetin-3-O-rhamnoside | BEH C18 (50 mm × 2.1 mm ID× 1.7 μm) column/ UV c, FLD d, ESI-MS e | A: water containing 0.1 %formic acid; B: acetonitrile; Elution profile: 0–0.25 min, 2% B; 0.25–10.70 min, 2–18% B; 10.70–18 min, 18–25% B; 18–20.70 min, 25–100% B; 20.70–22.5 min, 100% B; Flow rate: 0.80 ml/min; Injection volume: 2 μL | 50/22.5 | [243] |
| Bean | Ferulic acid, p-coumaric acid, sinapic acid, caffeic acid | RP C18 Luna (150 mm × 4.6 mm ID × 5 μm) column/ PDA b | A: water containing 0.1% formic acid; B: methanol; Elution profile: 0–50 min, 5–30% B; 50–65 min, 30% B; 65–75 min, 30–100% B; Flow rate: 0.7 mL/min; NM a | 25/65 | [244] |
| Green tea, green coffee, grapefruit | Catechin, epigallocatechin gallate, epicatechin gallate, epicatechine, gallocatechin, catechin gallate, gallic acid, caffeine | RP C18 Atlantis (100 mm × 4.6 mm ID × 3 μm)/ UV | A: water containing 0.1% formic acid; B: methanol containing 0.1% formic acid; Elution profile: 0–5 min, 10% B; 5–14 min, 10–20% B; 14–20 min, 20–50% B; 20–22 min, 50–90% B; 22–26 min, 90% B; 26–30 min, 90–10% B; Flow rate: 0.5 mL/min; Injection volume: 20 μL | 25/NM | [245] |
5.4. Other Assays for Separation and Quantification of Phenolics
6. Conclusions
References
- Jin, D.; Mumper, R.J. Plant phenolics: Extraction, analysis and their antioxidant and anticancer properties. Review. Molecules 2010, 15, 7313–7352. [Google Scholar] [CrossRef]
- Wijngaard, H.H.; Rößle, C.; Brunton, N. A survey of Irish fruit and vegetable waste and byproducts as a source of polyphenolic antioxidants. Food Chem. 2009, 116, 202–207. [Google Scholar] [CrossRef]
- Hoye, A.T.; Davoren, J.E.; Wipf, P.; Fink, M.P.; Kagan, V.E. Targeting mitochondria. Acc. Chem. Res. 2008, 41, 87–97. [Google Scholar] [CrossRef]
- Mohanlal, S.; Parvathy, R.; Shalini, V.; Mohanan, R.; Helen, A.; Jayalekshmy, A. Chemical indices, Antioxidant activity and anti-inflammatory effect of extracts of the medicinal rice “Njavara” and staple varieties. J. Food Biochem. 2012, 36, 1–12. [Google Scholar] [CrossRef]
- Zhang, L.; Ravipati, A.S.; Koyyalamudi, S.R.; Jeong, S.; Reddy, N.; Smith, P.T.; Bartlett, J.; Shanmugam, K.; Münch, G.; Wu, M.J. Antioxidant and anti-inflammatory activities of selected medicinal plants containing phenolic and flavonoid compounds. J. Agric. Food Chem. 2011, 59, 12361–12367. [Google Scholar]
- Jin, X.H.; Ohgami, K.; Shiratori, K.; Suzuki, Y.; Koyama, Y.; Yoshida, K.; Ilieva, I.; Tanaka, T.; Onoe, K.; Ohno, S. Effects of blue honeysuckle (Lonicera caerulea L.) extract on lipopolysaccharide-induced inflammation in vitro and in vivo. Exp. Eye Res. 2006, 82, 860–867. [Google Scholar] [CrossRef]
- Sawadogo, W.R.; Maciuk, A.; Banzouzi, J.T.; Champy, P.; Figadere, B.; Guissou, I.P.; Nacoulma, O.G. Mutagenic effect, Antioxidant and anticancer activities of six medicinal plants from Burkina Faso. Nat. Prod. Res. 2012, 26, 575–579. [Google Scholar] [CrossRef]
- Pieme, C.A.; Penlap, V.N.; Ngogang, J.; Costache, M. In vitro cytotoxicity and antioxidant activities of five medicinal plants of Malvaceae family from Cameroon. Environ. Toxicol. Pharmacol. 2010, 29, 223–228. [Google Scholar]
- Ramos, S. Cancer chemoprevention and chemotherapy: Dietary polyphenols and signaling pathways. Mol. Nutr. Food Res. 2008, 52, 507–526. [Google Scholar] [CrossRef]
- Slivova, V.; Zaloga, G.; DeMichele, S.J.; Mukerji, P.; Huang, Y.S.; Siddiqui, R.; Harvey, K.; Valachovicova, T.; Sliva, D. Green tea polyphenols modulate secretion of urokinase plasminogen activator (uPA) and inhibit invasive behavior of breast cancer cells. Nutr. Cancer 2005, 52, 66–73. [Google Scholar] [CrossRef]
- Kusirisin, W.; Srichairatanakool, S.; Lerttrakarnnon, P.; Lailerd, N.; Suttajit, M.; Jaikang, C.; Chaiyasut, C. Antioxidative activity, Polyphenolic content and anti-glycation effect of some Thai medicinal plants traditionally used in diabetic patients. Med. Chem. 2009, 5, 139–147. [Google Scholar] [CrossRef]
- Scalbert, A.; Manach, C.; Remesy, C.; Morand, C. Dietary polyphenols and the prevention of diseases. Crit. Rev. Food Sci. Nutr. 2005, 45, 287–306. [Google Scholar] [CrossRef]
- Pedreschi, R.; Cisneros-Zevallos, L. Antimutagenic and antioxidant properties of phenolic fractions from Andean purple corn (Zea mays L.). J. Agric. Food Chem. 2006, 54, 4557–4567. [Google Scholar] [CrossRef]
- Gomez-Cordoves, C.; Bartolome, B.; Vieira, W.; Virador, V.M. Effects of wine phenolics and sorghum tannins on tyrosinase activity and growth of melanoma cells. J. Agric. Food Chem. 2001, 49, 1620–1624. [Google Scholar] [CrossRef]
- Diaz Napal, G.N.; Defago, M.; Valladares, G.; Palacios, S. Response of Epilachna paenulata to two flavonoids, Pinocembrin and quercetin, in a comparative study. J. Chem. Ecol. 2010, 36, 898–904. [Google Scholar] [CrossRef]
- Kennedy, D.O.; Wightman, E.L. Herbal extracts and phytochemicals: Plant secondary metabolites and the enhancement of human brain function. Adv. Nutr. 2011, 2, 32–50. [Google Scholar] [CrossRef]
- Zulak, K.; Liscombe, D.; Ashihara, H.; Facchini, P. Alkaloids. Plant Secondary Metabolism in Diet and Human Health; Blackwell Publishing: Oxford, UK, 2006. [Google Scholar]
- Chung, I.M.; Park, M.R.; Chun, J.C.; Yun, S.J. Resveratrol accumulation and resveratrol synthase gene expression in response to abiotic stresses and hormones in peanut plants. Plant Sci. 2003, 164, 103–109. [Google Scholar] [CrossRef]
- Chirinos, R.; Betalleluz-Pallardel, I.; Huamán, A.; Arbizu, C.; Pedreschi, R.; Campos, D. HPLC-DAD characterisation of phenolic compounds from Andean oca (Oxalis tuberosa Mol.) tubers and their contribution to the antioxidant capacity. Food Chem. 2009, 113, 1243–1251. [Google Scholar] [CrossRef]
- Soto-Vaca, A.; Losso, J.N.; Xu, Z.; Finley, J.W. Review: Evolution of phenolic compounds from color and flavor problems to health benefits. J. Agric. Food Chem. 2012. Epub ahead of print. [Google Scholar]
- Ferreira, O.; Pinho, S.P. Solubility of flavonoids in pure solvents. Ind. Eng. Chem. Res. 2012, 51, 6586–6590. [Google Scholar] [CrossRef]
- Rong, T. Chemistry and biochemistry of dietary polyphenols, Review. Nutrients 2010, 2, 1231–1246. [Google Scholar] [CrossRef]
- Routray, W.; Orsat, V. Review: Microwave-assisted extraction of flavonoids. Food Bioprocess. Technol. 2012, 5, 409–424. [Google Scholar] [CrossRef]
- Stalikas, C.D. Review: Extraction, Separation, and detection methods for phenolic acids and flavonoids. J. Sep. Sci. 2007, 30, 3268–3295. [Google Scholar] [CrossRef]
- Pereira, D.M.; Valentão, P.; Pereira, J.A.; Andrade, P.B. Phenolics: From chemistry to biology. Molecules 2009, 2202–2211. [Google Scholar]
- Vanholme, R.; Demedts, B.; Morreel, K.; Ralph, J.; Boerjarr, W. Lignin biosynthesis and structure. Plant. Physiol. 2010, 153, 895–905. [Google Scholar] [CrossRef]
- Baucher, M.; Monties, B.; Van Montagu, M.; Boerjan, W. Biosynthesis and genetic engineering of lignin. Crit. Rev. Plant Sci. 1998, 17, 125–197. [Google Scholar] [CrossRef]
- Naczk, M.; Shahidi, F. Review: Extraction and analysis of phenolics in food. J. Chromatogr. A 2004, 1054, 95–111. [Google Scholar]
- Sejali, S.N.F.; Anuar, M.S. Effect of drying methods on phenolic contents of neem (Azadirachta indica) leaf powder. J. Herbs Spices Med. Plants 2011, 17, 119–131. [Google Scholar] [CrossRef]
- Giao, M.S.; Pereira, C.I.; Fonseca, S.C.; Pintado, M.E.; Malcata, F.X. Effect of particle size upon the extent of extraction of antioxidant power from the plants Agrimonia eupatoria, Salvia sp. and Satureja montana. Food Chem. 2009, 117, 412–416. [Google Scholar] [CrossRef]
- Weidner, S.; Powałka, A.; Karamać, M.; Amarowicz, R. Extracts of phenolic compounds from seeds of three wild grapevines—Comparison of their antioxidant activities and the content of phenolic compounds. Int. J. Mol. Sci. 2012, 13, 3444–3457. [Google Scholar] [CrossRef]
- Garcia-Salas, P.; Morales-Soto, A.; Segura-Carretero, A.; Fernández-Gutiérrez, A. Phenolic-compound-extraction systems for fruit and vegetable samples. Molecules 2010, 15, 8813–8826. [Google Scholar] [CrossRef]
- Agbangnan, P.C.; Tachon, D.C.; Dangou, J.; Chrostowska, A.; Fouquet, E.; Sohounhloue, D.C.K. Optimization of the extraction of sorghum’s polyphenols for industrial production by membrane processes. Res. J. Res. Sci. 2012, 1, 1–8. [Google Scholar]
- Verma, B.; Hucl, P.; Chibbar, R.N. Phenolic content and antioxidant properties of bran in 51 wheat cultivars. Cereal Chem. 2008, 85, 544–549. [Google Scholar] [CrossRef]
- Kalpana, K.; Kapil, S.; Harsh, P.S.; Bikram, S. Effects of extraction methods on phenolic contents and antioxidant activity in aerial parts of Potentilla atrosanguinea Lodd. and quantification of its phenolic constituents by RP-HPLC. J. Agric. Food Chem. 2008, 56, 10129–10134. [Google Scholar] [CrossRef]
- Casazza, A.A.; Aliakbarian, B.; Mantegna, S.; Cravotto, G.; Perego, P. Extraction of phenolics from Vitis vinifera wastes using non-conventional techniques. J. Food Eng. 2010, 100, 50–55. [Google Scholar] [CrossRef]
- Taha, F.S.; Mohamed, G.F.; Mohamed, S.H.; Mohamed, S.S.; Kamil, M.M. Optimization of the extraction of total phenolic compounds from sunflower meal and evaluation of the bioactives of chosen extract. Am. J. Food Technol. 2011, 6, 1002–1020. [Google Scholar] [CrossRef]
- Biesaga, M.; Pyrzynska, K. Stability of bioactive polyphenols from honey during different extraction methods. Food Chem. 2013, 136, 46–54. [Google Scholar] [CrossRef]
- Davidov-Pardo, G.; Arozarena Inigo Marın-Arroyo, M.R. Stability of polyphenolic extracts from grape seeds after thermal treatments. Eur. Food. Res. Technol. 2011, 232, 211–220. [Google Scholar] [CrossRef]
- Naczk, M.; Amarowicz, R.; Zadernowski, R.; Shahidi, F. Antioxidant Capacity of Phenolics from Canola Hulls as Affected by Different Solvents. In Phenolic Compounds in Foods and Natural Health Products; Shahidi, F., Ho, C.T., Eds.; Oxford University Press: New York, NY, USA, 2005; pp. 57–66. [Google Scholar]
- Al-Farsi, M.; Lee, C.Y. Review: Nutritional and functional properties of dates. Crit. Rev. Food Sci. Nutr. 2008, 48, 877–887. [Google Scholar] [CrossRef]
- Pinelo, M.; Zornoza, B.; Meyer, A.S. Selective release of phenols from apple skin: Mass transfer kinetics during solvent and enzyme-assisted extraction. Sep. Purif. Technol. 2008, 63, 620–627. [Google Scholar] [CrossRef]
- Haghi, G.; Hatami, A. Simultaneous quantification of flavonoids and phenolic acids in plant materials by a newly developed isocratic high-performance liquid chromatography approach. J. Agric. Food Chem. 2010, 58, 10812–10816. [Google Scholar] [CrossRef]
- Vichapong, J.; Maliwan, S.; Voranuch, S.; Prasan, S.; Supalax, S. High performance liquid chromatographic analysis of phenolic compounds and their antioxidant activities in rice varieties. LWT-Food Sci. Technol. 2010, 43, 1325–1330. [Google Scholar] [CrossRef]
- Ayumi, H.; Masatsune, M.; Seiichi, H. Analysis of free and bound phenolics in rice. Food Sci. Technol. Res. 1999, 5, 74–79. [Google Scholar]
- Nardini, M.; Cirillo, E.; Natella, F.; Mencarelli, D.; Comisso, A.; Scaccini, C. Detection of bound phenolic acidsprevention by ascorbic acid and ethylenediaminetetraacetic acid of degradation of phenolic acids during alkaline hydrolysis. Food Chem. 2002, 79, 119–124. [Google Scholar]
- Weidner, S.; Amarowicz, R.; Karamac, M.; Dabrowski, G. Phenolic acids in caryopses of two cultivars of wheat, rye and triticale that display different resistance to pre-harvest sprouting. Eur. Food Res. Technol. 1999, 210, 109–113. [Google Scholar] [CrossRef]
- Macdonald, I.O.; Oludare, A.S.; Olabiyi, A. Phytotoxic and anti-microbial activities of flavonoids in Ocimum gratissimum. Life Sci. 2010, 7, 45–48. [Google Scholar]
- Zhu, H.B.; Wang, Y.Z.; Liu, Y.X.; Xia, Y.L.; Tang, T. Analysis of flavonoids in Portulaca oleracea L. by UV-Vis spectrophotometry with comparative study on different extraction technologies. Food Anal. Meth. 2010, 3, 90–97. [Google Scholar] [CrossRef]
- Biesaga, M. Influence of extraction methods on stability of flavonoids. J. Chromatogr. A 2011, 1218, 2505–2512. [Google Scholar] [CrossRef]
- Tsimogiannis, D.; Samiotaki, M.; Panayotou, G.; Oreopoulou, V. Characterization of flavonoid subgroups and hydroxy substitution by HPLC-MS/MS. Molecules 2007, 12, 593–606. [Google Scholar] [CrossRef]
- Mattila, P.; Astola, J.; Kumpulainen, J. Determination of flavonoids in plant material by HPLC with diode-array and electro-array detections. J. Agric. Food Chem. 2000, 48, 5834–5841. [Google Scholar] [CrossRef]
- Wu, Y.; Wang, X.; Fan, E. Optimisation of ultrasound-assisted extraction of puerarin and total isoflavones from Puerariae lobatae Radix (Pueraria lobata (Wild.) Ohwi) with response surface methodology. Phytochem. Anal. 2012, 23, 513–519. [Google Scholar] [CrossRef]
- Patil, G.; Madhusudhan, M.C.; Ravindra Babu, B.; Raghavarao, K.S.M.S. Extraction, dealcoholization and concentration of anthocyanin from red radish. Chem. Eng. Process. 2009, 48, 364–369. [Google Scholar] [CrossRef]
- Amr, A.; Al-Tamimi, E. Stability of the crude extracts of Ranunculus asiaticus anthocyanins and their use as food colourants. Int. J. Food Sci. Tech. 2007, 42, 985–991. [Google Scholar] [CrossRef]
- Awika, J.M.; Rooney, L.W.; Waniska, R.D. Anthocyanins from black sorghum and their antioxidant properties. Food Chem. 2005, 90, 293–301. [Google Scholar] [CrossRef]
- Kong, J.M.; Chia, L.S.; Goh, N.K.; Chia, T.F.; Brouillard, R. Analysis and biological activities of anthocyanins. Phytochemistry 2003, 64, 923–933. [Google Scholar] [CrossRef]
- Castaneda-Ovando, A.; de Lourdes Pacheco-Hernandez, M.; Paez-Hernandez Ma, E.; RodriguezGalan-Vidal, J.A.; Galan-Vidal, C.A. Review: Chemical studies of anthocyanins. Food Chem. 2009, 113, 859–871. [Google Scholar] [CrossRef]
- Bridgers, E.N.; Chinn, M.S.; Truong, V.-D. Extraction of anthocyanins from industrial purple-fleshed sweetpotatoes and enzymatic hydrolysis of residues for fermentable sugars. Ind. Crop. Prod. 2010, 32, 613–620. [Google Scholar] [CrossRef]
- Lee, J.; Finn, C.E.; Wrolstad, R.E. Anthocyanin pigment and total phenolic content of three vaccinium species native to the Pacific Northwest of North America. Hortscience 2004, 39, 959–964. [Google Scholar]
- Kapasakalidis, P.G.; Rastall, R.A.; Gordon, M.H. Extraction of polyphenols from processed black currant (Ribes nigrum L.) residues. J. Agric. Food Chem. 2006, 54, 4016–4021. [Google Scholar] [CrossRef]
- Delgado-Vargas, F.; Paredes-Lopez, O. Natural Colorants for Food and Nutraceutical Uses; CRC Press: Boca Raton, FL, USA, 2002; p. 342. [Google Scholar]
- Ju, Z.Y.; Howard, L.R. Subcritical water and sulfured water extraction of anthocyanins and other phenolics from dried red grape skin. J. Food Sci. 2005, 70, 270–276. [Google Scholar]
- Cacace, J.E.; Mazza, G. Extraction of anthocyanins and other phenolics from black currants with sulfured water. J. Agric. Food Chem. 2002, 50, 5939–5946. [Google Scholar] [CrossRef]
- Nam-Young, K.; Min-Kyung, J.; Dong-Geun, L.; Ki, H.Y.; HyeJi, J.; Mihyang, K.; Sung, G.K.; Byung, H.Y.; Sang-Hyeon, L. Comparison of methods for proanthocyanidin extraction from pine (Pinus densiflora) needles and biological activities of the extracts. Nutr. Res. Pract. 2010, 4, 16–22. [Google Scholar] [CrossRef]
- Xiaoyun, X.; Bijun, X.; Siyi, P.; Erning, Y.; Kexin, W.; Stefan, C.; Arnie, W.H.; Shan, R. A new technology for extraction and purification of proanthocyanidins derived from sea buckthorn bark. J. Sci. Food Agric. 2006, 86, 486–492. [Google Scholar] [CrossRef]
- Hernández-Jiménez, A.; Kennedy, J.A.; Bautista-Ortín, A.B.; Gómez-Plaza, E. Effect of ethanol on grape seed proanthocyanidin extraction. Am. J. Enol. Vitic. 2012, 63, 57–61. [Google Scholar] [CrossRef]
- Vatai, T.; Skerget, M.; Knez, Z. Extraction of phenolic compounds from elder berry and different grape marc varieties using organic solvents and/or supercritical carbon dioxide. J. Food Eng. 2009, 90, 246–254. [Google Scholar] [CrossRef]
- Yang, L.; Sun, X.; Yang, F.; Zhao, C.; Zhang, L.; Zu, Y. Application of ionic liquids in the microwave-assisted extraction of proanthocyanidins from Larix gmelini bark. Int. J. Mol. Sci. 2012, 13, 5163–5178. [Google Scholar] [CrossRef]
- Castro-Vargas, H.I.; Rodríguez-Varela, L.I.; Ferreira, S.R.S.; Parada-Alfonso, F. Extraction of phenolic fraction from guava seeds (Psidium guajava L.) using supercritical carbon dioxide and co-solvents. J. Supercrit. Fluids 2010, 51, 319–324. [Google Scholar] [CrossRef]
- Santana, C.M.; Ferrera, Z.S.; Torres Padrón, M.E.; Santana Rodríguez, J.J. Methodologies for the extraction of phenolic compounds from environmental samples: New approaches. Molecules 2009, 14, 298–320. [Google Scholar] [CrossRef]
- Altuner, E.M.; Işlek, C.; Çeter, T.; Alpas, H. High hydrostatic pressure extraction of phenolic compounds from Maclura pomifera fruits. Afr. J. Biotechnol. 2012, 11, 930–937. [Google Scholar]
- Liazid, A.; Palma, M.; Brigui, J.; Barroso, C.G. Investigation on phenolic compounds stability during microwave-assisted extraction. J. Chromatogr. A 2007, 1140, 29–34. [Google Scholar] [CrossRef]
- Lutria, D.L. Application of green chemistry principles for extraction of polylipids and phenolic compounds. Ind. J. Chem. 2006, 45, 2291–2296. [Google Scholar]
- Aspé, E.; Fernández, K. The effect of different extraction techniques on extraction yield, total phenolic, and anti-radical capacity of extracts from Pinus radiata Bar. Ind. Crop. Prod. 2011, 34, 838–844. [Google Scholar] [CrossRef]
- Toma, M.; Vinatoru, M.; Paniwnyk, L.; Mason, T. Investigation of the effects of ultrasound on vegetal tissues during solvent extraction. Ultrason. Sonochem. 2001, 8, 137–142. [Google Scholar] [CrossRef]
- Vinatoru, M. An overview of the ultrasonically assisted extraction of bioactive principles from herbs. Ultrason. Sonochem. 2001, 8, 303–313. [Google Scholar] [CrossRef]
- Wang, L.; Weller, C.L. Recent advances in extraction of nutraceuticals from plants. Trends Food Sci. Technol. 2006, 17, 300–312. [Google Scholar] [CrossRef]
- Kivilompolo, M.; Hyotylainen, T. On-line coupled dynamic sonication-assisted extraction–liquid chromatography for the determination of phenolic acids in Lamiaceae herbs. J. Chromatogr. A 2009, 1216, 892–896. [Google Scholar] [CrossRef]
- Domeño, C.; Blasco, M.; Sánchez, C.; Nerín, C. A fast extraction technique for extracting polycyclic aromatic hydrocarbons (PAHs) from lichens samples used as biomonitors of air pollution: Dynamic sonication versus other methods. Anal. Chim. Acta 2006, 569, 103–112. [Google Scholar] [CrossRef]
- Blasco, M.; Domeño, C.; Nerín, C. Lichens biomonitoring as feasible methodology to assess air pollution in natural ecosystems: Combined study of quantitative PAHs analyses and lichen biodiversity in the Pyrenees Mountains. Anal. Bioanal. Chem. 2008, 391, 759–771. [Google Scholar] [CrossRef]
- Blasco, M.; Domeño, C.; Lopez, P.; Nerín, C. Behaviour of different lichen species as biomonitors of air pollution by PAHs in natural cosystems. J. Environ. Monit. 2011, 13, 2588–2596. [Google Scholar] [CrossRef]
- Luque-Garcia, J.L.; Luque de Castro, M.D. Where is microwave based analytical treatment for solid sample pre-treatment going? Trends Anal. Chem. 2003, 22, 90–99. [Google Scholar] [CrossRef]
- Lee, M.H.; Lin, C.C. Comparison of techniques for extraction of isoflavones from the root of Radix Puerariae: Ultrasonic and pressurized solvent extractions. Food Chem. 2007, 105, 223–228. [Google Scholar] [CrossRef]
- Lu, C.; Wang, H.; Lv, W.; Ma, C.; Lou, Z.; Xie, J.; Liu, B. Ionic liquid-based ultrasonic/microwave-assisted extraction combined with UPLC–MS–MS for the determination of tannins in Galla chinensis. Nat. Prod. Res. 2012, 26, 1842–1847. [Google Scholar] [CrossRef]
- Kamran Khan, M.; Abert-Vian, M.; Fabiano-Tixier, A.-S.; Dangles, O.; Chemat, F. Ultrasound-assisted extraction of polyphenols (flavanone glycosides) from orange (Citrus sinensis L.) peel. Food Chem. 2010, 119, 851–858. [Google Scholar] [CrossRef]
- Ma, Y.; Ye, X.; Fang, Z.; Chen, J.; Xu, G.; Liu, D. Phenolic compounds and antioxidant activity of extracts from ultrasonic treatment of Satsuma mandarin (Citrus unshiu Marc.) peels. J. Agric. Food Chem. 2008, 56, 5682–5690. [Google Scholar]
- Rostagno, A.; Palma, M.; Barroso, C. Ultrasound-assisted extraction of soy isoflavones. J. Chromatogr. A 2003, 1012, 119–128. [Google Scholar] [CrossRef]
- Zhang, H.F.; Yangb, X.H.; Wang, Y. Microwave assisted extraction of secondary metabolites from plants: Current status and future directions. Trends Food Sci. Technol. 2011, 22, 672–688. [Google Scholar] [CrossRef]
- Camel, V. Recent extraction techniques for solid matrices supercritical fluid extraction pressurised fluid extraction and microwave-assisted extraction: their potential and pitfalls. Analyst 2001, 126, 1182–1193. [Google Scholar] [CrossRef]
- Zuloaga, O.; Etxebarria, N.; Fernandez, L.A.; Madariaga, J.M. Optimization and comparison of microwave assisted extraction and Soxhlet extraction for the determination of polychlorinated biphenyls in soil samples using an experimental design approach. Talanta 1999, 50, 345–357. [Google Scholar] [CrossRef]
- Huie, C.W. A review of modern sample preparation techniques for the extraction and analysis of medicinal plants. Anal. Bioanal. Chem. 2002, 373, 23–30. [Google Scholar] [CrossRef]
- Mandal, V.; Mohan, Y.; Hemalatha, S. Microwave assisted extraction—An innovative and promising extraction tool for medicinal plant research. Pharmacogn. Rev. 2007, 1, 7–18. [Google Scholar]
- Eskilsson, C.S.; Björklund, E. Analytical-scale microwave-assisted extraction. J. Chromatogr. A 2000, 902, 227–250. [Google Scholar] [CrossRef]
- Witayasinthana, W.; Shotipruk, A. Recovery of anti-cancer damnacanthal from roots of morinda citrifolia by microwave-assisted extraction. Sep. Sci.Technol. 2009, 44, 2942–2955. [Google Scholar] [CrossRef]
- Ajila, C.M.; Brar, S.K.; Verma, M.; Tyagi, R.D.; Godbout, S.; Valero, J.R. Extraction and analysis of polyphenols: recent trends. Crit. Rev. Biotech. 2011, 31, 227–249. [Google Scholar] [CrossRef]
- Venkatesh, M.; Raghavan, G. An overview of microwave processing and dielectric properties of agri-food materials. Biosys. Eng. 2004, 88, 1–18. [Google Scholar] [CrossRef]
- Vasu, S.; Palaniyappan, V.; Badami, S. A novel microwave-assisted extraction for the isolation of andrographolide from Andrographis paniculata and its in vitro antioxidant activity. J. Nat. Prod. 2010, 24, 1560–1567. [Google Scholar] [CrossRef]
- Xiao, W.; Han, L.; Shi, B. Microwave-assisted extraction of flavonoids from Radix astragali. Sep. Purif. Technol. 2008, 62, 614–618. [Google Scholar] [CrossRef]
- Hemwimon, S.; Pavasant, P.; Shotipruk, A. Microwave-assisted extraction of antioxidative anthraquinones from roots of Morinda citrifolia. Sep. Purif. Technol. 2007, 54, 44–50. [Google Scholar] [CrossRef]
- Nkhili, E.; Valerie, T.; El Hajji, H.; El Boustani, E.S.; Chemat, F.; Dangles, O. Microwave-assisted water extraction of green tea polyphenols. Phytochem. Anal. 2009, 20, 408–415. [Google Scholar] [CrossRef]
- Wang, L.; Qin, P.; Hu, Y. Study on the microwave-assisted extraction of polyphenols from tea. Front. Chem. Eng. Chin. 2010, 4, 307–313. [Google Scholar] [CrossRef]
- Song, J.; Li, D.; Liu, C.; Zhang, Y. Optimized microwaveassisted extraction of total phenolics (TP) from Ipomoea batatas leaves and its antioxidant activity. Innovat. Food Sci. Emerg. Tech. 2011, 12, 282–287. [Google Scholar] [CrossRef]
- Sutivisedsak, N.; Cheng, H.N.; Willett, J.L.; Lesch, W.C.; Tangsrud, R.R.; Biswas, A. Microwave-assisted extraction of phenolics from bean (Phaseolus vulgaris L.). Food Res. Int. 2010, 43, 516–519. [Google Scholar] [CrossRef]
- Inglett, G.E.; Rose, D.J.; Chen, D.; Stevenson, D.G.; Biswas, A. Phenolic content and antioxidant activity of extracts from whole buckwheat (Fagopyrum esculentum Moench) with or without microwave irradiation. Food Chem. 2010, 119, 1216–1219. [Google Scholar] [CrossRef]
- Martino, E.; Ramaiola, I.; Urbano, M.; Bracco, F.; Collina, S. Microwave-assisted extraction of coumarin and related compounds from Melilotus officinalis (L.) Pallas as an alternative to Soxhlet and ultrasound-assisted extraction. J. Chromatogr. A 2006, 1125, 147–151. [Google Scholar] [CrossRef]
- Longares-Patron, A.; Canizares-Macıas, M.P. Focused microwaves-assisted extraction and simultaneous spectrophotometric determination of vanillin and p-hydroxybenzaldehyde from vanilla fragans. Talanta 2006, 69, 882–887. [Google Scholar] [CrossRef]
- Liu, Z.; Wang, J.; Shen, P.; Wang, C.; Shen, Y. Microwave-assisted extraction and high-speed counter-current chromatography purification of ferulic acid from Radix Angelicae sinensis. Sep. Purif. Technol. 2006, 52, 18–21. [Google Scholar] [CrossRef]
- Gao, M.; Song, B.-Z.; Liu, C.-Z. Dynamic microwave-assisted extraction of flavonoids from Saussurea medusa Maxim cultured cells. Biochem. Eng. J. 2006, 32, 79–83. [Google Scholar]
- Constance, C.; Lloyd, W.R.; Trust, B. Microwave-assisted extraction of bound phenolic acids in bran and flour fractions from sorghum and maize cultivars varying in hardness. J. Agric. Food Chem. 2012, 60, 4735–4742. [Google Scholar] [CrossRef]
- Gallo, M.; Ferracane, R.; Graziani, G.; Ritieni, A.; Fogliano, V. Microwave assisted extraction of phenolic compounds from four different spices. Molecules 2010, 15, 6356–6374. [Google Scholar]
- Jiao, Y.; Zuo, Y. Ultrasonic extraction and HPLC determination of anthraquinones, aloe-emodine, emodine, rheine, chrysophanol and physcione, in roots of Polygoni multiflori. Phytochem. Anal. 2009, 20, 272–278. [Google Scholar] [CrossRef]
- Rostagno, M.A.; Arrigo, M.D.; Martínez, J.A. Combinatory and hyphenated sample preparation for the determination of bioactive compounds in foods. TrAC-Trend. Anal. Chem. 2010, 29, 553–561. [Google Scholar] [CrossRef]
- Lou, Z.X.; Wang, H.X.; Zhu, S.; Zhang, M.; Gao, Y.; Ma, C.Y.; Wang, Z.P. Improved extraction and identification by ultra performance liquid chromatography tandem mass spectrometry of phenolic compounds in burdock leaves. J. Chromatogr. A 2010, 1217, 2441–2446. [Google Scholar] [CrossRef]
- Xiao-Lan, C.; Jin-Yi, W.; Ping, L.; Lian-Wen, Q. Ultrasonic/microwave assisted extraction and diagnostic ion filtering strategy by liquid chromatography–quadrupole time-of-flight mass spectrometry for rapid characterization of flavonoids in Spatholobus suberectus. J. Chromatogr. A 2011, 1218, 5774–5786. [Google Scholar] [CrossRef]
- Lianfu, Z.; Zelong, L. Optimization and comparison of ultrasound/microwave assisted extraction (UMAE) and ultrasonic assisted extraction (UAE) of lycopene from tomatoes. Ultrason. Sonochem. 2008, 15, 731–737. [Google Scholar] [CrossRef]
- Lou, Z.; Wang, H.; Zhu, S.; Chen, S.; Zhang, M.; Wang, Z. Ionic liquids based simultaneous ultrasonic and microwave assisted extraction of phenolic compounds from burdock leaves. Anal. Chim. Acta 2012, 716, 28–33. [Google Scholar] [CrossRef]
- Huang, L.Y.; Cao, Y.Y.; Chen, G.N. Purification of quercetin in Anoectochilu roxburghii (wall) Lindl using UMAE by high-speed countercurrent chromatography and subsequent structure identification. Sep. Purif. Technol. 2008, 64, 101–107. [Google Scholar] [CrossRef]
- Bleve, M.; Ciurlia, L.; Erroi, E.; Lionetto, G.; Longoc, L.; Rescioa, L.; Schettino, T.; Vaspollo, G. An innovative method for the purification of anthocyanins from grape skin extracts by using liquid and sub-critical carbon dioxide. Sep. Purif. Technol. 2008, 64, 192–197. [Google Scholar] [CrossRef]
- Ignat, I.; Volf, I.; Popa, V.I. A critical review of methods for characterisation of polyphenolic compounds in fruits and vegetables. Food Chem. 2011, 126, 1821–1835. [Google Scholar] [CrossRef]
- Nahar, L.; Sarker, S.D. Supercritical fluid extraction. Methods Biotechnol. 2005, 20, 47–76. [Google Scholar]
- Khosravi Darani, K.; Mozafari, M.R. A review: Supercritical fluids technology in bioprocess industries. J. Biochem. Tech. 2009, 2, 144–152. [Google Scholar]
- Wells, S.; DeSimone, J. CO2 technology platform: an important tools for environmental problem solving. Angew. Chem. Int. Ed. 2001, 40, 518–527. [Google Scholar] [CrossRef]
- Abbasi, H.; Rezaei, K.; Emamdjomeh, Z.; Ebrahimzadeh Mousavi, S.M. Effect of various extraction conditions on the phenolic contents of pomegranate seed oil. Eur. J. Lipid Sci. Technol. 2008, 110, 435–440. [Google Scholar] [CrossRef]
- Gelmez, N.; Kincal, N.S.; Yener, M.E. Optimization of supercritical carbon dioxide extraction of antioxidants from roasted wheat germ based on yield, total phenolic and tocopherol contents, and antioxidant activities of the extract. J. Supercrit. Fluids 2009, 48, 217–224. [Google Scholar] [CrossRef]
- Maróstica Junior, M.R.; Leite, A.V.; Vicente Dragano, N.R. Supercritical fluid extraction and stabilization of phenolic compounds from natural sources—Review (supercritical extraction and stabilization of phenolic compounds). Open Chem. Eng. J. 2010, 4, 51–60. [Google Scholar] [CrossRef]
- Holliday, D.L. Phenolic compounds and antioxidant activity of oat bran by various extraction methods. Masters Thesis, Louisiana State University, LA, USA, December 2010. [Google Scholar]
- Arlorio, M.; Coïsson, J.D.; Travaglia, F.; Varsaldi, F.; Miglio, G.; Lombardi, G.; Martelli, A. Antioxidant and biological activity of phenolic pigments from Theobroma cacao hulls extracted with supercritical CO2. Food Res. Int. 2005, 38, 1009–1014. [Google Scholar] [CrossRef]
- Leal, P.F.; Maia, N.B.; Carmello, Q.A.C.; Catharino, R.R.; Eberlin, M.N.; Meireles, M.A.A. Sweet basil (Ocimum basilicum) extracts obtained by supercritical fluid extraction (SFE): Global yields, Chemical composition, Antioxidant activity, and estimation of the cost of manufacturing. Food Bioprocess Technol. 2008, 1, 326–338. [Google Scholar] [CrossRef]
- Piantino, C.R.; Aquino, F.W.B.; Follegatti-Romero, L.A.; Cabral, F.A. Supercritical CO2 extraction of phenolic compounds from Baccharis dracunculifolia. J. Supercrit. Fluids 2008, 47, 209–214. [Google Scholar] [CrossRef]
- Goli, A.H.; Barzegar, M.; Sahari, M.A. Antioxidant activity and total phenolic Compounds of pistachio (Pistachia vera) hull extracts. Food Chem. 2005, 92, 521–525. [Google Scholar] [CrossRef]
- Wang, B.J.; Liu, C.T.; Tseng, C.Y.; Yu, Z.R. Antioxidant activity of Bupleurum kaoi Liu (Chao et Chuang) fractions fractionated by supercritical CO2. LWT-Food Sci. Technol. 2005, 38, 281–287. [Google Scholar] [CrossRef]
- Shan, B.; Xie, J.-H.; Zhu, J.-H.; Peng, Y. Ethanol modified supercritical carbon dioxide extraction of flavonoids from Momordica charantia L. and its antioxidant activity. Food Bioprod. Process. 2012, 90, 579–587. [Google Scholar] [CrossRef]
- Bimakr, M.; Abdul Rahman, R.; Saleena Taip, F.; Ganjloo, A.; Salleh, L.M.; Selamat, J.; Hamid, A.; Zaidul, I.S.M. Comparison of different extraction methods for the extraction of major bioactive flavonoid compounds from spearmint (Mentha spicata L.) leaves. Food Bioprod. Process. 2011, 89, 67–72. [Google Scholar] [CrossRef]
- Liza, M.S.; Abdul Rahman, R.; Mandana, B.; Jinap, S.; Rahmat, A.; Zaidul, I.S.M.; Hamid, A. Supercritical carbon dioxide extraction of bioactive flavonoid from Strobilanthes crispus (Pecah Kaca). 2010, 88, 319–326. [Google Scholar]
- Wang, L.; Yang, B.; Du, X.; Yi, C. Optimisation of supercritical extraction of flavonoids from Pueraria lobata. Food Chem. 2008, 108, 737–741. [Google Scholar] [CrossRef]
- Kronholm, J.; Hartonen, K.; Riekkola, M.L. Analytical extractions with water at elevated temperatures and pressures. TrAC–Trends Anal. Chem. 2007, 26, 396–412. [Google Scholar] [CrossRef]
- Herrero, M.; Cifuentes, A.; Ibanez, E. A review: Sub- and supercritical fluid extraction of functional ingredients from different natural sources: Plants, Food-byproducts, Algae and microalgae. Food Chem. 2006, 98, 136–148. [Google Scholar] [CrossRef]
- Moran, J.; Shapiro, H.N. Fundamentals of Engineering Thermodynamics, 5th ed; John Wiley & Sons Ltd: West Sussex, UK, 2006. [Google Scholar]
- Fernandez, D.P.; Goodwin, A.R.H.; Lemmon, E.W.; Levelt-Sengers, J.M.H.; Williams, R.C. A formulation for the static permittivity of water and steam at temperatures atures from 238 K to 873 K at pressures up to 1200 MPa, Including derivatives and Debye-Hückel coefficients. J. Phys. Chem. 1997, 26, 1126–1166. [Google Scholar]
- Miller, D.J.; Hawthorne, S.B. Solubility of liquid organic flavor and fragance compounds in subcritical (hot/liquid) water from 298 to 473 K. J. Chem. Eng. Data 2000, 45, 315–318. [Google Scholar] [CrossRef]
- Luque-Rodríguez, J.M.; Luque de Castro, M.D.; Pérez-Juan, P. Dynamic superheated liquid extraction of anthocyanins and other phenolics from red grape skins of wine making residues. Bioresource Technol. 2007, 98, 2705–2713. [Google Scholar]
- Eikani, M.H.; Golmohammad, F.; Rowshanzamir, S. Subcritical water extraction of essential oils from coriander seeds (Coriandrum sativum L.). J. Food Eng. 2006, 80, 735–740. [Google Scholar]
- Pongnaravane, B.; Goto, M.; Sasaki, M.; Anekpankul, T.; Pavasant, P.; Shotipruk, A. Extraction of anthraquinones from roots of Morinda citrifolia by pressurized hot water: Antioxidant activity of extracts. J. Supercrit. Fluids 2006, 37, 390–396. [Google Scholar] [CrossRef]
- He, L.; Zhang, X.; Xu, H.; Xu, C.; Yuan, F.; Knez, Z.E.; Novak, Z.; Gao, Y. Subcritical water extraction of phenolic compounds from pomegranate (Punica granatum L.) seed residues and investigation into their antioxidant activities with HPLC–ABTS•+ assay. Food. Bioprod. Process. 2012, 90, 215–223. [Google Scholar] [CrossRef]
- Khuwijitjaru, P.; Sayputikasikorn, N.; Samuhasaneetoo, S.; Penroj, P.; Siriwongwilaichat, P.; Adachi, S. Subcritical water extraction of flavoring and phenolic compounds from cinnamon bark (Cinnamomum zeylanicum). J. Oleo Sci. 2012, 61, 349–355. [Google Scholar] [CrossRef]
- Singh, P.P.; Saldaña, M.D.A. Subcritical water extraction of phenolic compounds from potato peel. Food. Res. Int. 2011, 44, 2452–2458. [Google Scholar] [CrossRef]
- Cynthia, F.; Ngoc, Y.T.-T.; Novy, S.K.; Yi-Hsu, J. Release of phenolic acids from defatted rice bran by subcritical water treatment. J. Sci. Food Agric. 2010, 90, 2576–2581. [Google Scholar] [CrossRef]
- Rangsriwong, P.; Rangkadilok, N.; Satayavivad, J.; Gotoc, M.; Shotipruk, A. Subcritical water extraction of polyphenolic compounds from Terminalia chebula Retz. fruits. Sep. Purif. Technol. 2009, 66, 51–56. [Google Scholar] [CrossRef]
- Budrat, P.; Shotipruk, A. Extraction of phenolic compounds from fruits of bitter melon (Momordica charantia) with subcritical water extraction and antioxidant activities of these extracts. Chiang Mai J. Sci. 2008, 35, 123–130. [Google Scholar]
- Rodríguez-Meizoso, I.; Marin, F.R.; Herrero, M.; Señorans, F.J.; Reglero, G.; Cifuentes, A.; Ibáñez, E. Subcritical water extraction of nutraceuticals with antioxidant activity from oregano. Chemical and functional characterization. J. Pharmaceut. Biomed. 2006, 41, 1560–1565. [Google Scholar] [CrossRef]
- Etoh, H.; Ohtaki, N.; Kato, H.; Kulkarni, A.; Morita, A. Sub-critical water extraction of residual green tea to produce a roasted green tea-like extract. Biosci. Biotechnol. Biochem. 2010, 74, 858–860. [Google Scholar] [CrossRef]
- Zhang, S.; Xi, J.; Wang, C. High hydrostatic pressure extraction of flavonoids from propolis. J. Chem. Technol. Biot. 2005, 80, 50–54. [Google Scholar] [CrossRef]
- Rastogi, N.K.; Angersbach, A.; Knorr, D. Food Preservation Technology; CRC Press: New York, NY, USA, 2003. [Google Scholar]
- Yan, H.X. Separation Engineering; China Petrochemical Press: Beijing, China, 2002. [Google Scholar]
- Richard, J.S. High Pressure Phase Behaviour of Multi Component Fluid Mixtures; Elsevier: Amsterdam, The Netherland, 1992. [Google Scholar]
- Smith, R.M. Extractions with superheated water. J. Chromatogr. A 2002, 975, 31–46. [Google Scholar] [CrossRef]
- Ramos, L.; Kristenson, E.M.; Brinkman, U.A.T. Current use of pressurised liquid extraction and subcritical water extraction in environmental analysis. J. Chromatogr. A 2002, 975, 3–29. [Google Scholar] [CrossRef]
- Jun, X.; Deji, S.; Shou, Z.; Bingbing, L.; Ye, L.; Zhang, R. Characterization of polyphenols from green tea leaves using a high hydrostatic pressure extraction. Int. J. Pharm. 2009, 382, 139–143. [Google Scholar] [CrossRef]
- Corrales, M.; Toepfl, S.; Butz, P.; Knorr, D.; Tauscher, B. Extraction of anthocyanins from grape by-products assisted by ultrasonics, high hydrostatic pressure or pulsed electric fields: A comparison. Innov. Food Sci. Emerg. Technol. 2008, 9, 85–91. [Google Scholar] [CrossRef]
- Queiroz, C.; Moreira, F.F.; Lavinas, F.C.; Lopes, M.L.M.; Fialho, E.; Valente-Mesquita, V.L. Effect of high hydrostatic pressure on phenolic compounds, ascorbic acid and antioxidant activity in cashew apple juice. High Pressure Res. 2010, 30, 507–513. [Google Scholar] [CrossRef]
- Puértolas, E.; Luengo, E.; Álvarez, I.; Raso, J. Improving mass transfer to soften tissues by pulsed electric fields: Fundamentals and applications. Annu. Rev. Food Sci. Technol. 2012, 3, 263–282. [Google Scholar] [CrossRef]
- Toepfl, S.; Mathys, A.; Heinz, V.; Knorr, D. Review: Potential of high hydrostatic pressure and pulsed electric fields for energy efficient and environmentally friendly food processing. Food Rev. Int. 2006, 22, 405–423. [Google Scholar] [CrossRef]
- Kannan, V. Extraction of bioactive compounds from whole red cabbage and beetroot using pulsed electric fields and evaluation of their functionality. Masters Thesis, University of Nebraska, Lincoln, NE, USA, May 2011. [Google Scholar]
- Odriozola-Serrano, I.; Soliva-Fortuny, R.; Martín-Belloso, O. Phenolic acids, flavonoids, vitamin C and antioxidant capacity of strawberry juices processed by high-intensity pulsed electric fields or heat treatments. Eur. Food Res. Technol. 2008, 228, 239–248. [Google Scholar] [CrossRef]
- López, N.; Puértolas, E.; Condón, S.; Álvarez, I.; Raso, J. Effects of pulsed electric fields on the extraction of phenolic compounds during the fermentation of must of Tempranillo grapes. Innov. Food Sci. Emerg. Technol. 2008, 9, 477–482. [Google Scholar] [CrossRef]
- Grimi, N.; Nikolai, I.L.; Eugene, V.; Jean, V. Effect of a pulsed electric field treatment on expression behavior and juice quality of chardonnay grape. Food Biophys. 2009, 4, 191–198. [Google Scholar] [CrossRef]
- Delsart, C.; Ghidossi, R.; Poupot, C.; Cholet, C.; Grimi, N.; Vorobiev, E.; Milisic, V.; Mietton, M.P. Enhanced extraction of phenolic compounds from Merlot grapes by pulsed electric field treatment. Am. J. Enol. Vitic. 2012, 63, 205–211. [Google Scholar] [CrossRef]
- Turk, M.F.; Baron, A.; Eugene, V. Effect of pulsed electric fields treatment and mash size on extraction and composition of apple juices. J. Agric. Food Chem. 2010, 58, 9611–9616. [Google Scholar] [CrossRef]
- Wibisono, R.; Zhang, J.; Saleh, Z.; Stevenson, D.E.; Joyce, N.I. Optimization of accelerated solvent extraction for screening of the health benefits of plant food materials. Health 2009, 1, 220–230. [Google Scholar] [CrossRef]
- Richter, B.E.; Jones, B.A.; Ezzell, J.L.; Porter, N.L.; Avdalovic, N.; Pohl, C. Accelerated solvent extraction: A technique for sample preparation. Ann. Chem. 1996, 68, 1033–1039. [Google Scholar] [CrossRef]
- Pinelo, M.; Meyer, A.S. Enzyme- assisted extraction of antioxidants: Release of phenols from vegetal matrixes. Elec. J. Env. Agric. Food Chem. 2008, 7, 3217–3220. [Google Scholar]
- Pinelo, M.; Arnous, A.; Meyer, A.S. Upgrading of grape skins: significance of plant cell-wall structural components and extraction techniques for phenol release. Trends Food Sci. Technol. 2006, 17, 579–590. [Google Scholar] [CrossRef]
- Le Bourvellec, C.; Guyot, S.; Renard, C.M.G.C. Interactions between apple (Malus x domestica Borkh.) polyphenol and cell walls modulate the extractability of polysaccharides. Carbohyd. Polym. 2009, 75, 251–561. [Google Scholar] [CrossRef]
- Laroze, L.; Soto, C.; Zúñiga, M.E. Phenolic antioxidants extraction from raspberry wastes assisted by-enzymes. Electron. J. Biotechnol. 2010, 13, 1–11. [Google Scholar]
- Maier, T.; Goppert, A.; Kammerer, D.R.; Schieber, A.; Carle, R. Optimization of a process for enzyme-assisted pigment extraction from grape (Vitis vinifera L.) pomace. Eur. Food Res. Technol. 2008, 227, 267–275. [Google Scholar] [CrossRef]
- Min, J.Y.; Kang, S.M.; Park, D.J.; Kim, Y.D.; Jung, H.N.; Yang, J.K.; Seo, W.T.; Kim, S.W.; Karigar, C.S.; Choi, M.S. Enzymatic release of ferulic acid from Ipomoea batatas L. (sweet potato) stem. Biotechnol. Bioproc. E 2006, 11, 372–376. [Google Scholar] [CrossRef]
- Hong, V.L.; Van Viet, M.L. Comparison of enzyme-assisted and ultrasound-assisted extraction of vitamin C and phenolic compounds from acerola (Malpighia emarginata DC.) fruit. Int. J. Food Sci. Tech. 2012, 47, 1206–1214. [Google Scholar] [CrossRef]
- Rong, T.; Yang, R. Optimization of a new mobile phase to know the complex and real polyphenolic composition: Towards a total phenolic index using high-performance liquid chromatography. J. Chromatogr. A 2003, 1018, 29–40. [Google Scholar] [CrossRef]
- Liu, Q.; Cai, W.; Shao, X. Determination of seven polyphenols in water by high performance liquid chromatography combined with preconcentration. Talanta 2008, 77, 679–683. [Google Scholar] [CrossRef]
- Lapornik, B.; Prosek, M.; Golc, W.A. Comparison of extracts prepared from plant by-products using different solvents and extraction time. J. Food Eng. 2005, 71, 214–222. [Google Scholar] [CrossRef]
- Naczk, M.; Shahidi, F. Phenolics in cereals, fruits and vegetables: Occurrence, extraction and analysis. J. Pharmaceut. Biomed. Anal. 2006, 41, 1523–1542. [Google Scholar] [CrossRef]
- Box, J.D. Investigation of the Folin-Ciocalteu phenol reagent for the determination of polyphenolic substances in natural waters. Water Res. 1983, 17, 511–525. [Google Scholar] [CrossRef]
- Huang, W.; Xue, A.; Niu, H.; Jia, Z.; Wang, J. Optimised ultrasonic-assisted extraction of flavonoids from Folium eucommiae and evaluation of antioxidant activity in multi-test systems in vitro. Food Chem. 2009, 114, 1147–1154. [Google Scholar] [CrossRef]
- Fernandes, A.J.D.; Ferreira, M.R.A.; Randau, K.P.; De Souza, T.P.; Soares, L.A.L. Total flavonoids content in the raw material and aqueous extractives from Bauhinia monandra Kurz (Caesalpiniaceae). Sci. World J. 2012, 2012, 1–7. [Google Scholar]
- Abeynayake, S.W.; Panter, S.; Mouradov, A.; Spangenberg, G. A high-resolution method for the localization of proanthocyanidins in plant tissues. Plant Method. 2011, 7, 1–6. [Google Scholar]
- Hartzfeld, P.W.; Forkner, R.; Hunter, M.D.; Hagerman, A.E. Determination of hydrolyzable tannins (gallotannins and ellagitannins) after reaction with potassium iodate. J. Agric. Food Chem. 2002, 50, 1785–1790. [Google Scholar] [CrossRef]
- Kobs, L. Dietary Polyphenolic Intake from Acrons and Acron Meal. Masters Thesis, The University of Georgia, GA, USA, May 2008. [Google Scholar]
- Porter, L.J.; Hrstich, L.N.; Chan, B.G. The conversion of procyanidins and prodelphinidins to cyanidin and delphinidin. Phytochemistry 1986, 25, 223–230. [Google Scholar]
- Hagerman, A.E.; Butler, L.G. Protein Precipitation Method for Quantitative-Determination of Tannins. J. Agric. Food Chem. 1978, 26, 809–812. [Google Scholar] [CrossRef]
- Sarneckis, C.; Dambergs, R.G.; Jones, P.; Mercurio, M.D.; Herderich, M.J.; Smith, P.A. Quantification of condensed tannins by precipitation with methyl cellulose: Development and validation of an optimized tool for grape and wine analysis. Aust. J. Grape Wine Res. 2006, 12, 39–49. [Google Scholar]
- Mercurio, M.D.; Dambergs, R.G.; Herderich, M.J.; Smith, P.A. High throughput analysis of red wine and grape phenolics—Adaption and validation of methyl cellulose precipitable tannin assay and modified somers color assay to a rapid 96 well plate format. J. Agric. Food Chem. 2007, 55, 4651–4657. [Google Scholar] [CrossRef]
- Inoue, K.H.; Hagerman, A.E. Determination of gallotannin with rhodanine. Anal. Biochem. 1988, 169, 363–369. [Google Scholar] [CrossRef]
- Wilson, T.C.; Hagerman, A.E. Quantitative determination of ellagic acid. J. Agric. Food Chem. 1990, 38, 1678–1683. [Google Scholar] [CrossRef]
- Jun, X.; Shouqin, Z. Antioxactivity of ethanolic extract of propolis by hydrostatic pressure extraction. Int. J. Food Sci. Tech. 2007, 42, 1350–1356. [Google Scholar] [CrossRef]
- Marghtas, L.A.; Mihai, C.M.; Chirila, F.; Dezmirean, D.S.; Fit, N. The study of the antimicrobial activity of Transylvanian (Romanian) propolis. Not. Bot. Hort. Agrobot. Cluj. 2010, 38, 40–44. [Google Scholar]
- Giusti, M.M.; Wrolstad, R.E. Acylated anthocyanins from edible sources and their applications in food systems. Biochem. Eng. J. 2003, 14, 217–225. [Google Scholar] [CrossRef]
- Welch, C.R.; Wu, Q.; Simon, J.E. Recent advances in anthocyanin analysis and characterization. Curr. Anal. Chem. 2008, 4, 75–101. [Google Scholar] [CrossRef]
- Martin, J.G.P.; Porto, E.; Corrêa, C.B.; De Alencar, S.M.; Da Gloria, E.M.; Cabral, I.S.R.; De Aquino, L.M. Antimicrobial potential and chemical composition of agro-industrial wastes. J. Nat. Prod. 2012, 5, 27–36. [Google Scholar]
- Shadkami, F.; Estevez, S.; Helleur, R. Analysis of catechins and condensed tannins by thermally assisted hydrolysis/methylation-GC/MS and by a novel two step methylation. J. Anal. Appl. Pyrolysis. 2009, 85, 54–65. [Google Scholar] [CrossRef]
- Proestos, C.; Boziaris, I.S.; Nychas, G.-J.E.; Komaitis, M. Analysis of flavonoids and phenolic acids in Greek aromatic plants: Investing ation of their antioxidant capacity and antimicrobial activity. Food Chem. 2006, 95, 664–671. [Google Scholar] [CrossRef]
- Liggins, J.; Bluck, L.J.C.; Coward, A.; Bingham, S.A. Extraction and quantification of daidzein and genistein in food. Anal. Biochem. 1998, 264, 1–7. [Google Scholar] [CrossRef]
- Siess, M.H.; Le Bon, A.M.; Canivenc-Lavier, M.C.; Amiot, M.J.; Sabatier, S.; Aubert, S.Y.; Suschetet, M. Flavonoids of honey and propolis: characterization and effects on hepatic drug-metabolizing enzymes and benzo[a]pyrene-DNA binding in rats. J. Agric. Food Chem. 1996, 44, 2297–2301. [Google Scholar] [CrossRef]
- Wang, L.F.; Kim, D.M.; Lee, C.Y. Effects of heat processing and storage on flavanols and sensory qualities of green tea beverage. J. Agric. Food Chem. 2000, 48, 4227–4232. [Google Scholar] [CrossRef]
- Smolarz, H.D. Application of GC-MS method for analysis of phenolic acids and their esters in chloroformic extracts from some taxons of Polygonum L. genus. Chem. Anal. 2001, 46, 439–444. [Google Scholar]
- Waksmundzka-Hajnos, M. Chromatographic separations of aromatic carboxylic acids. J. Chromatogr. B 1998, 717, 93–118. [Google Scholar] [CrossRef]
- Husek, P. Fast derivatization and GC analysis of phenolic acids. Chromatographia 1992, 34, 621–626. [Google Scholar] [CrossRef]
- Zadernowski, R.; Czaplicki, S.; Naczk, M. Phenolic acid profiles of mangosteen fruits (Garcinia mangostana). Food Chem. 2009, 112, 685–689. [Google Scholar] [CrossRef]
- Robbins, R.J. Phenolic acids in foods: An overview of analytical methodology. J. Agric. Food Chem. 2003, 51, 2866–2887. [Google Scholar] [CrossRef]
- Pereira, A.D.S.; Padilha, M.C.; Neto, F.R.D.A. Two decades of high temperature gas chromatography (1983-2003): What’S next? Microchem. J. 2004, 77, 141–149. [Google Scholar] [CrossRef]
- Deslaurien, I. Recovery, separation and characterization of phenolic compounds and flavonids from maple products. Masters Thesis, McGill University, Montreal, Québec, Canada, March 2000. [Google Scholar]
- Kalili, K.M.; de Villiers, A. Recent developments in the HPLC separation of phenolic compounds. J. Sep. Sci. 2011, 34, 854–876. [Google Scholar] [CrossRef]
- Flamini, R. Mass spectrometry in grape and wine chemistry. Part I: Polyphenols. Mass Spectrom. Rev. 2003, 22, 218–250. [Google Scholar] [CrossRef]
- Merken, H.M.; Beecher, G.R. Review: Measurement of food flavonoids by highperformance liquid chromatography. J. Agric. Food Chem. 2000, 48, 577–599. [Google Scholar] [CrossRef]
- Kuskoski, E.M.; Rios, J.J.; Bueno, J.M.; Fett, R.; Troncoso, A.M.; Asuero, A.G. Capillary gas chromatography-mass spectrometry (CGC-MS) analysis and antioxidant activities of phenolic and components of guarana and derivatives. Open Anal. Chem. J. 2012, 6, 1–8. [Google Scholar]
- Khallouki, F.; Haubner, R.; Erben, G.; Ulrich, C.M.; Owen, R.W. Phytochemical composition and antioxidant capacity of various botanical parts of the fruits of Prunus domestica L. from the Lorraine region of Europe. Food Chem. 2012, 133, 697–706. [Google Scholar] [CrossRef]
- Wang, C.; Zuo, Y. Ultrasound-assisted hydrolysis and gas chromatography–mass spectrometric determination of phenolic compounds in cranberry products. Food Chem. 2011, 128, 562–568. [Google Scholar] [CrossRef]
- Esmaeili, N.; Ebrahimzadeh, H.; Abdi, K.; Safarian, S. Determination of some phenolic compounds in Crocus sativus L. corms and its antioxidant activities study. Pharmacogn. Mag. 2011, 7, 74–80. [Google Scholar] [CrossRef]
- Salomão, K.; Dantas, A.P.; Borba, C.M.; Campos, L.C.; Machado, D.G.; Aquino Neto, F.R.; De Castro, S.L. Chemical composition and microbicidal activity of extracts from Brazilian and Bulgarian propolis. Appl. Microbiol. 2004, 38, 87–92. [Google Scholar] [CrossRef]
- Zarena, A.S.; Udaya Sankar, K. Phenolic acids, flavonoid profile and antioxidant activity in mangosteen (Garcina mangostana L.) pericarp. J. Food Biochem. 2011, 36, 627–633. [Google Scholar] [CrossRef]
- Qin, C.; Li, Y.; Niu, W.; Ding, Y.; Zhang, R.; Shang, X. Analysis and characterisation of anthocyanins in mulberry fruit. Czech J. Food Sci. 2010, 28, 117–126. [Google Scholar]
- Lee, J.; Rennaker, C.; Wrolstad, R.E. Correlation of two anthocyanin quantification methods: HPLC and spectrophotometric methods. Food Chem. 2008, 110, 782–786. [Google Scholar] [CrossRef]
- Diagone, C.A.; Colombo, R.; Lancas, F.M.; Yariwake, J.H. CZE/PAD and HPLC-UV/PAD profile of flavonoids from Maytenus aquifolium and Maytenus ilicifolia “espinheira santa” leaves extracts. Chromatogr. Res. Int. 2012, 2012. [Google Scholar] [CrossRef]
- Hoang, N.T.T.; Golding, J.B.; Wilkes, M.A. The effect of postharvest 1-MCP treatment and storage atmosphere on ‘Cripps Pink’ apple phenolics and antioxidant activity. Food Chem. 2011, 127, 1249–1256. [Google Scholar] [CrossRef]
- Simirgiotis, M.J.; Silva, M.; Becerra, J.; Schmeda-Hirschmann, G. Direct characterisation of phenolic antioxidants in infusions from four Mapuche medicinal plants by liquid chromatography with diode array detection (HPLC-DAD) and electrospray ionisation tandem mass spectrometry (HPLC-ESI–MS). Food Chem. 2012, 131, 318–327. [Google Scholar] [CrossRef]
- Rzeppa, S.; Von Bargen, C.; Bittner, K.; Humpf, H.-U. Analysis of flavan-3-ols and procyanidins in food samples by reversed phase high-performance liquid chromatography coupled to electrospray ionization tandem mass spectrometry (RP-HPLC-ESI-MS/MS). J. Agric. Food Chem. 2011, 59, 10594–10603. [Google Scholar] [CrossRef]
- Chen, S.; Wu, B.-H.; Fang, J.-B.; Liua, Y.-L.; Zhang, H.-H.; Fang, L.-C.; Guan, L.; Li, S.-H. Analysis of flavonoids from lotus (Nelumbo nucifera) leaves using high performance liquid chromatography/photodiode array detector tandem electrospray ionization mass spectrometry and an extraction method optimized by orthogonal design. J. Chromatogr. A 2012, 1227, 145–153. [Google Scholar] [CrossRef]
- Muller, D.; Schantz, M.; Richling, E. High performance liquid chromatography analysis of anthocyanins in bilberries (Vaccinium myrtillus L.), blueberries (Vaccinium corymbosum L.), and corresponding juices. J. Food Sci. 2012, 77, 340–345. [Google Scholar] [CrossRef]
- Jalili, A.; Sadeghzade, A. Comparative phenolic profile of Persian walnut (Juglans regia L.) leaves cultivars grown in Iran. Afr. J. Biochem. Res. 2012, 6, 33–38. [Google Scholar]
- Heinio, R.-L.; Liukkonena, K.-H.; Myllymaki, O.; Pihlavac, J.-M.; Adlercreutzd, H.; Heinonend, S.-M.; Poutanen, K. Quantities of phenolic compounds and their impacts on the perceived flavour attributes of rye grain. J. Cereal Sci. 2008, 47, 566–575. [Google Scholar] [CrossRef]
- Mousavinejad, G.; Emam-Djomeh, Z.; Rezaei, K.; Haddad Khodaparast, M.H. Identification and quantification of phenolic compounds and their effects on antioxidant activity in pomegranate juices of eight Iranian cultivars. Food Chem. 2009, 115, 1274–1278. [Google Scholar] [CrossRef]
- Kelebek, H.; Selli, S. Determination of volatile, phenolic, organic acid and sugar components in a Turkish cv. Dortyol (Citrus sinensis L. Osbeck) orange juice. J. Sci. Food. Agric. 2011, 91, 1855–1862. [Google Scholar] [CrossRef]
- Gómez-Caravaca, A.M.; Iafelice, G.; Lavini, A.; Pulvento, C.; Caboni, M.F.; Marconi, E. Phenolic compounds and saponins in quinoa samples (Chenopodium quinoa Willd.) grown under different saline and nonsaline irrigation regimens. J. Agric. Food Chem. 2012, 60, 4620–4627. [Google Scholar]
- Park, Y.S.; Jeon, M.H.; Hwang, H.J.; Park, M.R.; Lee, S.-H.; Kim, S.G.; Kim, M. Antioxidant activity and analysis of proanthocyanidins from pine (Pinus densiflora) needles. Nutr. Res. Pract. 2011, 5, 281–287. [Google Scholar] [CrossRef]
- Zitka, O.; Sochor, J.; Rop, O.; Skalickova, S.; Sobrova, P.; Zehnalek, J.; Beklova, M.; Krska, B.; Adam, V.; Kizek, R. Comparison of various easy-to-use procedures for extraction of phenols from apricot fruits. Molecules 2011, 16, 2914–2936. [Google Scholar] [CrossRef]
- Zimmermann, B.F.; Walch, S.G.; Tinzoh, L.N.; Stühlinger, W.; Lachenmeier, D.W. Rapid UHPLC determination of polyphenols in aqueous infusions of Salvia officinalis L. (sage tea). J. Chromatogr. B 2011, 879, 2459–2464. [Google Scholar] [CrossRef]
- Arraez-Roman, D.; Fu, S.; Sawalha, S.M.S.; Segura- Carretero, A.; Fernandez-Gutierrez, A. HPLC/CE-ESI-TOF-MS methods for the characterization of polyphenols in almond skin extracts. Electrophoresis 2010, 31, 2289–2296. [Google Scholar] [CrossRef]
- Pati, S.; Liberatore, M.T.; Gambacorta, G.; Antonacci, D.; La Notte, E. Rapid screening for anthocyanins and anthocyanin dimers in crude grape extracts by high performance liquid chromatography coupled with diode array detection and tandem mass spectrometry. J. Chromatogr. A 2009, 1216, 3864–3868. [Google Scholar] [CrossRef]
- Wei, S.-D.; Zhou, H.-C.; Lin, Y.-M. Antioxidant activities of fractions of polymeric procyanidins from stem bark of Acacia confuse. Int. J. Mol. Sci. 2011, 12, 1146–1160. [Google Scholar] [CrossRef]
- Ribas Agustí, A.; Gratacós Cubarsí, M.; Sárraga, C.; García Regueiro, J.A.; Castellari, M. Analysis of eleven phenolic compounds including novel p-coumaroyl derivatives in lettuce (Lactuca sativa L.) by ultra high performance liquid chromatography with photodiode array and mass spectrometry detection. Phytochem. Anal. 2011, 22, 555–563. [Google Scholar] [CrossRef]
- Hurst, W.J.; Stanley, B.; Glinski, J.A.; Davey, M.; Payne, M.J.; Stuart, D.A. Characterization of primary standards for use in the HPLC analysis of the procyanidin content of cocoa and chocolate containing products. Molecules 2009, 14, 4136–4146. [Google Scholar] [CrossRef]
- Barros, L.; Dueñas, M.; Ferreira, I.C.F.R.; Baptista, P.; Santos-Buelga, C. Phenolic acids determination by HPLC–DAD–ESI/MS in sixteen different Portuguese wild mushrooms species. Food. Chem. Toxicol. 2009, 47, 1076–1079. [Google Scholar] [CrossRef]
- Kalili, K.M.; de Villiers, A. Off-line comprehensive 2-dimensional hydrophilic interaction×reversed phase liquid chromatography analysis of procyanidins. J. Chromatogr. A 2009, 1216, 6274–6284. [Google Scholar] [CrossRef]
- Luthria, D.L.; Pastor-Corrales, M.A. Phenolic acids content of fifteen dry edible bean (Phaseolus vulgaris L.) varieties. J. Food Compos. Anal. 2006, 19, 205–211. [Google Scholar] [CrossRef]
- Colon, M.; Nerin, C. Role of catechins in the antioxidant capacity of an active film containing green tea, green coffee, and grapefruit extracts. J. Agric. Food Chem. 2012, 60, 9842–9849. [Google Scholar] [CrossRef]
- Antolovich, M.; Prenzler, P.; Robards, K.; Ryan, D. Sample preparation in the determination of phenolic compounds in fruits. Analyst 2000, 125, 989–1009. [Google Scholar] [CrossRef]
- Silva, E.M.; Pompeu, D.R.; Larondelle, Y.; Rogez, H. Optimisation of the adsorption of polyphenols from Inga edulis leaves on macroporous resins using an experimental design methodology. Sep. Purif. Technol. 2007, 53, 274–280. [Google Scholar] [CrossRef]
- Li, P.; Wang, Y.; Ma, R.; Zhang, X. Separation of tea polyphenol from green tea leaves by a combined CATUFM-adsorption resin process. J. Food Eng. 2005, 67, 253–260. [Google Scholar] [CrossRef]
- Scordino, M.; Di Muaro, A.; Passerini, A.; Maccarone, E. Adsorption of flavonoids on resins: cyanidin 3-glucoside. J. Agric. Food Chem. 2004, 52, 1965–1972. [Google Scholar] [CrossRef]
- Rostagno, M.A.; Palma, M.; Barroso, C.G. Solid-phase extraction of soy isoflavones. J. Chromatogr. A 2005, 1076, 110–117. [Google Scholar] [CrossRef]
- Michalkiewicz, A.; Biesaga, M.; Pyrzynska, K. Solid-phase extraction procedure for determination of phenolic acids and some flavonols in honey. J. Chromatogr. A 2008, 1187, 18–24. [Google Scholar] [CrossRef]
- Reichelt, K.V.; Peter, R.; Paetz, S.; Roloff, M.; Ley, J.P.; Krammer, G.E.; Engel, K.H. Characterization of flavor modulating effects in complex mixtures via high temperature liquid chromatography. J. Agric. Food Chem. 2010, 58, 458–464. [Google Scholar]
- Cooper, K.A.; Campos-Gimenez, E.; Jimenez-Alvarez, D.; Nagy, K.; Donovan, J.L.; Williamson, G. Rapid reversed phase ultra-performance liquid chromatography analysis of the major cocoa polyphenols and inter-relationships of their concentrations in chocolate. J. Agric. Food Chem. 2007, 55, 2841–2847. [Google Scholar]
- Takenaka, Y.; Morimoto, N.; Hamada, N.; Tanahashi, T. Phenolic compounds from the cultured mycobionts of Graphis proserpens. Phytochemistry 2011, 72, 1431–1435. [Google Scholar]
- Lopes-Lutz, D.; Dettmann, J.; Nimalaratne, C.; Schieber, A. Characterization and quantification of polyphenols in Amazon grape (Pourouma cecropiifolia Martius). Molecules 2010, 15, 8543–8552. [Google Scholar] [CrossRef]
- Klejdus, B.; Vacek, J.; Benesova, L.; Kopecky, J.; Lapcik, O.; Kuban, V. Rapid-resolution HPLC with spectrometric detection for the determination and identification of isoflavones in soy preparations and plant extracts. Anal. Bioanal. Chem. 2007, 389, 2277–2285. [Google Scholar] [CrossRef]
- Churchwell, M.I.; Twaddle, N.C.; Meeker, L.R.; Doerge, D.R. Improving LC–MS sensitivity through increases in chromatographic performance: Comparisons of UPLC–ES/MS/MS to HPLC–ES/MS/MS. J. Chromatogr. B 2005, 825, 134–143. [Google Scholar] [CrossRef]
- Cavaliere, C.; Foglia, P.; Gubbiotti, R.; Sacchetti, P.; Samperi, R.; Lagana, A. Rapid-resolution liquid chromatography/mass spectrometry for determination and quantitation of polyphenols in grape berries. Rapid Commun. Mass Spectrom. 2008, 22, 3089–3099. [Google Scholar] [CrossRef]
- Roggero, J.-P.; Archier, P.; Coen, S. Chromatography of Phenolics in Wine. In Wine: Nutritional and Therapeutic Benefits; American Chemical Society: Washington, DC, USA, 1997; pp. 6–11. [Google Scholar]
- de Villiers, A.; Kalili, K.M.; Malan, M.; Roodman, J. Improving HPLC separation of polyphenols. LCGC Eur. 2010, 23, 466–478. [Google Scholar]
- Cicchetti, E.; Chaintreau, A. Comparison of extraction techniques and modeling of accelerated solvent extraction for the authentication of natural vanilla flavors. J. Sep. Sci. 2009, 32, 3043–3052. [Google Scholar] [CrossRef]
- Selvaggini, R.; Servili, M.; Urbani, S.; Esposto, S.; Taticchi, A.; Montedoro, G. Evaluation of phenolic compounds in virgin olive oil by direct injection in high-performance liquid chromatography with fluorometric detection. J. Agric. Food Chem. 2006, 54, 2832–2838. [Google Scholar] [CrossRef]
- Kraushofer, T.; Sontag, G. Determination of some phenolic compounds in flax seed and nettle roots by HPLC with coulometric electrode array detection. Eur. Food Res. Technol. 2002, 215, 529–533. [Google Scholar] [CrossRef]
- de Pascual-Teresa, S.; Treutter, D.; Rivas-Gonzalo, J.C.; Santos-Buelga, C. Analysis of flavanols in beverages by high-performance liquid chromatography with chemical reactionm detection. J. Agric. Food Chem. 1998, 46, 4209–4213. [Google Scholar]
- Oh, Y.S.; Lee, J.H.; Yoon, S.H.; Oh, C.H.; Choi, D.S.; Choe, E.; Jung, M.Y. Characterization and quantification of anthocyanins in grape juices obtained from the grapes cultivated in Korea by HPLC/DAD, HPLC/MS, and HPLC/MS/MS. J. Food Sci. 2008, 73, 378–389. [Google Scholar]
- Moze, S.; Polak, T.; Gasperlin, L.; Koron, D.; Vanzo, A.; Poklar Ulrih, N.; Abram, V. Phenolics in Slovenian bilberries (Vaccinium myrtillus L.) and blueberries (Vaccinium corymbosum L.). J. Agric. Food Chem. 2011, 59, 6998–7004. [Google Scholar]
- do Amaral, F.P.; Napolitano, A.; Masullo, M.; dos Santos, L.C.; Festa, M.; Vilegas, W.; Pizza, C.; Piacente, S. HPLC-ESIMSn profiling, isolation, structural elucidation, and evaluation of the antioxidant potential of phenolics from Paepalanthus geniculatus. J. Nat. Prod. 2012, 75, 547–556. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, J.; Wang, Y.; Wu, D.; Xu, M. Phenolic extracts from Acacia mangium bark and their antioxidant activities. Molecules 2010, 15, 3567–3577. [Google Scholar] [CrossRef]
- Wei, S.-D.; Zhou, H.-C.; Lin, Y.-M.; Liao, M.-M.; Chai, W.-M. MALDI-TOF MS analysis of condensed tannins with potent antioxidant activity from the leaf, stem bark and root bark of Acacia confuse. Molecules 2010, 15, 4369–4381. [Google Scholar] [CrossRef]
- Edenharder, R.; Keller, G.; Platt, K.L.; Unger, K.K. Isolation and characterization of structurally novel antimutagenic flavonoids from spinach (Spinacia oleracea). J. Agric. Food Chem. 2001, 49, 2767–2773. [Google Scholar] [CrossRef]
- Bianco, A.; Buiarclli, F.; Cartoni, G.; Coccioli, F.; Muzzalupo, I.; Polidor, A.; Uccella, N. Analysis by HPLC-MS-MS of Biophenolics in olives and oils. Anal. Lett. 2001, 34, 1033–1051. [Google Scholar] [CrossRef]
- Chinnici, F.; Natall, N.; Spinabelli, U.; Riponi, C. Presence of polycyclic aromatic hydrocarbons in woody chips used as adjuvant in wines, vinegars and distillates. LWT-Food Sci. Technol. 2007, 40, 1587–1592. [Google Scholar] [CrossRef]
- Muanda, F.N.; Soulimani, R.; Dicko, A. Study on biological activities and chemical composition of extracts from Desmodium adscendens leaves. J. Nat. Prod. 2011, 4, 100–107. [Google Scholar]
- Pawlowska, A.M.; Oleszek, W.; Braca, A. Quali-quantitative analyses of flavonoids of Morus nigra L. and Morus alba L. (Moraceae) fruits. J. Agric. Food Chem. 2008, 56, 3377–3380. [Google Scholar] [CrossRef]
- Jandera, P. Stationary phases for hydrophilic interaction chromatography, their characterization and implementation into multidimensional chromatography concepts. J. Sep Sci. 2008, 31, 1421–1437. [Google Scholar] [CrossRef]
- Zeng, J.; Zhang, X.; Guo, Z.; Feng, J.; Zeng, J.; Xue, X.; Liang, X. Separation and identification of flavonoids from complex samples using off-line two-dimensional liquid chromatography tandem mass spectrometry. J. Chromatogr. A 2012, 1220, 50–56. [Google Scholar] [CrossRef]
- Liang, Z.; Li, K.; Wang, X.; Ke, Y.; Jin, Y.; Liang, X. Combination of off-line two-dimensional hydrophilic interaction liquid chromatography for polar fraction and two-dimensional hydrophilic interaction liquid chromatographyxreversed-phase liquid chromatography for medium-polar fraction in a traditional Chinese medicine. J. Chromatogr. A 2012, 1224, 61–69. [Google Scholar] [CrossRef]
- Roberts, E.A.H.; Wood, D.J. A study of the polyphenols in tea leaf by paper chromatography. Biochem. J. 1951, 49, 414–422. [Google Scholar]
- Nambiar, V.S.; Daniel, M.; Guin, P. Characterization of polyphenols from Coriander leaves (Coriandrum sativum), red Amaranthus (A. paniculatus) and green Amaranthus (A. frumetaceus) using paper chromatography: and their health implications. J. Herb Medic. Toxicol. 2010, 4, 173–177. [Google Scholar]
- Sajewicz, M.; Staszek, D.; Waksmundzka-Hajnos, M.; Kowalska, T. Comparison of TLC and HPLC fingerprints of phenolic acids and flavonoids fractions derived frome selected Sage (Salvia) species. J. Liq. Chrom. Rel. Technol. 2012, 35, 1388–1403. [Google Scholar]
- de Oliveira, C.B.; Comunello, L.N.; Lunardelli, A.; Amaral, R.H.; Pires, M.G.S.; da Silva, G.L.; Manfredini, V.; Vargas, C.R.; Gnoatto, S.C.B.; de Oliveira, J.R.; et al. Phenolic enriched extract of Baccharis trimera presents anti-inflammatory and antioxidant activities. Molecules 2012, 17, 1113–1123. [Google Scholar]
- Cao, X.; Wang, C.; Pei, H.; Sun, B. Separation and identification of polyphenols in apple pomace by high-speed counter-current chromatography and high-performance liquid chromatography coupled with mass spectrometry. J. Chromatogr. A 2009, 1216, 4268–4274. [Google Scholar] [CrossRef]
- Yang, Y.; Gu, D.; Wu, H.; Aisa, H.; Zhang, T.; Ito, Y. Application of preparative high-speed countercurrent chromatography for separation of elatine from Delphinium shawurense. J. Liq. Chrom. Rel. Technol. 2008, 31, 3012–3019. [Google Scholar] [CrossRef]
- Ito, Y. Golden rules and pitfalls in selecting optimum conditions for high-speed counter current chromatography. J. Chromatogr. A 2005, 1065, 145–168. [Google Scholar] [CrossRef]
- Lu, H.-T.; Jiang, Y.; Chen, F. Application of preparative high-speed counter-current chromatography for separation of chlorogenic acid from Flos lonicerae. J. Chromatogr. A 2004, 1026, 185–190. [Google Scholar] [CrossRef]
- Yanagida, A.; Shoji, A.; Shibusawa, Y.; Shindo, H.; Tagashira, M.; Ikeda, M.; Ito, Y. Analytical separation of tea catechins and food-related polyphenols by high-speed counter-current chromatography. J. Chromatogr. A 2006, 1112, 195–201. [Google Scholar] [CrossRef]
- Wang, J.; Gao, H.; Zhao, J.; Wang, Q.; Zhou, L.; Han, J.; Yu, Z.; Yang, F. Preparative separation of phenolic compounds from Halimodendron halodendron by high-speed counter-current chromatography. Molecules 2010, 15, 5998–6007. [Google Scholar] [CrossRef]
- He, D.; Gu, D.; Huang, Y.; Ayupbek, A.; Yang, Y.; Aisa, H.A.; Ito, Y. Separation and purification of phenolic acids and myricetin from black currant by high speed countercurrent chromatography. J. Liq. Chrom. Rel. Technol. 2009, 32, 3077–3088. [Google Scholar] [CrossRef]
- Caridi, D.; Trenerry, V.C.; Rochfort, S.; Duong, S.; Laugher, D.; Jones, R. Profiling and quantifying quercetin glucosides in onion (Allium cepa L.) varieties using capillary zone electrophoresis and high performance liquid chromatography. Food Chem. 2007, 105, 691–699. [Google Scholar] [CrossRef]
- Wang, S.C.; Zhang, Z.; He, L.C.; Li, H. Non-aqueous capillary electrophoresis for separation and simultaneous determination of magnoflorine, taspine, and caulophine in Caulophyllum robustum collected in different seasons. Anal. Lett. 2010, 43, 1534–1542. [Google Scholar] [CrossRef]
- Liu, E.-H.; Qi, L.-W.; Cao, J.; Li, P.; Li, C.-Y.; Peng, Y.-B. Review: Advances of modern chromatographic and electrophoretic methods in separation and analysis of flavonoids. Molecules 2008, 13, 2521–2544. [Google Scholar] [CrossRef]
- Fonseca, F.N.; Tavares, M.F.M.; C, H. Capillary electrochromatography of selected phenolic compounds of Chamomilla recutita. J. Chromatogr. A 2007, 1154, 390–399. [Google Scholar] [CrossRef]
- Chen, X.J.; Ji, H.; Wang, Y.T.; Li, S.P. Simultaneous determination of seven flavonoids in Epimedium using pressurized liquid extraction and capillary electrochromatography. J. Sep. Sci. 2008, 31, 881–887. [Google Scholar] [CrossRef]
- Cheung, R.H.F.; Marriott, P.J.; Small, D.M. CE methods applied to the analysis of micronutrient in foods. Electrophoresis 2007, 28, 3390–3413. [Google Scholar] [CrossRef]
- Rybarczyk, A.; Pegg, R.B.; Amarowic, R.Z. Capillary zone electrophoresis (CZE) of flaxseed phenolic constituent- A short report. Pol. J. Food Nutr. Sci. 2008, 58, 263–267. [Google Scholar]
- Wu, T.; Quan, Y.Q.; Ye, J.N. Determination of flavonoids and ascorbic acid in grapefruit peel and juice by capillary electrophoresis with electrochemical detection. Food Chem. 2007, 100, 1573–1579. [Google Scholar] [CrossRef]
- Zhang, S.; Dong, S.Q.; Chi, L.Z. Simultaneous determination of flavonoids in chrysanthemum by capillary zone electrophoresis with running buffer modifiers. Talanta 2008, 76, 780–784. [Google Scholar] [CrossRef]
- Yuan, G.; Liu, Y.; Li, T.; Wang, Y.; Sheng, Y.; Ming, G. Simultaneous and rapid determination of main lignans in different parts of Schisandra sphenanthera by micellar electrokinetic capillary chromatography. Molecules 2011, 16, 3713–3722. [Google Scholar] [CrossRef]
- Verardo, V.; Gomez-Caravaca, A.M.; Segura-Carretero, A.; Caboni, M.F.; Fernandez-Gutierrez, A. Development of a CE-ESI-microTOF-MS method for a rapid identification of phenolic compounds in buckwheat. Electrophoresis 2011, 32, 669–673. [Google Scholar] [CrossRef]
- Chester, T.L.; Pinkston, J.D.; Raynie, D.E. Supercritical fluid chromatography and extraction. Anal. Chem. 1994, 66, 106–130. [Google Scholar] [CrossRef]
- Miller, J.M. Chromatography: Concepts and Contrasts, 2nd ed; John Wiley and Sons.: Hoboken, NJ, USA, 2004. [Google Scholar]
- Bamba, T. Application of supercritical fluid chromatography to the analysis of hydrophobic metabolites. J. Sep. Sci. 2008, 31, 1274–1278. [Google Scholar] [CrossRef]
- Kamangerpour, A.; Ashraf-Khorassani, M.; Taylor, L.T.; McNair, H.M.; Chorida, L. Supercritical fluid chromatography of polyphenolic compounds in grape seed extract. Chromatographia 2002, 55, 417–421. [Google Scholar] [CrossRef]
- Liu, Z.; Zhao, S.; Wang, R. a.; Guanghua, Y. Separation of polyhydroxylflavonoids by packed-column supercritical fluid chromatography. J. Chromatogr. Sci. 1999, 37, 155–158. [Google Scholar]
- Sun, S.Y.; Jiang, W.G.; Zhao, Y.P. Comparison of aromatic and phenolic compounds in cherry wines with different cherry cultivars by HS-SPME-GC-MS and HPLC. Int. J. Food Sci. Technol. 2012, 47, 100–106. [Google Scholar] [CrossRef]
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Khoddami, A.; Wilkes, M.A.; Roberts, T.H. Techniques for Analysis of Plant Phenolic Compounds. Molecules 2013, 18, 2328-2375. https://doi.org/10.3390/molecules18022328
Khoddami A, Wilkes MA, Roberts TH. Techniques for Analysis of Plant Phenolic Compounds. Molecules. 2013; 18(2):2328-2375. https://doi.org/10.3390/molecules18022328
Chicago/Turabian StyleKhoddami, Ali, Meredith A. Wilkes, and Thomas H. Roberts. 2013. "Techniques for Analysis of Plant Phenolic Compounds" Molecules 18, no. 2: 2328-2375. https://doi.org/10.3390/molecules18022328
APA StyleKhoddami, A., Wilkes, M. A., & Roberts, T. H. (2013). Techniques for Analysis of Plant Phenolic Compounds. Molecules, 18(2), 2328-2375. https://doi.org/10.3390/molecules18022328






