Silica-Supported Polyphosphoric Acid in the Synthesis of 4-Substituted Tetrahydroisoquinoline Derivatives
Abstract
:1. Introduction
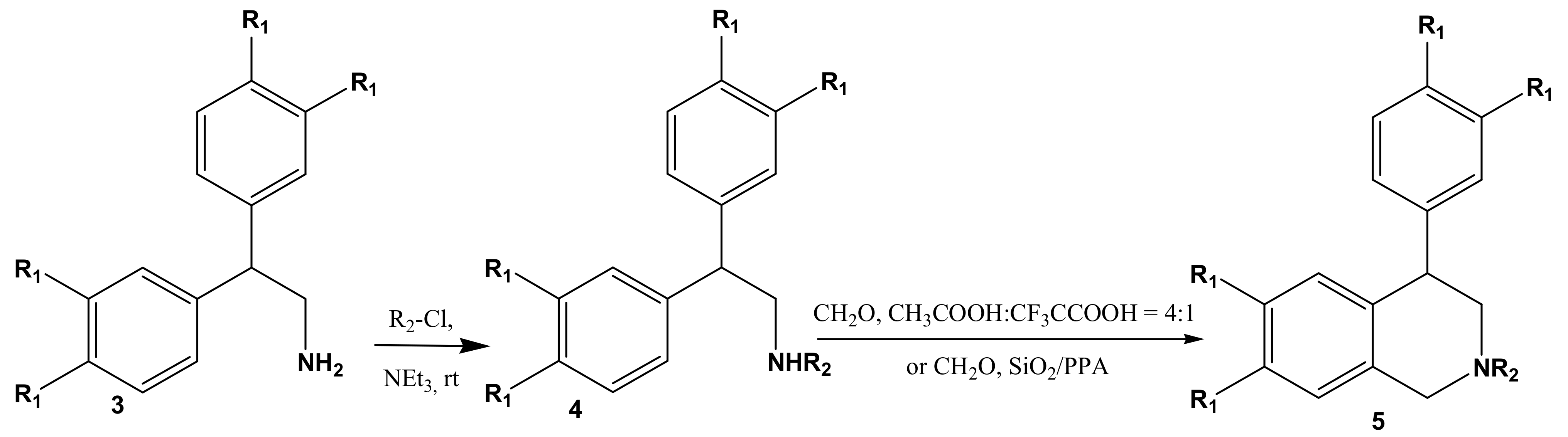
2. Results and Discussion
3. Experimental
3.1. General
3.2. Preparation of PPA/SiO2 Catalyst General Procedure
3.3. Typical Procedure for the Synthesis of 2-Amino-1,1-diphenylethanes 3
3.4. 2,2-Diphenylethanamine (3a): Known Compound [53]
3.5. Acylation of Amines 3: Typical Procedure for the Synthesis of Amides 4 and 6
3.6. Cyclization of Amides 4 and 6 in Acetic/Trifluoracetic Acid Milieu: Typical Procedure
3.7. Cyclization of Amides 4 and 6 in SiO2-Supported Milieu: Typical Procedure
4. Conclusions
Acknowledgments
References
- Brossi, A.; Grethe, G.; Tietel, S.; Wildman, W.C.; Bailey, D.T. Cherylline, a 4-phenyl-1,2,3,4-tetrahydroisoquinoline alkaloid. J. Org. Chem. 1970, 35, 1100–1104. [Google Scholar] [CrossRef]
- Jacob, J.N.; Nichols, D.E.; Kohli, J.D.; Glock, D. Dopamine agonist properties of N-alkyl-4-(3,4-dihydroxyphenyl)-1,2,3,4-tetrahydroisoquinolines. J. Med. Chem. 1981, 24, 1013–1015. [Google Scholar] [CrossRef] [PubMed]
- Zara-Kaczian, E.; Gyorgy, L.; Deak, G.; Seregi, A.; Doda, M. Synthesis and pharmacological evaluation of some new tetrahydroisoquinoline derivatives inhibiting dopamine uptake and/or possessing a dopaminomimetic property. J. Med. Chem. 1986, 29, 1189–1195. [Google Scholar] [CrossRef] [PubMed]
- Maryanoff, B.E.; Vaught, J.L.; Shank, R.P.; McComsey, D.F.; Costanzo, M.J.; Nortey, S.O. Pyrroloisoquinoline antidepressants. 3. A focus on serotonin. J. Med. Chem. 1990, 33, 2793–2797. [Google Scholar] [CrossRef] [PubMed]
- Kihara, M.; Ikeuchi, M.; Yamauchi, A.; Nukatsuka, M.; Matsumoto, H.; Toko, T. Synthesis and biological evaluation of 7-hydroxy-3,4-diphenyl-1,2-dihydroisoquinolines as new 4-hydroxytamoxifen analogues. Chem. Pharm. Bull. 1997, 45, 939–943. [Google Scholar] [CrossRef] [PubMed]
- Venkov, A.P.; Vodenicharov, D. A New Synthesis of 1,2,3,4-Tetrahydro-2-methyl-4-phenylisoquinolines. Synthesis 1990, 253–255. [Google Scholar] [CrossRef]
- Kihara, M.; Kashimoto, M.; Kobayashi, Y.; Kobayashi, S. A new intramolecular barbier reaction of N-(2-iodobenzyl)phenacylamines: A convenient synthesis of 1,2,3,4-tetrahydroisoquinomn-4-ols. Tetrahedron Lett. 1990, 31, 5347–5348. [Google Scholar] [CrossRef]
- Venkov, A.P.; Vodenicharov, D.M.; Ivanov, I.I. 4-Aryl-1,4-dihydro-3(2H)-isoquinolinones are obtained by oxidative cyclization of N-benzyl-N-alkylarylacetamides with lead tetra-acetate in acetic acid/trifluoroacetic acid. Synthesis 1991, 476–478. [Google Scholar] [CrossRef]
- Kihara, M.; Kashimoto, M.; Kobayashi, Y.; Nagao, Y. A new synthesis of 7,12-Dihydro-12-phenyl-5H-6,12-methanodibenz[c,f]azocines via N-Benzyl-1,2,3,4-tetrahydro-4-phenylisoquinolin-4-ols. Heterocycles 1992, 34, 747–756. [Google Scholar] [CrossRef]
- Coskun, N.; Sümengen, D. A new synthesis of N-Substituted-1,2,3,4-tetrahydro-4-phenylisoquinolin-3-ones. Synth. Commun. 1993, 23, 1393–1402. [Google Scholar] [CrossRef]
- Miller, R.B.; Svoboda, J.J. An efficient synthesis of 4-aryl-1,2,3,4-tetrahydroisoquinolines. Synth. Commun. 1994, 24, 1187–1194. [Google Scholar] [CrossRef]
- Meda, N.; Selvakumar, N.; Kraus, G.A. An efficient synthesis of 4-aryl kainic acid analogs. Tetrahedron 1999, 55, 943–954. [Google Scholar] [CrossRef]
- Kuster, G.J.; Kalmoua, F.; Scheeren, H.W.; de Gelder, R. A simple entry towards novel bi- and tricyclic N-oxy-β-lactams by high pressure promoted tandem [4+2]/[3+2] cycloadditions of enol ethers and β-nitrostyrene. Chem. Commun. 1999, 855–856. [Google Scholar] [CrossRef]
- Nolellino, L.; D’Ischia, M.; Prota, G. Expedient synthesis of 5,6-dihydroxyindole and derivatives via an improved Zn(II)-assisted 2,β-dinitrostyrene approach. Synthesis 1999, 793–796. [Google Scholar] [CrossRef]
- Kihara, M.; Andoh, J.I.; Yoshida, C. New reduction reaction of benzylic alcohols with acid and proof of the intermolecular hydride shift mechanism. Heterocycles 2000, 53, 359–372. [Google Scholar] [CrossRef]
- Labrana, J.; Machocho, A.K.; Kricsfalusy, V.; Brun, R.; Codina, C.; Viladomat, F.; Bastida, J. Alkaloids from Narcissus angustifolius subsp. transcarpathicus (Amaryllidaceae). Phytochemistry 2002, 60, 847–856. [Google Scholar] [CrossRef]
- Elgorashi, E.E.; Stafford, G.J.; Jäger, A.K.; van Staden, J. Inhibition of [3H] citalopram binding to the rat brain serotonin transporter by amaryllidaceae alkaloids. Planta Med. 2006, 72, 470–473. [Google Scholar] [CrossRef] [PubMed]
- Dong, H.; Sheng, J.Z.; Lee, C.M.; Wong, T.M. Calcium antagonistic and antiarrhythmic actions of CPU-23, a substituted tetrahydroisoquinoline. Br. J. Pharmacol. 1993, 109, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Bernan, V.S.; Montenegro, D.A.; Korshalla, J.D.; Maiese, W.M.; Steinberg, D.A.; Greenstein, M. Bioxalomycins, new antibiotics produced by the marine Streptomyces sp. LL-31F508: Toxonomy and fermentation. J. Antibiot. 1994, 47, 1417–1424. [Google Scholar] [CrossRef] [PubMed]
- Ratsimamanga-Urverg, S.; Rasoanaivo, P.; Rafatro, H.; Robijaona, B.; Rakoto-Ratsimamanga, A. In vitro antiplasmodial activity and chloroquine-potentiating action of three new isoquinoline alkaloid dimers isolated from Hernandia voyronii Jumelle. Ann. Trop. Med. Parasitol. 1994, 88, 271–277. [Google Scholar] [CrossRef] [PubMed]
- Riggs, R.M.; Nichols, D.E.; Foreman, M.M.; Truex, L.L. Effect of β-alkyl substitution on D-1 dopamine agonist activity: Absolute configuration of β-methyldopamine. J. Med. Chem. 1987, 30, 1887–1897. [Google Scholar] [CrossRef] [PubMed]
- Chesworth, R.; Zawistoski, M.P.; Lefker, B.A.; Cameron, K.O.; Day, R.F.; Mangano, F.M.; Rosati, R.L.; Colella, S.; Petersen, D.N.; Brault, A.; et al. Tetrahydroisoquinolines as subtype selective estrogen agonists/antagonists. Bioorg. Med. Chem. Lett. 2004, 14, 2729–2733. [Google Scholar] [CrossRef] [PubMed]
- Keith, J.M.; Barbier, A.J.; Wilson, S.J.; Miller, K.; Boggs, J.D.; Fraser, I.C.; Mazur, C.; Lovenberg, T.W.; Carruthers, N.I. Dual serotonin transporter inhibitor/histamine H3 antagonists: Development of rigidified H3 pharmacophores. Bioorg. Med. Chem. Lett. 2007, 17, 5325–5329. [Google Scholar] [CrossRef] [PubMed]
- Brossi, A.; Teitel, S. Total synthesis of racemic cherylline. Tetrahedron Lett. 1970, 11, 417–419. [Google Scholar] [CrossRef]
- Ruchirawat, S.; Tontoolarug, S.; Sahakitpichan, P. Synthesis of 4-aryltetrahydroiso-quinolines: Application to the synthesis of cherylline. Heterocycles 2001, 55, 635–640. [Google Scholar] [CrossRef]
- Takano, S.; Akiyama, M.; Ogasawara, K. Total synthesis of (±)- and (+)-latifine. J. Chem. Soc. Perkin Trans. 1 1985, 2447–2453. [Google Scholar] [CrossRef]
- Couture, A.; Deniau, E.; Lebrun, S.; Grandclaudon, P. Total syntheses of (+/−)-cherylline and (+/−)-latifine. J. Chem. Soc. Perkin Trans. 1 1999, 789–794. [Google Scholar] [CrossRef]
- Cuevas, J.-C.; Snieckus, V. α′-Silylated tertiary benzamides as dual ortho- and α′-carbanion synthons. Carbodesilylative routes to isoquinoline and dibenzoquinolizidine derivatives. Tetrahedron Lett. 1989, 30, 5837–5840. [Google Scholar] [CrossRef]
- Philippe, N.; Denivet, F.; Vasse, J.-L.; Sopkova-de Olivera Santos, J.; Levacher, V.; Dupas, G. Highly stereoselective Friedel–Crafts type cyclization. Facile access to enantiopure 1,4-dihydro-4-phenyl isoquinolinones. Tetrahedron 2003, 59, 8049–8056. [Google Scholar] [CrossRef]
- Hara, H.; Shirai, R.; Hoshino, O.; Umezawa, B. A Facile Synthesis of 4-aryl-1,2,3,4-tetrahydroisoquinolines: A total synthesis of (±)-cherylline. Heterocycles 1983, 20, 1945–1950. [Google Scholar]
- Toda, J.; Sonobe, A.; Ichikava, T.; Saitoh, T.; Horiguchi, Y.; Sano, T. Synthesis of 4-aryl-2-methyl-1,2,3,4-tetrahydroisoquinolines via Pummerer-type cyclization of N-(arylmethyl)-N-methyl-2-aryl-2-(phenylsulfinyl) acetamides. Arcivoc 2000, 1, 165–180. [Google Scholar]
- Hara, H.; Shirai, R.; Hoshino, O.; Umezawa, B. Studies on tetrahydroisoquinolines. XXV. A synthesis of 4-aryl-1,2,3,4-tetrahydroisoquinolines; Total synthesis of (±)-cherylline. Chem. Pharm. Bull 1985, 33, 3107–3112. [Google Scholar] [CrossRef]
- Couture, A.; Deniau, E.; Grandclaudon, P.; Lebrun, S. Asymmetric synthesis of (+)- and (−)-latifine. Asymmetry 2003, 14, 1309–1316. [Google Scholar] [CrossRef]
- Seijas, J.A.; Vázquer-Tato, M.P.; Martínez, M.M.; Pizzolatti, M.G. Oxazoline as a useful tool in organic synthesis: Preparation of 4-aryl-1,2,3,4-tetrahydroisoquinoline alkaloid skeleton. Tetrahedron Lett. 2005, 46, 5827–5830. [Google Scholar] [CrossRef]
- Irie, H.; Shiina, A.; Fushimi, T.; Katakawa, J.; Fujii, N.; Yajima, H. New synthesis of isoquinoline alkaloids, thalifoline, corypalline, and cherylline. Chem. Lett. 1980, 875–878. [Google Scholar] [CrossRef]
- Katakawa, J.; Yoshimatsu, H.; Yoshida, M.; Zhang, Y.; Irie, H.; Yajima, H. Synthesis of Amaryllidaceae Alkaloids, (±)-Cherylline and (±)-Latifine. Chem. Pharm. Bull. 1988, 36, 3928–3932. [Google Scholar] [CrossRef]
- Kessar, V.; Singh, P.; Chawla, R.; Kumar, P. Cyclization of ortho-halogenated N-acylbenzylamines: A formal synthesis of (±)-cherylline. Chem. Soc. Chem. Commun. 1981, 1074–1075. [Google Scholar] [CrossRef]
- Schwartz, M.A.; Scott, S.W. Biogenetically patterned synthesis of (+−)-cherylline. J. Org. Chem. 1971, 36, 1827–1829. [Google Scholar] [CrossRef]
- Kametani, T.; Takahashi, K.; Van Loc, C. Studies on the syntheses of heterocyclic compounds—DXCI: Total synthesis of (±)-cherylline and corgoine through quinonoid intermediates. Tetrahedron 1975, 31, 235–238. [Google Scholar] [CrossRef]
- Honda, T.; Namiki, H.; Saton, F. Palladium-catalyzed intramolecular δ-lactam formation of aryl halides and amide-enolates: Syntheses of cherylline and latifine. Org. Lett. 2001, 3, 631–633. [Google Scholar] [CrossRef] [PubMed]
- Freter, K.; Dubois, E.; Thomas, A. A new tetrahydroisoquinoline synthesis. J. Heterocycl. Chem. 1970, 7, 159–169. [Google Scholar] [CrossRef]
- Lebrun, S.; Couture, A.; Deniau, E.; Grandclaudon, P. A new synthesis of (+)- and (−)-cherylline. Org. Biomol. Chem. 2003, 1, 1701–1706. [Google Scholar] [CrossRef] [PubMed]
- Bonacorso, H.G.; Andrighetto, R.; Zanatta, N.; Martins, M.A.P. The unexpected cyclization routes of N,N′-bis(oxotrifluoroalkenyl)-1,3-phenylenediamines in polyphosphoric acid medium. Tetraheron Lett. 2010, 51, 3752–3755. [Google Scholar] [CrossRef]
- Ivanov, I.; Nikolova, S.; Statkova-Abeghe, S. Efficient one-pot Friedel-Crafts acylation of benzene and its derivatives with unprotected aminocarboxylic acids in polyphosphoric acid. Synth. Commun. 2006, 36, 1405–1411. [Google Scholar] [CrossRef]
- Ivanov, I.; Nikolova, S.; Statkova-Abeghe, S. A simple method for the synthesis of 1-substituted beta-carboline derivatives from tryptamine and carboxylic acids in polyphosphoric acid. Heterocycles 2005, 65, 2483–2492. [Google Scholar] [CrossRef]
- Aoyama, T.; Takido, T.; Kodomari, M. Silica gel-supported polyphosphoric acid (PPA/SiO2) as an efficient and reusable catalyst for conversion of carbonyl compounds into oxathioacetals and dithioacetals. Synlett 2004, 13, 2307–2310. [Google Scholar] [CrossRef]
- Itoh, K.-I.; Aoyama, T.; Satoh, H.; Fujii, Y.; Sakamaki, H.; Takido, T.; Kodomari, M. Application of silica gel-supported polyphosphoric acid (PPA/SiO2) as a reusable solid acid catalyst to the synthesis of 3-benzoylisoxazoles and isoxazolines. Tetrahedron Lett. 2011, 52, 6892–6895. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, S.; Deng, Y. Recent advances in ionic liquid catalysis. Green. Chem. 2011, 13, 2619–2637. [Google Scholar] [CrossRef]
- Ansari, M.I.; Hussain, M.K.; Yadav, N.; Gupta, P.K.; Hajela, K. Silica supported perchloric acid catalysed rapid N-formylation under solvent-free conditions. Tetrahedron Lett. 2012, 53, 2063–2065. [Google Scholar] [CrossRef]
- Verma, R.S. Solvent-free organic syntheses using supported reagents and microwave irradiation. Green. Chem. 1999, 1, 43–55. [Google Scholar] [CrossRef]
- Corma, A.; Garcia, H. Silica-bound homogenous catalysts as recoverable and reusable catalysts in organic synthesis. Adv. Synth. Catal. 2006, 348, 1391–1412. [Google Scholar] [CrossRef]
- Kantevari, S.; Bantu, R.; Nagarapu, L. HClO4–SiO2 and PPA–SiO2 catalyzed efficient one-pot Knoevenagel condensation, Michael addition and cyclo-dehydration of dimedone and aldehydes in acetonitrile, aqueous and solvent free conditions: Scope and limitations. J. Mol. Catal. A-Chem. 2007, 269, 53–57. [Google Scholar] [CrossRef]
- Klumpp, D.A.; Sanchez, G.V., Jr.; Aguirre, S.L.; Zhang, Y.; de Leon, S. Chemistry of dicationic electrophiles: Superacid-catalyzed reactions of amino acetals. J. Org. Chem. 2002, 67, 5028–5031. [Google Scholar] [CrossRef] [PubMed]
- Khojastehnezhad, A.; Moeinpour, F.; Davoodnia, A. PPA–SiO2 catalyzed efficient synthesis of polyhydroquinoline derivatives through Hantzsch multicomponent condensation under solvent-free conditions. Chin. Chem. Lett. 2011, 22, 807–810. [Google Scholar] [CrossRef]
- Vittorio, F.; Santagati, N.A.; Duro, R.; Duro, F.; Caruso, A.; Amico Roxas, M.; Trombadore, S. Alkyl derivatives of isoquinoline. III. Synthesis and pharmacologic activity of dialkylaminoalkyl amides of 1-chloro- and 1-methoxy-3-carboxy-4-methylisoquinoline and of 3-carboxy-2,4-dimethyl-1,2-dihydro-1-oxoisoquinoline. Farmaco Sci. 1984, 39, 229–245. [Google Scholar] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
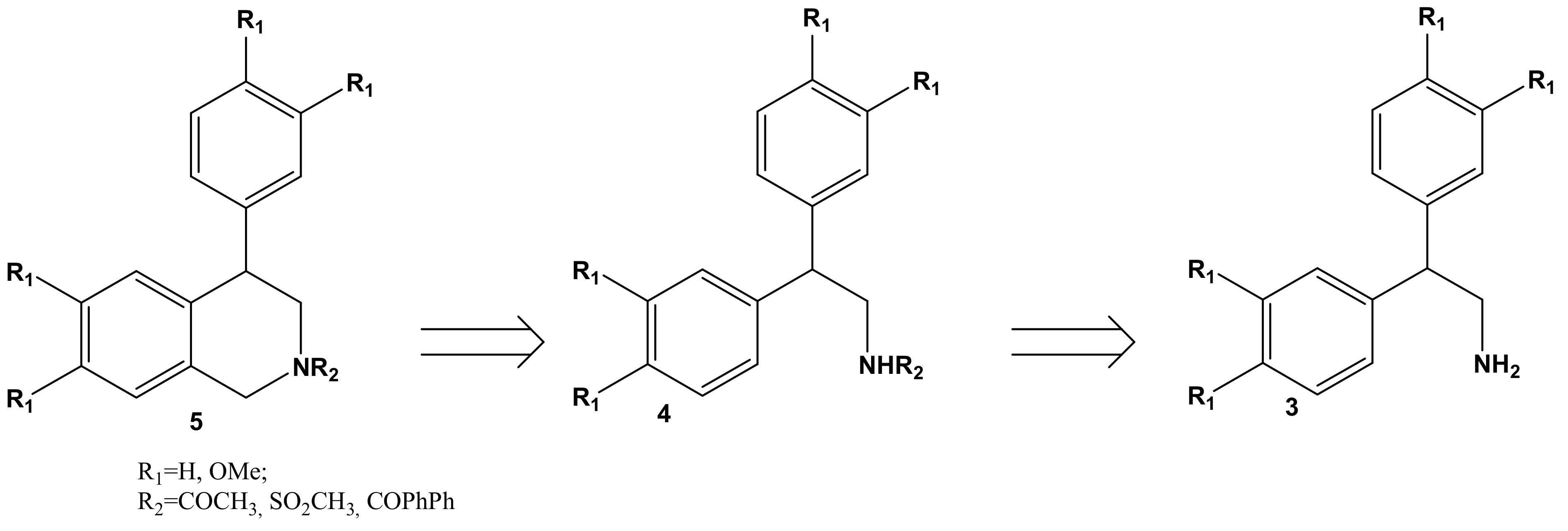
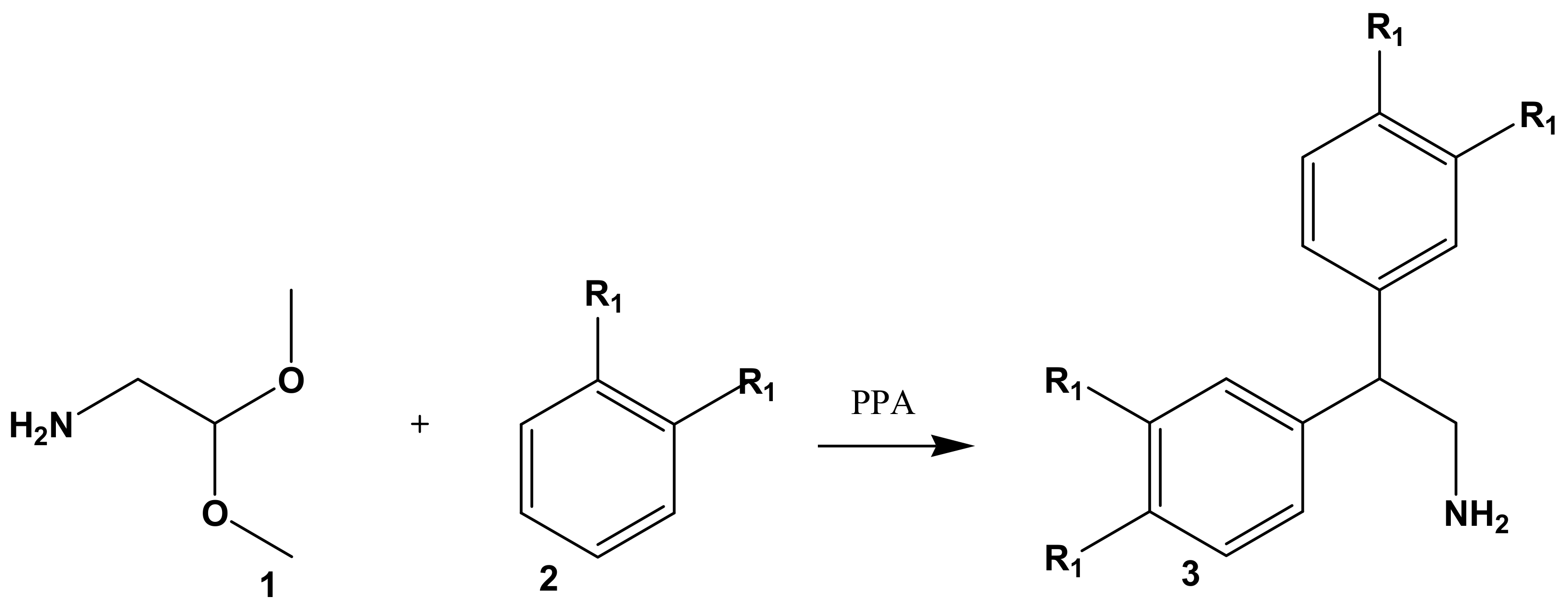
| 3 | R1 | Yield, % | M.p., °C | |
|---|---|---|---|---|
| a | H | H | 42 | 47–49 |
| b | OCH3 | OCH3 | 82 | oil |
| 4 | R | R1 | R2 | Yield, % | M.p., °C |
|---|---|---|---|---|---|
| a | H | H | COCH3 | 80 | 85–86 |
| b | H | H | SO2CH3 | 98 | 138–139 |
| c | H | H | COPhPh | 94 | 192–193 |
| d | OCH3 | OCH3 | COCH3 | 91 | 126–128 |
| e | OCH3 | OCH3 | SO2CH3 | 90 | 49–52 |
| f | OCH3 | OCH3 | COPhPh | 97 | 200–202 |
| 5 | R | R1 | R2 | Yield, % (CH3COOH:CF3COOH = 4:1) | Yield, % (SiO2/PPA) |
|---|---|---|---|---|---|
| a | H | H | COCH3 | 90 | 94 |
| b | H | H | SO2CH3 | 80 | 91 |
| c | H | H | COPhPh | 84 | 96 |
| d | OCH3 | OCH3 | COCH3 | 85 | 96 |
| e | OCH3 | OCH3 | SO2CH3 | 87 | 97 |
| f | OCH3 | OCH3 | COPhPh | 87 | 98 |
| 6 | R2 | Yield, % | M.p., °C |
| a | COCH3 | 98 | Oil |
| b | SO2CH3 | 98 | Oil |
| c | COPhPh | 98 | 156–157 |
| 7 | R2 | Yield, % | m.p., °C |
| a | COCH3 | 89 | Oil |
| b | SO2CH3 | 93 | 66–67 |
| c | COPhPh | 96 | Oil |
© 2013 by the authors. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Manolov, S.; Nikolova, S.; Ivanov, I. Silica-Supported Polyphosphoric Acid in the Synthesis of 4-Substituted Tetrahydroisoquinoline Derivatives. Molecules 2013, 18, 1869-1880. https://doi.org/10.3390/molecules18021869
Manolov S, Nikolova S, Ivanov I. Silica-Supported Polyphosphoric Acid in the Synthesis of 4-Substituted Tetrahydroisoquinoline Derivatives. Molecules. 2013; 18(2):1869-1880. https://doi.org/10.3390/molecules18021869
Chicago/Turabian StyleManolov, Stanimir, Stoyanka Nikolova, and Iliyan Ivanov. 2013. "Silica-Supported Polyphosphoric Acid in the Synthesis of 4-Substituted Tetrahydroisoquinoline Derivatives" Molecules 18, no. 2: 1869-1880. https://doi.org/10.3390/molecules18021869
APA StyleManolov, S., Nikolova, S., & Ivanov, I. (2013). Silica-Supported Polyphosphoric Acid in the Synthesis of 4-Substituted Tetrahydroisoquinoline Derivatives. Molecules, 18(2), 1869-1880. https://doi.org/10.3390/molecules18021869







