Effects of Aronia melanocarpa Constituents on Biofilm Formation of Escherichia coli and Bacillus cereus
Abstract
:1. Introduction
2. Results and Discussion
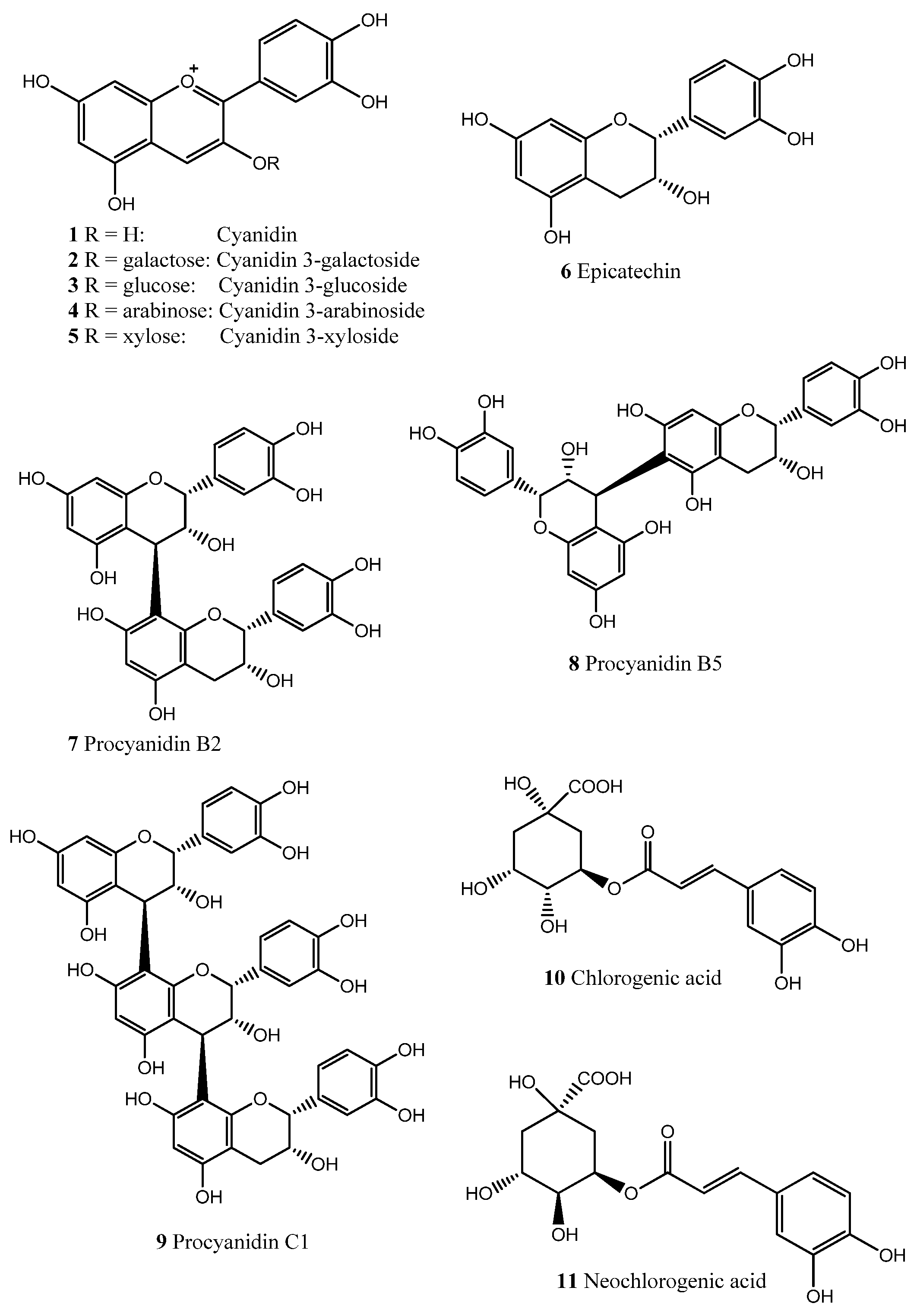
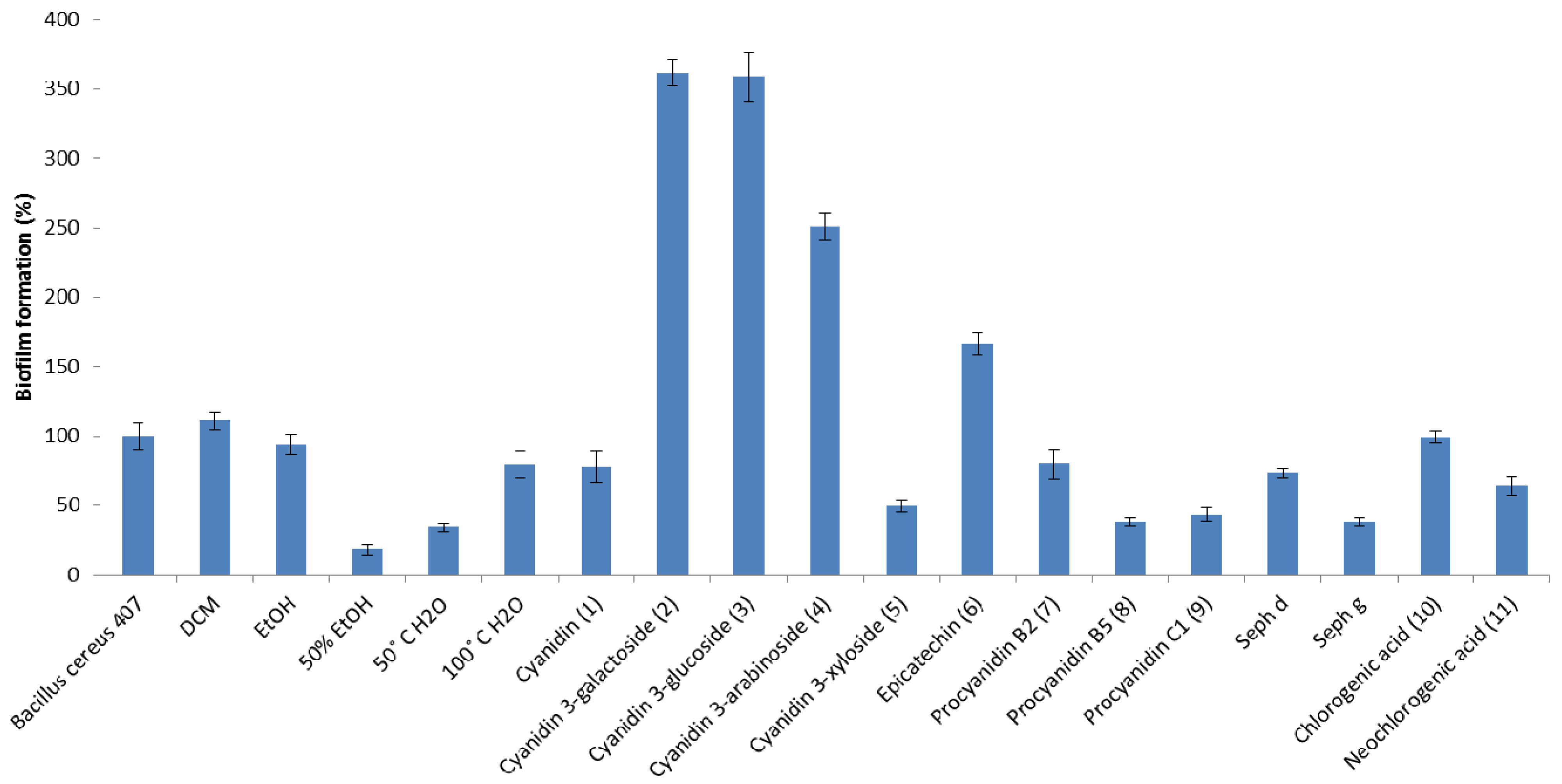
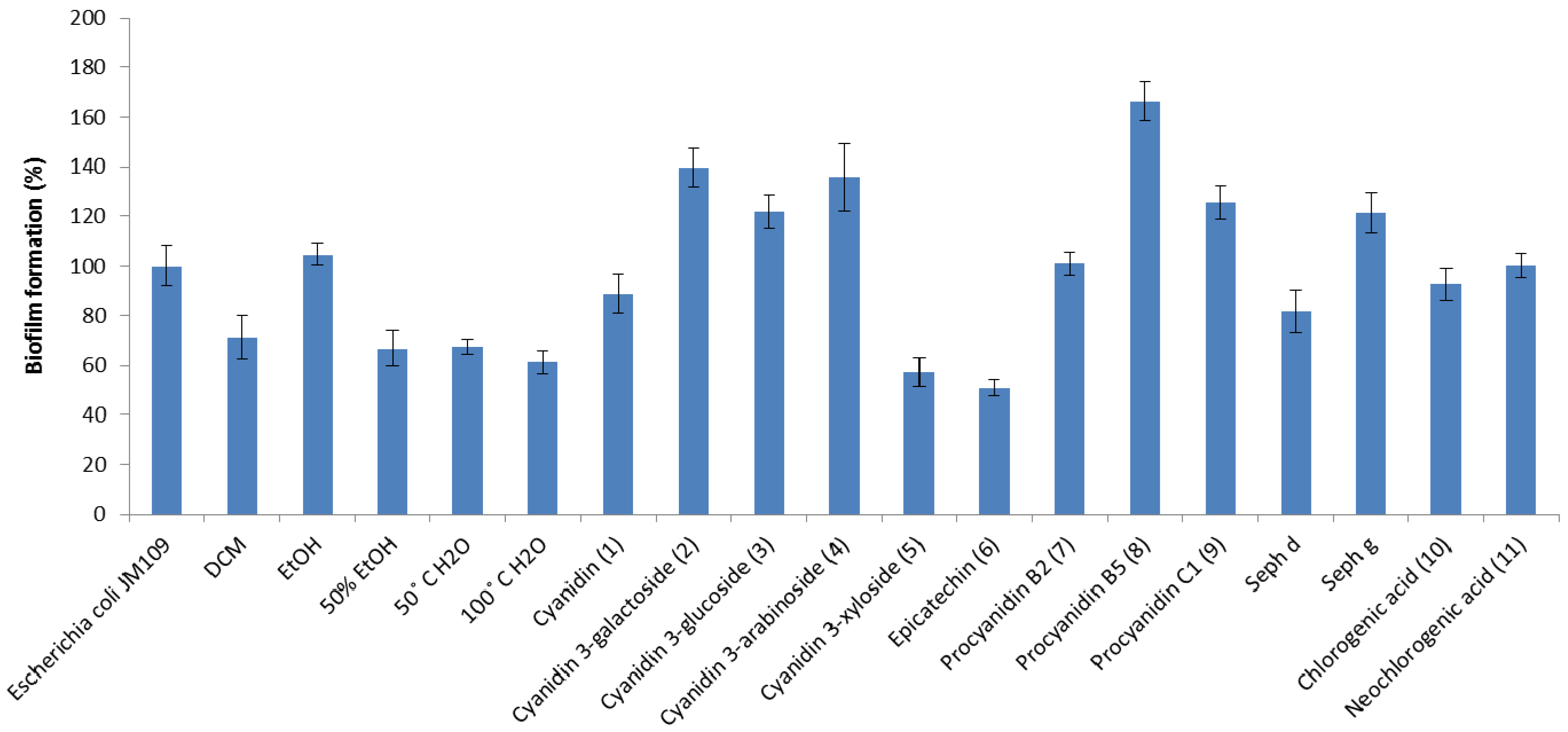
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction, Fractionation and Isolation
3.4. Microtiter Plate Biofilm Formation Assay
3.5. Antimicrobial Disk Susceptibility Test
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Ren, D.; Zuo, R.; González Barrios, A.F.; Bedzyk, L.A.; Eldridge, G.R.; Pasmore, M.E.; Wood, T.K. Differential gene expression for investigation of Escherichia coli biofilm inhibition by plant extract ursolic acid. Appl. Environ. Microbiol. 2005, 71, 4022–4034. [Google Scholar] [CrossRef]
- Carneiro, V.A.; dos Santos, H.S.; Arruda, F.V.S.; Bandeira, P.N.; Albuquerque, M.R.J.R.; Pereira, M.O.; Henriques, M.; Cavada, B.S.; Teixeira, E.H. Casbane diterpene as a promising natural antimicrobial agent against biofilm-associated infections. Molecules 2011, 16, 190–201. [Google Scholar]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef]
- Auger, S.; Krin, E.; Aymerich, S.; Gohar, M. Autoinducer 2 affects biofilm formation by Bacillus cereus. Appl. Environ. Microbiol. 2006, 72, 937–941. [Google Scholar] [CrossRef]
- Peng, J.S.; Tsai, W.C.; Chou, C.C. Inactivation and removal of Bacillus cereus by sanitizer and detergent. Int. J. Food Microbiol. 2002, 77, 11–18. [Google Scholar] [CrossRef]
- Bottone, E.J. Bacillus cereus, a volatile human pathogen. Clin. Microbiol. Rev. 2010, 23, 382–398. [Google Scholar] [CrossRef]
- Nickel, J.C.; Ruseska, I.; Wright, J.B.; Costerton, J.W. Tobramycin resistance of Pseudomonas aeruginosa cells growing as a biofilm on urinary catheter material. Antimicrob. Agents Chemother. 1985, 27, 619–624. [Google Scholar] [CrossRef]
- Potera, C. Forging a link between biofilms and disease. Science 1999, 283, 1837–1839. [Google Scholar] [CrossRef]
- Estrela, A.B.; Abraham, W.R. Combining biofilm-controlling compounds and antibiotics as a promising new way to control biofilm infections. Pharmaceuticals 2010, 3, 1374–1393. [Google Scholar] [CrossRef]
- Hentzer, M.; Riedel, K.; Rasmussen, T.B.; Heydorn, A.; Andersen, J.B.; Parsek, M.R.; Rice, S.A.; Eberl, L.; Molin, S.; Høiby, N.; et al. Inhibition of quorum sensing in Pseudomonas aeruginosa biofilm bacteria by a halogenated furanone compound. Microbiology 2002, 148, 87–102. [Google Scholar]
- Donlan, R.M. Biofilm elimination on intravascular catheters: Important considerations for the infectious disease practitioner. Clin. Infect. Dis. 2011, 52, 1038–1045. [Google Scholar] [CrossRef]
- Kokotkiewicz, A.; Jaremicz, Z.; Luczkiewicz, M. Aronia plants: A review of traditional use, biological activities, and perspectives for modern medicine. J. Med. Food 2010, 13, 255–269. [Google Scholar] [CrossRef]
- Oszmiański, J.; Wojdylo, A. Aronia melanocarpa phenolics and their antioxidant activity. Eur. Food Res. Technol. 2005, 221, 809–813. [Google Scholar] [CrossRef]
- Kulling, S.E.; Rawel, H.M. Chokeberry (Aronia melanocarpa)—A review on the characteristic components and potential health effects. Planta Med. 2008, 74, 1625–1634. [Google Scholar] [CrossRef]
- Slimestad, R.; Torskangerpoll, K.; Nateland, H.S.; Johannesen, T.; Giske, N.H. Flavonoids from black chokeberries, Aronia melanocarpa. J. Food Comp. Anal. 2005, 18, 61–68. [Google Scholar] [CrossRef]
- Valcheva-Kuzmanova, S.V.; Belcheva, A. Current knowledge of Aronia melanocarpa as a medicinal plant. Folia Med. 2006, 48, 11–17. [Google Scholar]
- Bräunlich, M.; Christensen, H.; Johannesen, S.; Slimestad, R.; Wangensteen, H.; Malterud, K.E.; Barsett, H. In vitro inhibition of cytochrome P450 3A4 by Aronia melanocarpa constituents. Planta Med. 2013, 79, 137–141. [Google Scholar]
- Bräunlich, M.; Slimestad, R.; Wangensteen, H.; Brede, C.; Malterud, K.E.; Barsett, H. Extracts, anthocyanins and procyanidins from Aronia melanocarpa as radical scavengers and enzyme inhibitors. Nutrients 2013, 5, 663–678. [Google Scholar] [CrossRef]
- O’Toole, G.A.; Pratt, L.A.; Watnick, P.I.; Newman, D.K.; Weaver, V.B.; Kolter, R. Genetic approaches to study of biofilms. Methods Enzymol. 1999, 310, 91–109. [Google Scholar]
- O’Toole, G.A. Microtiter Dish Biofilm Formation Assay. Available online: http://www.jove.com/details.php?id=2437/ (accessed on 27 November 2013).
- Økstad, O.A. Cyclic Diguanylate (c-di-GMP) Regulates Biofilm Formation in Bacillus cereus. In Presented at the Bacillus ACT 2013—International Conference on Bacillus anthracis, B. cereus, and B. thuringiensis, Victoria, BC, Canada, 4 September 2013.
- Pinzόn-Arango, P.A.; Holguin, K.; Camesano, T.A. Impact of cranberry juice and proanthocyanidins on the ability of Escherichia coli to form biofilms. Food Sci. Biotechnol. 2011, 20, 1315–1321. [Google Scholar] [CrossRef]
- Majtán, J.; Majtánova, L.; Xu, M.; Majtán, V. In vitro effect of subinhibitory concentrations of antibiotics on biofilm formation by clinical strains of Salmonella enterica serovar Typhimurium isolated in Slovakia. J. Appl. Microbiol. 2008, 104, 1294–1301. [Google Scholar] [CrossRef]
- Koo, H.; Duarte, S.; Murata, R.M.; Scott-Anne, K.; Gregoire, S.; Watson, G.E.; Singh, A.P.; Vorsa, N. Influence of cranberry proanthocyanidins on formation of biofilms by Streptococcus mutans on saliva-coated apatitic surface and on dental caries development in vivo. Caries Res. 2010, 44, 116–126. [Google Scholar] [CrossRef]
- Shmuely, H.; Ofek, I.; Weiss, E.I.; Rones, Z.; Huori-Haddad, Y. Cranberry components for the therapy of infectious disease. Curr. Opin. Biotechnol. 2012, 23, 148–152. [Google Scholar] [CrossRef]
- Leshem, R.; Maharshak, I.; Jacob, E.B.; Ofek, I.; Kremer, I. The effect of nondialyzable material (NDM) cranberry extract on formation of contact lens biofilm by Staphylococcus epidermidis. Invest. Ophthalmol. Vis. Sci. 2011, 52, 4929–4934. [Google Scholar] [CrossRef]
- Di Martino, P.; Agniel, R.; Gaillard, J.L.; Denys, P. Effects of cranberry juice on uropathogenic Escherichia coli in vitro biofilm formation. J. Chemother. 2005, 17, 563–565. [Google Scholar]
- Tao, Y.; Pinzόn-Arango, P.A.; Howell, A.B.; Camesano, T.A. Oral consumption of cranberry juice cocktail inhibits molecular-scale adhesion of clinical uropathogenic Escherichia coli. J. Med. Food 2011, 14, 739–745. [Google Scholar] [CrossRef]
- LaPlante, K.L.; Sarkisian, S.A.; Woodmansee, S.; Rowley, D.C.; Seeram, N.P. Effects of cranberry extracts on growth and biofilm production of Escherichia coli and Staphylococcus species. Phytother. Res. 2012, 26, 1371–1374. [Google Scholar] [CrossRef]
- Manner, S.; Skogman, M.; Goeres, D.; Vuorela, P.; Fallarero, A. Systematic exploration of natural and synthetic flavonoids for the inhibition of Staphylococcus aureus biofilms. Int. J. Mol. Sci. 2013, 14, 19434–19451. [Google Scholar] [CrossRef]
- Houry, A.; Briandet, R.; Aymerich, S.; Gohar, M. Involvement of motility and flagella in Bacillus cereus biofilm formation. Microbiology 2010, 156, 1009–1018. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute. Performance Standards for Antimicrobial Disk Susceptibility Tests: Approved Standard, 9th ed.; CLSI: Wayne, PA, USA, 2006. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bräunlich, M.; Økstad, O.A.; Slimestad, R.; Wangensteen, H.; Malterud, K.E.; Barsett, H. Effects of Aronia melanocarpa Constituents on Biofilm Formation of Escherichia coli and Bacillus cereus. Molecules 2013, 18, 14989-14999. https://doi.org/10.3390/molecules181214989
Bräunlich M, Økstad OA, Slimestad R, Wangensteen H, Malterud KE, Barsett H. Effects of Aronia melanocarpa Constituents on Biofilm Formation of Escherichia coli and Bacillus cereus. Molecules. 2013; 18(12):14989-14999. https://doi.org/10.3390/molecules181214989
Chicago/Turabian StyleBräunlich, Marie, Ole A. Økstad, Rune Slimestad, Helle Wangensteen, Karl E. Malterud, and Hilde Barsett. 2013. "Effects of Aronia melanocarpa Constituents on Biofilm Formation of Escherichia coli and Bacillus cereus" Molecules 18, no. 12: 14989-14999. https://doi.org/10.3390/molecules181214989
APA StyleBräunlich, M., Økstad, O. A., Slimestad, R., Wangensteen, H., Malterud, K. E., & Barsett, H. (2013). Effects of Aronia melanocarpa Constituents on Biofilm Formation of Escherichia coli and Bacillus cereus. Molecules, 18(12), 14989-14999. https://doi.org/10.3390/molecules181214989



