Anti- and Pro-Lipase Activity of Selected Medicinal, Herbal and Aquatic Plants, and Structure Elucidation of an Anti-Lipase Compound
Abstract
:1. Introduction
2. Results and Discussion
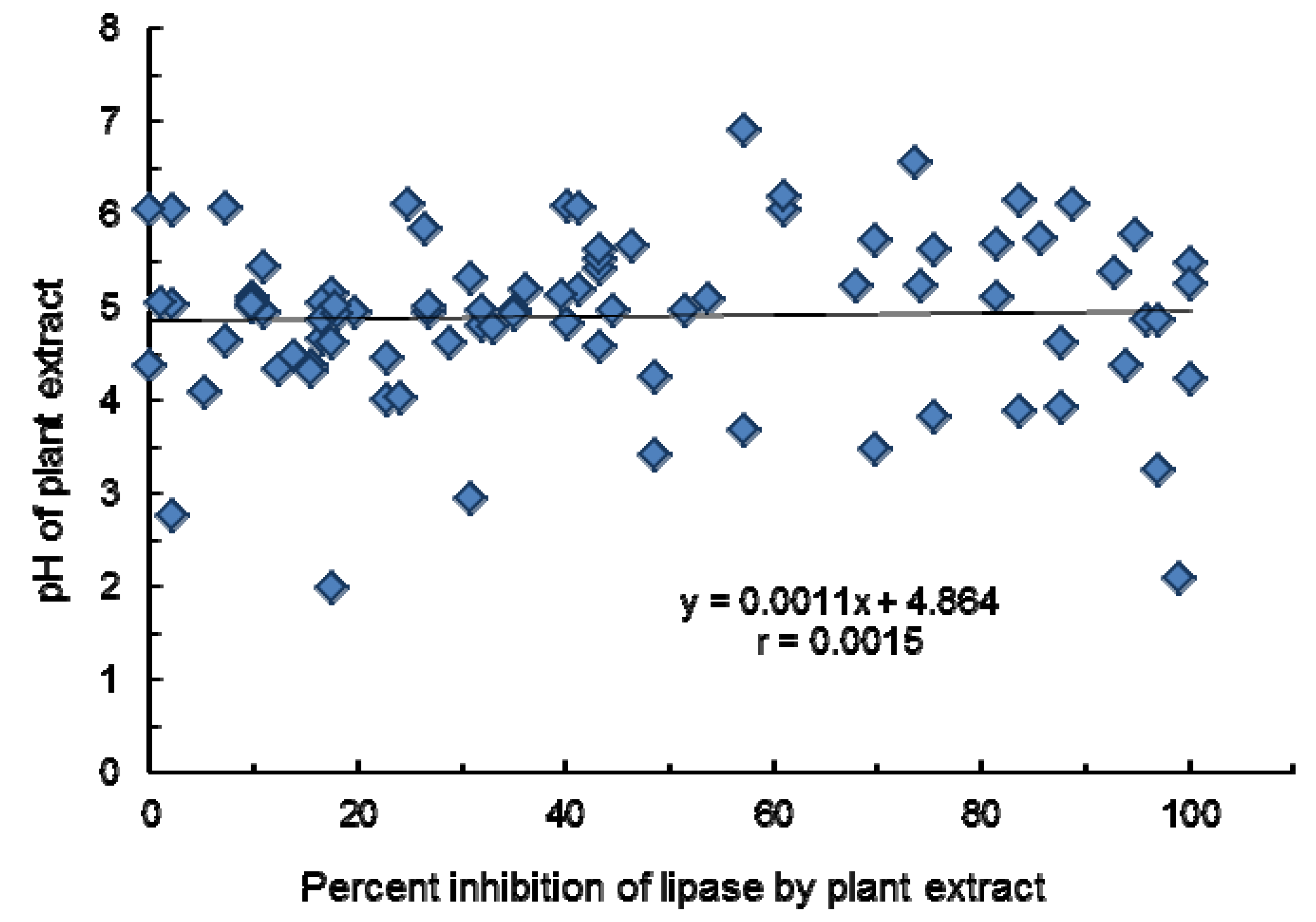
2.1. Effect of Methanol Extract of Medicinal, Herbs, and Aquatic Plants on Porcine Pancreatic Lipase
2.2. Isolation of Lipase Inhibitory Compound Using Bio-Assay Guided Isolation Protocol
2.2.1. Fractionation of Extract Residue of the Active C. cauliflora L. Leaves
| No. | Scientific Name | Local Name | Part Used | Family | Percent Inhibition | Orlistat Equiv. (µg/mL) | pH of Extract |
|---|---|---|---|---|---|---|---|
| 1 | Adenanthera bicolor Moon | Daun tajam | L | Acanthaceae | 26.4 | 0.03 | 5.84 |
| 2 | Adenanthera bicolor Moon | Daun tajam | SD | Acanthaceae | 26.8 | 0.03 | 4.95 |
| 3 | Aleurites moluccana L. Willd | Buah keras (candle nut) | L | Euphocynacea | 100.0 | 0.11 | 5.47 |
| 4 | Allamanda cathartica L. | Alamanda | F | Aphobiaceae | 0.0 | 0.00 | 4.38 |
| 5 | Allium cepa L. | Bawang merah (shallot) | RT | Liliaceae | 73.5 | 0.08 | 6.56 |
| 6 | Allium sativum | Bawang putih (garlic) | RT | Apiaceae | 44.6 | 0.05 | 4.96 |
| 7 | Alocasia macrorrhizos L. (G) Don | Keladi ganyong (yam) | L | Araceae | 12.4 | 0.01 | 4.34 |
| 8 | Amomun cardamomum L. Maton | Pelaga (cardamom) | SD | Zingiberaceae | 36.1 | 0.04 | 5.18 |
| 9 | Anacardium occidentale L. | Gajus (cashew) | L | Anacardiaceae | 88.7 | 0.10 | 6.11 |
| 10 | Andrographi panicutata (Burmf.)Wall | Hempedu bumi | L | Acanthacea | 69.8 | 0.08 | 3.47 |
| 11 | Archidendron jiringa (Jack) I.CNielsen | Jering | F | Falaceae | 100.0 | 0.11 | 5.25 |
| 12 | Asystasia gangetica L. T. Anderson | Akar ruas-ruas (Chinese violet) | L | Acanthaceae | 17.5 | 0.02 | 5.15 |
| 13 | Averrhoa carambola L. | Belimbing besi (starfruit) | FR | Oxalidaceae | 100.0 | 0.11 | 4.23 |
| 14 | Averrhoa carambola L. | Belimbing besi (starfruit) | L | Oxalidaceae | 94.8 | 0.10 | 5.78 |
| 15 | Azadirachta indica A. Juss | Semambu (neem) | L | Meliaceae | 93.8 | 0.10 | 4.38 |
| 16 | Barringtononia racemaso L. Spreng | Putat kampong | L | Lecythidaceae | 95.9 | 0.10 | 4.86 |
| 17 | Brassica oleracea L. | Kobis (cabbage) | L | Brassicaceae | 46.4 | 0.05 | 5.67 |
| 18 | Brassica sp. | Sawi hijau | WP | Brassicaceae | 7.2 | 0.01 | 6.04 |
| 19 | Citrus hystrix DC. | Limau purut | L | Rutaceae | 1.0 | 0.00 | 4.45 |
| 20 | Coleus amboinicus Lour. | Bangun-bangun | L | Labiateae | 9.6 | 0.01 | 4.83 |
| 21 | Colubrina asiatica L. Brongn | Peria pantai | L | Rhamnaceae | 10.3 | 0.01 | 4.81 |
| 22 | Coriandrum sativum L. | Ketumbar (coriander) | SD | Apiaceae | 16.5 | 0.02 | 5.03 |
| 23 | Cosmos caudatus Kunth | Ulam raja | WP | Asteraceae | 35.1 | 0.04 | 4.25 |
| 24 | Cuminum cyminum L. | Jintan putih (cumin) | SD | Apiaceae | 44.6 | 0.05 | 5.41 |
| 25 | Curcuma longa L. | Kunyit (tumeric) | RT | Zingerberaceae | 62.9 | 0.07 | 4.95 |
| 26 | Curcuma longa L. | Kunyit (tumeric) | L | Zingerberaceae | 52.7 | 0.06 | 3.24 |
| 27 | Curcuma xanthorrhiza Roxb | Temu lawak | L | Zingerberaceae | 16.9 | 0.02 | 4.62 |
| 28 | Cynometra cauliflora L. | Nam-nam | L | Falaceae | 100.0 | 0.11 | 4.65 |
| 29 | Cynometra cauliflora L. | Nam-nam | FR | Falaceae | 97.9 | 0.11 | 3.88 |
| 30 | Emblica officinalis | Melaka | L | Euphobiceae | 76.3 | 0.08 | 5.51 |
| 31 | Euginia michelii L. | Cermai belanda | ST | Mytaceae | 90.7 | 0.10 | 4.97 |
| 32 | Euginia michelii L. | Cermai belanda | L | Mytaceae | 92.8 | 0.10 | 4.38 |
| 33 | Ficus deltoidea Jackvar. kunstleri (King Corner) | Mas cotek | FR | Moraceae | 22.7 | 0.02 | 3.83 |
| 34 | Hibiscus esculentus L. | Kacang bendi (okra) | L | Malvaceae | 10.3 | 0.01 | 5.20 |
| 35 | Hibiscus sabdariffa L. | Asam susur (roselle) | L | Malvaceae | 60.9 | 0.07 | 6.20 |
| 36 | Illicium verum hook fil | Bunga lawang (star anise) | SD | Illiciaceae | 22.7 | 0.02 | 4.00 |
| 37 | Ipomonea reptans Forsk | Kangkung (water convolvulus) | L | Convolvulaceae | 40.2 | 0.04 | 6.09 |
| 38 | Labisia sp. | Halia bara (red ginger) | RT | Myrsinaceae | 32.0 | 0.03 | 4.97 |
| 39 | Lawasonia inermis L. | Inai (henna) | L | Lythraceae | 2.1 | 0.00 | 2.75 |
| 40 | Leea indica | Memali | L | Leeaceae | 48.5 | 0.05 | 3.41 |
| 41 | Leucaena leucocephala (Lam) Dewit | Petai belalang | L | Fabaceae | 43.3 | 0.05 | 4.58 |
| 42 | Limnocharis flava L. Buchenau | Emparut | L | Limnocha-ritaceae | 10.9 | 0.01 | 5.43 |
| 43 | Melastoma melabathricum | Senduduk | L | Melastomataceae | 96.9 | 0.10 | 4.86 |
| 44 | Melastoma melabathricum | Senduduk | FR | Melastomataceae | 87.6 | 0.09 | 3.93 |
| 45 | Mentha piperita L. (Pro. Sp) | Pudina (mint) | L | Lamiaceae | 16.5 | 0.02 | 5.04 |
| 46 | Momordica charantia L. | Peria katak | FR | Cucurbitaceae | 83.6 | 0.09 | 6.14 |
| 47 | Monochoria hastata L. Solms | Keladi agas | L | Pontederiaceae | 43.3 | 0.05 | 5.61 |
| 48 | Morinda citrifolia L. | Mengkudu (noni) | FR | Rubiceae | 35.1 | 0.04 | 4.92 |
| 49 | Morinda citrifolia L. | Mengkudu (noni) | L | Rubiceae | 15.5 | 0.02 | 4.31 |
| 50 | Moringa oleifera Lam | Kelor | L | Moringaceae | 75.4 | 0.08 | 5.62 |
| 51 | Murraya koerugii Spreng. | Kari | L | Rutaceae | 41.2 | 0.04 | 6.07 |
| 52 | Musa acuminate Colla | Pisang (banana) | L | Musaceae | 16.5 | 0.02 | 4.87 |
| 53 | Musa acuminate Colla | Jantung pisang | FR | Musaceae | 19.6 | 0.02 | 4.95 |
| 54 | Nigella sativa L. | Jintan hitam | SD | Ranunculaceae | 37.1 | 0.04 | 5.12 |
| 55 | Oenanthe javanica (Blume) DC. | Selom | L | Apiaceae | 39.5 | 0.04 | 5.01 |
| 56 | Ormosia bancana (miq.) Merr. | Saga dengkol | SD | Fabaceae | 26.8 | 0.03 | 6.10 |
| 57 | Orthosiphon spicatus BBS | Misai kucing | WP | Acanthaceae | 24.8 | 0.03 | 5.23 |
| 58 | Pakia speciosa Hassk | Petai | ST | Fabaceae | 74.2 | 0.09 | 5.09 |
| 59 | Pakia speciosa Hassk | Petai | P | Fabaceae | 53.6 | 0.06 | 4.92 |
| 60 | Pakia speciosa Hassk | Petai | L | Fabaceae | 18.0 | 0.02 | 4.62 |
| 61 | Pandanus amaryllifolius | Pandan | L | Pandanaceae | 17.5 | 0.02 | 5.72 |
| 62 | Peperomia pellucid L. Kunth | Ketumpangan air | WP | Piperales | 69.8 | 0.08 | 4.64 |
| 63 | Pereskia sacharosa Griseb | Jarum tujuh bilas | L | Cactaceae | 7.2 | 0.01 | 5.37 |
| 64 | Persea americana Mill | Avocado | L | Lauraceae | 92.8 | 0.10 | 6.07 |
| 65 | Phyllanthus minima | Letup-letup | L | Solanaceae | 7.2 | 0.01 | 5.68 |
| 66 | Phyllanthus niruri L. | Dukung anak | WP | Euphorbiaceae | 81.4 | 0.09 | 5.11 |
| 67 | Piper battle L. | Sirih | WP | Piperaceae | 9.9 | 0.01 | 5.01 |
| 68 | Piper nigrum L. | Lada putih (white pepper) | SD | Piperaceae | 24.1 | 0.03 | 4.03 |
| 69 | Piper sarmentosum Roxb | Kadok | L | Piperaceae | 2.1 | 0.00 | 6.04 |
| 70 | Pisonia grandis R.Br. | Mengkudu siam | L | Nyctaginaceae | 1.0 | 0.00 | 5.05 |
| 71 | Pista statiotes L. | Selada air | WP | Araceae | 57.2 | 0.06 | 6.91 |
| 72 | Premma cordiflora | Buas-buas | L | Verbenaceae | 30.9 | 0.03 | 2.94 |
| 73 | Premma cordiflora | Buas-buas | SD | Verbenaceae | 13.9 | 0.02 | 4.48 |
| 74 | Premma cordiflora | Buas-buas | RT | Verbenaceae | 17.5 | 0.02 | 1.98 |
| 75 | Psidium guajava L. | Jambu (guava) | L | Myrtaceae | 99.0 | 0.11 | 2.09 |
| 76 | Saricocalix crispus | Pecah kaca | L | Acanthaceae | 30.9 | 0.03 | 5.31 |
| 77 | Sauropus androgynus L. Merr. | Cekur manis | RT | Euphorbiaceae | 9.9 | 0.01 | 5.06 |
| 78 | Solanum mammosum L. | Terung susu kambing (nipplefruit) | L | Solanaceae | 33.0 | 0.04 | 4.79 |
| 79 | Spermacoce latifolia Aubl | Kutu tahang (buttonweed) | L | Rubiaceae | 57.2 | 0.06 | 3.68 |
| 80 | Syzygium polyanthum | Serai kayu/daun salam | L | Olacaceae | 5.2 | 0.01 | 4.09 |
| 81 | Syzygium malaccense L. | Jambu air | L | Myrtaceae | 85.6 | 0.09 | 5.74 |
| 82 | Tamarindus indica L. | Asam jawa (tamarind) | L | Fabaceae | 9.9 | 0.01 | 5.03 |
| 83 | Tamarindus indica L. | Asam jawa (tamarind) | FR | Fabaceae | 68.0 | 0.07 | 5.24 |
| 84 | Tinospora crispa (Miers) | Patawali | WP | Menispermaceae | 28.9 | 0.03 | 4.62 |
| 85 | Trigonella foenumgraecum L. | Halba (fenugreek) | SD | Fabales | 0.0 | 0.00 | 6.04 |
| 86 | Vitex sp. | Lemuni hitam | L | Verbenaceae | 51.5 | 0.06 | 4.97 |
| No. | Scientific Name | Local Name | Part Used | Family | Percent Activation | pH of Extract |
|---|---|---|---|---|---|---|
| 1 | Capsium frutescens L. | Cili api | FR | Solanaceae | 125.8 | 5.21 |
| 2 | Cymbopogen citrates DC. | Serai (lemon grass) | WP | Graminae | 104.6 | 6.04 |
| 3 | Emilia sonchifilia | Tambak-tambak | WP | Asteraceae | 131.6 | 3.10 |
| 4 | Gynura procumbens (Lour) Merr | Sambung nyawa | WP | Compositae | 120.6 | 6.87 |
| 5 | Kaempferia galanga L. | Kencur (aromatic ginger) | RT | Zingiberaceae | 122.3 | 5.47 |
| 6 | Lindernia crustacean L. | Unknown | L | Scrophulariaceae | 105.2 | 6.39 |
| 7 | Monochoria hastata L. Solms | Keladi agas | RT | Pontederiaceae | 103.1 | 6.37 |
| 8 | Ocimum basilicum L. | Selasih (basil) | L | Lamiaceae | 103.1 | 5.81 |
| 9 | Pimpinella anisum L. | Jintan manis (aniseed) | SD | Apiaceae | 186.5 | 6.41 |
| 10 | Plunchea indica L. Less | Beluntas | L | Compositae | 110.3 | 5.37 |
| 11 | Psophocarpus tetragonolobus L. | Kacang botor (winged bean) | FR | Falacea | 109.3 | 4.07 |
| 12 | Typha angustifolia L. | Lembang | WP | Typhaceae | 106.9 | 4.17 |
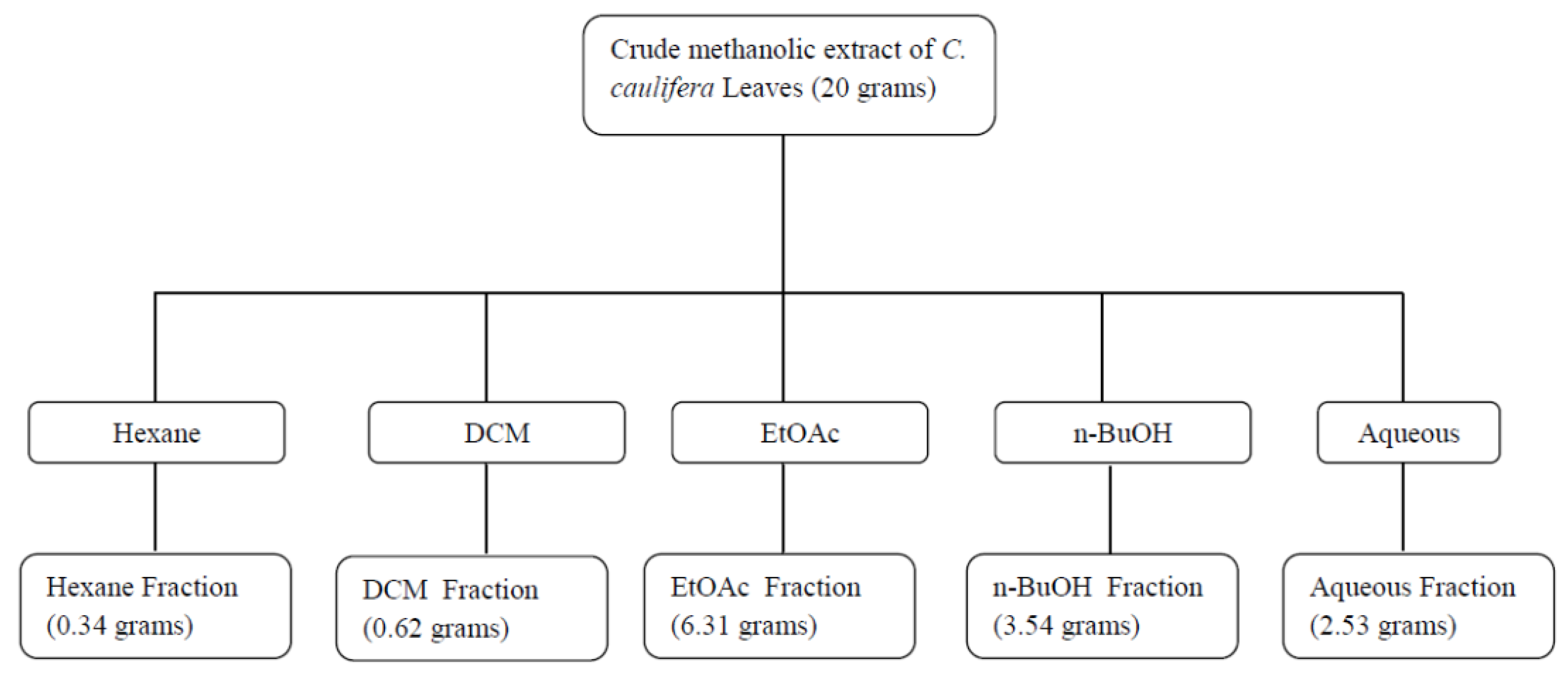
2.2.2. Effect of C. cauliflora Fractions on Porcine Pancreatic Lipase
| Fraction | Percent Inhibition | Orlistat Equiv. (µg/mL) |
|---|---|---|
| Hexane fraction | 14.8 | 1.84 |
| DCM fraction | 17.8 | 2.21 |
| EtOAc fraction | 85.3 | 10.59 |
| n-BuOH fraction | 71.5 | 8.88 |
| Aqueous fraction | 54.7 | 6.79 |
2.3. Column Chromatography and Isolation of Anti-Lipase Compound from EtOAc Fraction of C. cauliflora Leaves
| Sub-Fraction | Percent Inhibition | Orlistat Equiv. (µg/mL) |
|---|---|---|
| A1 | 31.5 | 3.91 |
| A2 | 26.1 | 3.24 |
| A3 | 51.8 | 6.43 |
| A4 | 23.5 | 2.91 |
| A5 | 13.9 | 1.72 |
| A6 | 36.1 | 4.48 |
| Sub-Fraction | Percent Inhibition | Orlistat Equiv. (µg/mL) |
|---|---|---|
| A3-1 | 11.4 | 1.42 |
| A3-2 | 27.5 | 3.41 |
| A3-3 | 45.1 | 5.59 |
| A4-4 | 13.2 | 1.64 |
| A3-1 | 11.4 | 1.42 |
| A3-2 | 27.5 | 3.41 |
2.4. HPLC Profiling of Compounds from Sub-Fractions A3 and A3-3
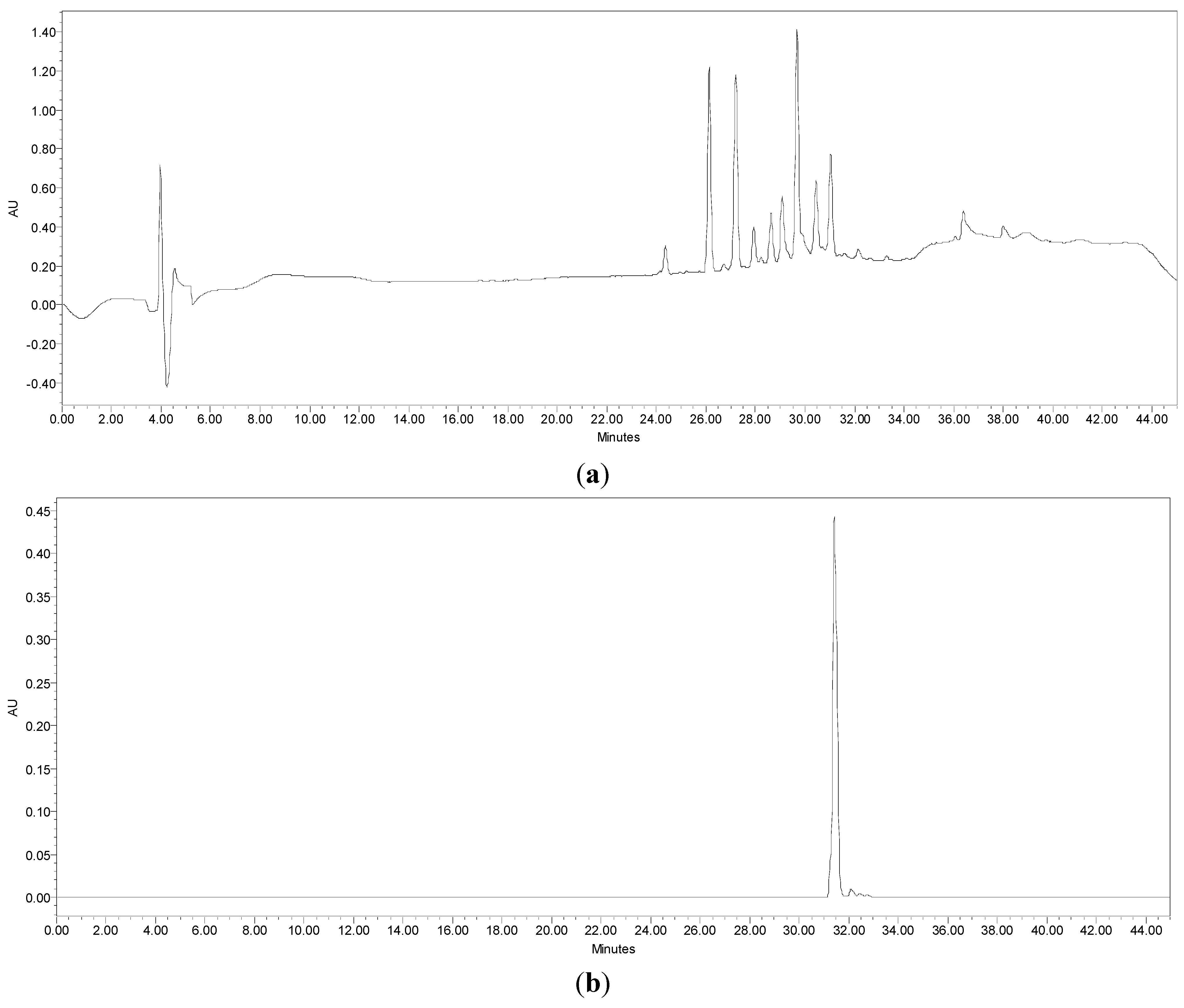
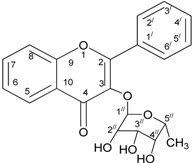
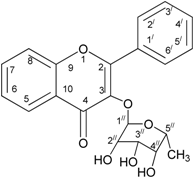
2.5. Structure Elucidation of Anti-Lipase Compound
| Position | δH | δC Reported Values | δC Experimental Values | COSY |
|---|---|---|---|---|
| 1 | - | - | - | - |
| 2 | 155.7 | 157.5 | ||
| 3 | 134.6 | 135.0 | ||
| 4 | 177.9 | 178.4 | ||
| 5 | 160.9 | 160.4 | ||
| 6 | 6.20 | 99.3 | 98.8 | |
| 7 | 161.2 | 164.9 | ||
| 8 | 6.37 | 94.4 | 93.6 | |
| 9 | 155.9 | 158.0 | ||
| 10 | 104.9 | 103.0 | ||
| 1' | 120.4 | 121.5 | ||
| 2' | 7.77 | 130.6 | 130.7 | 7.77, 6.95 |
| 3' | 3.31 | 115.3 | 115.4 | 7.77, 6.95 |
| 4' | 160.0 | 162.1 | ||
| 5' | 6.94 | 115.3 | 115.6 | 7.77, 6.95 |
| 6' | 7.77 | 130.6 | 130.7 | 7.77, 6.95 |
| Rhamnoside C-3 | ||||
| 1" | 5.39 | 101.9 | 102.3 | 5.39, 4.22 |
| 2" | 4.22 | 70.1 | 70.7 | |
| 3" | 3.72 | 70.6 | 70.9 | 3.72, 4.22 |
| 4" | 3.34 | 73.8 | 72.0 | |
| 5" | 3.32 | 70.2 | 70.9 | 3.32, 3.34 |
| 6" | 0.98 | 17.8 | 16.5 |
| Position | Number of Correlated Proton δ H with C | Reported Values δC | Experimental Values δC |
|---|---|---|---|
| 1 | - | - | - |
| 2 | 155.7 | 157.5 | |
| 3 | 5.39 | 134.6 | 135.0 |
| 4 | - | 177.9 | 178.4 |
| 5 | 6.95, 7.77 | 160.9 | 160.4 |
| 6 | 6.37 | 99.3 | 98.8 |
| 7 | 6.37, 6.20 | 161.2 | 164.9 |
| 8 | 6.02 | 94.4 | 93.6 |
| 9 | 6.37, 7.77 | 155.9 | 158.0 |
| 10 | 6.37, 6.20 | 104.9 | 103.0 |
| 1' | 6.95 | 120.4 | 121.5 |
| 2' | 7.77 | 130.6 | 130.7 |
| 3' | 6.95, 7.77 | 115.3 | 115.4 |
| 4' | 6.95, 6.20 | 160.0 | 162.1 |
| 5' | 6.95 | 115.3 | 115.6 |
| 6' | - | 130.6 | 130.7 |
3. Discussion
4. Experimental
4.1. Materials
4.2. Extraction of Plant Metabolites
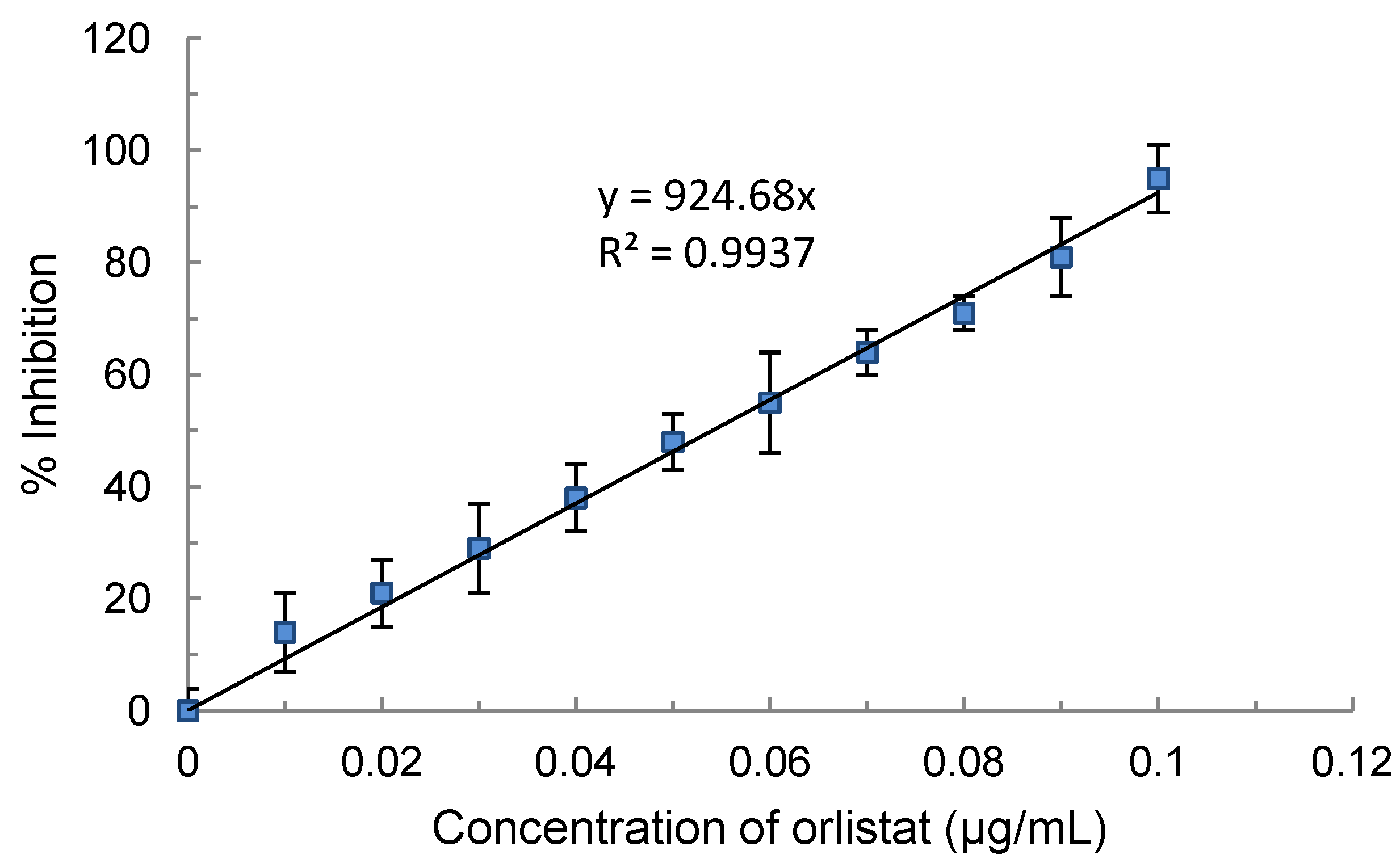
4.3. Assay for Pancreatic Lipase Activity
4.4. Effect of Orlistat on Lipase Activity and Orlistat Equivalent
4.5. Column Chromatography of EtOAc Fraction of C. cauliflora Leaves
Thin Layer Chromatography (TLC)
4.6. Dyeing Reagent for TLC
Preparation of Vanillin-Sulfuric Acid Solution
4.7. Instrumentation
Nuclear Magnetic Resonance (NMR)
5. Conclusions
Acknowledgments
Conflicts of Interest
References
- Chantre, P.; Lairon, D. Recent findings of green tea extract AR25 (Exolise) and its activity for the treatment of obesity. J. Phytomed. 2002, 9, 3–8. [Google Scholar] [CrossRef]
- W.H.O. Obesity and Overweight, Fact sheet N°311. Available online: http://www.who.int/mediacentre/factsheets/fs311/en/index.html (accessed on 2 September 2013).
- Robaayah, Z. Trends in cardiovascular diseases and risk factors in Malaysia. Int. Congr. Ser. 2004, 1262, 446–449. [Google Scholar] [CrossRef]
- Fouad, M.F.; Rastam, S.; Ward, K.D.; Maziak, W. Prevalence of obesity and its associated factors in Aleppo Syria. Prev. Control 2006, 2, 85–94. [Google Scholar]
- Dzulkifli, A.R. Comment: Ban or No, Let’s Keep a Lid on the Other “Silent Killer”. Available online: http://www.nst.com.my/Current_News/NST/Sunday/Focus/20070310105354/Article/index_html (accessed on 8 May 2008).
- Rampal, L.; Rampal, S.; Azhar, M.Z.; Rahman, A.R. Prevalence, awareness, treatment and control of hypertension in Malaysia: A national study of 16,440 subjects. J. R. Inst. Publ. Health 2007, 122, 11–18. [Google Scholar]
- Ogden, C.L.; Carroll, M.D.; Kit, B.K.; Flegal, K.M. Prevalence of obesity in the United States 2009–2010. NCHS Data Brief 2012, No. 82. [Google Scholar]
- Han, L.; Zheng, Y.; Yoshikawa, M.; Okuda, H.; Kimura, Y. Anti-obesity effects of chikusetsusaponins isolated from Panax japonicus rhizomes. BMC Complement. Altern. Med. 2005, 5, 9–18. [Google Scholar]
- Sharma, N.; Sharma, V.K.; Seo, S. Screening of some medicinal plants for anti-lipase activity. J. Ethnopharmacol. 2005, 97, 453–456. [Google Scholar] [CrossRef]
- Kang, J.G.; Park, C.-Y. Anti-obesity drugs: A review about their effects and safety. Diabetes Metab. J. 2012, 36, 13–25. [Google Scholar] [CrossRef]
- Mohammadi, A.; Haririan, I.; Rezanour, N.; Ghiasi, L.; Walker, R.B. A stability indicating high performance liquid chromatographic assay for the determination of orlistat in capsules. J. Chromatogr. A 2006, 1116, 153–157. [Google Scholar] [CrossRef]
- Al-Suwailem, A.K.; Al-Tamimi, A.S.; Al-Omar, M.A.; Al-Suhibani, M.S. Safety and mechanism of action of orlistat (tetrahydrolipstatin) as the first local antiobesity drug. J. Appl. Sci. Res. 2006, 2, 205–208. [Google Scholar]
- Faridah, A.; Nordin, H.L.; Israf, D.A.; Khozirah, S.; Umi Kalsom, Y. Antioxidant and nitric oxide inhibition activities of selected Malay traditional vegetables. Food Chem. 2005, 95, 566–573. [Google Scholar]
- Moreno, D.A.; Ilic, N.; Poulev, A.; Brasaemle, D.L.; Fried, S.K.; Raskin, I. Inhibitory effects of grape seed extract on lipases. J. Nutr. 2003, 19, 876–879. [Google Scholar]
- Thomas, S.; Patil, D.A.; Patil, A.G.; Chandra, N. Pharmacognostic evaluation and physicochemical analysis of Averrhoa carambola L. fruit. J. Herbal Med. Toxicol. 2008, 2, 51–54. [Google Scholar]
- Kim, J.H.; Kim, H.J.; Kim, C.; Jung, H.; Kim, Y.O.; Ju, J.Y.; Shin, C.S. Development of lipase inhibitors from various derivatives of monascus pigment produced by Monascus fermentation. J. Food Chem. 2007, 101, 357–364. [Google Scholar] [CrossRef]
- Liu, W.; Zheng, Y.; Han, L.; Wang, H.; Saito, M.; kimura, Y.; Feng, Y. Saponins (Ginsenosides) from stems and leaves of Panax quinquefolium prevented high-fat diet-induced obesity in mice. J. Phytomed. 2008a, 15, 1140–1145. [Google Scholar]
- McDougall, G.J.; Kulkarni, N.N.; Stewart, D. Berry polyphenols inhibit pancreatic lipase activity in vitro. J. Food Chem. 2009, 115, 193–199. [Google Scholar] [CrossRef]
- Han, K.L.; Zheng, N.Y.; Xu, J.B.; Okuda, H.; Kimura, Y. Saponins from Platycodi radix ameliorate high fat diet-induced obesity in mice. J. Nutr. Metabol. 2002, 132, 2241–2245. [Google Scholar]
- Birari, R.B.; Bhutani, K.K. Pancreatic lipase inhibitors from natural sources: Unexplored potential. Drugs Discov. Today 2007, 12, 879–889. [Google Scholar] [CrossRef]
- Mizuno, M.; Kyotani, Y.; Iinuma, M.; Tanaka, T.; Iwatsuki, K. Kaempferol 3-rhamnoside-7-[6-feruloylglucosyl (13) rhamnoside] from Asplenium prolongatum. J. Phytochem. 1999, 29, 2742–2743. [Google Scholar]
- Liu, H.; Di, Y.; Yang, J.; Teng, F.; Lu, Y.; Ni, W.; Chen, C.; Hao, X. Three novel 3,4-seco-podocarpane trinorditerpenoids from Aleurites moluccana. Tetrahedron Lett. 2008b, 49, 5150–5151. [Google Scholar]
- Mackeen, M.M.; Ali, A.M.; Abdullah, M.A.; Nasir, R.M.; Mat, N.B.; Razak, A.R.; Kawazu, K. Antinematodal activity of some Malaysian plant extracts against the pine wood nematode, Bursaphelenchus xylophilus. Pestic. Sci. 1997, 51, 165–170. [Google Scholar]
- Charungchitrak, S.; Petsoma, A.; Sangvanich, P.; Karnchanatat, A. Antifungal and antibacterial activities of lectin from the seeds of Archidendron jiringa Nielsen. J. Food Chem. 2011, 126, 1025–1032. [Google Scholar] [CrossRef]
- Ikrama, E.H.K.; Khoo, H.E.; Jalil, A.M.; Ismail, A.; Idris, S.; Azlan, A.; Nazri, H.S.M.; Diton, N.A.M.; Mokhtar, R.A.M. Antioxidant capacity and total phenolic content of Malaysian underutilized fruits. J. Food Compos. Anal. 2009, 22, 388–393. [Google Scholar] [CrossRef]
- Gülçin, I.; Oktay, M.; Kireçci, E.; Küfrevioğlu, Ö.Í. Screening of antioxidant and antimicrobial activities of anise (Pimpinella anisum L.) seed extracts. J. Food Chem. 2003, 83, 371–382. [Google Scholar] [CrossRef]
- Boskabady, M.H.; Ramazani, M. Relaxant effect of Pimpinella anisum on isolated guinea pig tracheal chains and its possible mechanism(s). J. Ethnopharmacol. 2001, 74, 83–88. [Google Scholar] [CrossRef]
- De la Garza, A.; Milagro, F.I.; Boque, N.; Campión, J.; Martínez, J.A. Natural inhibitors of pancreatic lipase as new players in obesity treatment. Planta Med. 2011, 77, 773–785. [Google Scholar] [CrossRef]
- Won, S.; Kim, S.; Kim, Y.; Lee, P.; Ryu, J.; Kim, J. Licochalcone A: A lipase inhibitor from the roots of Glycyrrhiza uralensis. Food Rev. Int. 2007, 40, 1046–1050. [Google Scholar]
- Raghavendra, M.P.; Prakash, V. Phenylboronic acid—a potent inhibitor of lipase from Oryza sativa. J. Agric. Food Chem. 2002, 50, 6037–6041. [Google Scholar] [CrossRef]
- Ninomiya, K.; Matsuda, H.; Shimoda, H.; Nishida, N.; Kasajima, N.; Yoshino, T. Carnosic acid, a new class of lipid absorption inhibitor from sage. Bioorg. Med. Chem. Lett. 2004, 14, 1943–1946. [Google Scholar] [CrossRef]
- Calderón-Montaño, J.M.; Burgos-Morón, E.; Pérez-Guerrero, C.; López-Lázaro, M. A review on the dietary flavonoid kaempferol. Mini Rev. Med. Chem. 2011, 11, 298–344. [Google Scholar] [CrossRef]
- Nazemiyeh, H.; Kazemi, E.M.; Zare, K.; Jodari, M.; Nahar, L.; Sarker, S.D. Free radical scavengers from the aerial parts of Euphorbia petiolata. J. Nat. Med. 2009, 64, 187–190. [Google Scholar]
- Xu, H.X.; Lee, S.F. Activity of plant flavonoids against antibiotic-resistant bacteria. Phytother. Res. 2001, 15, 39–43. [Google Scholar]
- Sample Availability: Contact the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ado, M.A.; Abas, F.; Mohammed, A.S.; Ghazali, H.M. Anti- and Pro-Lipase Activity of Selected Medicinal, Herbal and Aquatic Plants, and Structure Elucidation of an Anti-Lipase Compound. Molecules 2013, 18, 14651-14669. https://doi.org/10.3390/molecules181214651
Ado MA, Abas F, Mohammed AS, Ghazali HM. Anti- and Pro-Lipase Activity of Selected Medicinal, Herbal and Aquatic Plants, and Structure Elucidation of an Anti-Lipase Compound. Molecules. 2013; 18(12):14651-14669. https://doi.org/10.3390/molecules181214651
Chicago/Turabian StyleAdo, Muhammad Abubakar, Faridah Abas, Abdulkarim Sabo Mohammed, and Hasanah M. Ghazali. 2013. "Anti- and Pro-Lipase Activity of Selected Medicinal, Herbal and Aquatic Plants, and Structure Elucidation of an Anti-Lipase Compound" Molecules 18, no. 12: 14651-14669. https://doi.org/10.3390/molecules181214651
APA StyleAdo, M. A., Abas, F., Mohammed, A. S., & Ghazali, H. M. (2013). Anti- and Pro-Lipase Activity of Selected Medicinal, Herbal and Aquatic Plants, and Structure Elucidation of an Anti-Lipase Compound. Molecules, 18(12), 14651-14669. https://doi.org/10.3390/molecules181214651