Anti-Tumor Effects of Atractylenolide I Isolated from Atractylodes macrocephala in Human Lung Carcinoma Cell Lines
Abstract
:1. Introduction
2. Results and Discussion
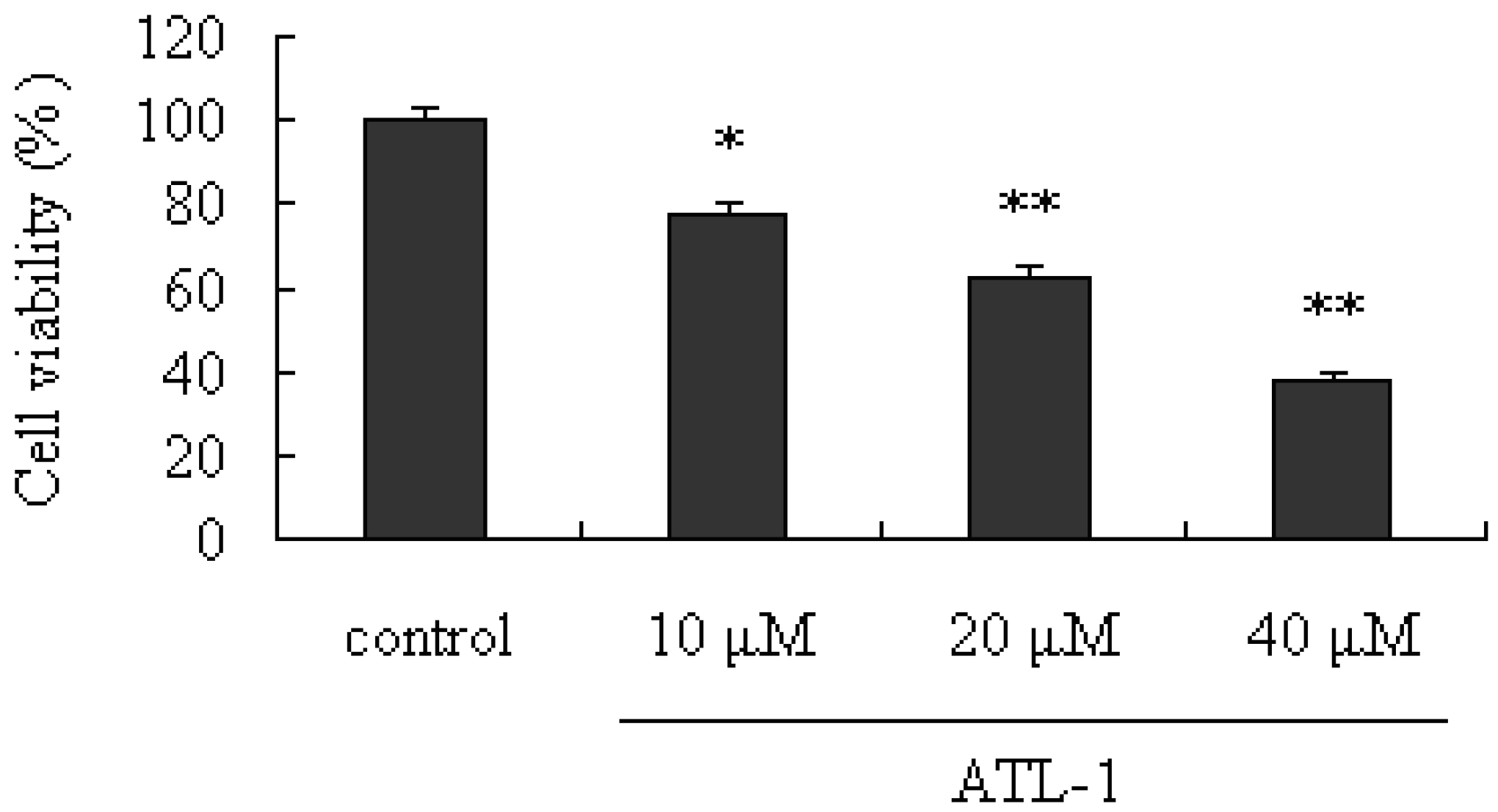
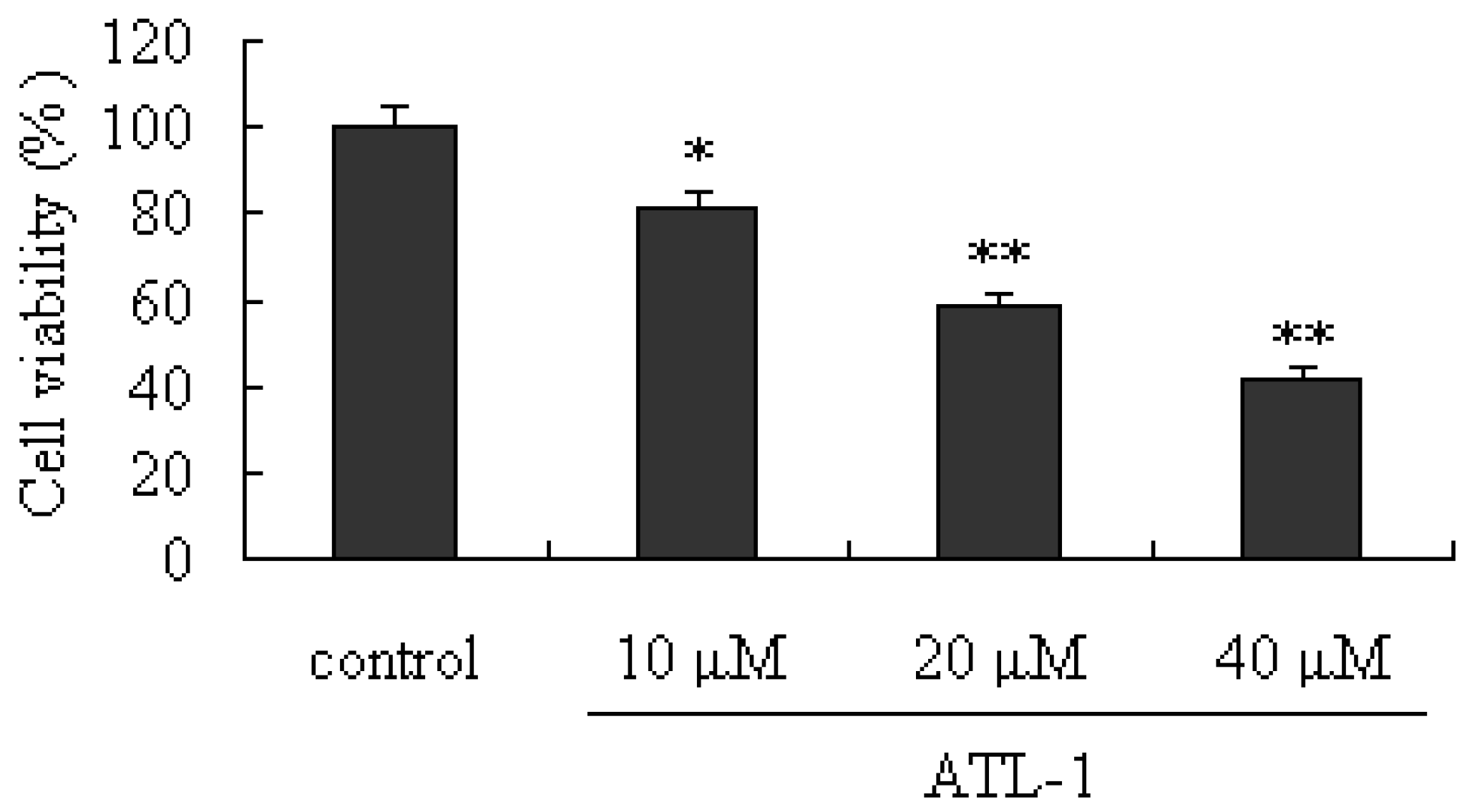
2.1. Cytotoxic Effect of ATL-I on A549 Cells
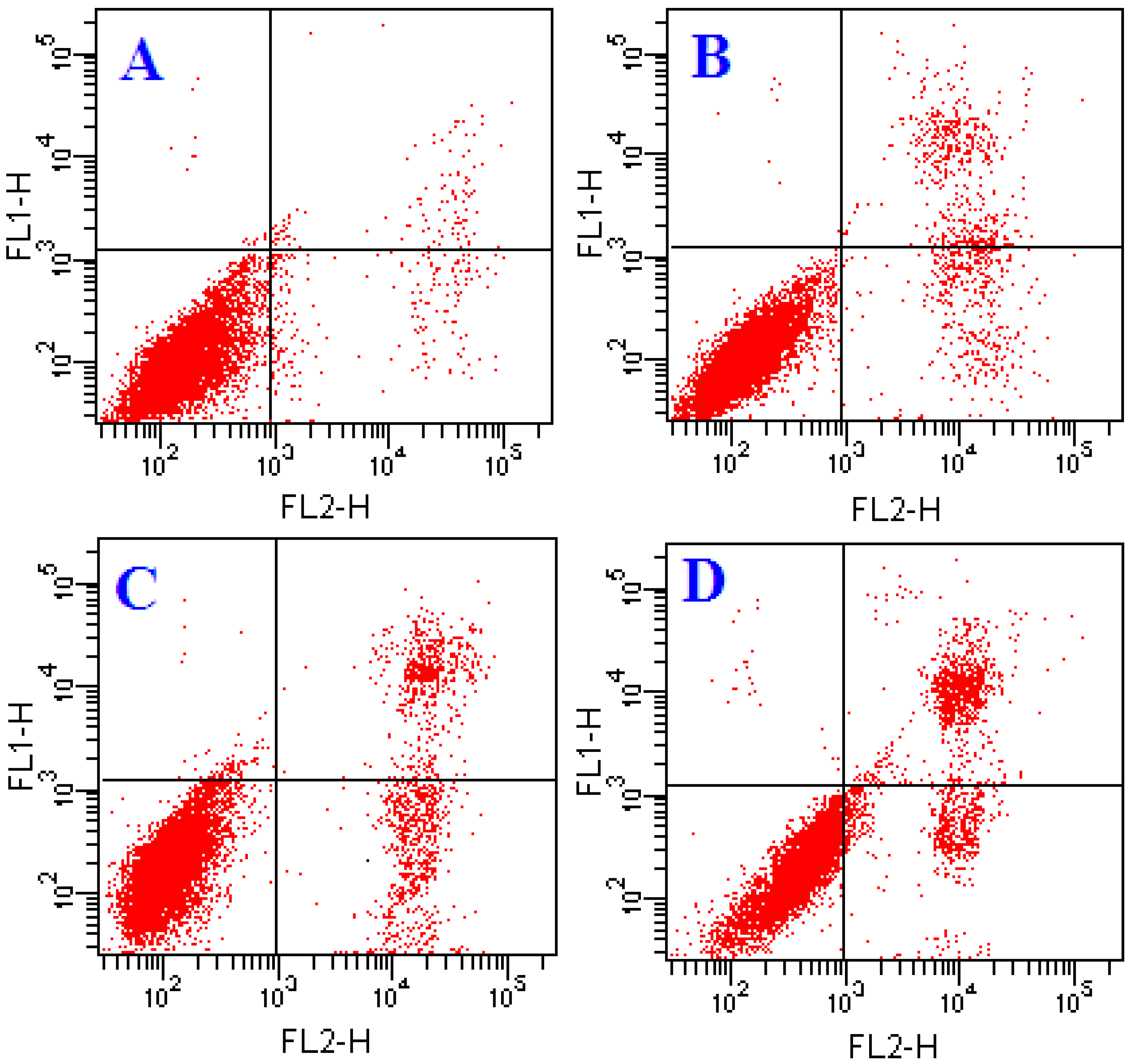
2.2. Effects of ATL-1 on Apoptosis in A549 Cells

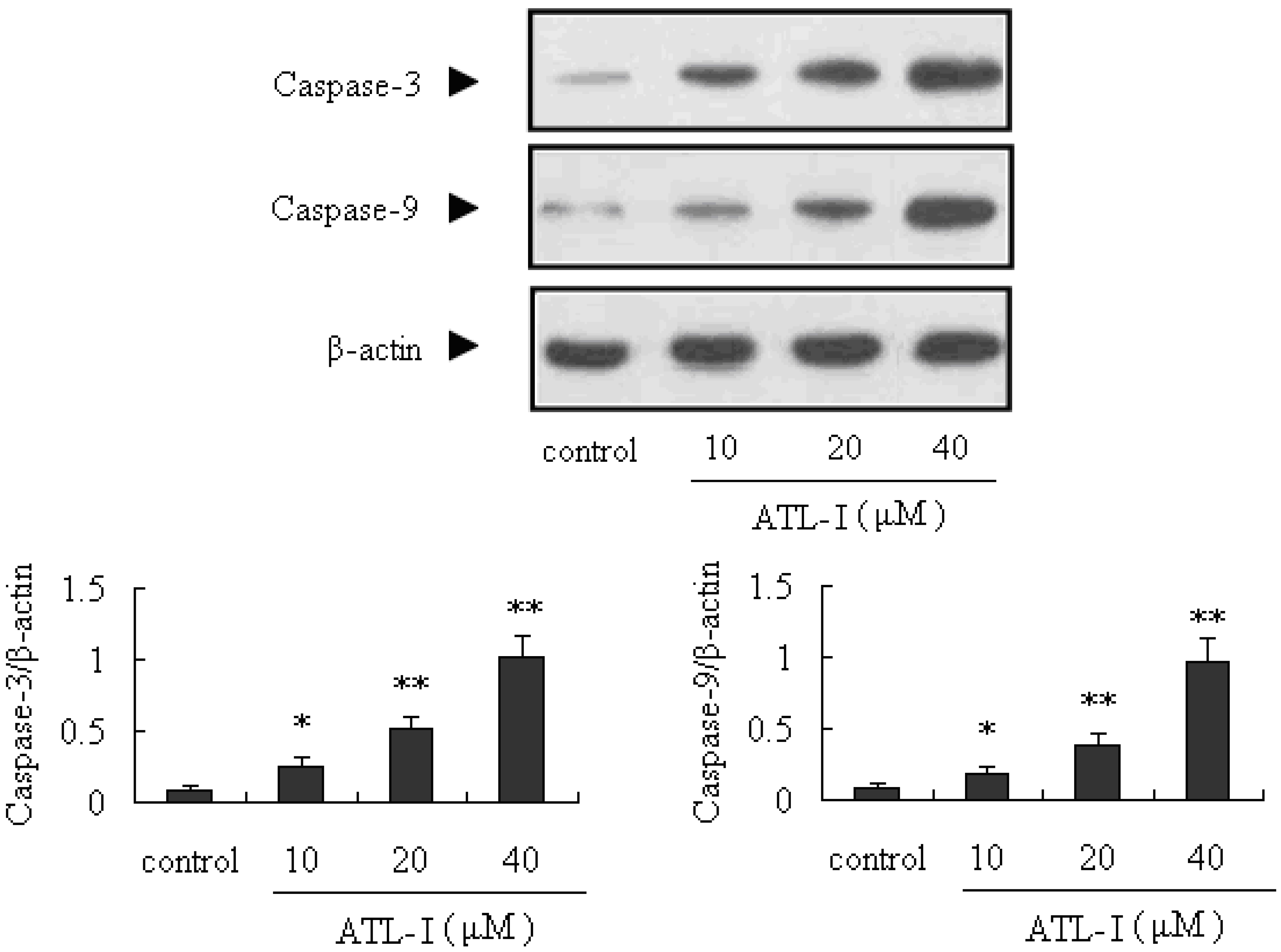
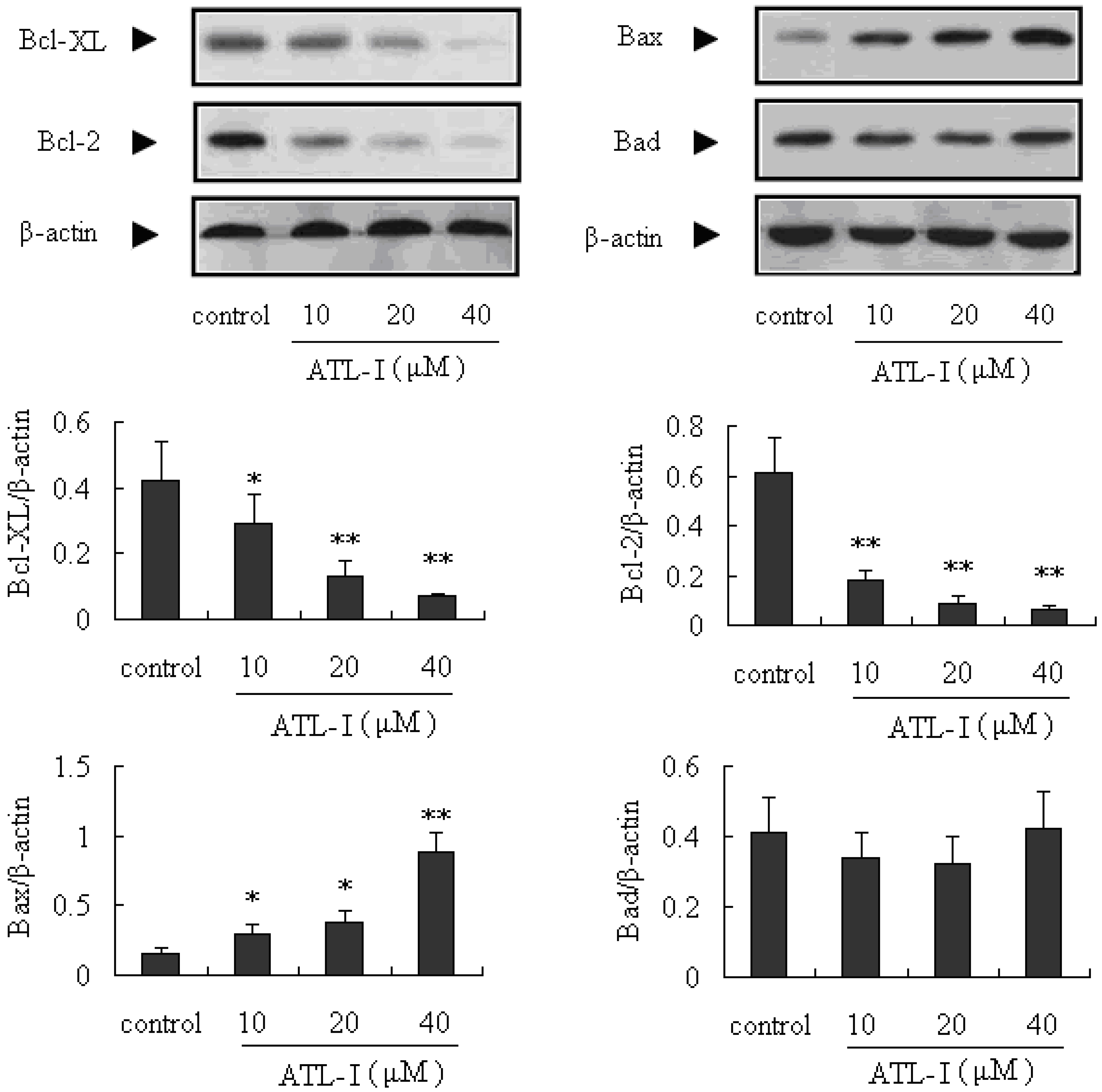
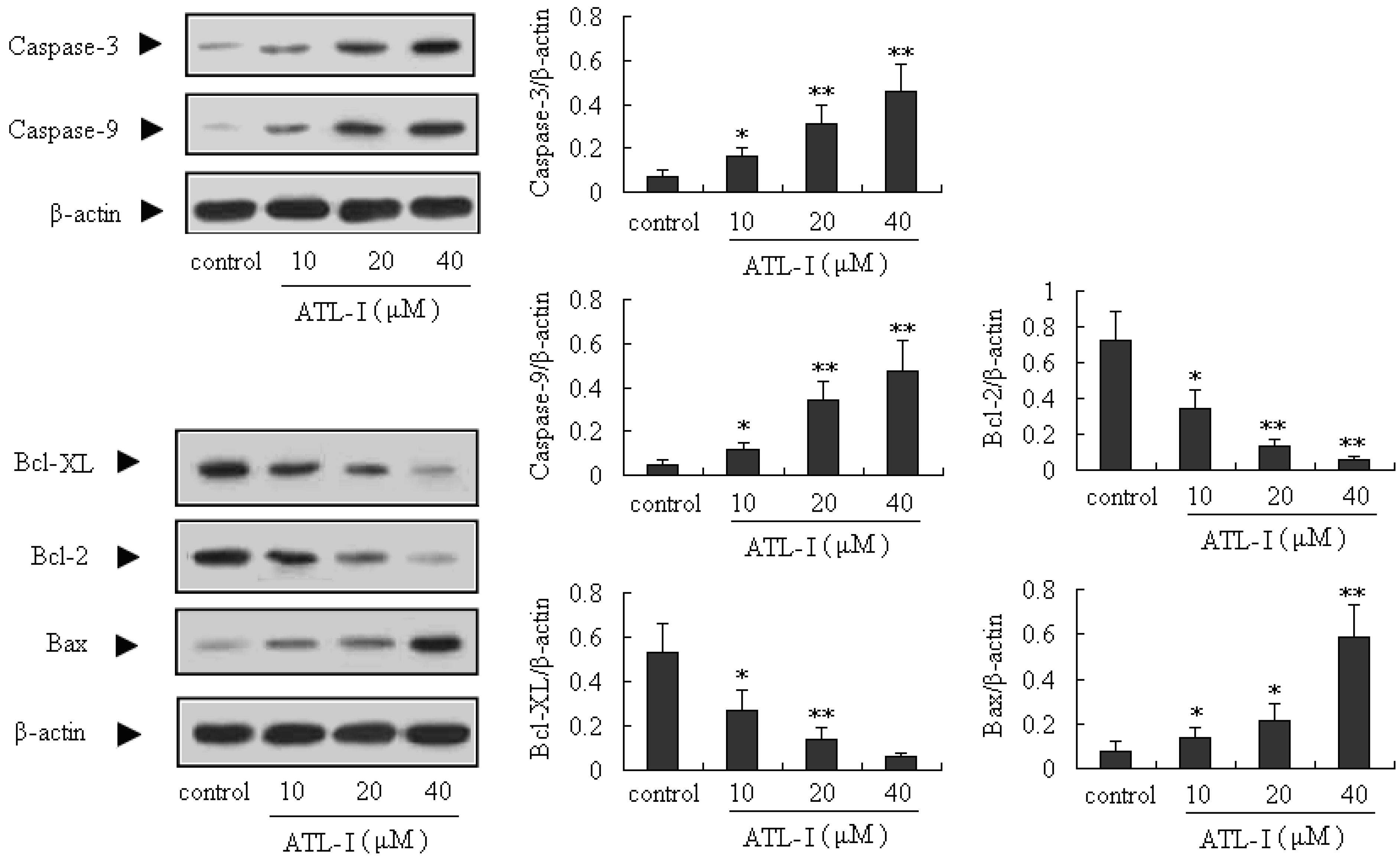
2.3. ATL-I Induces Apoptosis in A549 and HCC827 Cells via a Mitochondria-Mediated Apoptosis Pathway
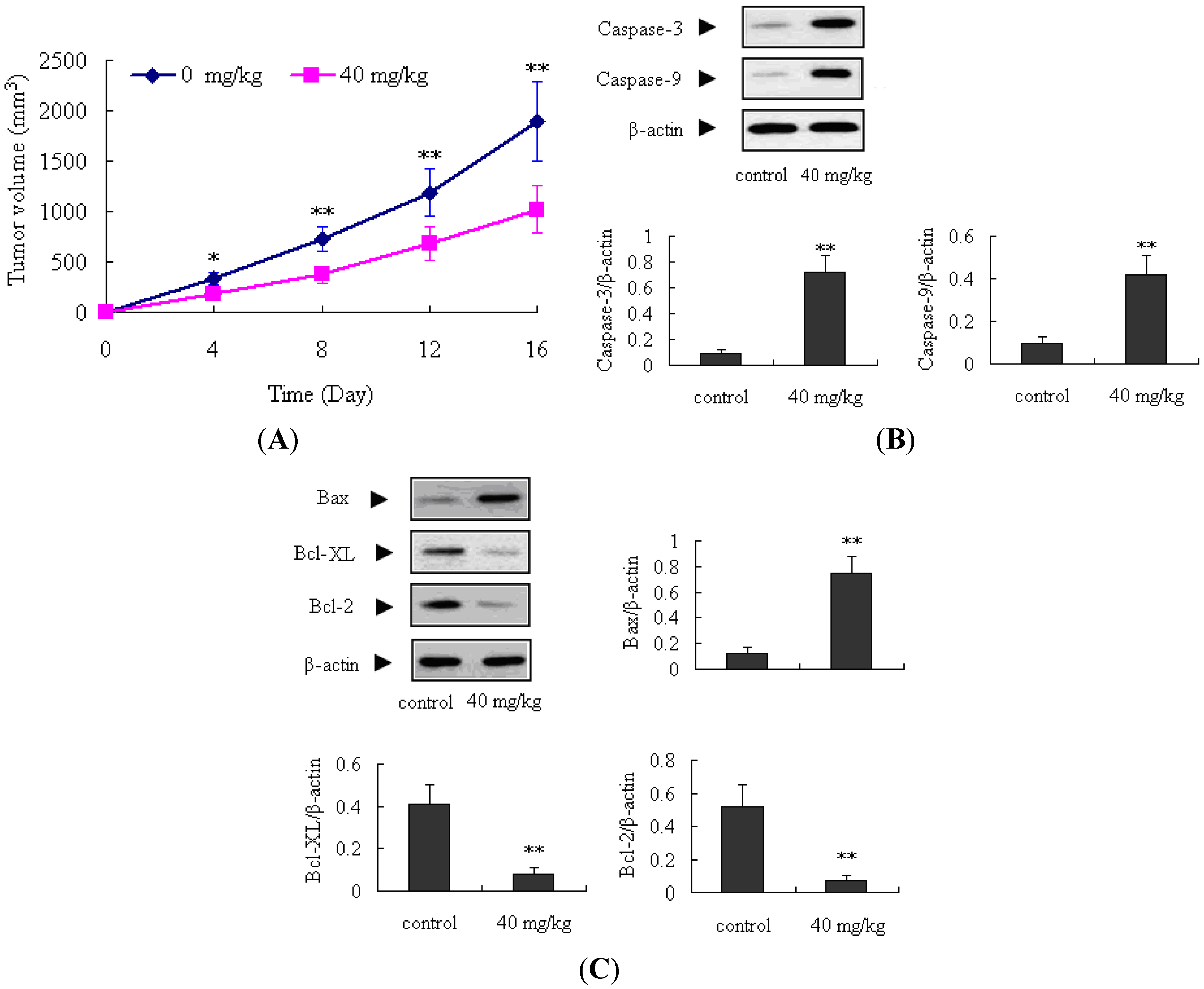
2.4. Antitumor Activity of ATL-1 on Nude Mice
3. Experimental
3.1. Chemicals
3.2. Animals
3.3. Isolation and Preparation of ATL-1
3.4. Cell culture
3.5. MTT Reduction Assay
3.6. Apoptosis Assay
3.7. Western Blot Analysis
3.8. In Vivo Antitumor Efficacy Study
4. Conclusions
Conflicts of Interest
References
- Siegel, R.; Naishadham, D.; Jemal, A. Cancer statistics, 2012. CA Cancer J. Clin. 2012, 62, 10–29. [Google Scholar] [CrossRef]
- Gao, W.M.; Mady, H.H.; Yu, G.Y.; Siegfried, J.M.; Luketich, J.D.; Melhem, M.F.; Keohavong, P. Comparison of p53 mutations between adenocarcinoma and squamous cell carcinoma of the lung: Unique spectra involving G to A transitions and G to T transversions in both histologic types. Lung Cancer 2003, 40, 141–150. [Google Scholar] [CrossRef]
- Tamura, A.; Hebisawa, A.; Hayashi, K.; Sagara, Y.Z.; Fukushima, K.; Kurashima, A.; Yotsumoto, H.; Mori, M.; Komatsu, H. Prognostic significance of thrombomodulin expression and vascular invasion in stage I squamous cell carcinoma of the lung. Lung Cancer 2001, 34, 375–382. [Google Scholar] [CrossRef]
- Kang, T.H.; Bang, J.Y.; Kim, M.H.; Kang, I.C.; Kim, H.M.; Jeong, H.J. Atractylenolide III, a sesquiterpenoid, induces apoptosis in human lung carcinoma A549 cells via mitochondria-mediated death pathway. Food Chem. Toxicol. 2011, 49, 514–519. [Google Scholar] [CrossRef]
- Liao, N.; Ao, M.Z.; Zhang, P.; Yu, L.J. Extracts of Lycoris aurea Induce Apoptosis in Murine Sarcoma S180 Cells. Molecules 2012, 17, 3723–3735. [Google Scholar] [CrossRef]
- Peng, W.; Han, T.; Xin, W.B.; Zhang, X.G.; Zhang, Q.Y.; Jia, M.; Qin, L.P. Comparative research of chemical constituents and bioactivities between petroleum ether extracts of the aerial part and the rhizome of Atractylodes macrocephala. Med. Chem. Res. 2011, 20, 146–151. [Google Scholar] [CrossRef]
- Peng, W.; Han, T.; Wang, Y.; Xin, W.B.; Zheng, C.J.; Qin, L.P. Chemical constituents of the aerial part of Atractylodes macrocephala. Chem. Nat. Comp. 2011, 46, 959–960. [Google Scholar] [CrossRef]
- Endo, K.; Taguchi, T. Antiinflammatory principle of Atractylodes rhizomes. Chem. Pharm. Bull. 1979, 27, 2954–2958. [Google Scholar] [CrossRef]
- Chen, Q.F.; Liu, Z.P.; Wang, F.P. Natural sesquiterpenoids as cytotoxic anticancer agents. Mini Rev. Med. Chem. 2011, 11, 1153–1164. [Google Scholar] [CrossRef]
- Kim, K.H.; Noh, H.J.; Choi, S.U.; Park, K.M,; Seok, S.J.; Lee, K.R. Lactarane sesquiterpenoids from Lactarius subvellereus and their cytotoxicity. Bioorg. Med. Chem. Lett. 2010, 20, 5385–5388. [Google Scholar] [CrossRef]
- Narender, T.; Sukanya, P.; Sharma, K.; Bathula, S.R. Preparation of novel antiproliferative emodin derivatives and studies on their cellcycle arrest, caspase dependent apoptosis and DNA binding interaction. Phytomedicine 2013, 20, 890–896. [Google Scholar] [CrossRef]
- Ma, Y.S.; Weng, S.W.; Lin, M.W.; Lu, C.C.; Chiang, J.H.; Yang, J.S. Antitumor effects of emodin on LS1034 human colon cancer cells in vitro and in vivo: Roles of apoptotic cell death and LS1034 tumor xenografts model. Food Chem. Toxicol. 2012, 50, 1271–1278. [Google Scholar] [CrossRef]
- Su, Y.T.; Chang, H.L.; Shyue, S.K.; Hsu, S.L. Emodin induces apoptosis in human lung adenocarcinoma cells through a reactive oxygen species-dependent mitochondrial signaling pathway. Biochem. Pharmacol. 2005, 70, 229–241. [Google Scholar] [CrossRef]
- Strasser, A.; Huang, D.C.; Vaux, D.L. The role of the bcl-2/ced-9 gene family in cancer and general implications of defects in cell death control for tumourigenesis and resistance to chemotherapy. Biochim. Biophys. Acta 1997, 1333, 151–178. [Google Scholar]
- Sun, N.; Hao, J.R.; Li, X.Y.; Yin, X.H.; Zong, Y.Y.; Zhang, G.Y.; Gao, C. GluR6-FasL-Trx2 mediates denitrosylation and activation of procaspase-3 in cerebral ischemia/reperfusion in rats. Cell Death Dis. 2013, 4, e771. [Google Scholar] [CrossRef]
- Hu, W.; Kavanagh, J.J. Anticancer therapy targeting the apoptotic pathway. Lancet Oncol. 2003, 4, 721–729. [Google Scholar] [CrossRef]
- Gao, N.; Budhraja, A.; Cheng, S.; Yao, H.; Zhang, Z.; Shi, X. Induction of apoptosis in human leukemia cells by grape seed extract occurs via activation of c-Jun NH2-terminal kinase. Clin. Cancer Res. 2009, 15, 140–149. [Google Scholar] [CrossRef]
- Antonsson, B. Bax and other pro-apoptotic Bcl-2 family ‘‘killerproteins’’ and their victim the mitochondrion. Cell. Tissue Res. 2001, 306, 347–361. [Google Scholar] [CrossRef]
- Huang, B.S.; Sun, J.S.; Chen, Z.L. Isolation and identification of atractylenolide IV from Atractylodes macrocephala Koidz. Acta Botanica. Sin. 1992, 31, 614–617. [Google Scholar]
- Zhou, T; Li, G; Cao, B; Li, L; Cheng, Q; Kong, H; Shan, C; Huang, X; Chen, J; Gao, N. Downregulation of Mcl-1 through inhibition of translation contributes to benzyl isothiocyanate induced cell cycle arrest and apoptosis in human leukemia cells. Cell. Death Dis. 2013, 4, e515. [Google Scholar] [CrossRef]
- Sample Availability: Contact the authors.
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, H.; Zhu, Y.; Zhang, T.; Zhao, Z.; Zhao, Y.; Cheng, P.; Li, H.; Gao, H.; Su, X. Anti-Tumor Effects of Atractylenolide I Isolated from Atractylodes macrocephala in Human Lung Carcinoma Cell Lines. Molecules 2013, 18, 13357-13368. https://doi.org/10.3390/molecules181113357
Liu H, Zhu Y, Zhang T, Zhao Z, Zhao Y, Cheng P, Li H, Gao H, Su X. Anti-Tumor Effects of Atractylenolide I Isolated from Atractylodes macrocephala in Human Lung Carcinoma Cell Lines. Molecules. 2013; 18(11):13357-13368. https://doi.org/10.3390/molecules181113357
Chicago/Turabian StyleLiu, Huanyi, Yajie Zhu, Tao Zhang, Zhenguo Zhao, Yu Zhao, Peng Cheng, Hua Li, Hui Gao, and Xiaomei Su. 2013. "Anti-Tumor Effects of Atractylenolide I Isolated from Atractylodes macrocephala in Human Lung Carcinoma Cell Lines" Molecules 18, no. 11: 13357-13368. https://doi.org/10.3390/molecules181113357
APA StyleLiu, H., Zhu, Y., Zhang, T., Zhao, Z., Zhao, Y., Cheng, P., Li, H., Gao, H., & Su, X. (2013). Anti-Tumor Effects of Atractylenolide I Isolated from Atractylodes macrocephala in Human Lung Carcinoma Cell Lines. Molecules, 18(11), 13357-13368. https://doi.org/10.3390/molecules181113357



