Supercritical Carbon Dioxide Extraction of Seed Oil from Winter Melon (Benincasa hispida) and Its Antioxidant Activity and Fatty Acid Composition
Abstract
:1. Introduction
2. Results and Discussion
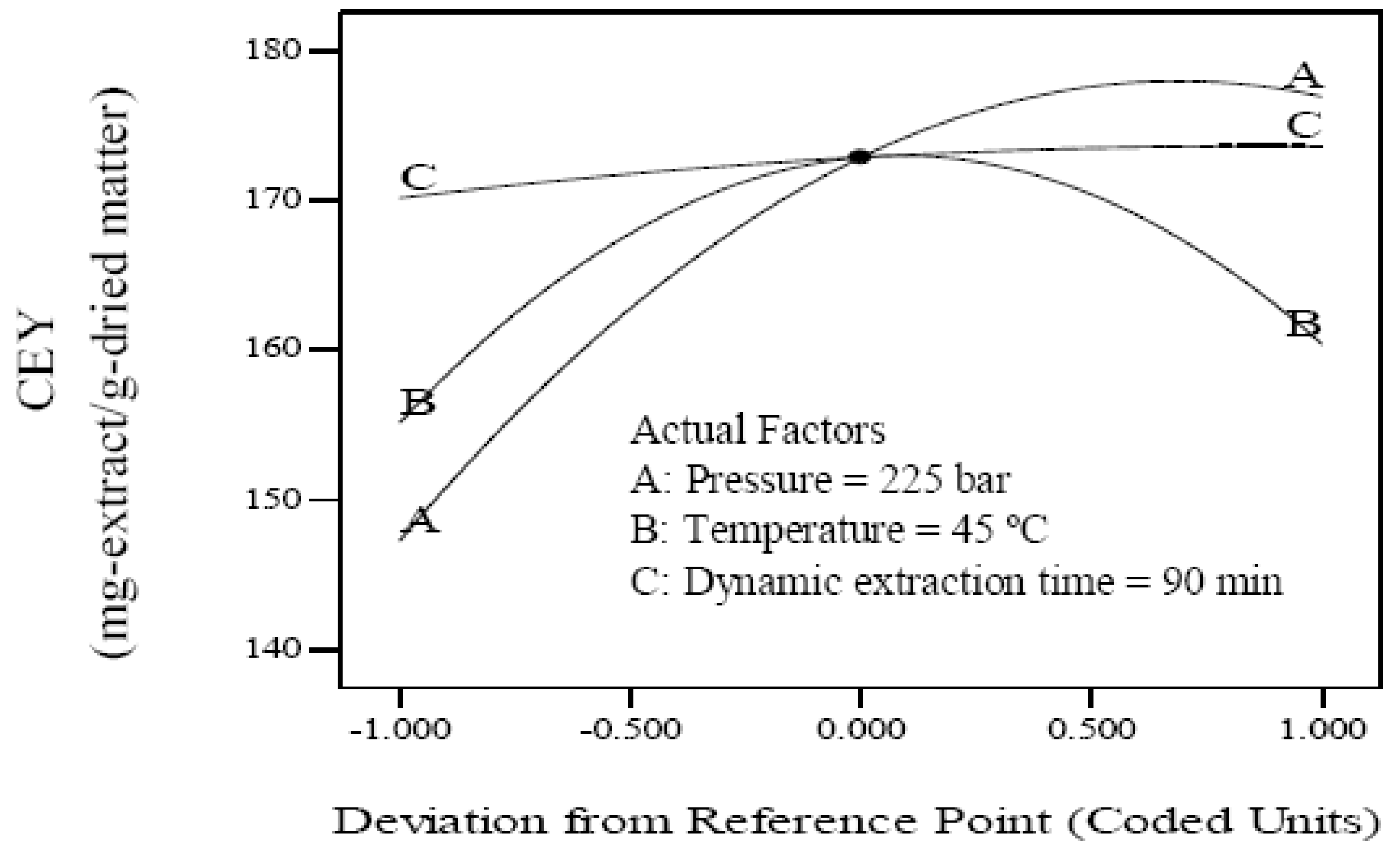
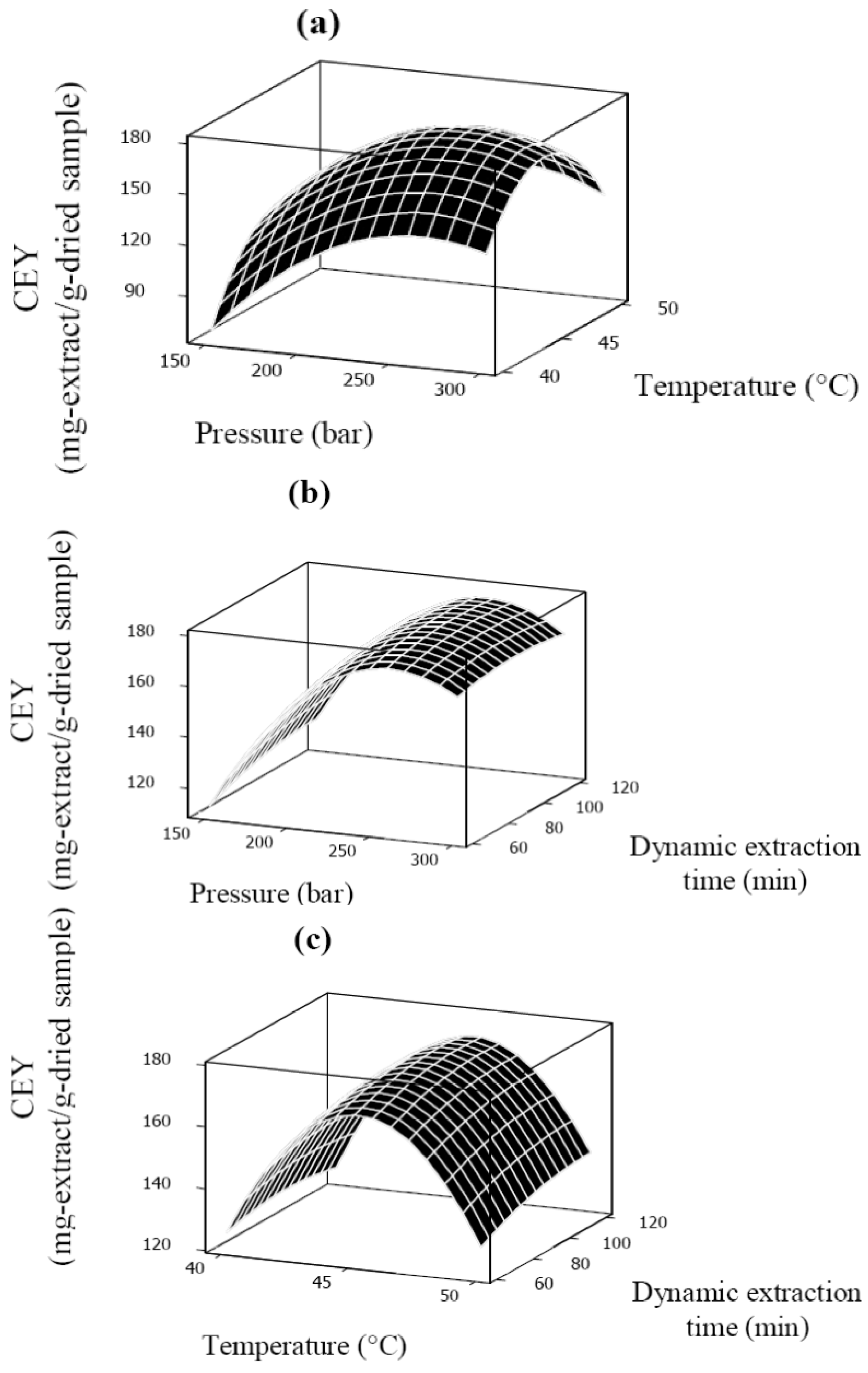
2.1. Effect of the Process Variables on Crude Extraction Yield
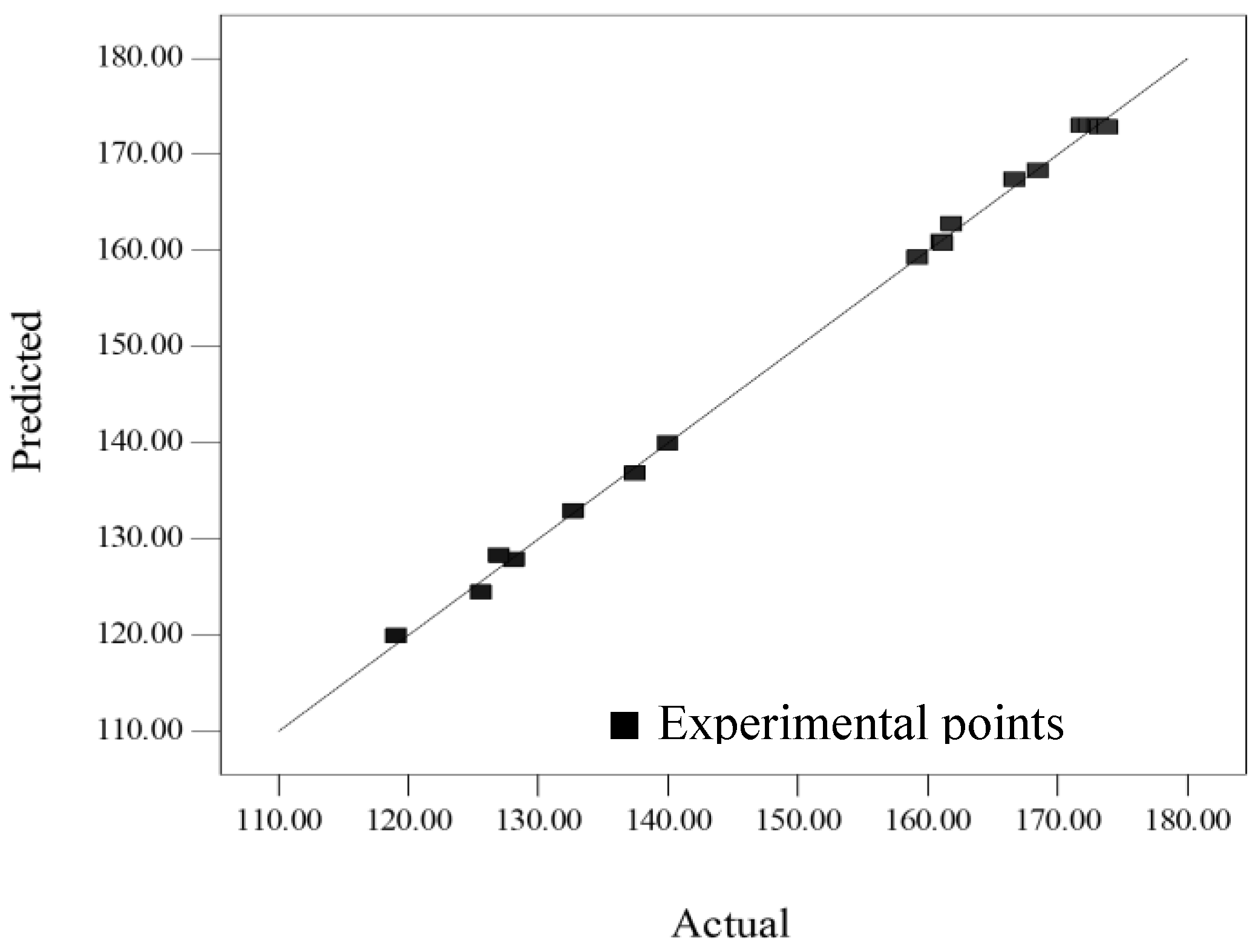
2.2. Optimization of SC-CO2 Extraction Conditions and Verification of the Model
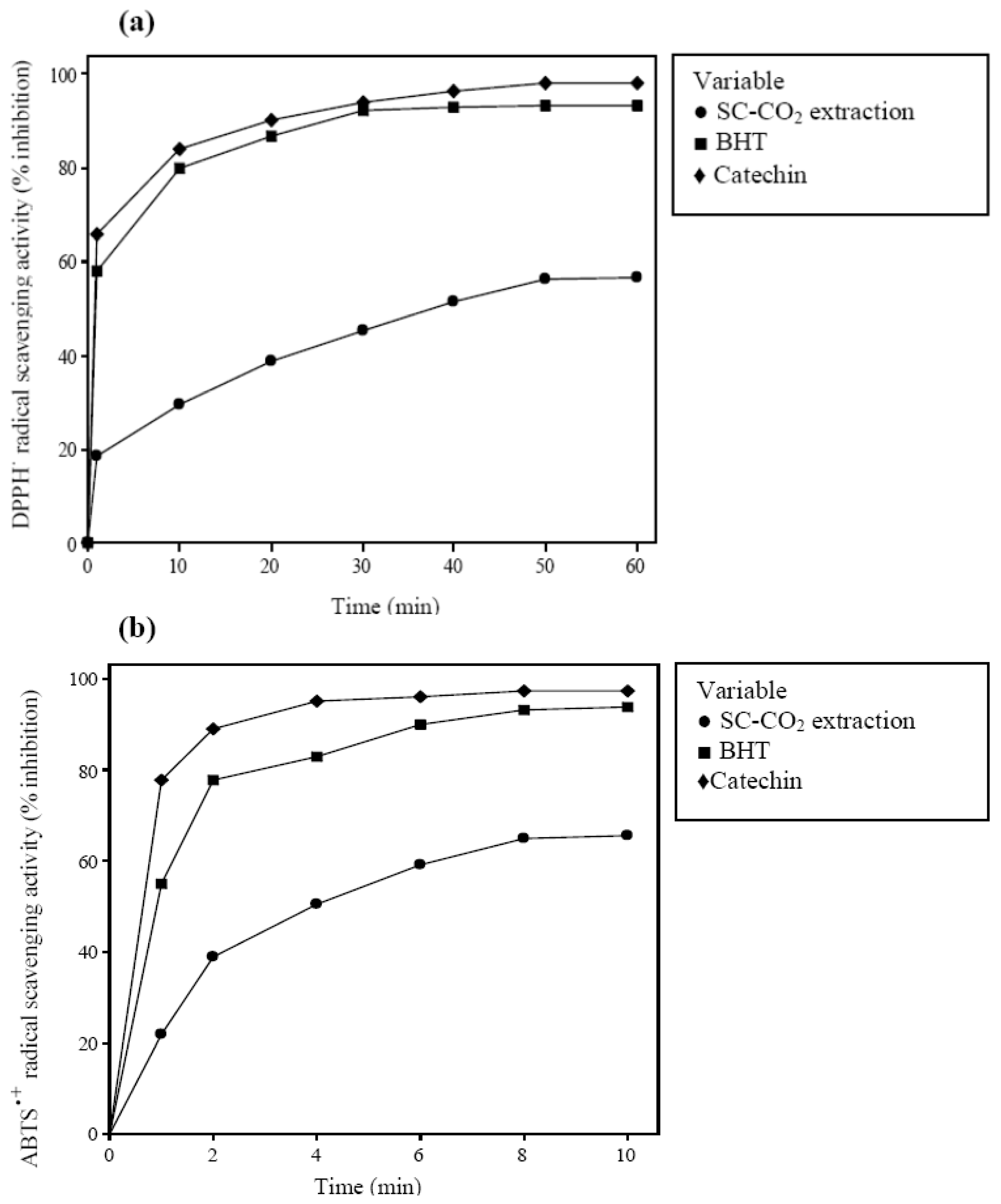
2.3. Antioxidant Activity of Extracted Crude Oil
2.4. Identification and Quantification of Fatty Acid Composition
2.5. Comparison with Other Extraction Techniques
3. Experimental
3.1. Material and Reagents
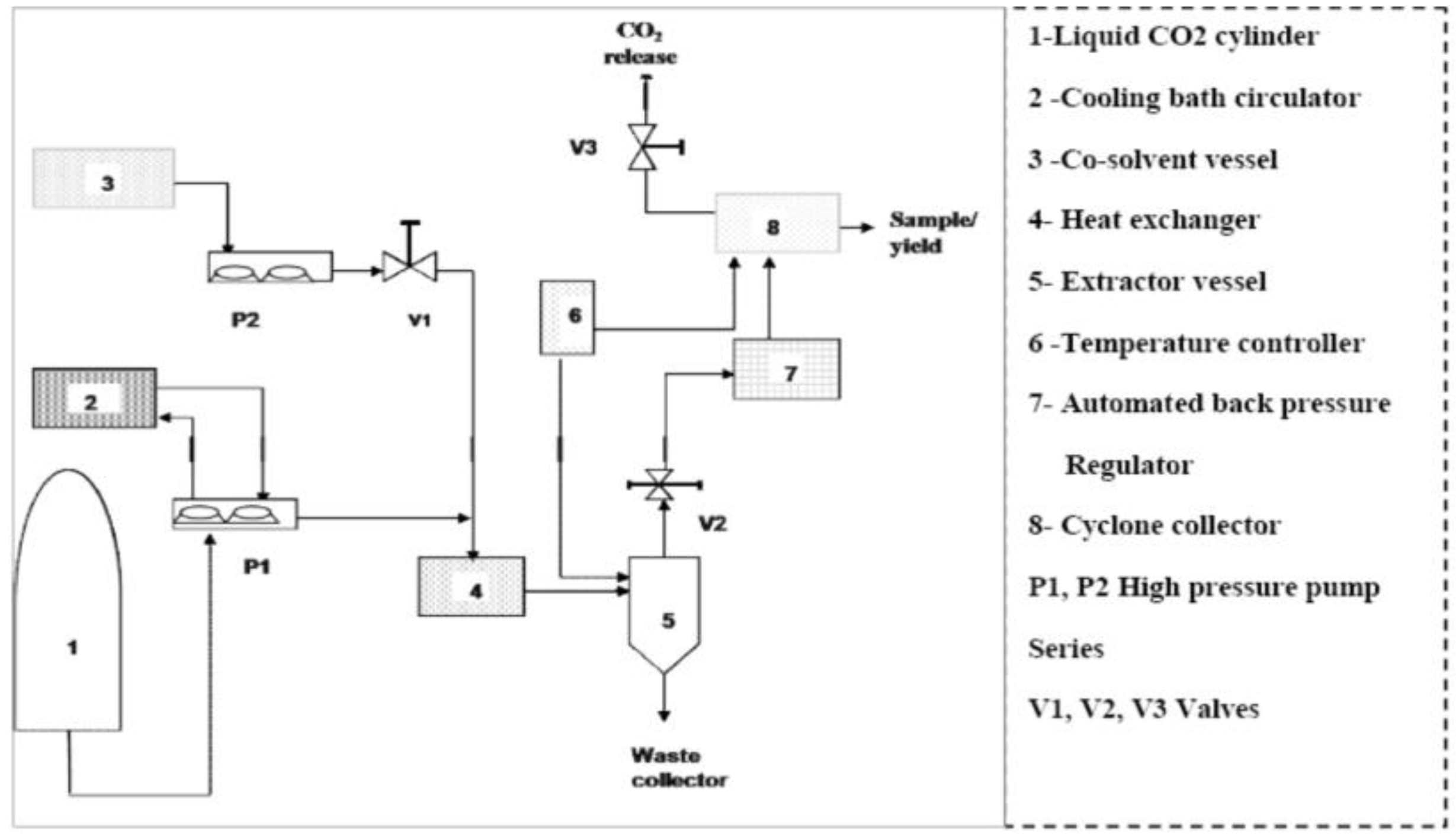
3.2. Supercritical Carbon Dioxide Extraction
3.3. Crude Extraction Yield Measurement
3.4. Determination of Radical Scavenging Activity
3.4.1. Determination of DPPH˙ Radical Scavenging Activity
3.4.2. Determination of ABTS˙+ Radical Scavenging Activity
3.5. Preparation of Fatty Acid Methyl Esters
3.6. Gas Chromatography Analysis
3.7. Experimental Design and Statistical Analysis
4. Conclusions
Acknowledgments
References
- Valko, M.; Leibfritz, D.; Moncol, J.; Cronin, M.T.D.; Mazur, M.; Telser, J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007, 39, 44–84. [Google Scholar] [CrossRef] [PubMed]
- Dalle-Donne, I.; Rossi, R.; Colombo, R.; Giustarini, D.; Milzani, A. Biomarkers of oxidative damage in human disease. Clin. Chem. 2006, 52, 601–623. [Google Scholar] [CrossRef] [PubMed]
- Simopoulos, A. Omega-3 fatty acids and antioxidants in edible wild plants. Biol. Res. 2004, 37, 263–277. [Google Scholar] [CrossRef] [PubMed]
- Erkan, N.; Ayranci, G.; Ayranci, E. Antioxidant activities of rosemary (Rosmarinus Officinalis L.) extract, blackseed (Nigella sativa L.) essential oil, carnosic acid, rosmarinic acid and sesamol. Food Chem. 2008, 110, 76–82. [Google Scholar] [CrossRef] [PubMed]
- Dawidowicz, A.L.; Wianowska, D.; Baraniak, B. The antioxidant properties of alcoholic extracts from Sambucus nigra L. (antioxidant properties of extracts). LWT Food Sci. Technol. 2006, 39, 308–315. [Google Scholar] [CrossRef]
- Mohd Zaini, N.A.; Anwar, F.; Abdul Hamid, A.; Saari, N. Kundur [Benincasa hispida (Thunb.) Cogn.]: A potential source for valuable nutrients and functional foods. Food Res. Intern. 2011, 14, 2368–2376. [Google Scholar] [CrossRef]
- Morton, J.F. The wax gourd, a year-round Florida vegetable with unusual keeping quality. Proc. FL State Hortic. Soc. 1971, 84, 104–109. [Google Scholar]
- Raveendra, R.K.; Martin, P. Ethnomedicinal Plants; Agrobios: Jodhpur, India, 2006; p. 108. [Google Scholar]
- Yagnik, B.; Jitendra, V.; Nurudin, J.; Nilesh, K.; Rameshvar, P.; Natavarlal, P. Antioxidant activity of Benincasa hispida on renal ischemia/reperfusion injury. Pharm. Online 2009, 1, 44–49. [Google Scholar]
- Stevigny, C.; Rolle, L.; Valentini, N.; Zeppa, G. Optimization of extraction of phenolic content from hazelnut shell using response surface methodology. J. Sci. Food Agric. 2007, 87, 2817–2822. [Google Scholar] [CrossRef]
- Bimakr, M.; Rahman, R.A.; Ganjloo, A.; Taip, F.S.; Saleh, L.M.; Sarker, Z.I. Optimization of supercritical carbon dioxide extraction of bioactive flavonoid compounds from spearmint (Mentha spicata L.) leaves by using response surface methodology. Food Bioprocess Technol. 2012, 5, 912–920. [Google Scholar] [CrossRef]
- Sajfrtová, M.; Ličková, I.; Wimmerová, M.; Sovová, H.; Zdeněk, W. β-sitosterol: Supercritical carbon dioxide extraction from sea buckthorn (Hippophae rhamnoides L.) seeds. Int. J. Mol. Sci. 2010, 11, 1842–1850. [Google Scholar] [CrossRef] [PubMed]
- Gomes, P.B.; Mata, V.G.; Rodrigues, A.E. Production of rose geranium oil using supercritical fluid extraction. J. Supercrit. Fluids 2007, 41, 50–60. [Google Scholar] [CrossRef]
- Liu, W.; Yu, Y.; Yang, R.; Wan, C.; Xu, B.; Cao, S. Optimization of total flavonoid compound extraction from Gynura medica leaf using response surface methodology and chemical composition analysis. Int. J. Mol. Sci. 2010, 11, 4750–4763. [Google Scholar] [CrossRef] [PubMed]
- Liza, M.S.; Abdul Rahman, R.A.; Mandana, B.; Jinap, S.; Rahmat, A.; Zaidul, I.; Hamid, A. Supercritical carbon dioxide extraction of bioactive flavonoid from Strobilanthes crispus (Pecah Kaca). Food Bioprod. Process 2010, 88, 319–326. [Google Scholar] [CrossRef]
- Gomez, A.M.; Lopez, C.P.; de la Ossa, E.M. Recovery of grape seed oil by liquid and supercritical carbon dioxide extraction: A comparison with conventional solvent extraction. Chem. Eng. J. 1996, 61, 227–231. [Google Scholar]
- Gerard, D.; May, P. Herb and spice carbon dioxide extracts-versatile, safe ingredients for premium food and health food. Food Technol. 2002, 2, 1–5. [Google Scholar]
- Bernado-Gil, M.G.; Grenha, J.; Santos, J.; Gardos, P. Supercritical fluid extraction and characterisation of oil from hazelnut. Eur. J. Lipid Sci. Tech. 2002, 104, 402–409. [Google Scholar] [CrossRef]
- Cao, X.; Ito, Y. Supercritical fluid extraction of grape seed oil and subsequent separation of free fatty acids by high-speed counter-current chromatography. J. Chromatogr. A 2003, 1021, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Vaughn Katherine, L.S.; Clausen Edgar, C.; King Jerry, W.; Howard Luke, R.; Julie, C.D. Extraction conditions affecting supercritical fluid extraction (SFE) of lycopene from watermelon. Bioresour. Technol. 2008, 99, 7835–7841. [Google Scholar] [CrossRef] [PubMed]
- Benelli, P.; Riehl, C.A.S.; Smânia, A., Jr.; Smânia, E.F.A.; Ferreira, S.R.S. Bioactive extracts of orange (Citrus sinensis L. Osbeck) pomace obtained by SFE and low pressure techniques: Mathematical modeling and extract composition. J. Supercrit. Fluids 2010, 55, 132–141. [Google Scholar] [CrossRef]
- Herrero, M.; Mendiola, J.A.; Cifuentes, A.; Ibáñez, E. Supercritical fluid extraction: Recent advances and applications. J. Chromatogr. A 2010, 1217, 2495–2511. [Google Scholar] [CrossRef] [PubMed]
- Kviecinski, M.R.; Benelli, P.; Felipe, K.B.; Correia, J.F.G.; Pich, C.T.; Ferreira, S.R.S.; Pedrosa, R.C. SFE from Bidens pilosa Linné to obtain extracts rich in cytotoxic polyacetylenes with antitumor activity. J. Supercrit. Fluids 2011, 56, 243–248. [Google Scholar] [CrossRef]
- Orio, L.; Alexandru, L.; Cravotto, G.; Mantegna, S.; Barge, A. UAE, MAE, SFE-CO2 and classical methods for the extraction of Mitragyna speciosa leaves. Ultrason. Sonochem. 2012, 19, 591–595. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Vicente, Y.; Cabañas, A.; Renuncio, J.A.R.; Pando, C. The supercritical fluid extraction of peach (Prunus persica) seed oil using carbon dioxide and ethanol. J. Supercrit. Fluids 2009, 49, 167–173. [Google Scholar] [CrossRef]
- Ollanketo, M.; Peltoketo, A.; Hartonen, K.; Hiltunen, R.; Riekkola, H. Extraction of sage (Salvia officinalis L.) by pressurized hot water and conventional methods: Antioxidant activity of the extracts. Eur. Food Res. Technol. 2002, 215, 158–163. [Google Scholar] [CrossRef]
- Mónica, R.; García-Risco, E.J.H.; Vicente, G.; Fornari, T.; Señoráns, F.J.; Reglero, G. Kinetic study of pilot-scale supercritical CO2 extraction of rosemary (Rosmarinus officinalis) leaves. J. Supercrit. Fluids 2011, 55, 971–976. [Google Scholar]
- Veggi, P.C.; Cavalcanti, R.N.; Angela, M.; Meireles, A. Modifier effects on supercritical fluid extraction (SFE) of some Brazilian plants: antioxidant activity and economical evaluation. Procedia Food Sci. 2011, 1, 1717–1724. [Google Scholar] [CrossRef]
- Bas, D.; Boyaci, I.H. Modeling and optimization: Usability of response surface methodology. J. Food Eng. 2007, 78, 836–845. [Google Scholar] [CrossRef]
- Gottipati, R.; Mishra, S. Application of response surface methodology for optimization of Cr(III) and Cr(VI) adsorption on commercial activated carbons. Res. J. Chem. Sci. 2012, 2, 40–48. [Google Scholar]
- Rezaei, K.; Temelli, F. Using supercritical fluid chromatography to determine the binary diffusion coefficient of lipids in supercritical CO2. J. Supercrit. Fluids 2000, 17, 35–44. [Google Scholar] [CrossRef]
- Luengthanaphol, S.; Mongkholkhajornsilp, D.; Douglas, S.; Douglas, P.L.; Pengsopa, L.; Pongamphai, S. Extraction of antioxidants from sweet Thai tamarind seed coat, preliminary experiments. J. Food Eng. 2004, 63, 247–252. [Google Scholar] [CrossRef]
- Wei, Z.J.; Liao, M.A.; Zhang, H.X.; Liu, J.; Jiang, S.H. Optimization of supercritical carbon dioxide extraction of silkworm pupal oil applying the response surface methodology. Bioresour. Technol. 2009, 100, 4214–4219. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Fu, Y.J.; Zu, Y.G.; Tong, M.H.; Wu, N.; Liu, X.L. Supercritical carbon dioxide extraction of seed oil from Opuntia dillenii Haw. and its antioxidant activity. Food Chem. 2009, 114, 334–339. [Google Scholar] [CrossRef]
- Wang, L.; Yang, B.; Du, X.; Yi, C. Optimisation of supercritical fluid extraction of Flavonoids from Pueraria lobata. Food Chem. 2008, 108, 737–741. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lin, S.; Wang, Z.; Wang, C.; Wang, E.; Zhang, Y. Supercritical fluid extraction of flavonoids from Maydis stigma and its nitrite-scavenging ability. Food Bioprod. Process 2011, 89, 333–339. [Google Scholar] [CrossRef]
- Thana, P.; Machmudah, S.; Goto, M.; Sasaki, M.; Pavasant, P.; Shotipruk, A. Response surface methodology to supercritical carbon dioxide extraction of astaxanthin from Haematococcus pluvialis. Bioresour. Technol. 2008, 99, 3110–3115. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Weller, C.L.; Schlegel, V.L.; Carr, T.P.; Cuppett, S.L. Supercritical CO2 extraction of lipids from grain sorghum dried distillers grains with solubles. Bioresour. Technol. 2008, 99, 1373–1382. [Google Scholar] [CrossRef] [PubMed]
- Mitra, P.; Ramaswamy, H.S.; Chang, K.S. Pumpkin (Cucurbita maxima) seed oil extraction using supercritical carbon dioxide and physicochemical properties of the oil. J. Food Eng. 2009, 208, 208–213. [Google Scholar] [CrossRef]
- Bimakr, M.; Russly, A.R.; Farah, S.T.; Noranizan, M.A.; Zaidul, I.S.; Ali, G. Optimization of ultrasound-assisted extraction of crude oil from winter melon (Benincasa hispida) seed using response surface methodology and evaluation of its antioxidant activity, total phenolic content and fatty acid composition. Molecules 2012, 17, 11748–11762. [Google Scholar] [CrossRef] [PubMed]
- Bimakr, M.; Russly, A.R.; Farah, S.T.; Noranizan, M.A.; Zaidul, I.S.; Ali, G. Antioxidant activity of winter melon (Benincasa hispida) seeds using conventional Soxhlet extraction technique. Int. Food Res. J. 2012, 19, 229–234. [Google Scholar]
- Bimakr, M.; Russly, A.R.; Ali, G.; Farah, S.T.; Liza, S.; Jinap, S.; Hamid, A. Comparison of different extraction methods for the extraction of major bioactive flavonoid compounds from spearmint (Mentha spicata l.) leaves. Food Bioprod. Process 2011, 89, 67–72. [Google Scholar] [CrossRef]
- Rajaei, A.; Barzegar, M.; Yamini, Y. Supercritical fluid extraction of tea seed oil and its comparison with solvent extraction. Eur. Food Res. Technol. 2005, 220, 401–405. [Google Scholar] [CrossRef]
- Zengin, G.; Cakmak, Y.S.; Guler, G.O.; Aktumsek, A. In vitro antioxidant capacities and fatty acid compositions of three Centaurea species collected from Central Anatolia region of Turkey. Food Chem. Toxicol. 2010, 48, 2638–2641. [Google Scholar] [CrossRef] [PubMed]
- Cai, Y.; Luo, Q.; Sun, M.; Corke, H. Antioxidant activity and phenolic compounds of 112 traditional Chinese medicinal plants associated with anticancer. Life Sci. 2004, 74, 2157–2184. [Google Scholar] [CrossRef] [PubMed]
- Chua, S.C.; Tan, C.P.; Mirhosseini, H.; Lai, O.M.; Long, K.; Baharin, B.S. Optimization of ultrasound extraction condition of phospholipids from palm-pressed fiber. J. Food Eng. 2009, 92, 403–409. [Google Scholar] [CrossRef]
- Deng, Y.; Zhao, Y. Effect of pulsed vacuum and ultrasound osmo pretreatments on glass transition temperature, texture, microstructure and calcium penetration of dried apples (Fuji). LWT Food Sci. Technol. 2008, 41, 1575–1585. [Google Scholar] [CrossRef]
Sample Availability: Samples of the winter melon oil, extracted by SC-SO2 and Soxhlet method, are available from the authors. |
| Run | Block | Process variables | CEY (mg-extract/g-dried sample) | ||
|---|---|---|---|---|---|
| Pressure (bar) | Temperature (°C) | Dynamic extraction time (min) | |||
| 1 | 1 | 271 (+1) | 48 (+1) | 72 (−1) | 159.24 |
| 2 | 1 | 271 (+1) | 48 (+1) | 108 (+1) | 161.88 |
| 3 (Cp) | 1 | 225 (0) | 45 (0) | 90 (0) | 172.43 |
| 4 (Cp) | 1 | 225 (0) | 45 (0) | 90 (0) | 172.85 |
| 5 | 1 | 179 (-1) | 48 (+1) | 72 (−1) | 132.73 |
| 6 | 1 | 179 (-1) | 42(−1) | 108 (+1) | 128.18 |
| 7 (Cp) | 1 | 225 (0) | 45 (0) | 90 (0) | 173.22 |
| 8 | 1 | 179 (-1) | 42 (−1) | 72 (−1) | 125.62 |
| 9 | 1 | 179 (-1) | 48 (+1) | 108 (+1) | 140.00 |
| 10 (Cp) | 1 | 225 (0) | 45 (0) | 90 (0) | 171.87 |
| 11 | 1 | 271 (+1) | 42 (−1) | 72 (−1) | 161.12 |
| 12 | 1 | 271 (+1) | 42 (−1) | 108 (+1) | 161.20 |
| 13 | 2 | 225 (0) | 45 (0) | 120 (+1.63) | 173.10 |
| 14 (Cp) | 2 | 225 (0) | 45 (0) | 90 (0) | 173.33 |
| 15 | 2 | 225 (0) | 50 (+1.63) | 90 (0) | 137.50 |
| 16 (Cp) | 2 | 225 (0) | 45 (0) | 90 (0) | 173.87 |
| 17 | 2 | 150 (−1.63) | 45 (0) | 90 (0) | 119.11 |
| 18 | 2 | 225 (0) | 40 (−1.63) | 90 (0) | 127.00 |
| 19 | 2 | 300 (+1.63) | 45 (0) | 90 (0) | 168.53 |
| 20 | 2 | 225 (0) | 45 (0) | 60 (−1.63) | 166.70 |
| Source | df a | CEY(mg-extract/g-dried sample) | ||
|---|---|---|---|---|
| Coefficient | Sum of squares | p-Value | ||
| Model | 9 | 172.88 | 7,392.83 | <0.0001 |
| X1 | 1 | 14.82 | 2,928.80 | <0.0001 |
| X2 | 1 | 2.62 | 91.23 | <0.0001 |
| X3 | 1 | 1.73 | 39.68 | 0.0001 |
| X12 | 1 | −10.77 | 1,532.34 | <0.0001 |
| X22 | 1 | −15.11 | 3,015.37 | <0.0001 |
| X32 | 1 | −0.99 | 12.99 | 0.0052 |
| X1X2 | 1 | −2.52 | 50.65 | <0.0001 |
| X1X3 | 1 | −0.89 | 6.32 | 0.0307 |
| X2X3 | 1 | 0.91 | 6.61 | 0.0280 |
| Residual | 9 | 8.68 | ||
| Lack of fit | 5 | 7.53 | 0.0673 | |
| Pure error | 4 | 1.15 | ||
| Total | 19 | 7,401.60 | ||
| R2 | 0.998 | |||
| Adjusted- R2 | 0.997 | |||
| C.V.% | 0.630 | |||
| E (%) | 0.350 | |||
| Extraction Mode | CEY (mg-extract/g-dried sample) | Antioxidant activity | Fatty acid composition * | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| %DPPHsc | %ABTSsc | C14:00 a | C16:00 b | C16:01 c | C18:00 d | C18:01 e | C18:02 f | C18:03 g | ΣSFA h | ΣUFA i | ||
| CSE (EtOH, 99.5%) | 250.00 ± 1.30 | 28.70 ± 0.70 | 27.00 ± 0.90 | 1.60 ± 0.11 | 15.30 ± 0.15 | 0.68 ± 0.27 | 7.40 ± 0.18 | 14.10 ± 0.15 | 60.60 ± 0.13 | - | 24.30 | 75.38 |
| UAE (EtOH, 99.5%) | 108.62 ± 0.78 | 35.84 ± 0.42 | 43.10 ± 0.63 | 1.30 ± 0.11 | 10.80 ± 0.25 | 0.80 ± 0.33 | 5.70 ± 0.14 | 14.40 ± 0.22 | 66.20 ± 0.10 | 0.60 ± 0.12 | 17.80 | 82.00 |
| SCE (SC-CO2+EtOH) | 175.60 ± 0.33 | 53.20 ± 0.54 | 62.22 ± 0.25 | 1.07 ± 0.18 | 9.83 ± 0.33 | 0.95 ± 0.13 | 5.07 ± 0.21 | 14.73 ± 0.10 | 67.17 ± 0.15 | 0.68 ± 0.12 | 15.97 | 83.53 |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Bimakr, M.; Rahman, R.A.; Taip, F.S.; Adzahan, N.M.; Sarker, M.Z.I.; Ganjloo, A. Supercritical Carbon Dioxide Extraction of Seed Oil from Winter Melon (Benincasa hispida) and Its Antioxidant Activity and Fatty Acid Composition. Molecules 2013, 18, 997-1014. https://doi.org/10.3390/molecules18010997
Bimakr M, Rahman RA, Taip FS, Adzahan NM, Sarker MZI, Ganjloo A. Supercritical Carbon Dioxide Extraction of Seed Oil from Winter Melon (Benincasa hispida) and Its Antioxidant Activity and Fatty Acid Composition. Molecules. 2013; 18(1):997-1014. https://doi.org/10.3390/molecules18010997
Chicago/Turabian StyleBimakr, Mandana, Russly Abdul Rahman, Farah Saleena Taip, Noranizan Mohd Adzahan, Md. Zaidul Islam Sarker, and Ali Ganjloo. 2013. "Supercritical Carbon Dioxide Extraction of Seed Oil from Winter Melon (Benincasa hispida) and Its Antioxidant Activity and Fatty Acid Composition" Molecules 18, no. 1: 997-1014. https://doi.org/10.3390/molecules18010997
APA StyleBimakr, M., Rahman, R. A., Taip, F. S., Adzahan, N. M., Sarker, M. Z. I., & Ganjloo, A. (2013). Supercritical Carbon Dioxide Extraction of Seed Oil from Winter Melon (Benincasa hispida) and Its Antioxidant Activity and Fatty Acid Composition. Molecules, 18(1), 997-1014. https://doi.org/10.3390/molecules18010997




