(3S)-1,2,3,4-Tetrahydro-β-carboline-3-carboxylic Acid from Cichorium endivia. L Induces Apoptosis of Human Colorectal Cancer HCT-8 Cells
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structure Elucidation of Compound 1
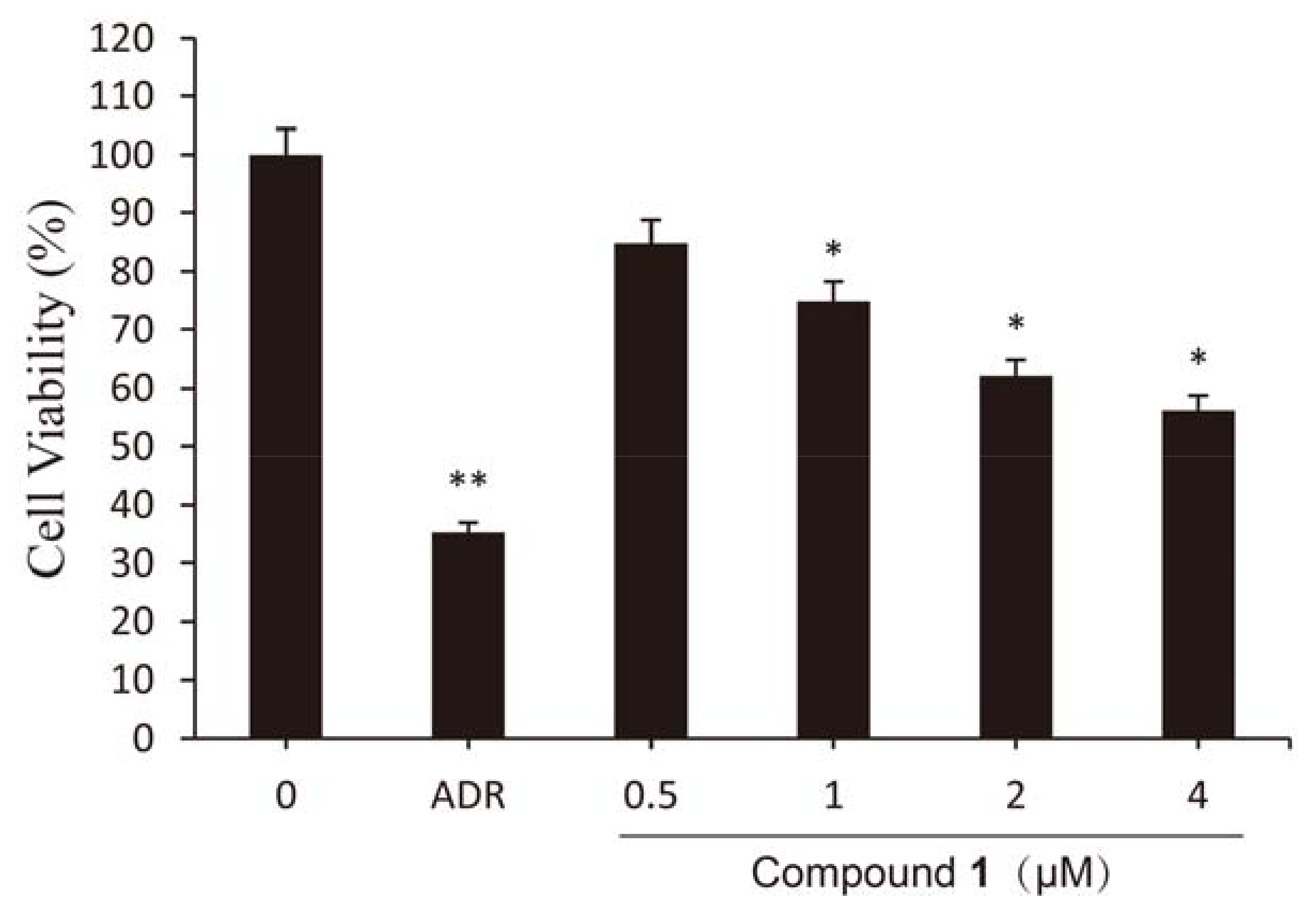
2.2. Effect of Compound 1 on Viability of HCT-8 Cells
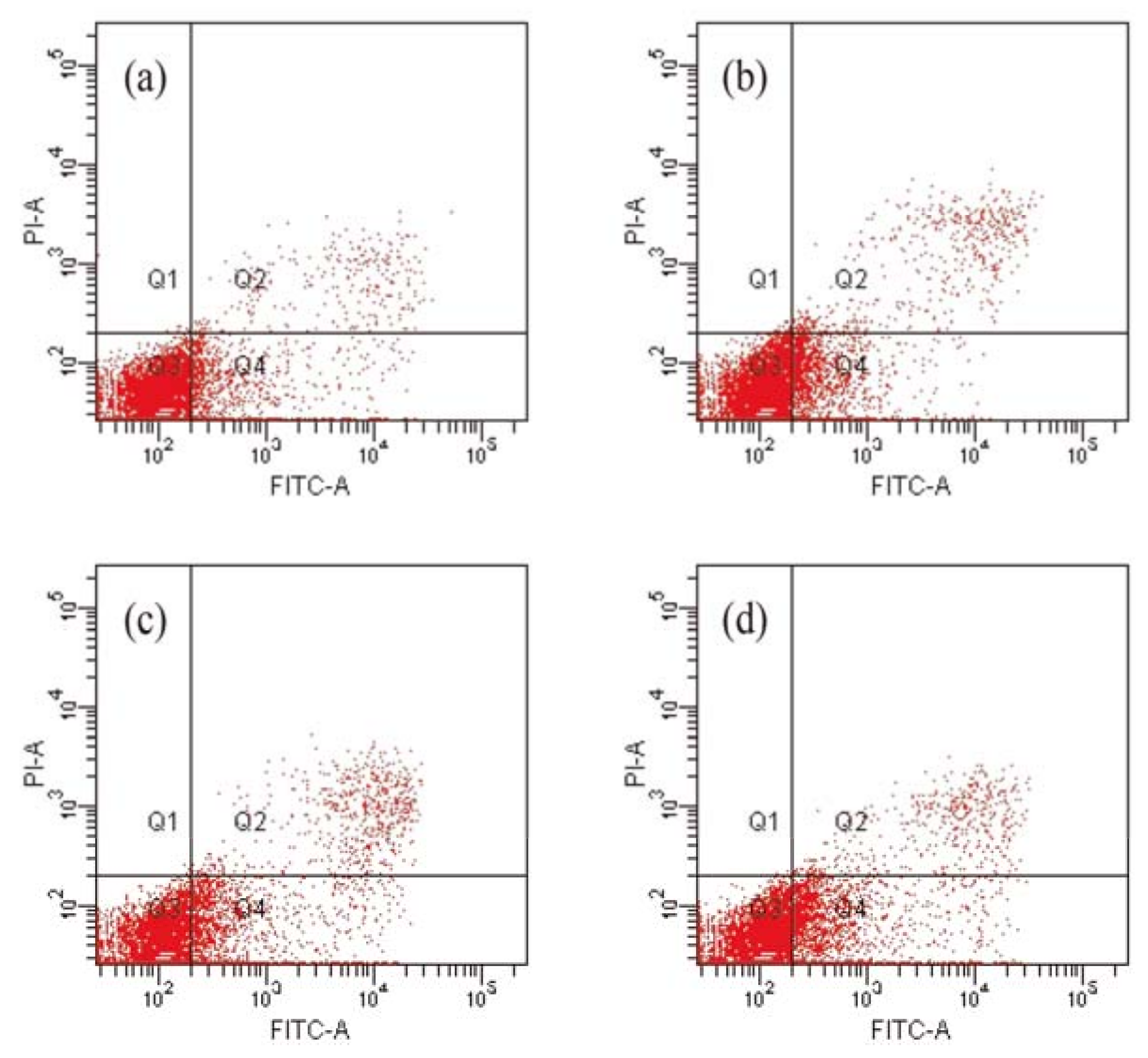
2.3. Compound 1 Induced the Apoptosis of HCT-8 Cells
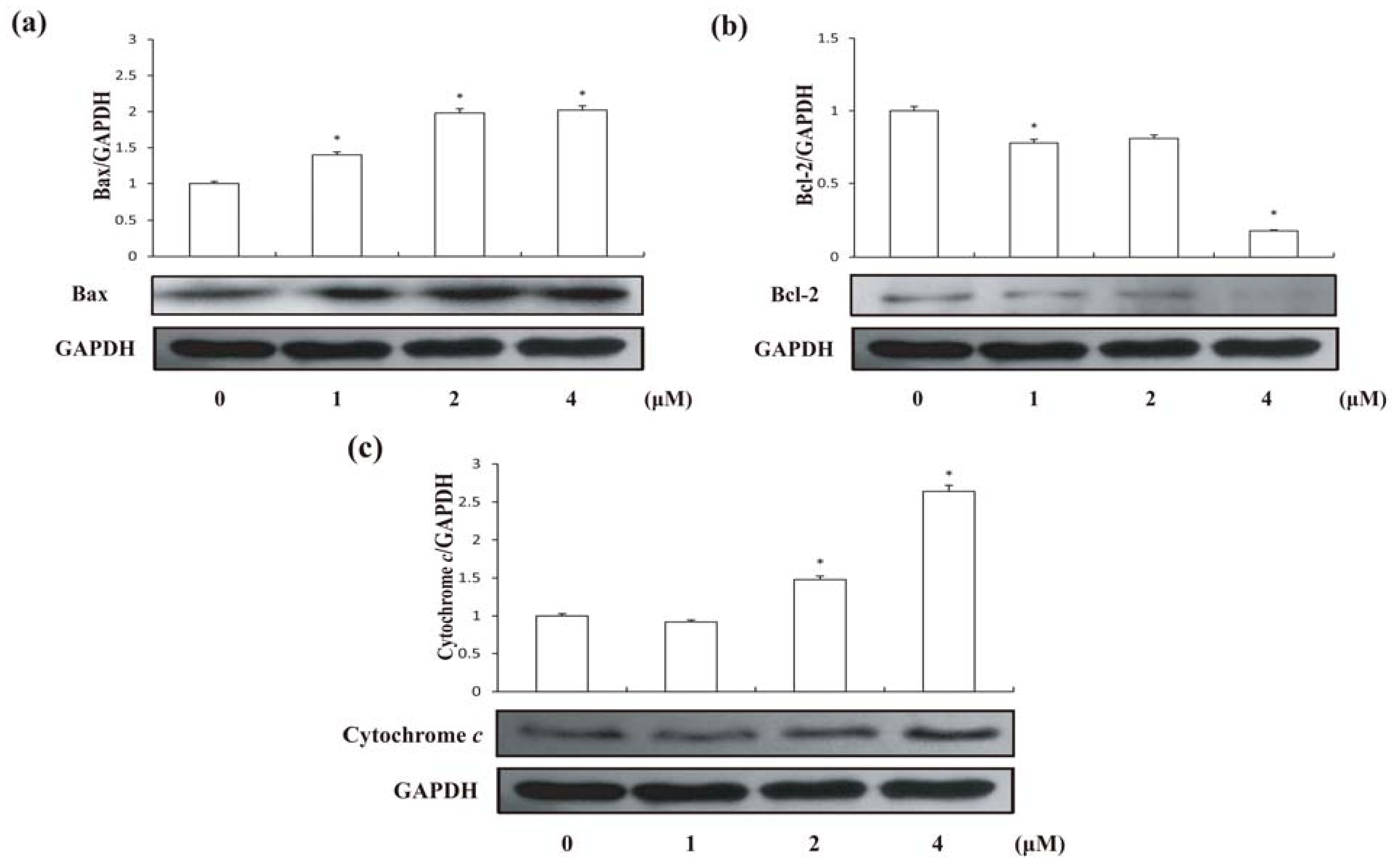
2.4. Effects of Compound 1 on Expression of Bax and Bcl-2 Proteins in HCT-8 Cells
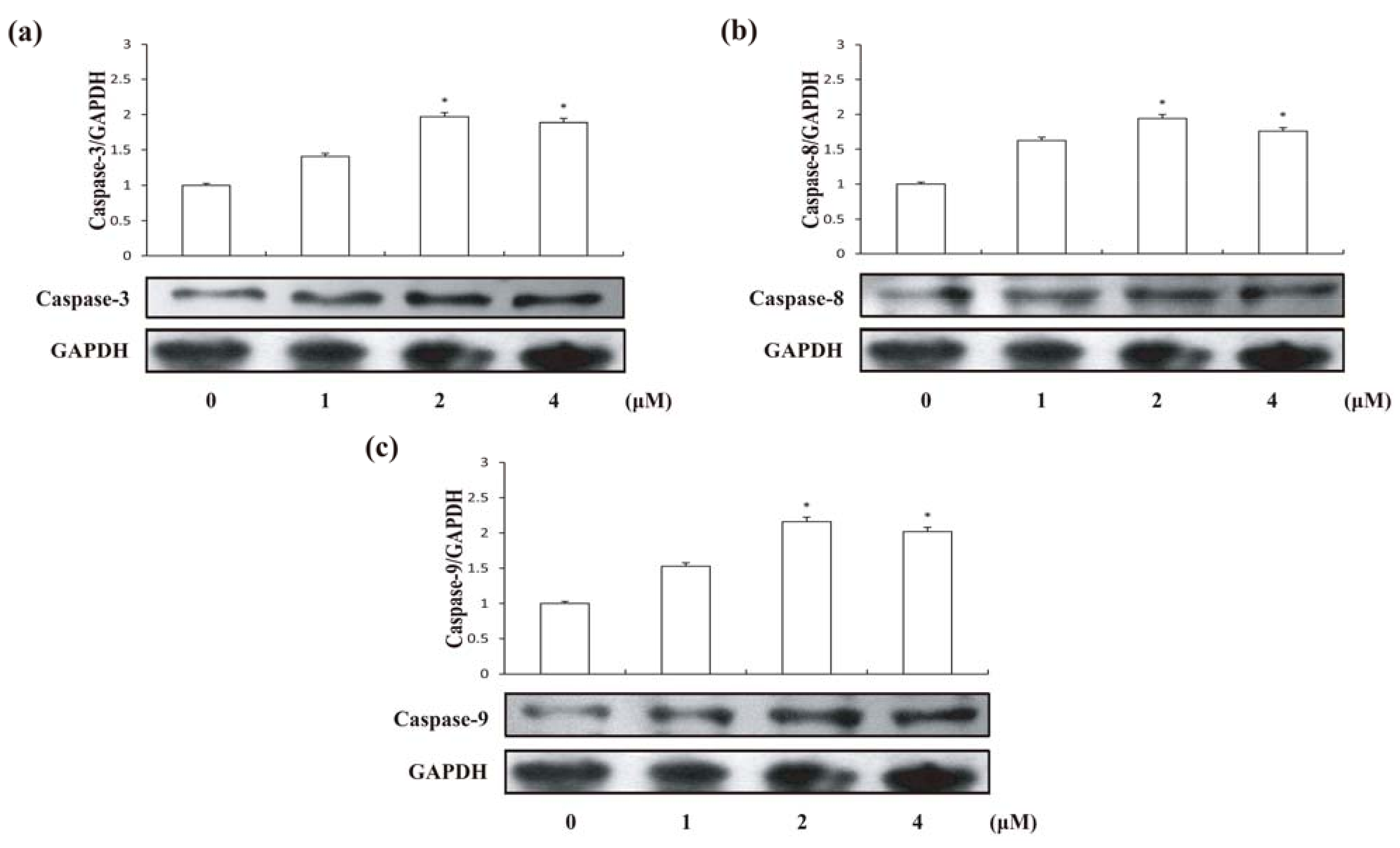
2.5. Effects of Compound 1 on Caspase Activities
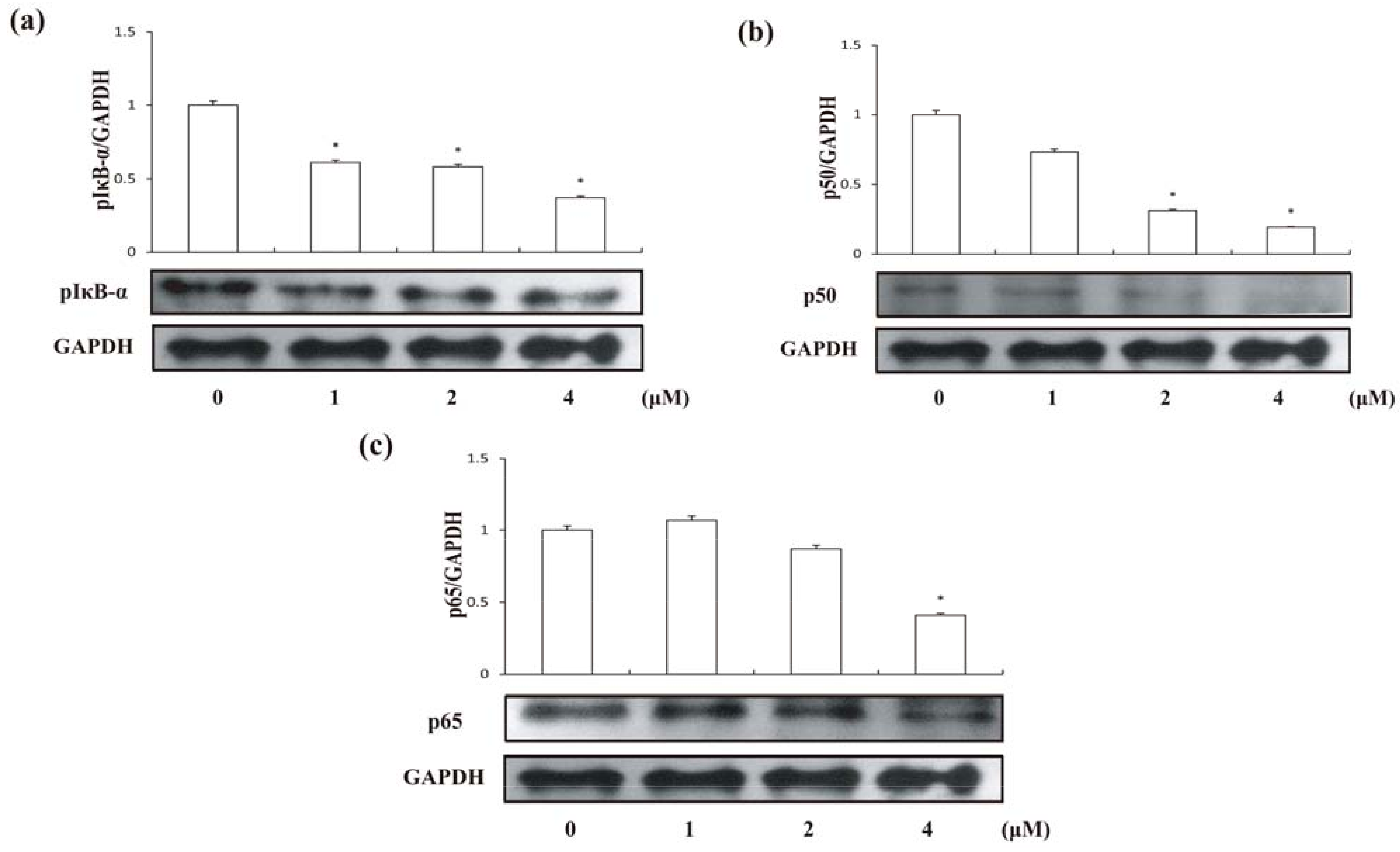
2.6. Compound 1 suppressed the Activation of NF-κB
3. Experimental
3.1. Materials
3.2. Cell Culture
3.3. Cell Viability Test (SRB assay)
3.4. Assessment of Apoptosis on HCT-8 Cells by Annexin V/PI Staining
3.5. Analysis of Mitochondrial Membrane Potential
3.6. Western Blot Analysis
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Koudela, M.; Petříková, K. Nutritional composition and yield of endive cultivars—Cichorium endivia L. Hort. Sci. 2007, 34, 6–10. [Google Scholar] [CrossRef]
- DuPont, M.S.; Mondin, Z.; Williamson, G.; Price, K.R. Effect of variety, processing, and storage on the flavonoid glycoside content and composition of lettuce and endive. J. Agric. Food Chem. 2000, 48, 3957–3964. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.J.; Deng, A.J.; Liu, C.; Shi, R.; Qin, H.L.; Wang, A.P. Hepatoprotective activity of Cichorium endivia L. extract and its chemical constituents. Molecules 2011, 16, 9049–9066. [Google Scholar] [CrossRef] [PubMed]
- Seto, M.; Miyase, T.; Umehara, K.; Ueno, A.; Hirano, Y.; Otani, N. Sesquiterpene lactones from Cichorium endivia L. and C. intybus L. and cytotoxic activity. Chem. Pharm. Bull. 1988, 36, 2423–2429. [Google Scholar] [CrossRef] [PubMed]
- Kisiel, W.; Michalska, K. Sesquiterpenoids and phenolics from roots of Cichorium endivia var. crispum. Fitoterapia 2006, 77, 354–357. [Google Scholar] [CrossRef] [PubMed]
- Papetti, A.; Daglia, M.; Aceti, C.; Sordelli, B.; Spini, V.; Carazzone, C.; Gazzani, G. Hydroxycinnamic acid derivatives occurring in Cichorium endivia vegetables. J. Pharm. Biomed. 2008, 48, 472–476. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Siegel, R.; Ward, E.; Hao, Y.; Xu, J.; Murray, T.; Thun, M.J. Cancer statistics, 2008. CA Cancer J. Clin. 2008, 58, 71–96. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.J.; Tang, W.Y.; Hsu, C.W.; Tsai, Y.T.; Wu, J.F.; Lin, C.W.; Cheng, Y.M.; Hsu, Y.C. Apoptosis Induction in Primary Human Colorectal Cancer Cell Lines and Retarded Tumor Growth in SCID Mice by Sulforaphane. Evid. Based Complement. Alternat. Med. 2012, 2012, 415231. [Google Scholar] [CrossRef] [PubMed]
- Traka, M.H.; Spinks, C.A.; Doleman, J.F.; Melchini, A.; Ball, R.Y.; Mills, R.D.; Mithen, R.F. The dietary isothiocyanate sulforaphane modulates gene expression and alternative gene splicing in a PTEN null preclinical murine model of prostate cancer. Mol. Cancer 2010, 9, 189. [Google Scholar] [CrossRef] [PubMed]
- Elmore, S. Apoptosis: A review of programmed cell death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef] [PubMed]
- Wongsirisin, P.; Yodkeeree, S.; Pompimon, W.; Limtrakul, P. Induction of G1 Arrest and Apoptosis in Human Cancer Cells by Crebanine, an Alkaloid from Stephania venosa. Chem. Pharm. Bull. 2012, 60, 1283–1289. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Singh, S.V. D,L-Sulforaphane causes transcriptional repression of androgen receptor in human prostate cancer cells. Mol. Cancer Ther. 2009, 8, 1946–1954. [Google Scholar] [CrossRef] [PubMed]
- Mao, L.; Wang, H.D.; Wang, X.L.; Qiao, L.; Yin, H.X. Sulforaphane attenuates matrix metalloproteinase-9 expression following spinal cord injury in mice. Ann. Clin. Lab. Sci. 2010, 40, 354–360. [Google Scholar] [PubMed]
- Yahara, S.; Uda, N.; Yoshio, E.; Yae, E. Steroidal alkaloid glycosides from tomato (Lycopersicon esculentum). J. Nat. Prod. 2004, 67, 500–502. [Google Scholar] [CrossRef] [PubMed]
- Campisi, J. Cellular senescence and apoptosis: How cellular responses might influence aging phenotypes. Exp. Gerontol. 2003, 38, 5–11. [Google Scholar] [CrossRef]
- Fischer, U.; Schulze-Osthoff, K. Apoptosis-based therapies and drug targets. Cell Death Differ. 2005, 12, 942–961. [Google Scholar] [CrossRef] [PubMed]
- Gross, A.; McDonnell, J.M.; Korsmeyer, S.J. BCL-2 family members and the mitochondria in apoptosis. Genes Dev. 1999, 13, 1899–1911. [Google Scholar] [CrossRef] [PubMed]
- Green, D.R.; Reed, J.C. Mitochondria and apoptosis. Science 1998, 281, 1309–1312. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, E.; Armour, S.M.; Harris, M.H.; Thompson, C.B. Mitochondrial membrane potential regulates matrix configuration and cytochrome c release during apoptosis. Cell Death Differ. 2003, 10, 709–717. [Google Scholar] [CrossRef] [PubMed]
- Kim, R. Recent advances in understanding the cell death pathways activated by anticancer therapy. Cancer 2005, 103, 1551–1560. [Google Scholar] [CrossRef] [PubMed]
- Hsu, W.H.; Lee, B.H.; Pan, T.M. Effects of red mold dioscorea on oral carcinogenesis in DMBA-induced hamster animal model. Food Chem. Toxicol. 2011, 49, 1292–1297. [Google Scholar] [CrossRef] [PubMed]
- Stennicke, H.R.; Jurgensmeier, J.M.; Shin, H.; Deveraux, Q.; Wolf, B.B.; Yang, X.; Zhou, Q.; Ellerby, H.M.; Ellerby, L.M.; Bredesen, D.; et al. Pro-caspase-3 is a major physiologic target of caspase-8. J. Biol. Chem. 1998, 273, 27084–27090. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P.; Camandola, S. NF-kappaB in neuronal plasticity and neurodegenerative disorders. J. Clin. Invest. 2001, 107, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Zhang, X.; Li, J.J. The role of NF-kappaB in the regulation of cell stress responses. Int. Immunopharmacol. 2002, 2, 1509–1520. [Google Scholar] [CrossRef]
- Osborn, L.; Kunkel, S.; Nabel, G.J. Tumor necrosis factor alpha and interleukin 1 stimulate the human immunodeficiency virus enhancer by activation of the nuclear factor kappa B. Proc. Natl. Acad. Sci. USA 1989, 86, 2336–2340. [Google Scholar] [CrossRef] [PubMed]
- Schreck, R.; Rieber, P.; Baeuerle, P.A. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO J. 1991, 10, 2247–2258. [Google Scholar] [CrossRef] [PubMed]
- Perera, P.Y.; Qureshi, N.; Vogel, S.N. Paclitaxel (Taxol)-induced NF-kappaB translocation in murine macrophages. Infect. Immun. 1996, 64, 878–884. [Google Scholar] [PubMed]
- Zhong, H.; SuYang, H.; Erdjument-Bromage, H.; Tempst, P.; Ghosh, S. The transcriptional activity of NF-kappaB is regulated by the IkappaB-associated PKAc subunit through a cyclic AMP-independent mechanism. Cell 1997, 89, 413–424. [Google Scholar] [CrossRef]
- Scheinman, R.I.; Cogswell, P.C.; Lofquist, A.K.; Baldwin, A.S., Jr. Role of transcriptional activation of I kappa B alpha in mediation of immunosuppression by glucocorticoids. Science 1995, 270, 283–286. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
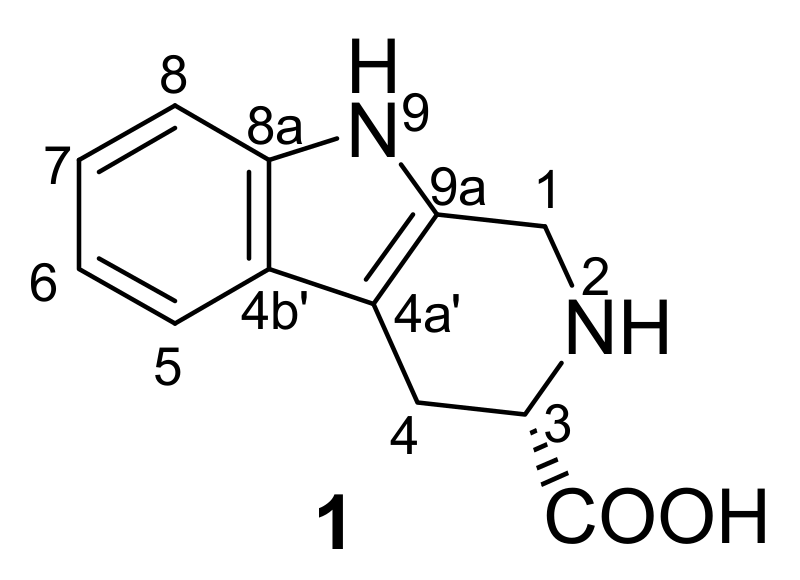

| No. | δH ab | δC ac | δH de |
|---|---|---|---|
| 1a | 4.38 d (15.6) | 40.5 t | 4.15 d (15.3) |
| 1b | 4.54 d (15.6) | 4.23 d (15.9) | |
| 3 | 4.27 dd (10.4, 5.6) | 55.0 d | 3.60 m |
| 4a | 3.13 dd (10.8, 16.4) | 21.7 t | 2.81 dd-like |
| 4b | 3.38 dd (5.6, 16.4) | 3.13 br d-like | |
| 4a′ | 104.9 s | ||
| 4b′ | 125.6 f s | ||
| 5 | 7.53 d (8.0) | 118.2 d | 7.32 d (7.5) |
| 6 | 7.13 t (7.6) | 120.0 d | 7.06 t (7.5) |
| 7 | 7.22 t (8.0) | 122.9 d | 6.97 t (7.8) |
| 8 | 7.43 d (8.0) | 111.9 d | 7.43 d (7.5) |
| 8a | 136.7 s | ||
| 9a | 125.4 fs | ||
| COOH | 171.3 s | 10.91 s | |
| 9-NH |
© 2013 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, F.-X.; Deng, A.-J.; Li, M.; Wei, J.-F.; Qin, H.-L.; Wang, A.-P. (3S)-1,2,3,4-Tetrahydro-β-carboline-3-carboxylic Acid from Cichorium endivia. L Induces Apoptosis of Human Colorectal Cancer HCT-8 Cells. Molecules 2013, 18, 418-429. https://doi.org/10.3390/molecules18010418
Wang F-X, Deng A-J, Li M, Wei J-F, Qin H-L, Wang A-P. (3S)-1,2,3,4-Tetrahydro-β-carboline-3-carboxylic Acid from Cichorium endivia. L Induces Apoptosis of Human Colorectal Cancer HCT-8 Cells. Molecules. 2013; 18(1):418-429. https://doi.org/10.3390/molecules18010418
Chicago/Turabian StyleWang, Fu-Xin, An-Jun Deng, Ming Li, Jin-Feng Wei, Hai-Lin Qin, and Ai-Ping Wang. 2013. "(3S)-1,2,3,4-Tetrahydro-β-carboline-3-carboxylic Acid from Cichorium endivia. L Induces Apoptosis of Human Colorectal Cancer HCT-8 Cells" Molecules 18, no. 1: 418-429. https://doi.org/10.3390/molecules18010418
APA StyleWang, F.-X., Deng, A.-J., Li, M., Wei, J.-F., Qin, H.-L., & Wang, A.-P. (2013). (3S)-1,2,3,4-Tetrahydro-β-carboline-3-carboxylic Acid from Cichorium endivia. L Induces Apoptosis of Human Colorectal Cancer HCT-8 Cells. Molecules, 18(1), 418-429. https://doi.org/10.3390/molecules18010418




