Novel Coumarin Derivatives Containing 1,2,4-Triazole, 4,5-Dicyanoimidazole and Purine Moieties: Synthesis and Evaluation of Their Cytostatic Activity
Abstract
:1. Introduction
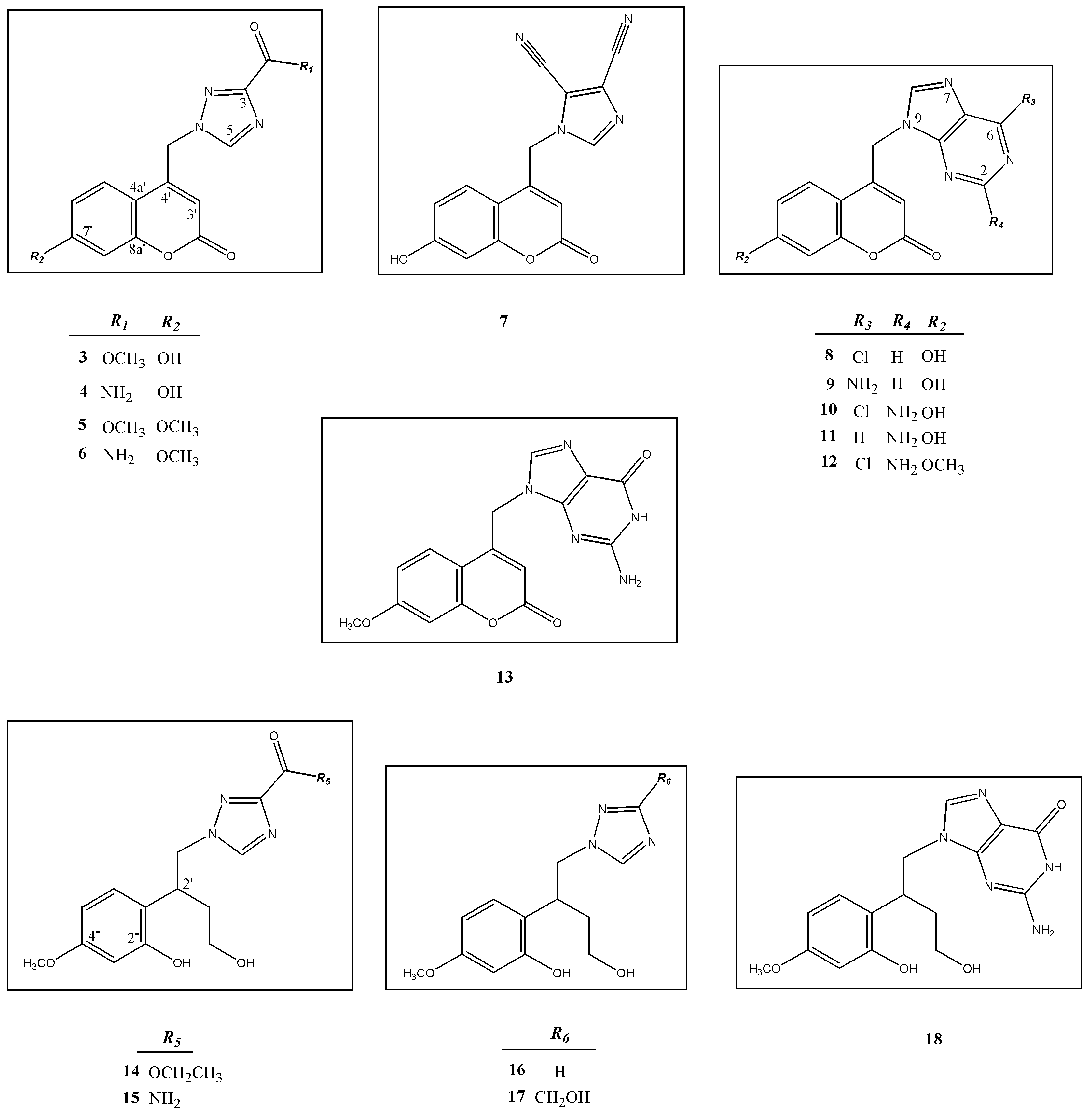
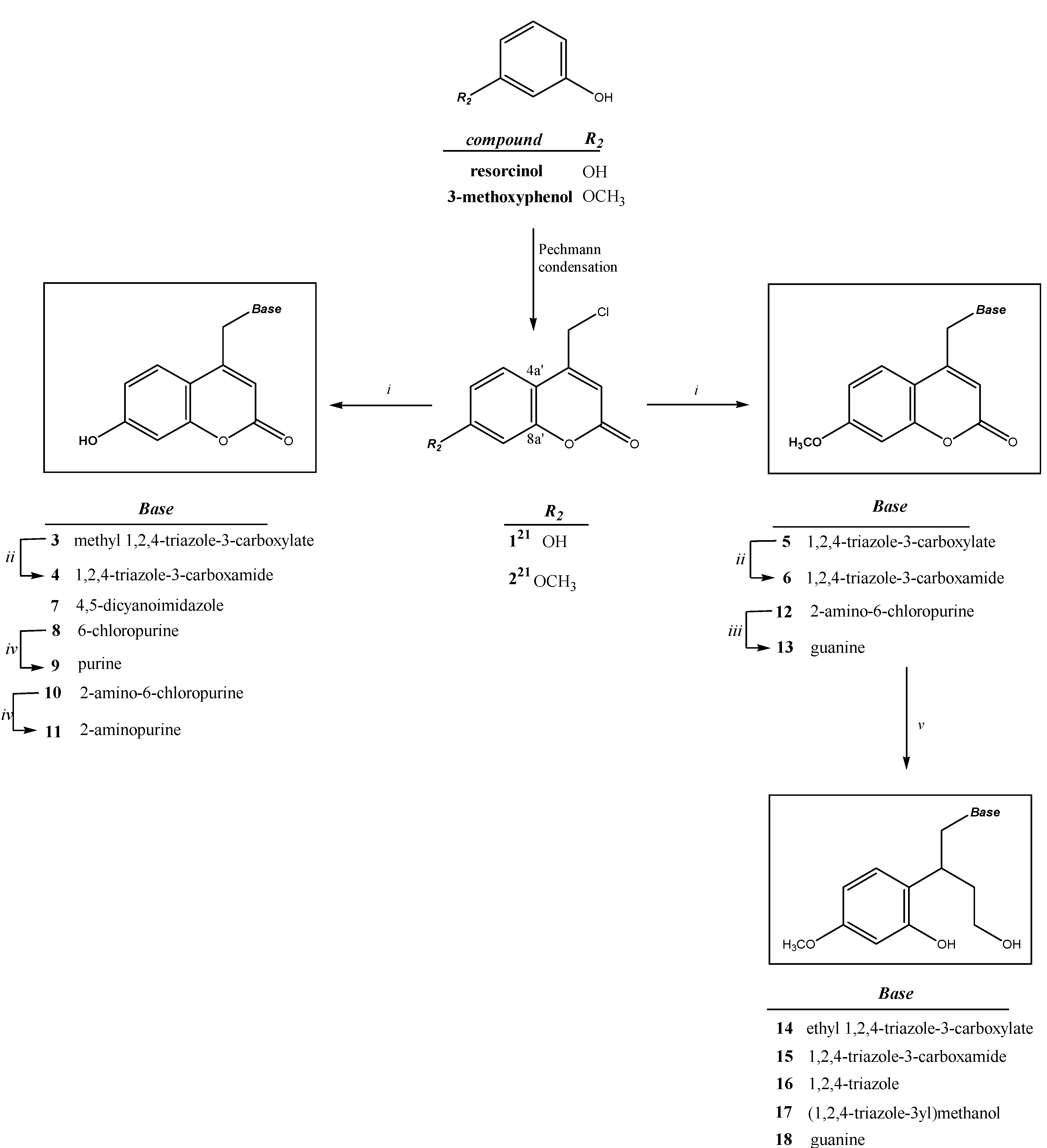
2. Results and Discussion
2.1. Chemistry
2.2. NMR Assignments
2.3. Antiproliferative Effects
| OH-7' | H-5 | H-8 | H-5' | H-6' | H-8' | NH2-2 | CH2-N | H-3' | OMe-7' | |
|---|---|---|---|---|---|---|---|---|---|---|
| 3 a | 10.72 (s, 1H) | 8.34 (s, 1H) | / | 7.72 (d, 1H, J = 8.8) | 6.87 (AB, dd, 1H, J = 2.3, 8.7) | 6.78 (d, 1H, J = 2.2) | / | 6.01 (s, 2H) | 5.30 (s, 1H) | / |
| 4 b | 10.72 (s, 1H) | 8.25 (s, 1H) | / | 7.74 (d, 1H, J = 8.7) | 6.85 (AB, dd, 1H, J = 2.0, 8.6) | 6.77 (d, 1H, J = 2.0) | / | 6.06 (s, 2H) | 5.26 (s, 1H) | / |
| 5 c | / | 8.09 (s, 1H) | / | 7.77 (d, 1H, J = 8.9) | 7.05 (AB, dd, 1H, J = 2.5, 8.6) | 7.01 (AB, dd, 1H, J = 2.6, 8.8) | / | 5.84 (s, 2H) | 5.76 (s, 1H) | 3.85 (s, 3H) |
| 6 d | / | 8.83 (s, 1H) | / | 7.73 (d, 1H, J = 8.9) | 7.07 (AB, dd, 1H, J = 2.0, 8.6) | 7.02 (AB, dd, 1H, J = 2.0, 8.8) | / | 5.81 (s, 2H) | 5.79 (s, 1H) | 3.87 (s, 3H) |
| 7 | 10.76 (s, 1H) | 8.45 (s, 1H) | / | 7.66 (d, 1H, J = 8.7) | 6.87 (AB, dd, 1H, J = 2.2, 8.7) | 6.80 (d, 1H, J = 2.1) | / | 5.82 (s, 2H) | 5.76 (s, 1H) | / |
| 8 d | 10.72 (s, 1H) | / | 8.82 (s, 1H) | 7.80 (d, 1H, J = 8.7) | 6.87 (AB, dd, 1H, J = 2.0, 8.6) | 6.78 (d, 1H, J = 2.1) | / | 5.81 (s, 2H) | 5.61 (s, 1H) | / |
| 9 f | 10.72 (s, 1H) | / | 8.10 (s, 1H) | 7.77 (d, 1H, J = 8.7) | 6.89 (AB, dd, 1H, J = 2.3, 8.7) | 6.81 (d, 1H, J = 2.2) | / | 5.65 (s, 2H) | 5.36 (s, 1H) | / |
| 10 | 10.72 (s, 1H) | / | 8.22 (s, 1H) | 7.79 (d, 1H, J = 8.7) | 6.87 (AB, dd, 1H, J = 2.2, 8.7) | 6.78 (d, 1H, J = 2.2) | 6.76 (s, 2H) | 5.55 (s, 2H) | 5.44 (s, 1H) | / |
| 11 g | 10.01 (s, 1H) | / | 7.78 (s, 1H) | 7.74 (d, 1H, J = 8.8) | 6.84 (AB, dd, 1H, J = 1.9, 8.7) | 6.76 (d, 1H, J = 2.2) | 6.75 (s, 2H) | 5.42 (s, 2H) | 5.23 (s, 1H) | / |
| 12 | / | / | 8.20 (s, 1H) | 7.85 (d, 1H, J = 8.8) | 7.01 (AB, dd, 1H, J = 2.0, 8.7) | 7.05 (d, 1H, J = 1.9) | 6.97 (s, 2H) | 5.56 (s, 2H) | 5.54 (s, 1H) | 3.87 (s, 3H) |
| 13 h | / | / | 7.78 (s, 1H) | 7.86 (d, 1H, J = 8.8) | 7.02 (AB, dd, 1H, J = 1.9, 8.7) | 7.06 (d, 1H, J = 1.9) | 6.53 (s, 2H) | 5.46 (s, 2H) | 5.41 (s, 1H) | 3.88 (s, 3H) |
| OH-2" | H-8 | H-5 | H-6" | H-5" | H-3" | OH-4' | OMe-4" | H-1' | H-2' | H-3' | H-4' | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 14 a | 9.64 (s, 1H) | / | 8.11 (s, 1H) | 6.88 (AB, dd, 1H, J= 3.27, 8.43) | 6.36 (AB, dd, 1H, J = 2.34, 8.36) | 6.36 (d, 1H, J = 8.30) | 5.19 (t, 1H, J = 6.03) | 3.65 (s, 3H) | 3.17–3.24 (m, 2H) | 2.52–2.56 (m, 1H) | 1.80–1.61 (m, 2H) | 4.15–4.18 (m, 2H) |
| 15 b | 9.49 (s, 1H) | / | 8.25 (s, 1H) | 6.91 (d, 1H, J = 8.37) | 6.35 (d, 1H, J = 2.31) | 6.30 (AB, dd, 1H, J= 2.28, 8.34) | 5.11 (t, 1H, J = 6.01) | 3.65 (s, 3H) | 3.16–3.18 (m, 2H) | 2.32–2.35 (m, 1H) | 1.79–1.61 (m, 2H) | 4.37–4.40 (m, 2H) |
| 16 c | 9.47 (s, 1H) | / | 8.17 (s, 1H) | 6.87 (d, 1H, J = 8.40) | 6.34 (d, 1H, J = 2.28) | 6.28 (AB, dd, 1H, J = 2.28, 8.40) | 5.14 (t, 1H, J = 5.96) | 3.65 (s, 3H) | 3.25–3.17 (m, 2H) | 2.84–2.87 (m, 1H) | 1.79–1.61 (m, 2H) | 4.27–4.29 (m, 2H) |
| 17 d | 9.42 (s, 1H) | / | 8.07 (s, 1H) | 6.91 (d, 1H, J = 8.40) | 6.36 (d, 1H, J = 2.46) | 6.31 (AB, dd, 1H, J = 2.40, 8.34) | 5.11 (t, 1H, J = 6.06) | 3.66 (s, 3H) | 3.21–3.13 (m, 2H) | 2.33–2.37 (m, 1H) | 1.79–1.61 (m, 2H) | 4.34–4.37 (m, 2H) |
| 18 e | 9.46 (s, 1H) | 7.2 (s, 1H) | / | 6.92 (d, 1H, J = 8.34) | 6.32 (d, 1H, J = 1.95) | 6.29 (AB, dd, 1H, J = 2.35, 8.19) | 4.97 (t, 1H, J = 6.04) | 3.66 (s, 3H) | 3.26–3.17 (m, 2H) | 3.04–3.07 (m, 1H) | 1.82–1.62 (m, 2H) | 4.13– 4.16 (m, 2H) |
| IC50a (μM) | |||||
|---|---|---|---|---|---|
| Substance No. | Cell lines | ||||
| HeLa | MCF-7 | HepG2 | SW620 | BJ | |
| 3 | >100 | >100 | >100 | >100 | >100 |
| 4 | >100 | >100 | >100 | >100 | >100 |
| 5 | >100 | >100 | >100 | >100 | >100 |
| 6 | 35.5 ± 13.5 | >100 | >100 | >100 | >100 |
| 7 | >100 | >100 | >100 | >100 | >100 |
| 8 | >100 | >100 | >100 | >100 | >100 |
| 9 | >100 | >100 | >100 | >100 | >100 |
| 10 | 34 ± 8.4 | >100 | 25.6 ± 12.6 | 35.4 ± 3.7 | >100 |
| 11 | >100 | >100 | >100 | >100 | >100 |
| 12 | >100 | >100 | >100 | >100 | >100 |
| 13 | >100 | >100 | >100 | >100 | >100 |
| 14 | >100 | >100 | >100 | >100 | >100 |
| 15 | >100 | >100 | >100 | >100 | >100 |
| 16 | >100 | >100 | >100 | >100 | >100 |
| 17 | >100 | >100 | >100 | >100 | >100 |
| 18 | >100 | >100 | >100 | >100 | >100 |
3. Experimental
3.1. General
3.2. Procedures for the Preparation of Compounds
3.2.1. Compounds 3–13
3.2.2. Compound Data
3.2.3. Compounds 14–18
3.2.4. Compound Data
3.2.5. Cytostatic Activity Assay
4. Conclusions
Acknowledgements
References
- Egan, D.; O’kennedy, R.; Moran, E.; Cox, D.; Prosser, E.; Thornes, R.D. The Pharmacology, Metabolism, Analysis, and Applications of Coumarin and Coumarin-Related Compounds. Drug Metab. Rev. 1990, 22, 503–529. [Google Scholar] [CrossRef]
- Borges, F.; Roleira, F.; Milhazes, N.; Santana, L.; Uriarte, E. Simple Coumarins and Analogues in Medicinal Chemistry: Occurrence, Synthesis and Biological Activity. Curr. Med. Chem. 2005, 12, 887–916. [Google Scholar] [CrossRef]
- Harvey, R.G.; Cortez, C.; Ananthanarayan, T.P.; Schmolka, S. A new coumarin synthesis and its utilization for the synthesis of polycyclic coumarin compounds with anticarcinogenic properties. J. Org. Chem. 1988, 53, 3936–3943. [Google Scholar]
- Kostova, I.; Raleva, S.; Genova, P.; Argirova, R. Structure-Activity Relationships of Synthetic Coumarins as HIV-1 Inhibitors. Bioinorg. Chem. Appl. 2006. [Google Scholar] [CrossRef]
- Moffet, R.S. Central Nervous System Depressants. VII.1 Pyridyl Coumarins. J. Med. Chem. 1964, 7, 446–449. [Google Scholar] [CrossRef]
- Al-Haiza, M.A.; Mostafa, M.S.; El-Kady, M.Y. Synthesis and Biological Evaluation of Some New Coumarin Derivatives. Molecules 2003, 8, 275–286. [Google Scholar] [CrossRef]
- Musicki, B.; Periers, A.-M.; Laurin, P.; Ferroud, D.; Benedetti, Y.; Lachaud, S.; Chatreaux, F.; Haesslein, J.L.; Iltis, A.; Pierre, C.; et al. Improved antibacterial activities of coumarin antibiotics bearing 5',5'-dialkylnoviose: Biological activity of RU79115. Bioorg. Med. Chem. Lett. 2000, 10, 1695–1699. [Google Scholar]
- Fylaktakidou, K.C.; Hadjipavlou-Litina, D.J.; Litinas, K.E.; Nicolaides, D.N. Natural and Synthetic Coumarin Derivatives with Anti-Inflammatory/Antioxidant Activities. Curr. Pharm. Des. 2004, 10, 3813–3833. [Google Scholar]
- Bucolo, C.; Cuzzocrea, S.; Mazzon, E.; Caputi, A.P. Effects of Cloricromene, a Coumarin Derivative, on Endotoxin-Induced Uveitis in Lewis Rats. Invest. Ophthalmol. Vis. Sci. 2003, 44, 1178–1184. [Google Scholar] [CrossRef]
- Bucolo, C.; Ward, K.W.; Mazzon, E.; Cuzzocrea, S.; Drago, F. Protective Effects of a Coumarin Derivative in Diabetic Rats. Invest. Ophthalmol. Vis. Sci. 2009, 50, 3846–3852. [Google Scholar] [CrossRef]
- Payá, M.; Halliwell, B.; Hoult, J.R. Interactions of a series of coumarins with reactive oxygen species: Scavenging of superoxide, hypochlorous acid and hydroxyl radicals. Biochem. Pharmacol. 1992, 44, 205–214. [Google Scholar]
- Marshall, M.E.; Ryles, M.; Butler, K.; Weiss, L. Treatment of advanced renal cell carcinoma (RCC) with coumarin and cimetidine: long-term follow-up of patients on a phase I trial. J. Cancer Res. Clin. Oncol. 1994, 120, 535–538. [Google Scholar]
- Marshall, M.E.; Mohler, J.L.; Edmonds, K.; Williams, B.; Butler, K.; Ryles, M.; Weiss, L.; Urban, D.; Bueschen, A.; Markiewicz, M. An updated review of the clinical development of coumarin (1,2-benzopyrone) and 7-hydroxycoumarin. J. Cancer Res. Clin. Oncol. 1994, 120, S39–S42. [Google Scholar] [CrossRef]
- Stanchev, S.; Momekov, G.; Jensen, F.; Manolov, I. Synthesis, computational study and cytotoxic activity of new 4-hydroxycoumarin derivatives. Eur. J. Med. Chem. 2008, 43, 694–706. [Google Scholar] [CrossRef]
- Thornes, R.D.; Daly, L.; Lynch, G.; Breslin, B.; Browne, H.; Browne, H.Y.; Corrigan, T.; Daly, P.; Edwards, G.; Gaffney, E.; et al. Treatment with coumarin to prevent or delay recurrence of malignant melanoma. J. Cancer Res. Clin. Oncol. 1994, 120, S32–S34. [Google Scholar] [CrossRef]
- Marshall, M.E.; Butler, K.; Fried, A. Phase I evaluation of coumarin (1,2-benzopyrone) and cimetidine in patients with advanced malignancies. Mol. Biother. 1991, 3, 170–178. [Google Scholar]
- Mohler, J.L.; Gomella, L.G.; Crawford, E.D.; Glode, L.M.; Zippe, C.D.; Fair, W.R.; Marshall, M.E. Phase II evaluation of coumarin (1,2-benzopyrone) in metastatic prostatic carcinoma. Prostate 1992, 20, 123–131. [Google Scholar] [CrossRef]
- Jung, J.-C.; Kim, J.-C.; Park, O.-S. Simple and cost-effective syntheses of 4-hydroxycoumarins. Synth. Commun. 1999, 29, 3587–3595. [Google Scholar] [CrossRef]
- Jacquot, Y.; Bermont, L.; Giorgi, H.; Refouvelet, B.; Adessi, G.; Daubrosse, E.; Xicluna, A. Substituted benzopyranobenzothiazinones. Synthesis and estrogenic activity on MCF-7 breast carcinoma cells. Eur. J. Med. Chem 2001, 36, 127–136. [Google Scholar] [CrossRef]
- Budzisz, E.; Brzezinska, E.; Krajewska, U.; Rozalski, M. Cytotoxic effects, alkylating properties and molecular modelling of coumarin derivatives and their phosphonic analogues. Eur. J. Med. Chem. 2003, 38, 597–603. [Google Scholar] [CrossRef]
- Musa, M.A.; Cooperwood, J.S.; Khan, M.O.F. A Review of Coumarin Derivatives in Pharmacotherapy of Breast Cancer. Curr. Med. Chem. 2008, 15, 2664–2679. [Google Scholar] [CrossRef]
- Steffen, U.S.; Weber, B.; Siegers, C. Antitumor-activities of coumarin, 7-hydroxy-coumarin and its glucuronide in several human tumor cell lines. Res. Commun. Mol. Pathol. Pharmacol. 1998, 99, 193–206. [Google Scholar]
- Zagotto, G.; Gia, O.; Baccichetti, F.; Uriarte, E.; Palumbo, M. Synthesis and Photobiological Properties of 4-Hydroxymethyl-4'-methylpsoralen Derivatives. Photochem. Photobiol. 1993, 58, 486–491. [Google Scholar]
- McCulloch, P.; George, W.D. Warfarin inhibits metastasis of Mtln3 rat mammary carcinoma without affecting primary tumour growth. Br. J.Cancer 1989, 59, 179–183. [Google Scholar] [CrossRef]
- Itokawa, H.; Yun, J.S.; Morita, H.; Takeya, K. Cytotoxic coumarins from roots of Angelica gigas Nakai. Nat. Med. 1994, 48, 334–335. [Google Scholar]
- Kolodziej, H.; Kayser, O.; Woerdenbag, H.J.; Van Uden, W.; Pras, N. Examination for anti-Human Immunodeficienvy Virus—Type 1(HIV-1) effect of three 4-hydroxycoumarin (4-hc) derivatives. Z.Naturforsch. C 1997, 52, 240–244. [Google Scholar]
- Zhao, H.; Neamati, N.; Hong, H.; Mazumder, A.; Wang, S.; Sunder, S.; Milne, G.W.A.; Pommier, Y.; Burke, T.R. Coumarin-Based Inhibitors of HIV Integrase. J. Med. Chem. 1997, 40, 242–248. [Google Scholar] [CrossRef]
- Singhal, N.; Sharma, P.K.; Dudhe, R.; Kumar, N. Recent advancement of triazole derivatives and their biological significance. J. Chem. Pharm. Res. 2011, 3, 126–133. [Google Scholar]
- Amr, A.E.; Nermien, M.S.; Abdulla, M.M. Synthesis, reactions, and anti-inflammatory activity of heterocyclic systems fused to a thiophene moiety using citrazinic acid as synthon. Monatsh. Chem. 2007, 138, 699–707. [Google Scholar]
- Fujiwara, N.; Nakajima, T.; Ueda, Y.; Fujita, H.; Kawakami, H. Novel piperidinylpyrimidine derivatives as inhibitors of HIV-1 LTR activation. Bioorg. Med. Chem. 2008, 16, 9804–9816. [Google Scholar] [CrossRef]
- Ballell, L.; Field, R.A.; Chung, G.A.C.; Young, R.J. New thiopyrazolo[3,4-d]pyrimidine derivatives as anti-mycobacterial agents. Bioorg. Med. Chem. Lett. 2007, 17, 1736–1740. [Google Scholar] [CrossRef]
- Wagner, E.; Al-Kadasi, K.; Zimecki, M.; Sawka-Dobrowolska, W. Synthesis and pharmacological screening of derivatives of isoxazolo[4,5-d]pyrimidine. Eur. J. Med. Chem. 2008, 43, 2498–2504. [Google Scholar] [CrossRef]
- Završnik, D.; Muratović, S.; Makuc, D.; Plavec, J.; Cetina, M.; Nagl, A.; DeClercq, E.; Balzarini, J.; Mintas, M. Benzylidene-bis-(4-hydroxycoumarin) and benzopyranocoumarin derivatives: synthesis, 1H/13C-NMR conformational and X-ray crystal structure studies and in vitro antiviral activity evaluations. Molecules 2011, 16, 6023–6040. [Google Scholar] [CrossRef]
- Al-Soud, Y.A.; Al-Masoudi, I.A.; Saeed, B.; Beifuß, U.; Al-Masoudi, N.A. Synthesis of new 1H-1,2,4-triazolylcoumarins and theirantitumor and anti-HIVactivities. Chem. Heterocycl. Comp. 2006, 42, 583–590. [Google Scholar]
- Moon, H.R.; Kim, H.O.; Lee, S.K.; Choi, W.J.; Chun, M.W.; Jeong, L.S. Synthesis and biological evaluation of novel thiapio dideoxynucleosides. Bioorg. Med. Chem. 2002, 10, 1499–1507. [Google Scholar]
- Mosmann, T. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Gazivoda, T.; Raić-Malić, S.; Krištafor, V.; Makuc, D.; Plavec, J.; Bratulic, S.; Kraljević Pavelić, S.; Pavelić, K.; Naesens, L.; Andrei, G.; et al. Synthesis, Cytostatic and Anti-HIV Evaluations of the New Unsaturated Acyclic C-5 Pyrimidine Nucleoside Analogues. Bioorg. Med. Chem. 2008, 16, 5624–5634. [Google Scholar] [CrossRef]
- Sample Availability: Samples of all compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Benci, K.; Mandić, L.; Suhina, T.; Sedić, M.; Klobučar, M.; Kraljević Pavelić, S.; Pavelić, K.; Wittine, K.; Mintas, M. Novel Coumarin Derivatives Containing 1,2,4-Triazole, 4,5-Dicyanoimidazole and Purine Moieties: Synthesis and Evaluation of Their Cytostatic Activity. Molecules 2012, 17, 11010-11025. https://doi.org/10.3390/molecules170911010
Benci K, Mandić L, Suhina T, Sedić M, Klobučar M, Kraljević Pavelić S, Pavelić K, Wittine K, Mintas M. Novel Coumarin Derivatives Containing 1,2,4-Triazole, 4,5-Dicyanoimidazole and Purine Moieties: Synthesis and Evaluation of Their Cytostatic Activity. Molecules. 2012; 17(9):11010-11025. https://doi.org/10.3390/molecules170911010
Chicago/Turabian StyleBenci, Krešimir, Leo Mandić, Tomislav Suhina, Mirela Sedić, Marko Klobučar, Sandra Kraljević Pavelić, Krešimir Pavelić, Karlo Wittine, and Mladen Mintas. 2012. "Novel Coumarin Derivatives Containing 1,2,4-Triazole, 4,5-Dicyanoimidazole and Purine Moieties: Synthesis and Evaluation of Their Cytostatic Activity" Molecules 17, no. 9: 11010-11025. https://doi.org/10.3390/molecules170911010
APA StyleBenci, K., Mandić, L., Suhina, T., Sedić, M., Klobučar, M., Kraljević Pavelić, S., Pavelić, K., Wittine, K., & Mintas, M. (2012). Novel Coumarin Derivatives Containing 1,2,4-Triazole, 4,5-Dicyanoimidazole and Purine Moieties: Synthesis and Evaluation of Their Cytostatic Activity. Molecules, 17(9), 11010-11025. https://doi.org/10.3390/molecules170911010




