An Improved Synthesis of 1,2-Diarylethanols under Conventional Heating and Ultrasound Irradiation
Abstract
:1. Introduction

2. Results and Discussion
| Entry | Base | Solvent | Temperature | Time | Yield (%) a |
|---|---|---|---|---|---|
| °C | h | ||||
| 1 | K2CO3 | EtOH | 40 | 5 | 0 |
| 2 | NaOH | EtOH | 40 | 5 | 5 |
| 3 | DBU | EtOH | 40 | 5 | 8 |
| 4 | NaOEt | EtOH | 40 | 5 | 22 |
| 5 | NaOEt | DMSO | 40 | 5 | 44 |
| 6 | NaOEt | EtOH:DMSO (6:1) | 40 | 8 | 53 |
| 7 | NaOEt | EtOH:DMSO (6:1) | 50 | 8 | 62 |
| 8 | NaOEt | EtOH:DMSO (6:1) | 60 | 8 | 56 |
| 9 | NaOEt | EtOH:DMSO (6:1) | 70 | 8 | 28 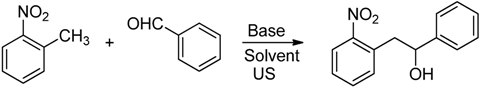 |
| Entry | Compound 1 | Compound 2 | Product 3 | Time (h) | Yield (%) a |
|---|---|---|---|---|---|
| a | 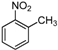 | 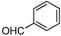 | 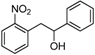 | 8 | 62 |
| b | 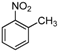 | 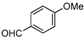 | 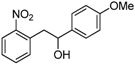 | 8 | 65 |
| c | 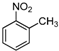 | 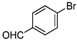 | 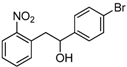 | 12 | 60 |
| d | 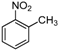 |  |  | 12 | 49 |
| e | 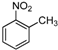 | 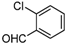 |  | 12 | 58 |
| f | 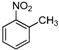 |  | 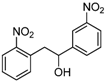 | 12 | 46 |
| g | 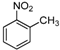 | 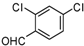 |  | 8 | 44 |
| h |  | 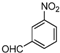 | 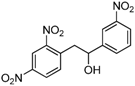 | 12 | 45 |
| i | 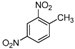 | 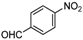 |  | 8 | 44 |
| j |  | 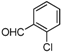 | 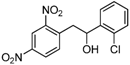 | 8 | 35 |
| k | 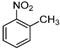 | 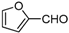 | 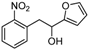 | 8 | 53
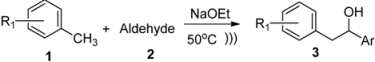 |
3. Experimental
3.1. General
3.2. General Procedure
4. Conclusions
Acknowledgments
References
- Baur, J.A.; Sinclair, D.A. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef]
- Cirla, A.; Mann, J. Combretastatins: From natural products to drug discovery. Nat. Prod. Rep. 2003, 20, 558–564. [Google Scholar] [CrossRef]
- Singh, S.B.; Pettit, G.R. Synthesis of (±)-isocombretasstatins A-C. Synth. Commun. 1987, 17, 877–892. [Google Scholar] [CrossRef]
- Pettit, G.R.; Singh, S.B.; Cragg, G.M. Antineoplastic agents. 113. Synthesis of natural (−)-combretastatin. J. Org. Chem. 1985, 50, 3404–3406. [Google Scholar]
- Hamel, E.; Lin, C.M. Interactions of combretastatin, a new plant-derived antimitotic agent, with tubulin. Biochem. Pharmacol. 1983, 32, 3864–3867. [Google Scholar] [CrossRef]
- Barrero, A.F.; Herrador, M.M.; Quílez del Moral, J.F.; Arteaga, P.; Akssira, M.; El Hanbali, F.; Arteaga, J.F.; Diéguez, H.R.; Sánchez, E.M. Couplings of Benzylic Halides Mediated by Titanocene Chloride: Synthesis of Bibenzyl Derivatives. J. Org. Chem. 2007, 72, 2251–2254. [Google Scholar]
- Murata, S.; Tsubone, Y.; Kawai, R.; Eguchi, D.; Tomioka, H. Mechanistic studies of intramolecular C-H insertion reaction of arylnitrenes: Isotope effect, configurational purity and radical clock studies. J. Phys. Org. Chem. 2005, 18, 9–20. [Google Scholar]
- Yadav, J.S.; Reddy, B.V.S.; Reddy, K.S. Ultrasound-accelerated synthesis of chiral allylic alcohols promoted by indium metal. Tetrahedron 2003, 59, 5333–5336. [Google Scholar] [CrossRef]
- Wang, S.Y.; Ji, S.J.; Su, X.M. A Meldrum’s acid catalyzed synthesis of bis(indolyl)methanes in water under ultrasonic condition. Chin. J. Chem. 2008, 26, 22–24. [Google Scholar] [CrossRef]
- Pasha, M.A.; Jayashankara, V.P. Reduction of arylnitro compounds to azoarenes and/or arylamines by Al/NaOH in methanol under ultrasonic conditions. Ultrason. Sonochem 2005, 12, 433–435. [Google Scholar] [CrossRef]
- Li, J.-T.; Li, X.-L.; Li, T.-S. Synthesis of oximes under ultrasound irradiation. Ultrason. Sonochem. 2006, 13, 200–202. [Google Scholar] [CrossRef]
- Disselkamp, R.S.; Hart, T.R.; Williams, A.M.; White, J.F.; Peden, C.H.F. Ultrasound-assisted hydrogenation of cinnamaldehyde. Ultrason. Sonochem. 2005, 12, 319–324. [Google Scholar] [CrossRef]
- Grönroos, A.; Aittokallio, N.; Kolehmainen, E. Ultrasound accelerated esterification of bile acids. Ultrason. Sonochem. 2004, 11, 161–165. [Google Scholar] [CrossRef]
- Mason, T.J.; Peters, D. Practical Sonochemistry; Ellis Horwood: New York, NY, USA, 1991. [Google Scholar]
- Mason, T.J. Sonochemistry and the environment—Providing a “green” link between chemistry, physics and engineering. Ultrason. Sonochem. 2007, 14, 476–483. [Google Scholar] [CrossRef]
- Luche, J.L. Organic Sonochemistry; Plenum Press: New York, NY, USA, 1998. [Google Scholar]
- Bühler, S.; Lagoja, I.; Giegrich, H.; Stengele, K.-P.; Pfleiderer, W. New Types of Very Efficient Photolabile Protecting Groups Based upon the [2-(2-Nitrophenyl)propoxy]carbonyl (NPPOC) Moiety. Helv. Chim. Acta 2004, 87, 620–659. [Google Scholar] [CrossRef]
- Pizzotti, M.; Cenini, S.; Quici, S.; Tollari, S. Role of Alkali Halides in the Synthesis of Nitrogen Containing Heterocycles by Reductive Carbonylation of Aromatic Nitro-Derivatives Catalysed by Ru3(CO)12. J. Chem. Soc. Perkin Trans. 2 1994, 913–917. [Google Scholar]
- Sample Availability: Samples of the compounds 3a-k are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gao, D.-M.; Ma, W.-L.; Li, T.-R.; Huang, L.-Z.; Du, Z.-T. An Improved Synthesis of 1,2-Diarylethanols under Conventional Heating and Ultrasound Irradiation. Molecules 2012, 17, 10708-10715. https://doi.org/10.3390/molecules170910708
Gao D-M, Ma W-L, Li T-R, Huang L-Z, Du Z-T. An Improved Synthesis of 1,2-Diarylethanols under Conventional Heating and Ultrasound Irradiation. Molecules. 2012; 17(9):10708-10715. https://doi.org/10.3390/molecules170910708
Chicago/Turabian StyleGao, Dong-Mei, Wei-Li Ma, Tian-Rui Li, Liang-Zhu Huang, and Zhen-Ting Du. 2012. "An Improved Synthesis of 1,2-Diarylethanols under Conventional Heating and Ultrasound Irradiation" Molecules 17, no. 9: 10708-10715. https://doi.org/10.3390/molecules170910708
APA StyleGao, D.-M., Ma, W.-L., Li, T.-R., Huang, L.-Z., & Du, Z.-T. (2012). An Improved Synthesis of 1,2-Diarylethanols under Conventional Heating and Ultrasound Irradiation. Molecules, 17(9), 10708-10715. https://doi.org/10.3390/molecules170910708




