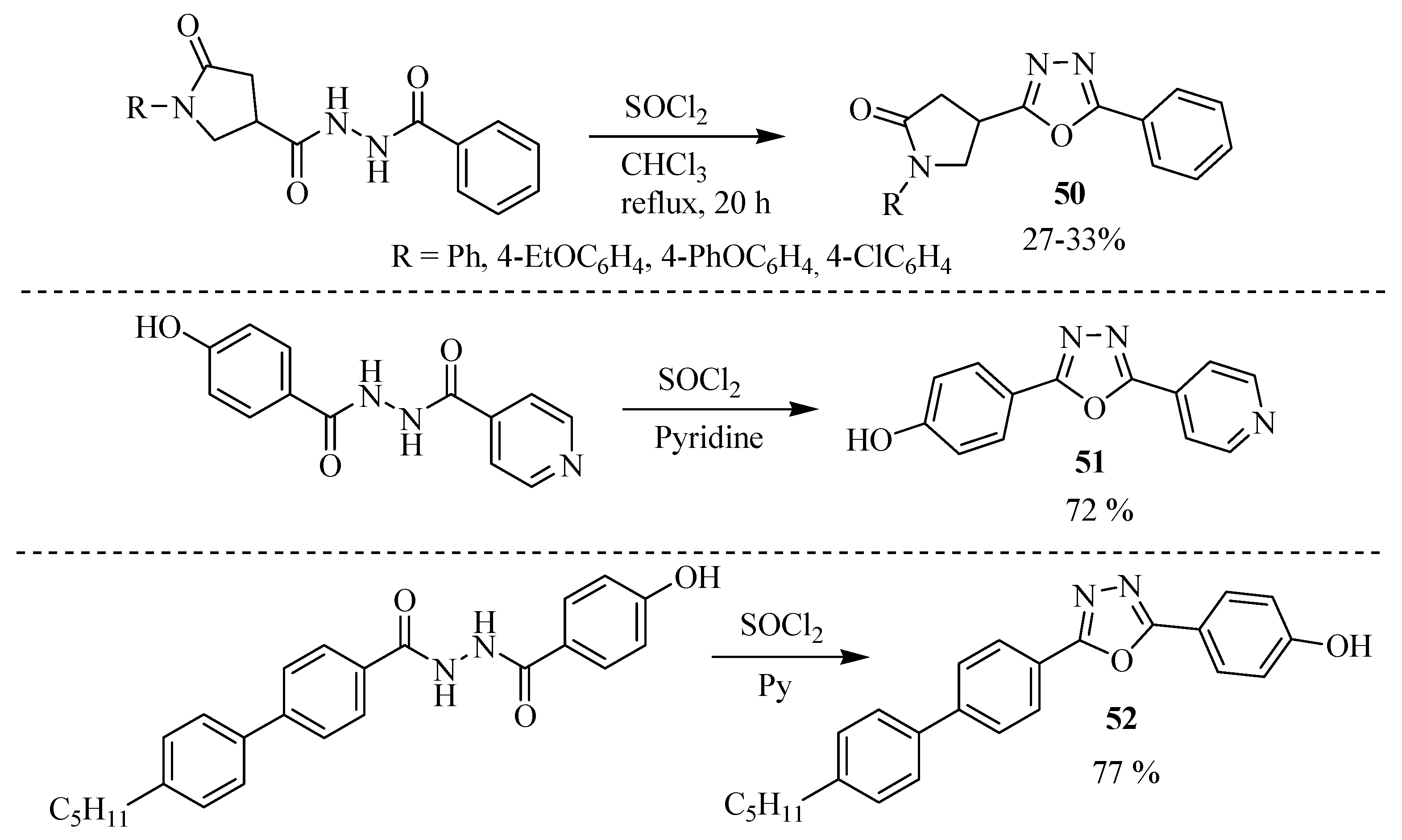
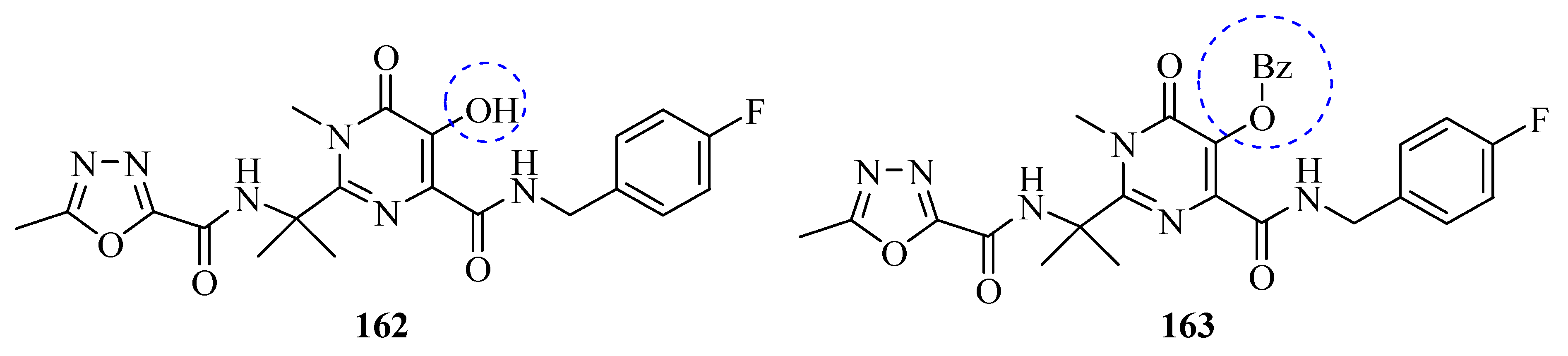
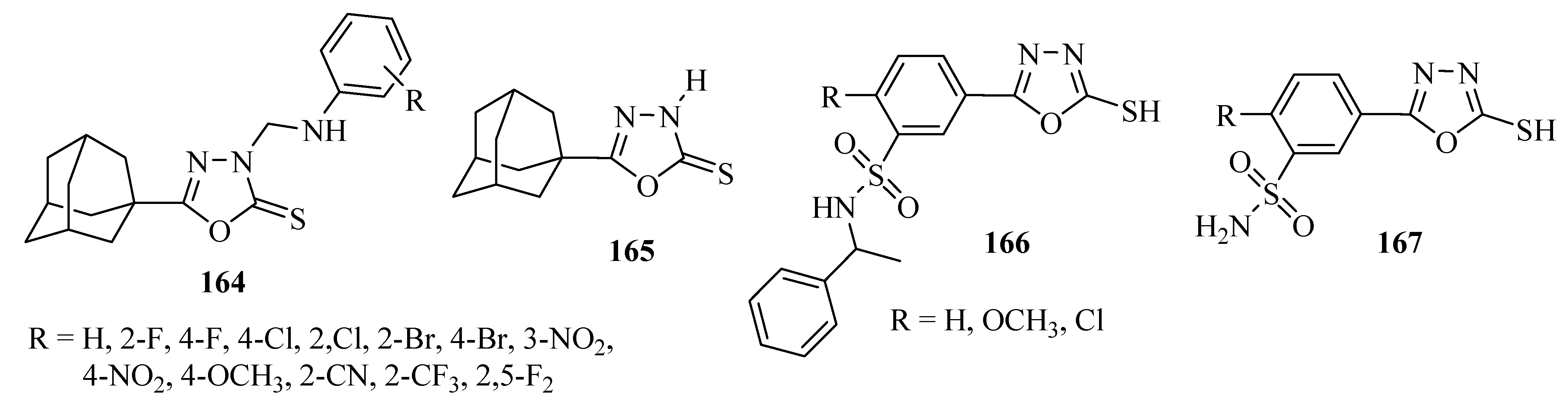
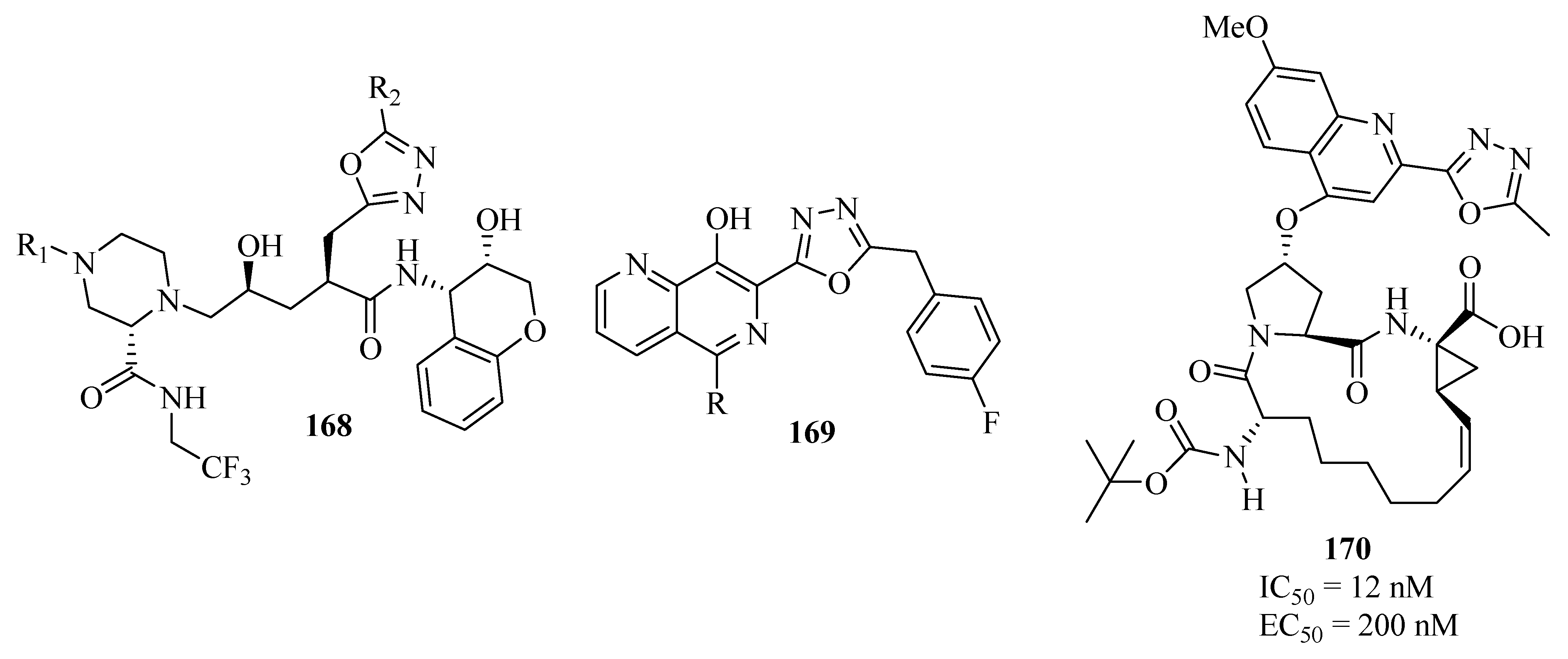
Synthetic Approaches and Pharmacological Activity of 1,3,4-Oxadiazoles: A Review of the Literature from 2000–2012
Abstract
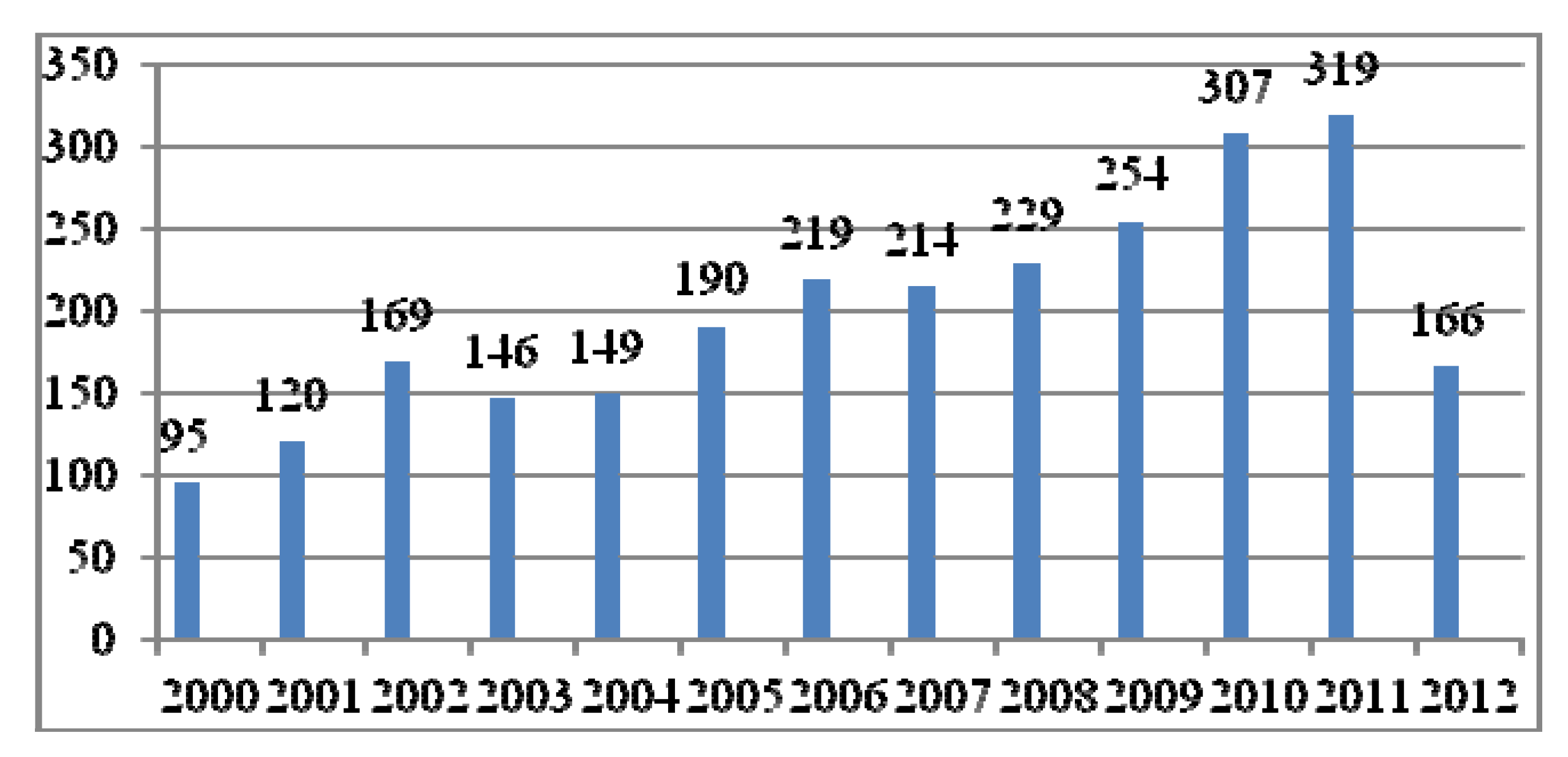
:1. Introduction
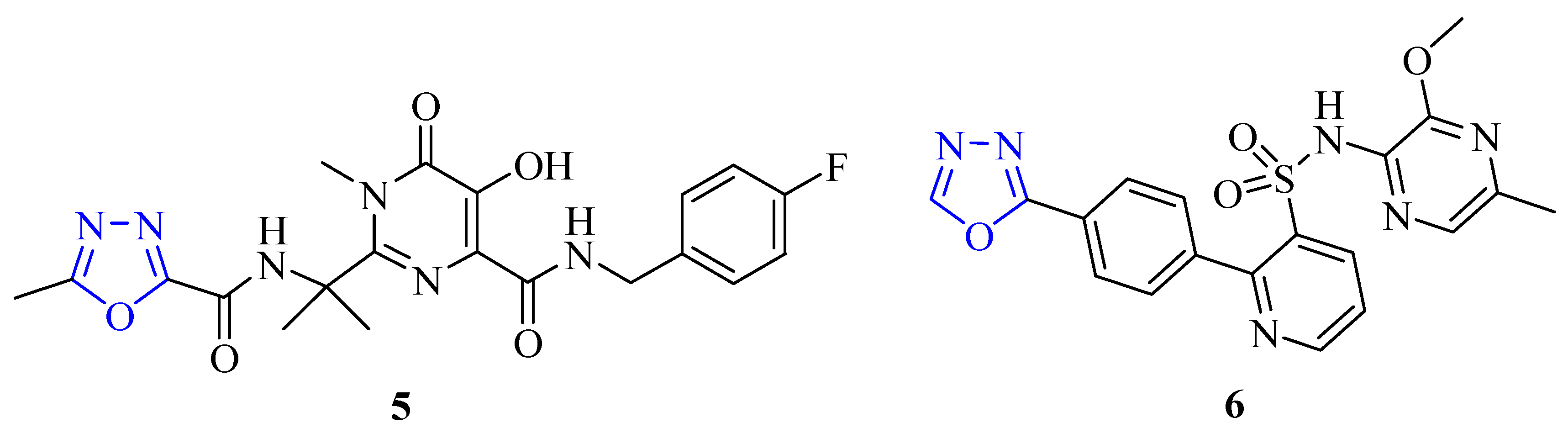
2. Methods of Synthesis for 2,5-Disubstituted-1,3,4-oxadiazoles
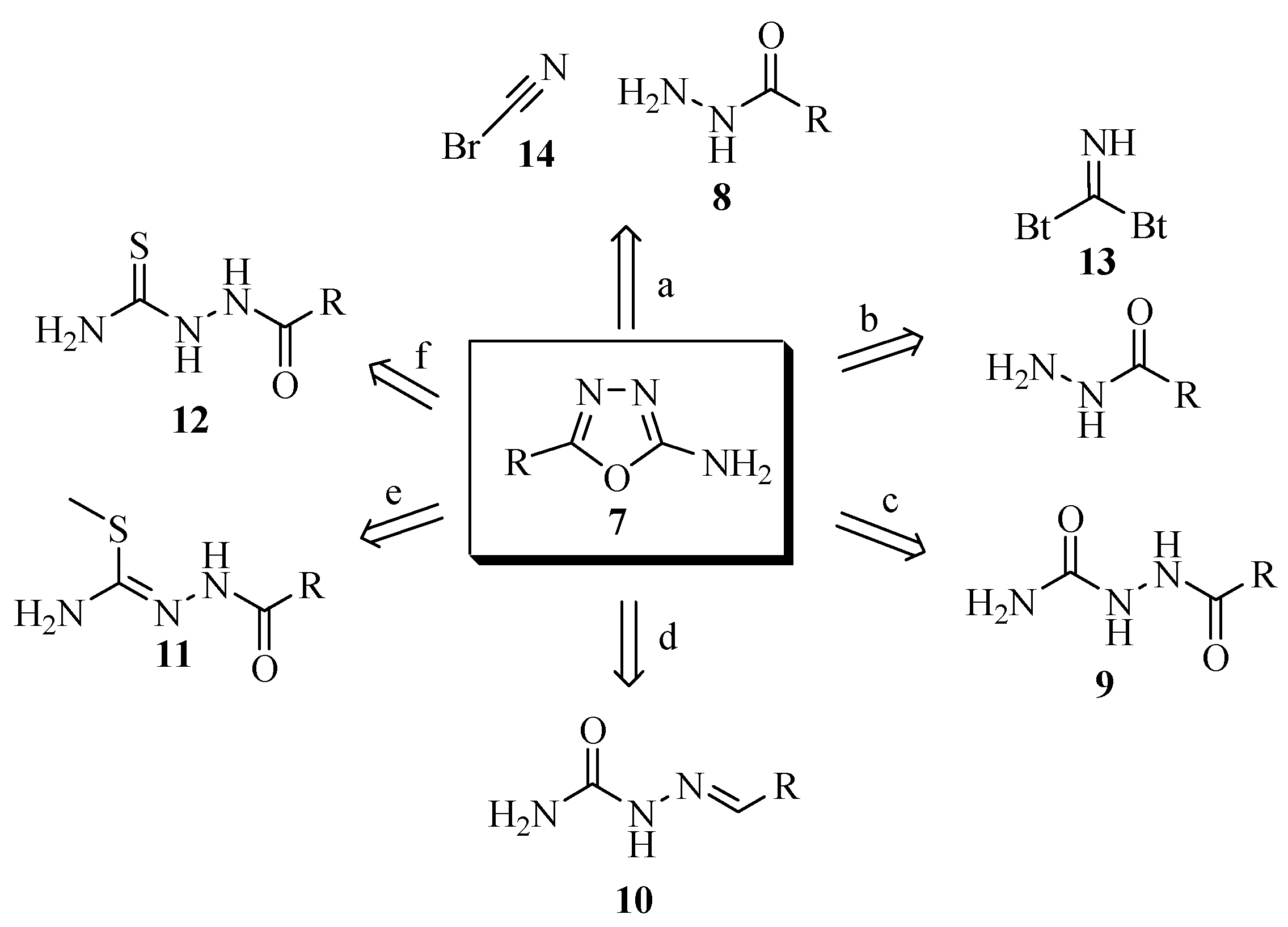
2.1. Methods of Synthesis for 5-Substituted-2-amino-1,3,4-oxadiazoles
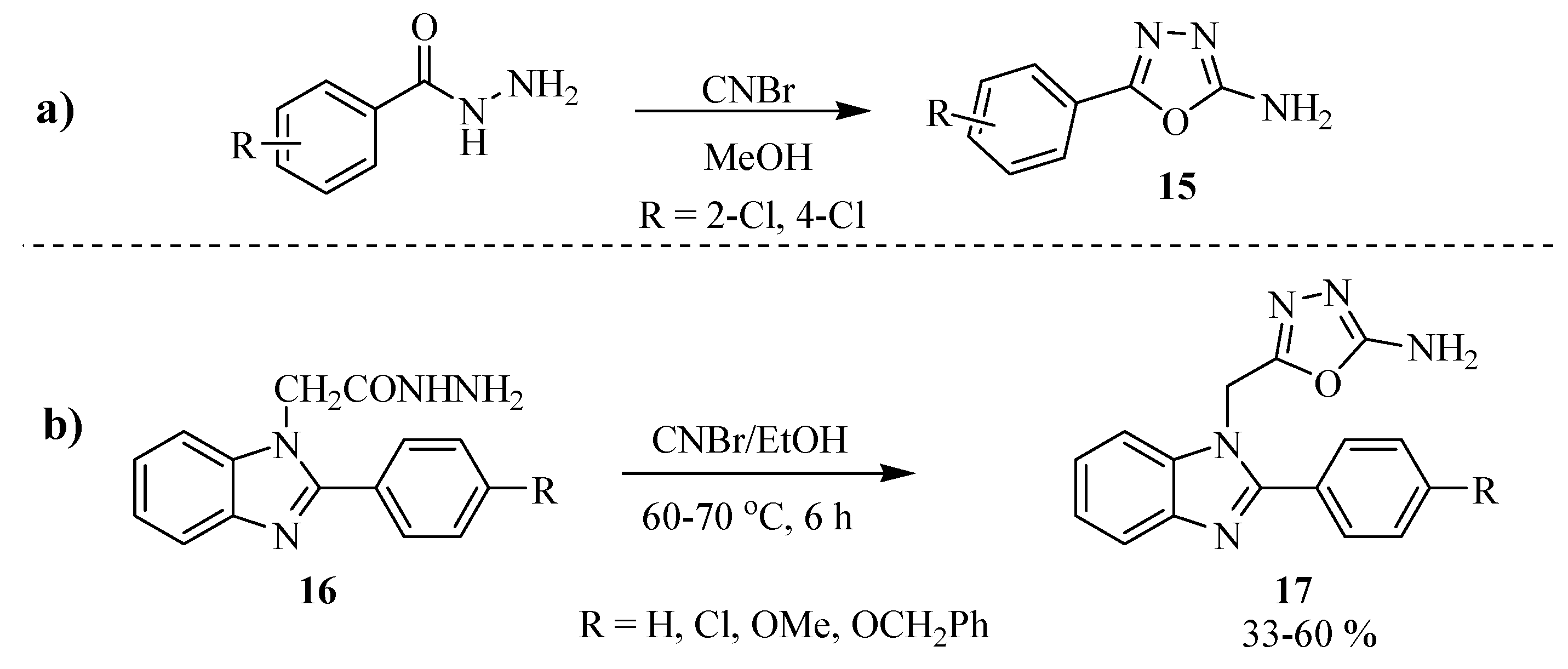
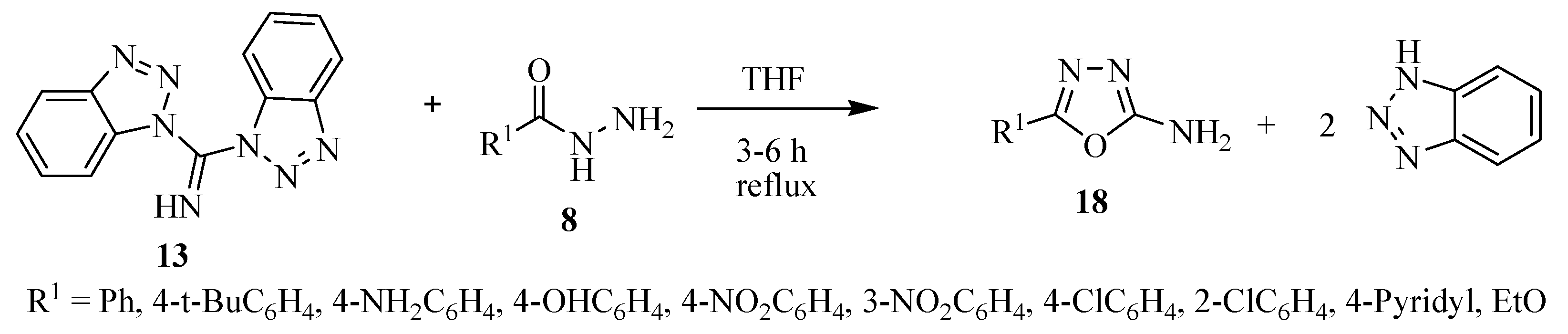
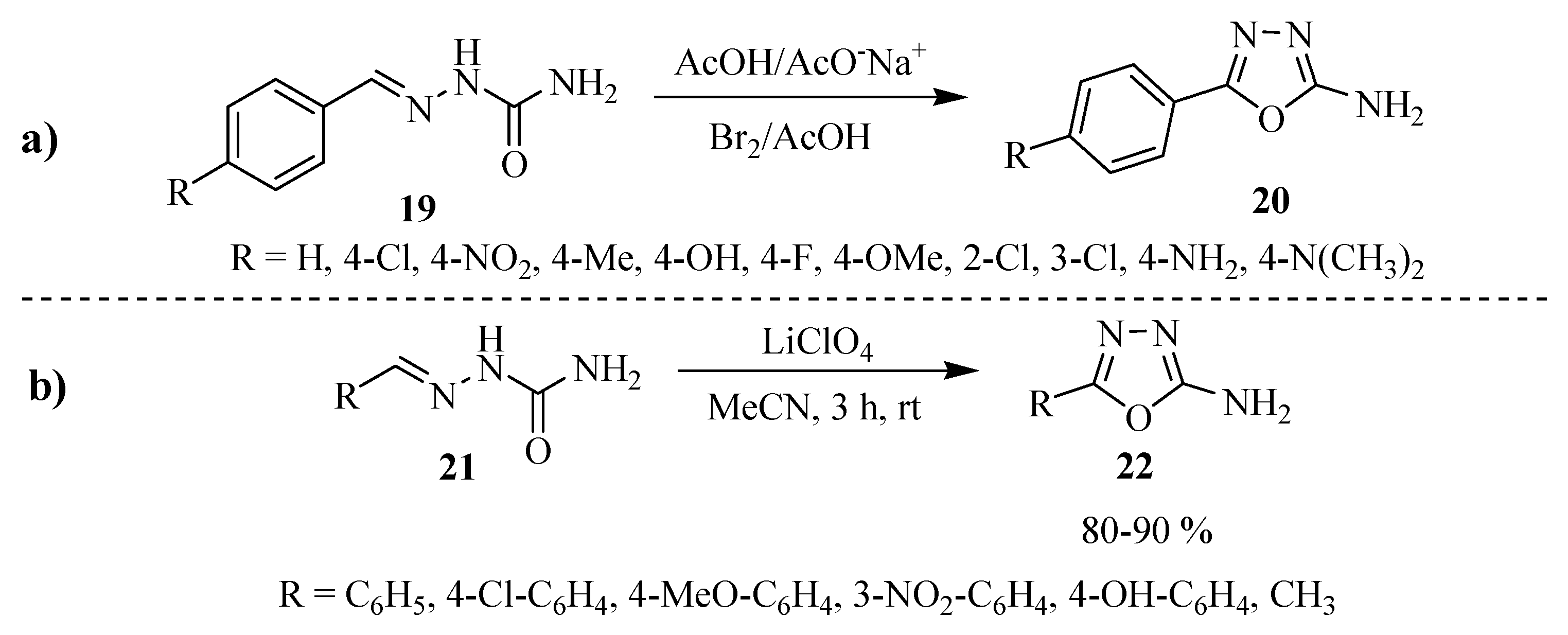
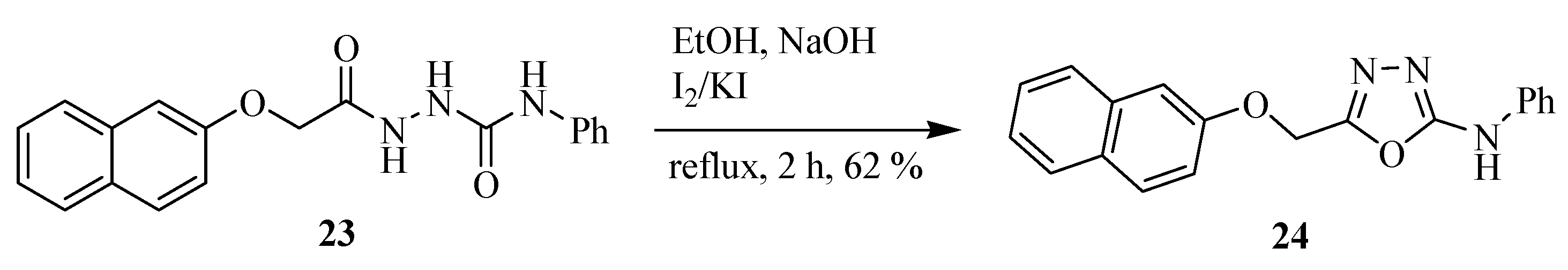
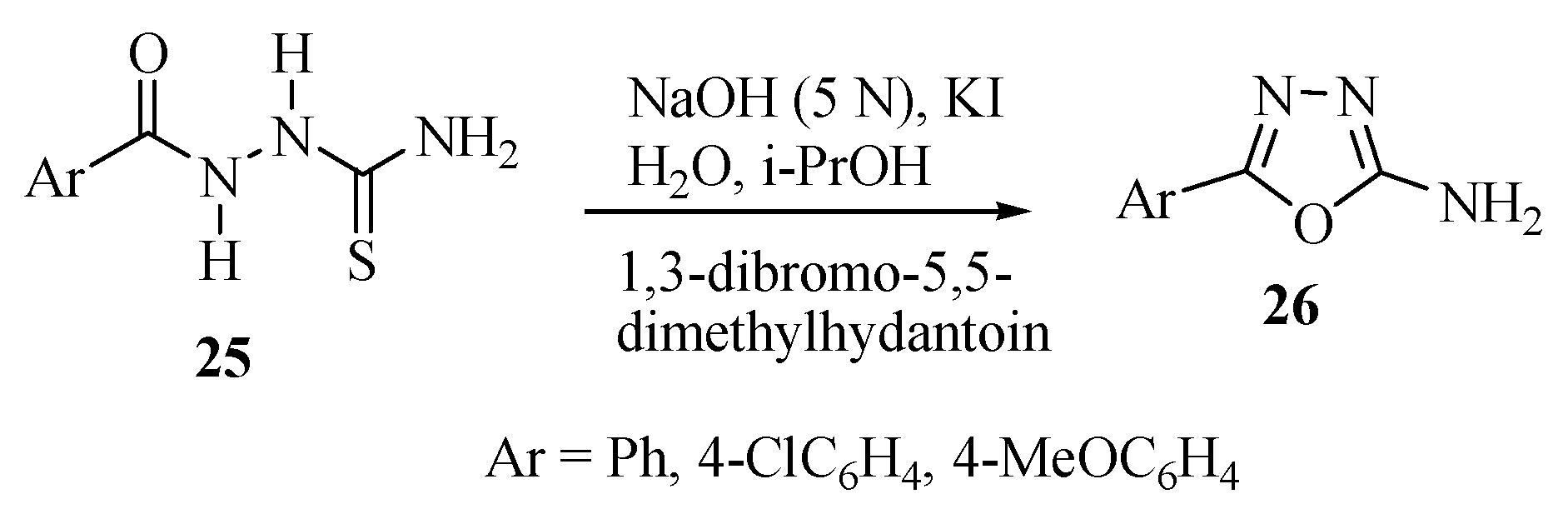
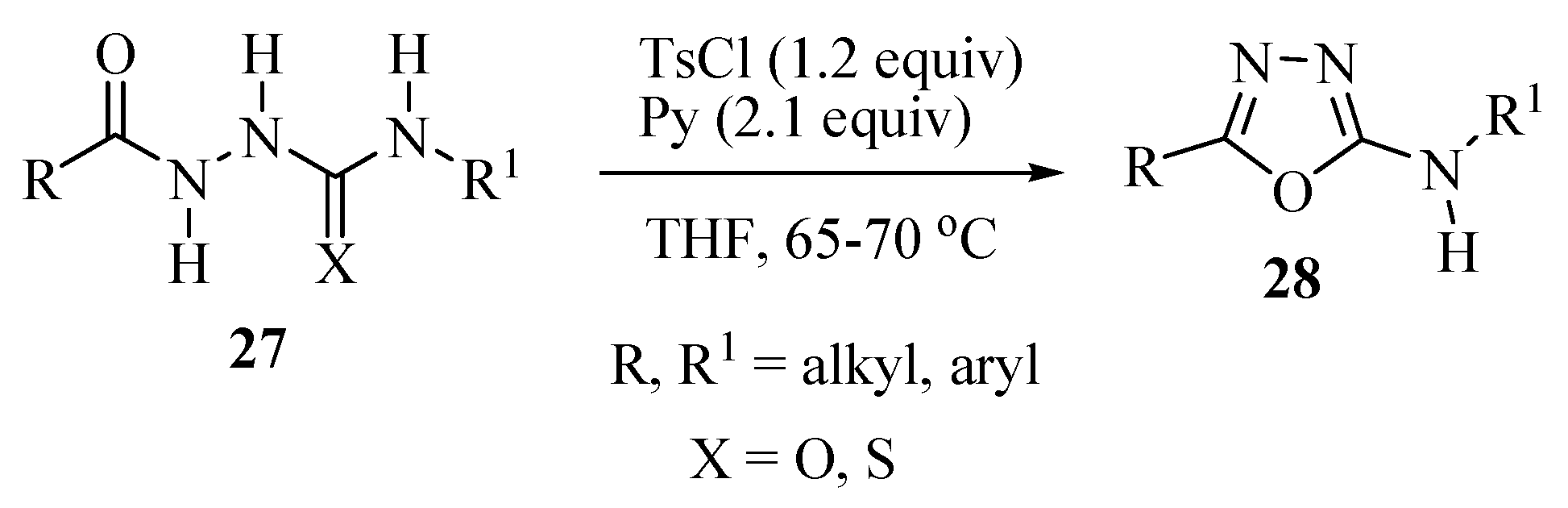
2.2. Methods of Synthesis for 5-Substituted-1,3,4-oxadiazole-2-thiols
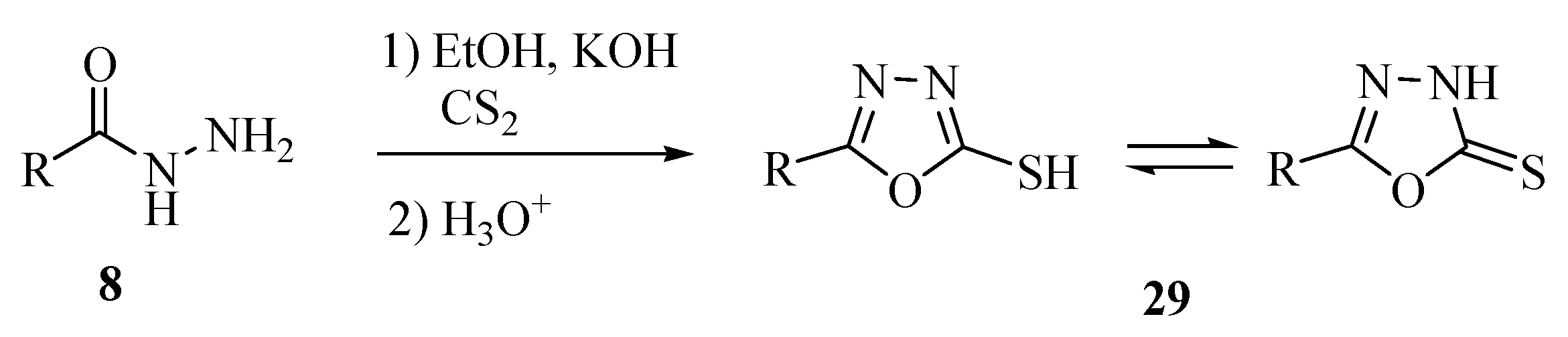
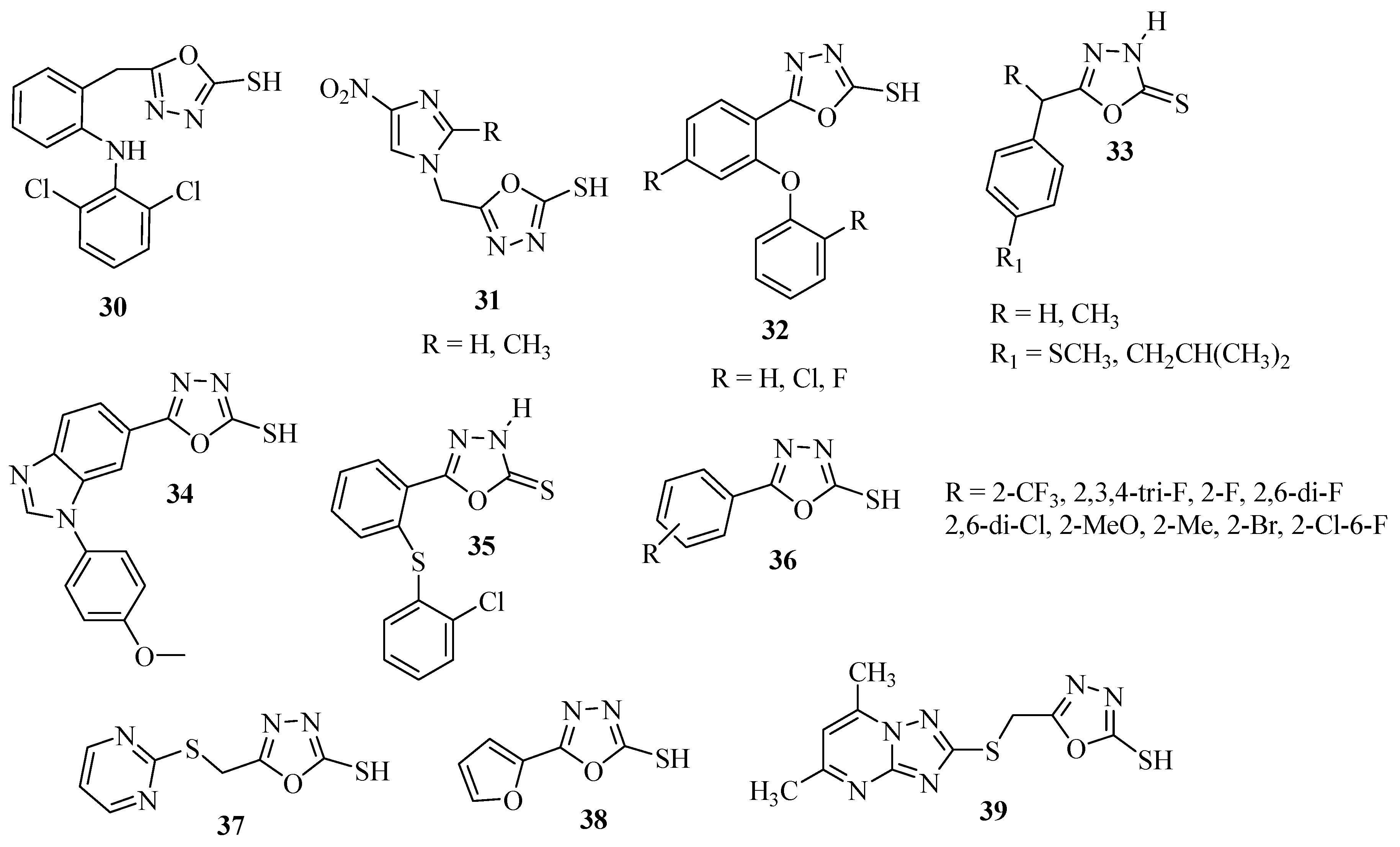
2.3. Methods of Synthesis for 2,5-Diaryl(alkyl)-1,3,4-oxadiazole
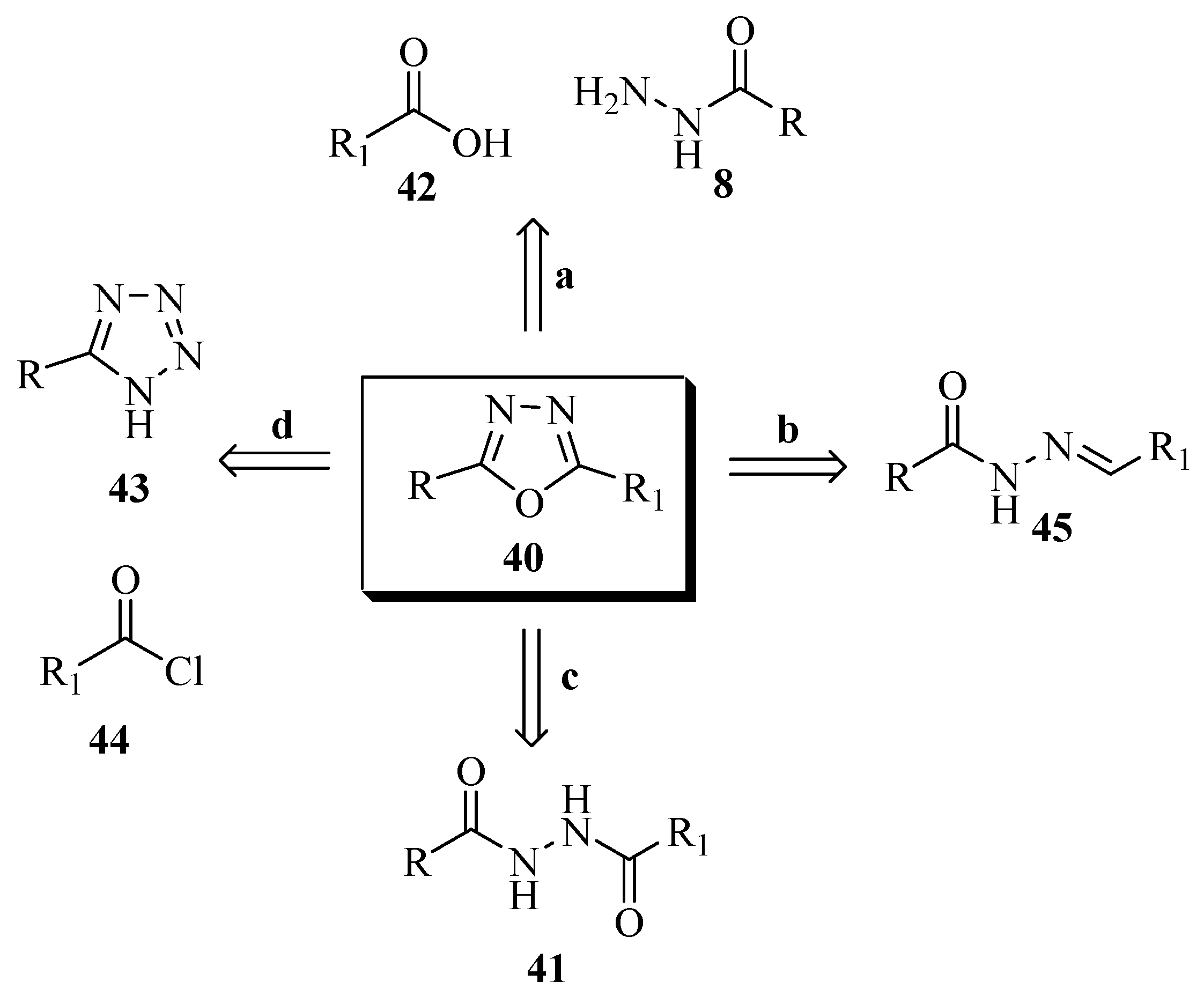
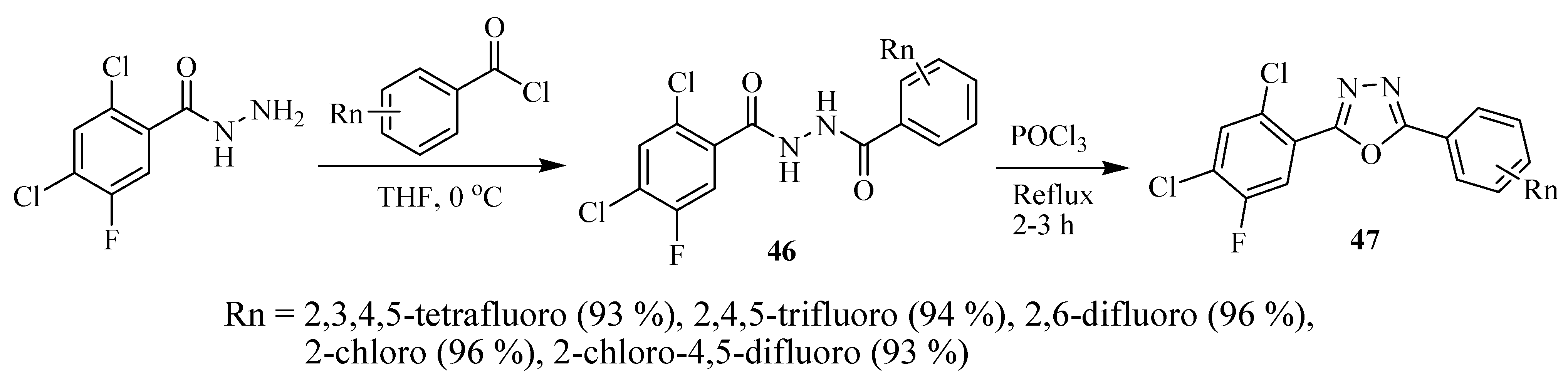

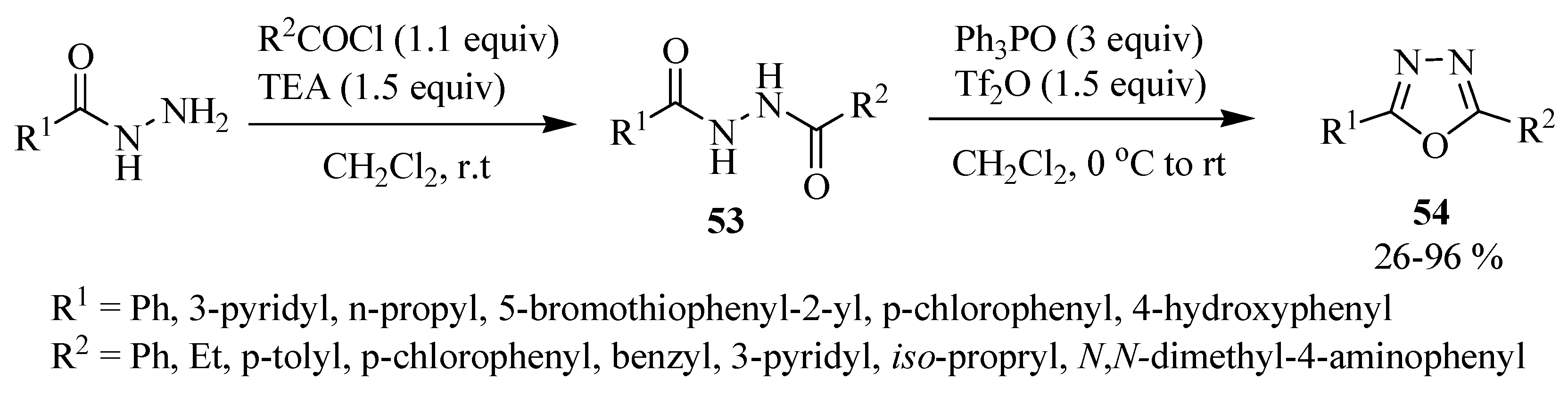
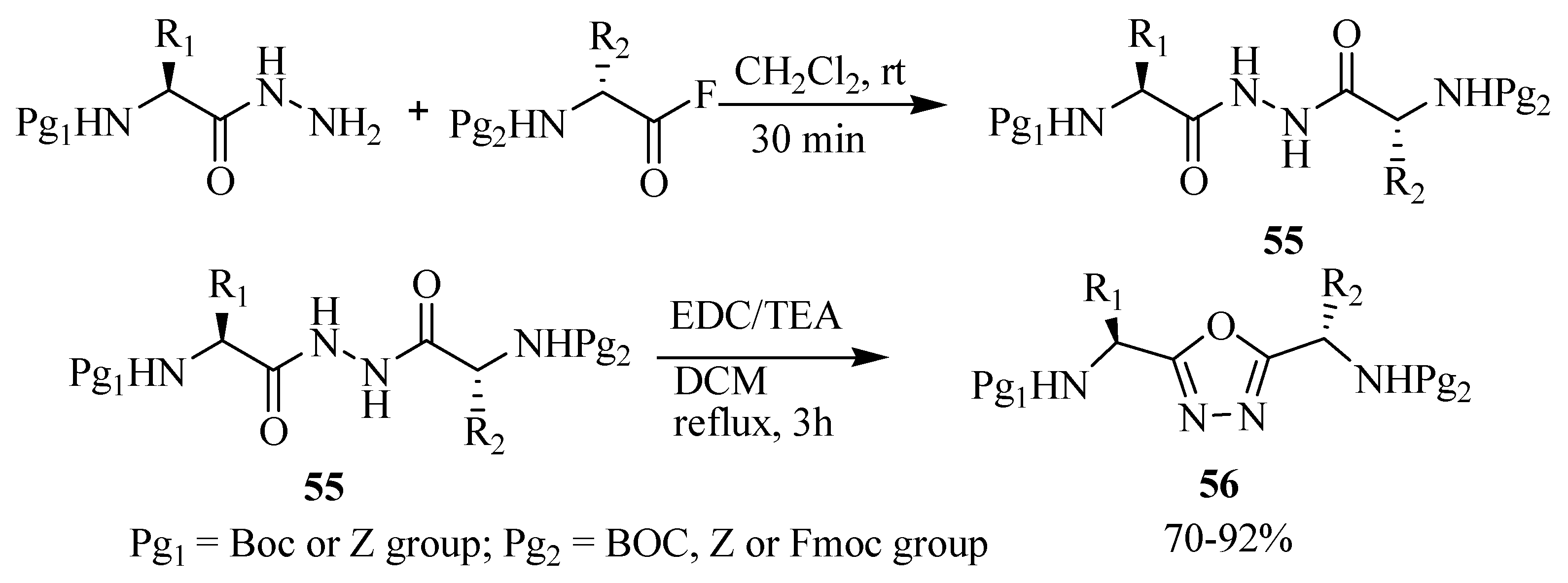
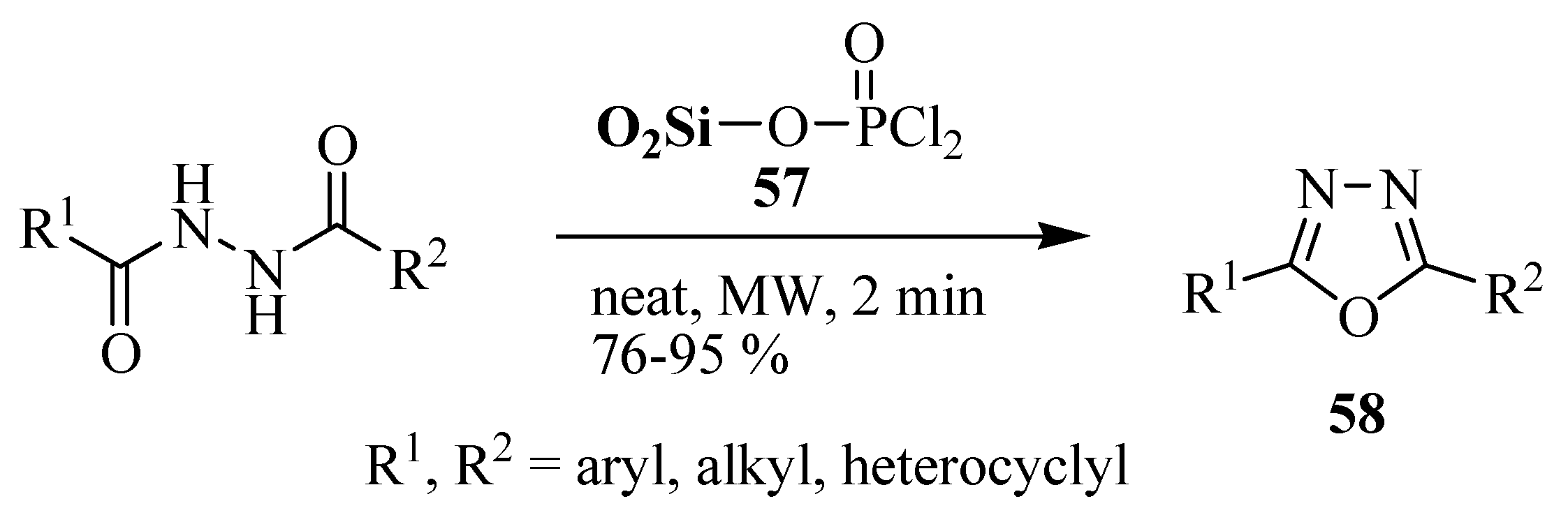
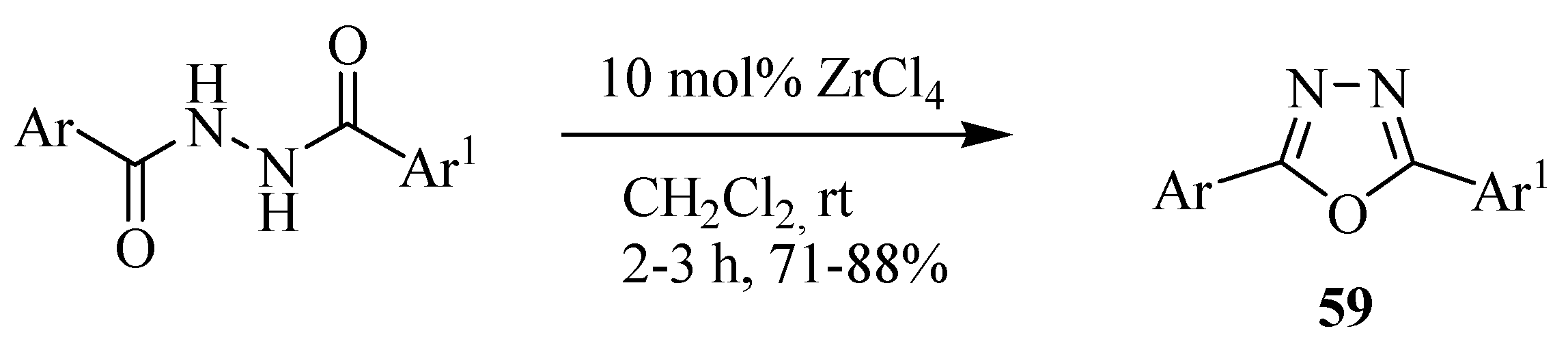
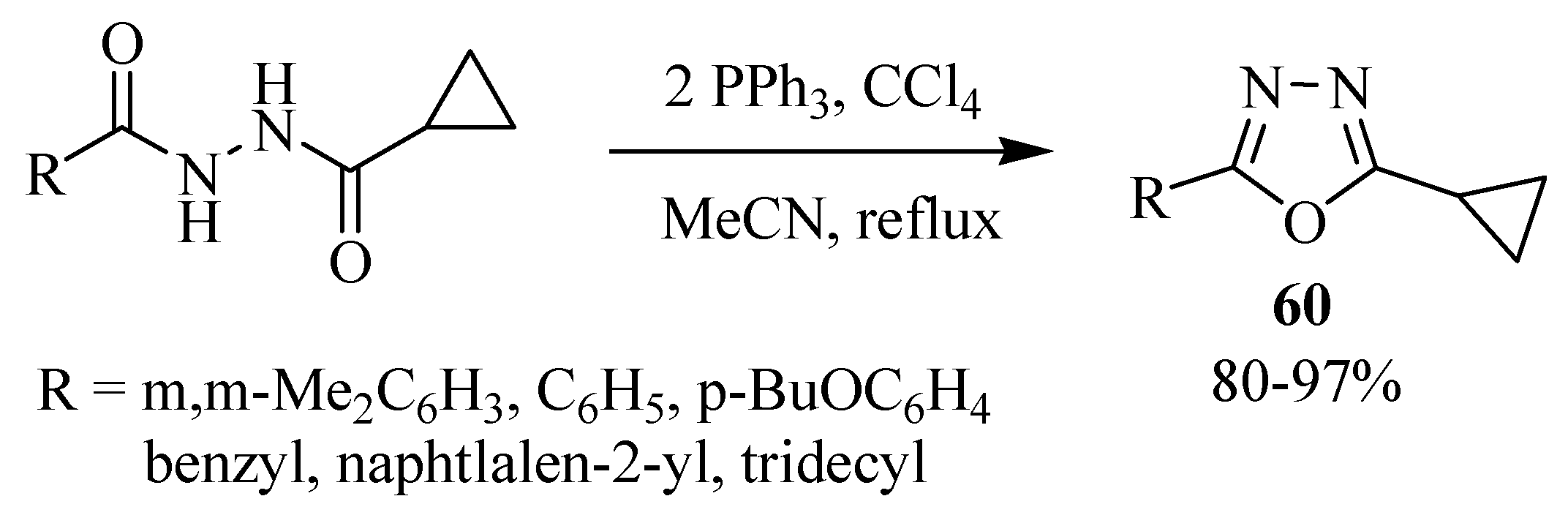
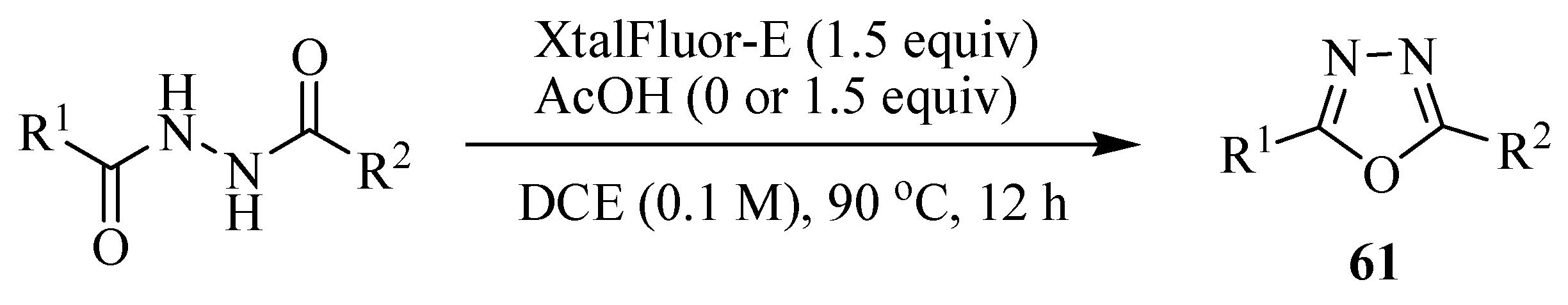

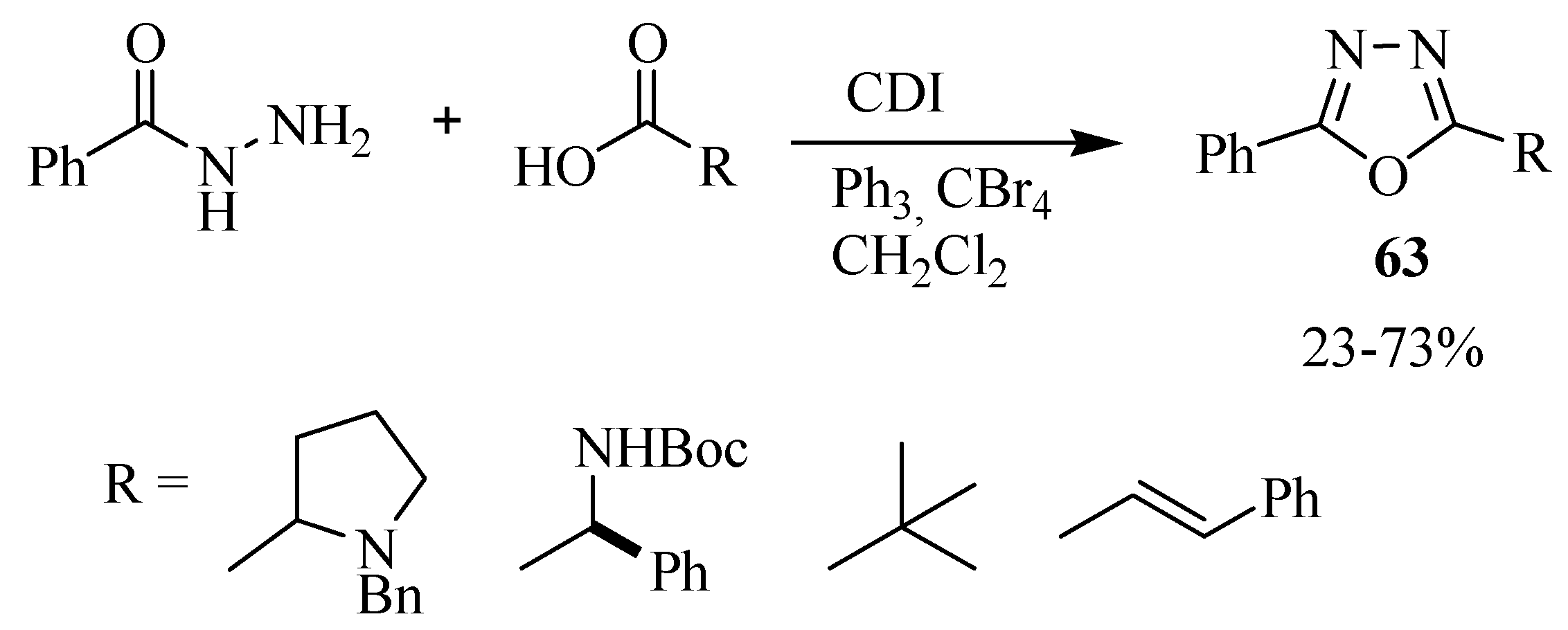
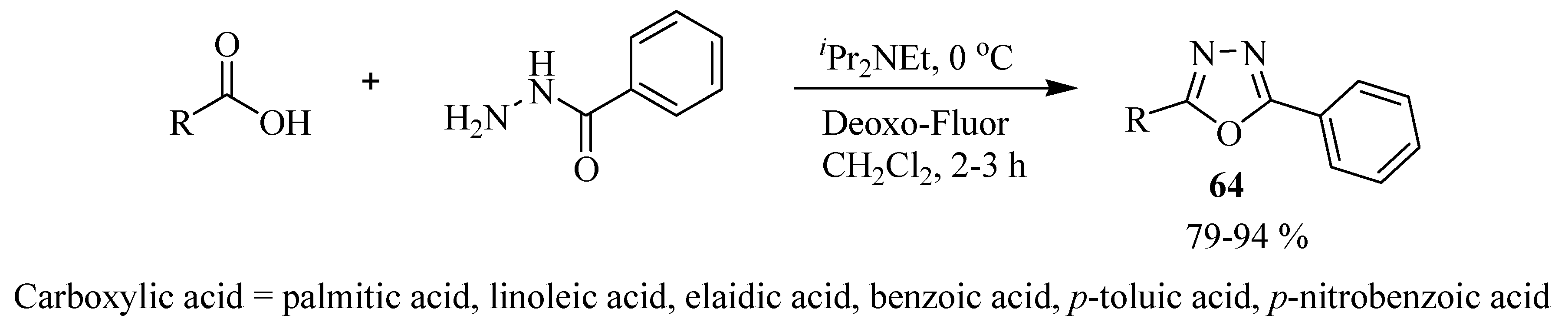
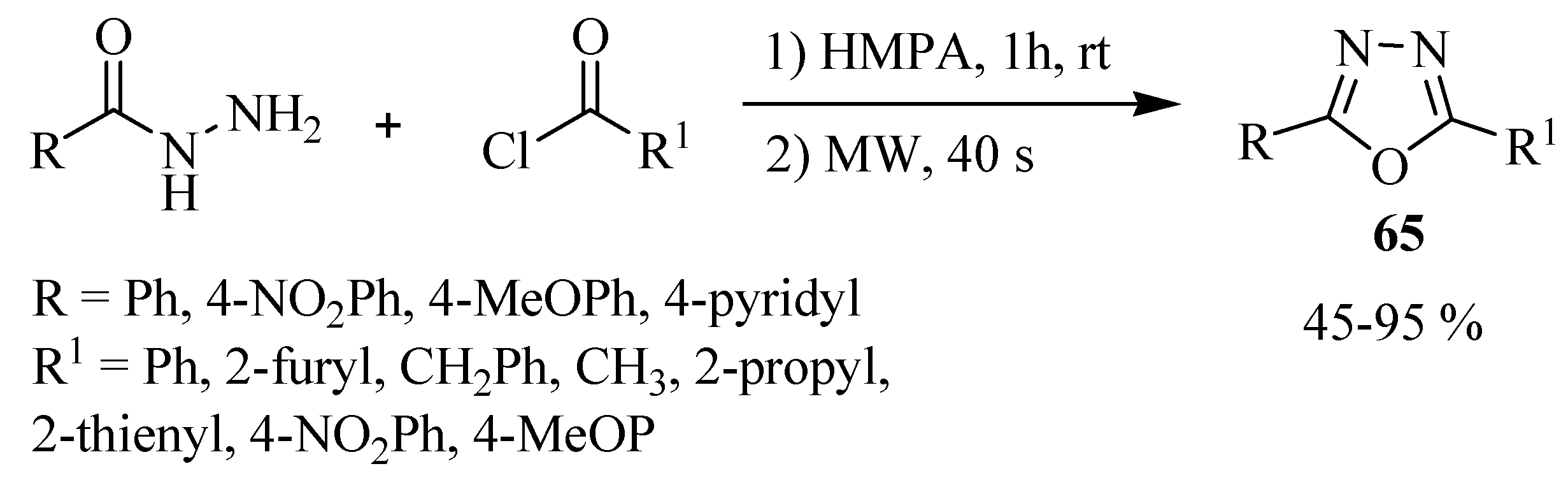
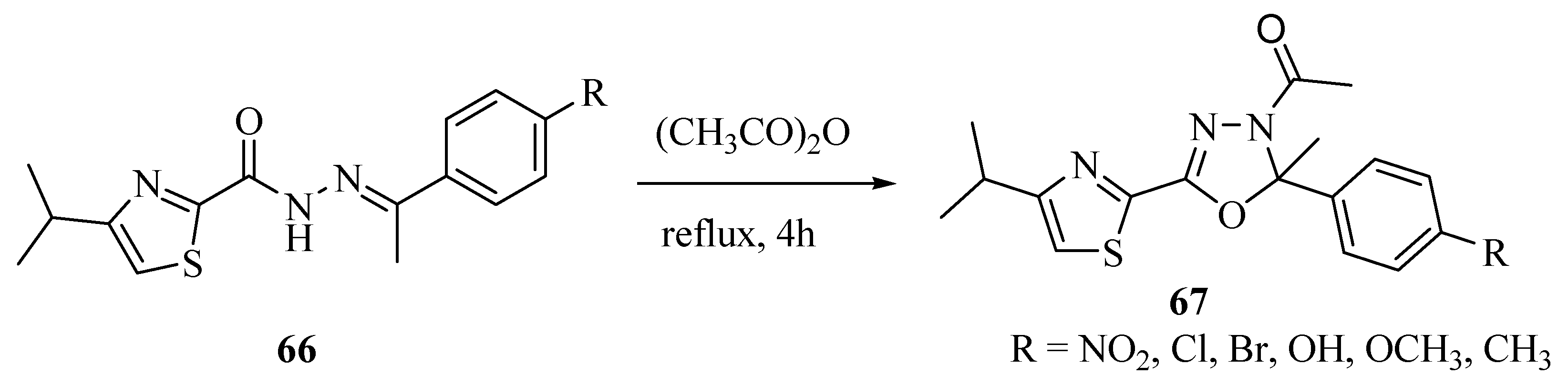
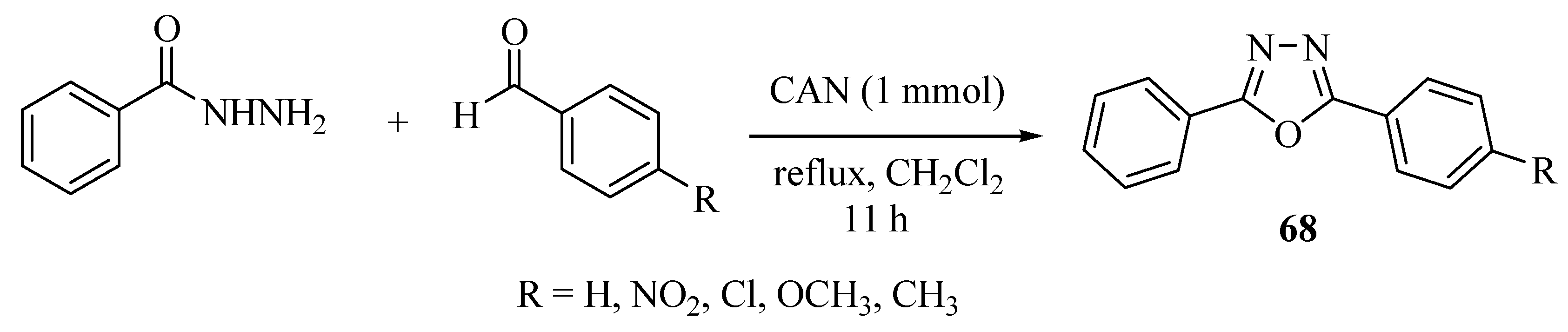
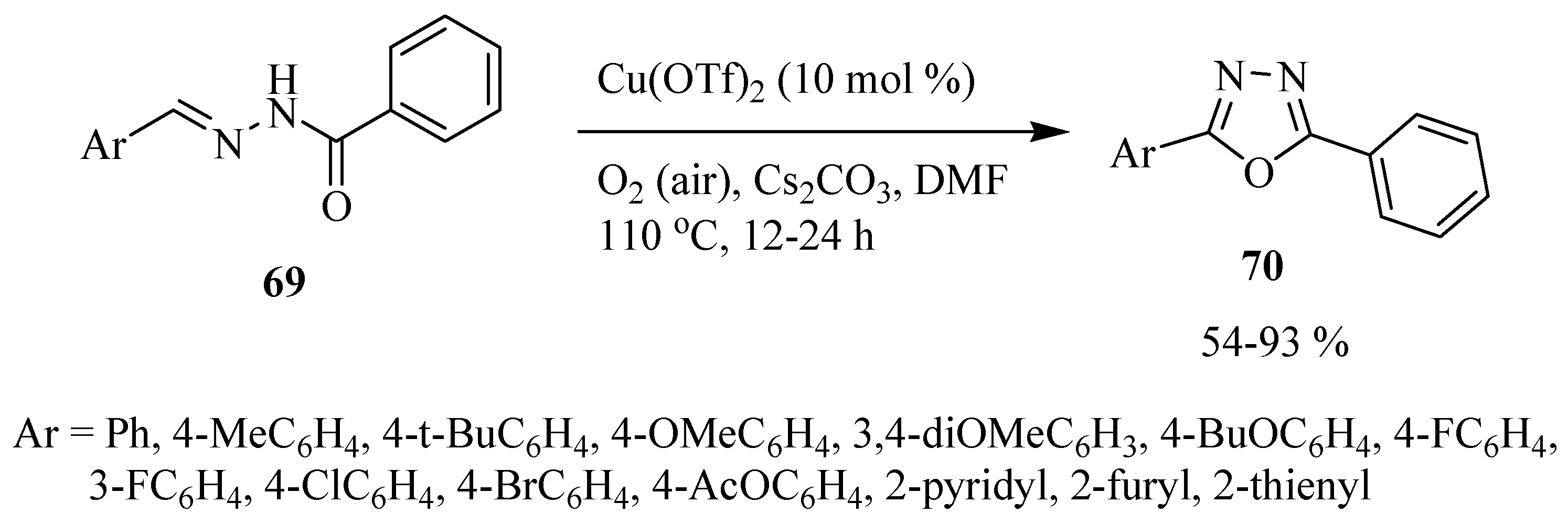
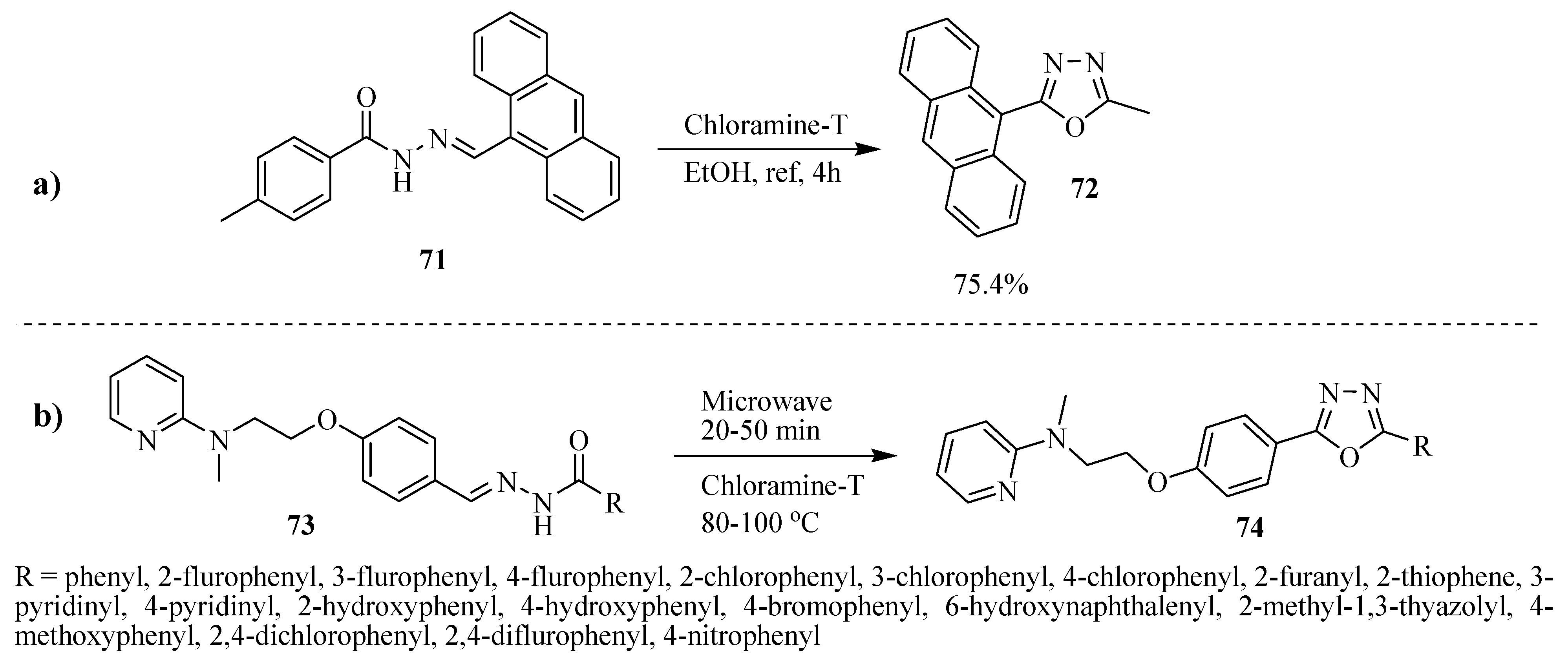
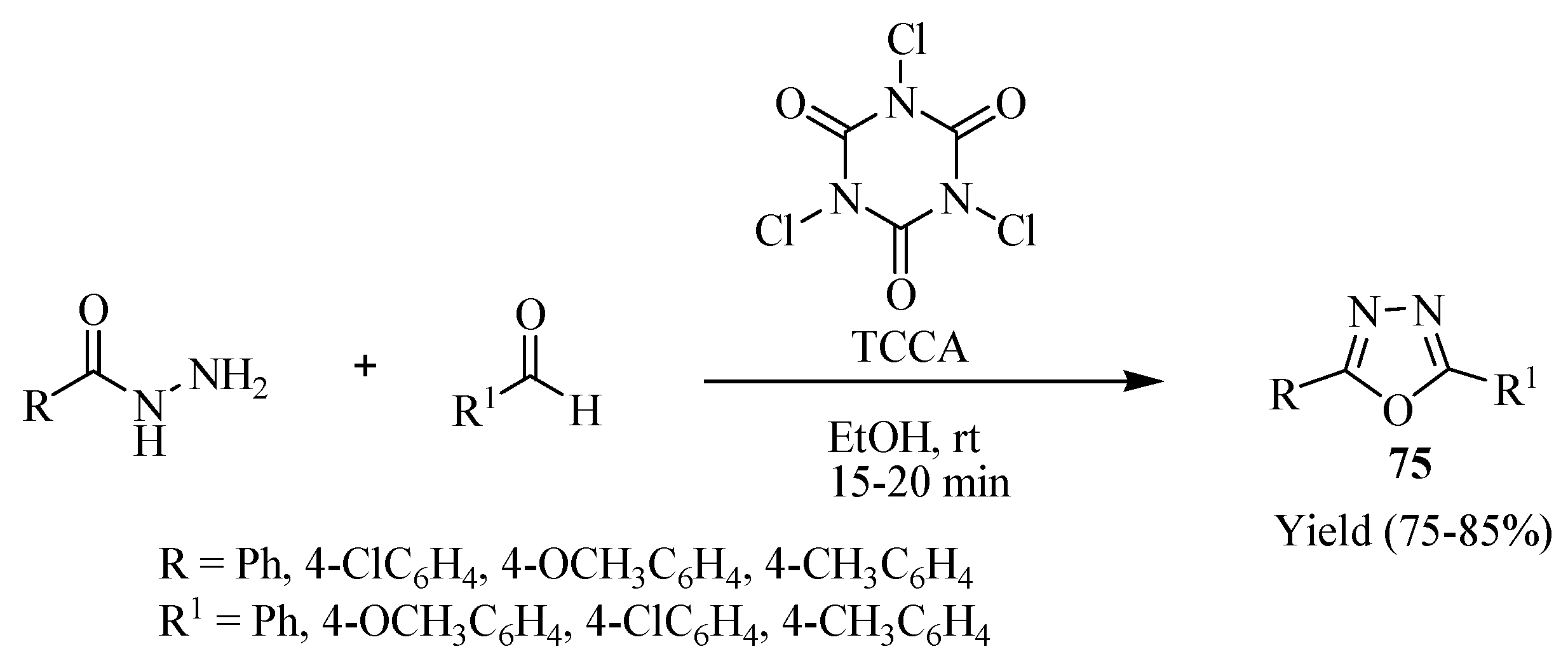
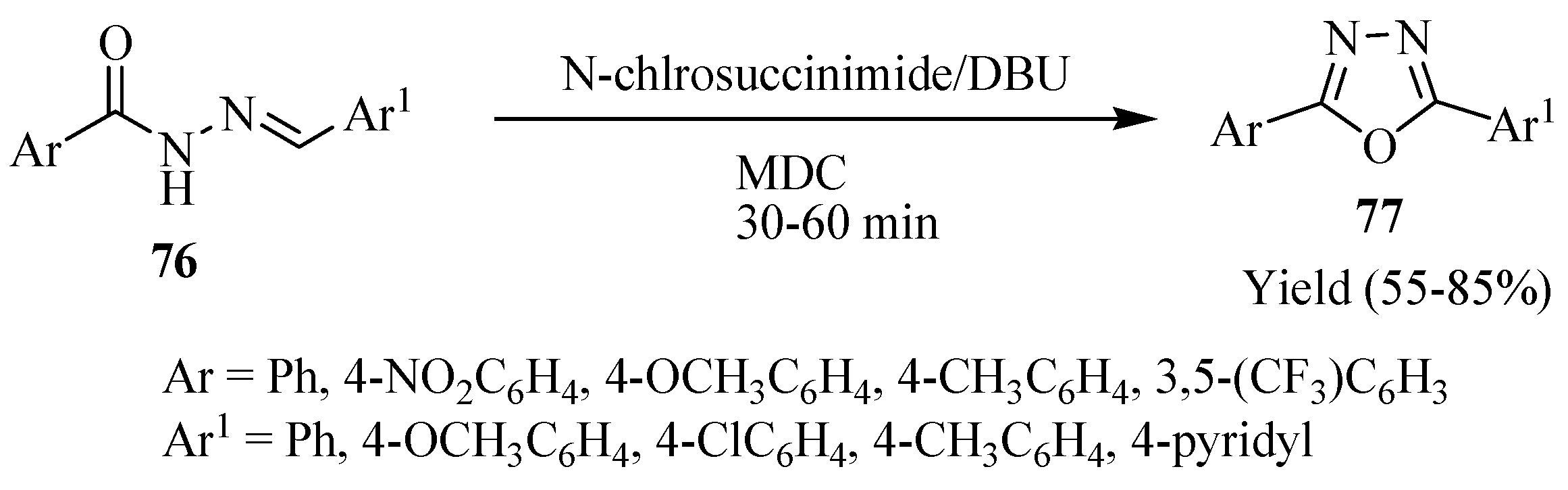
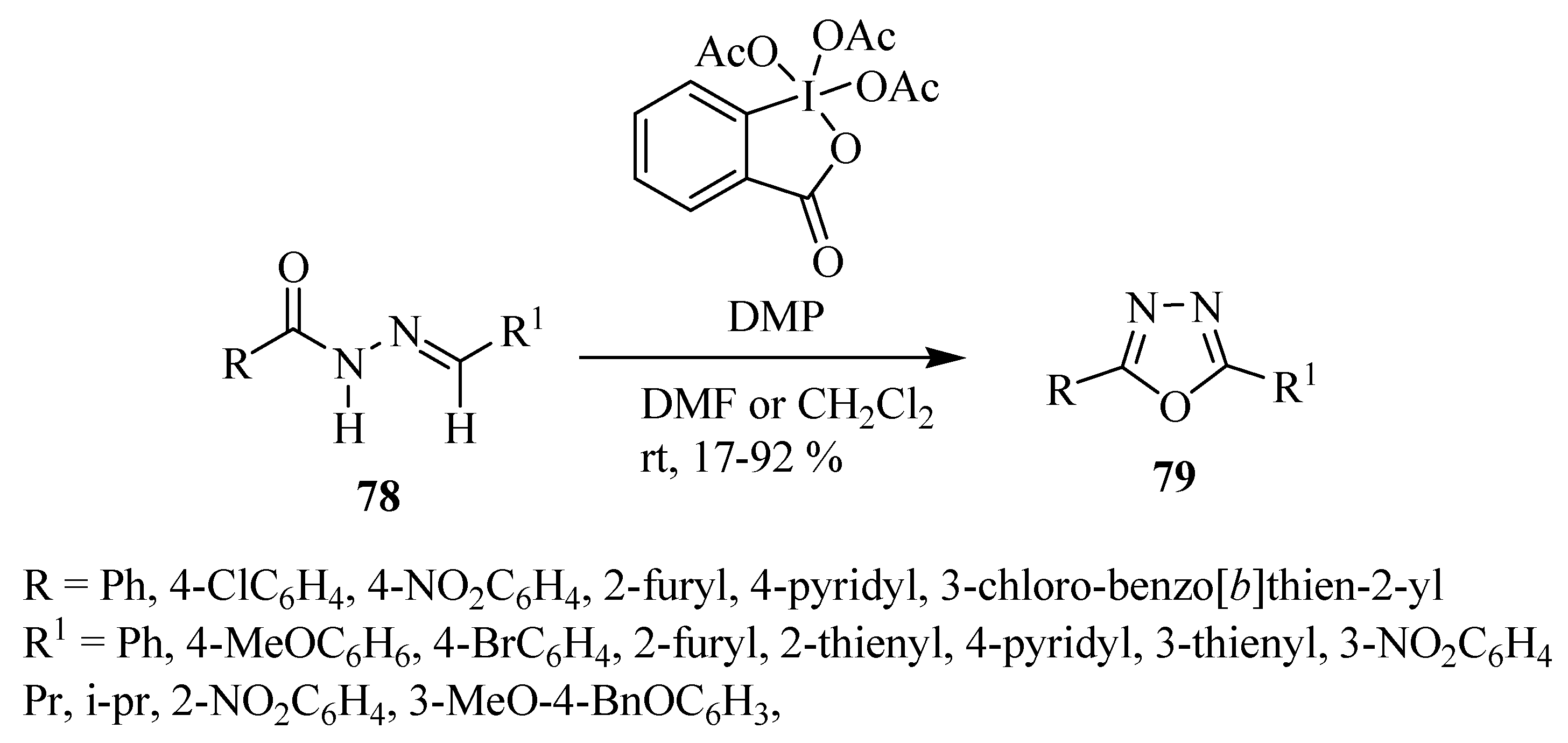
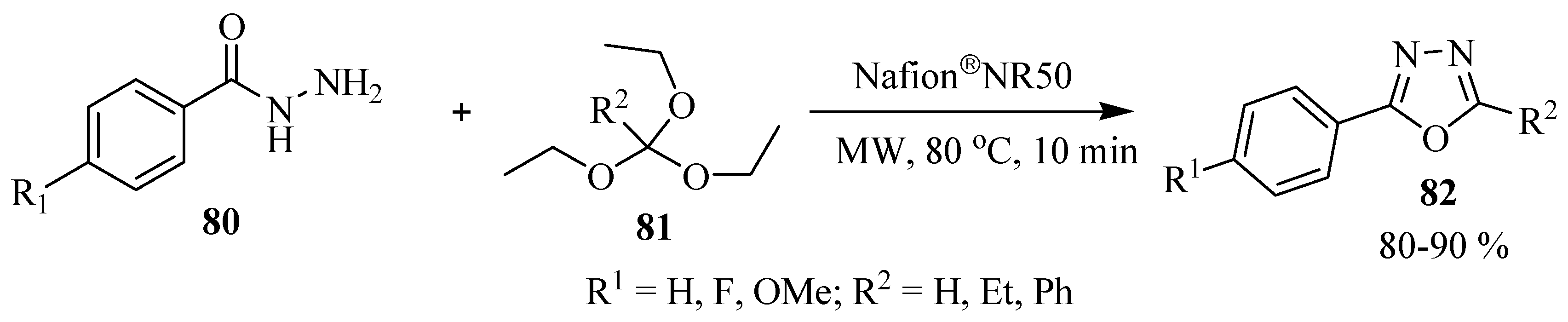
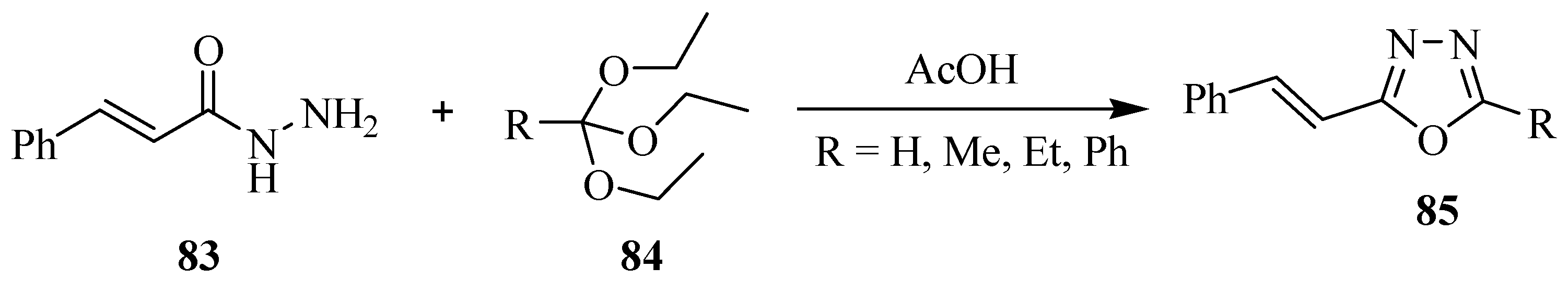
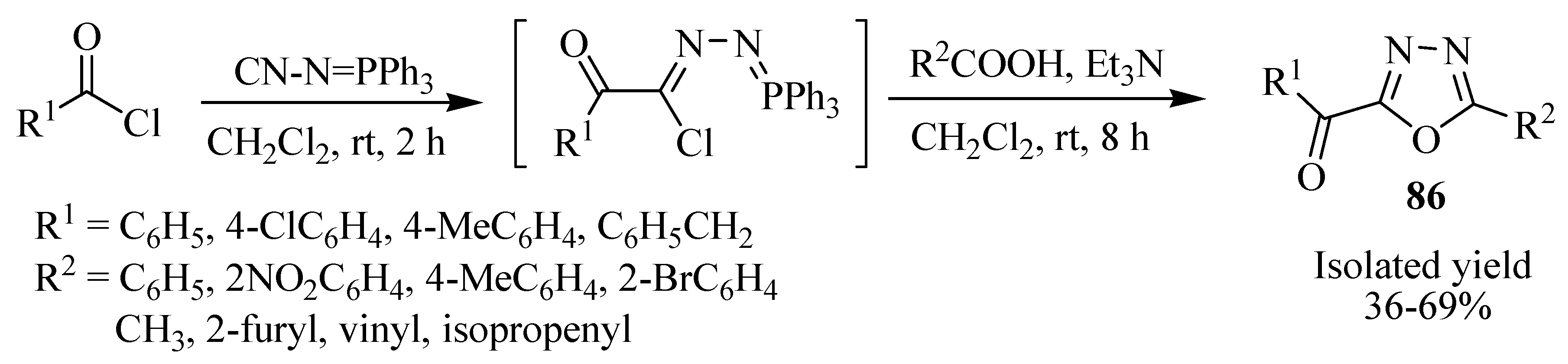
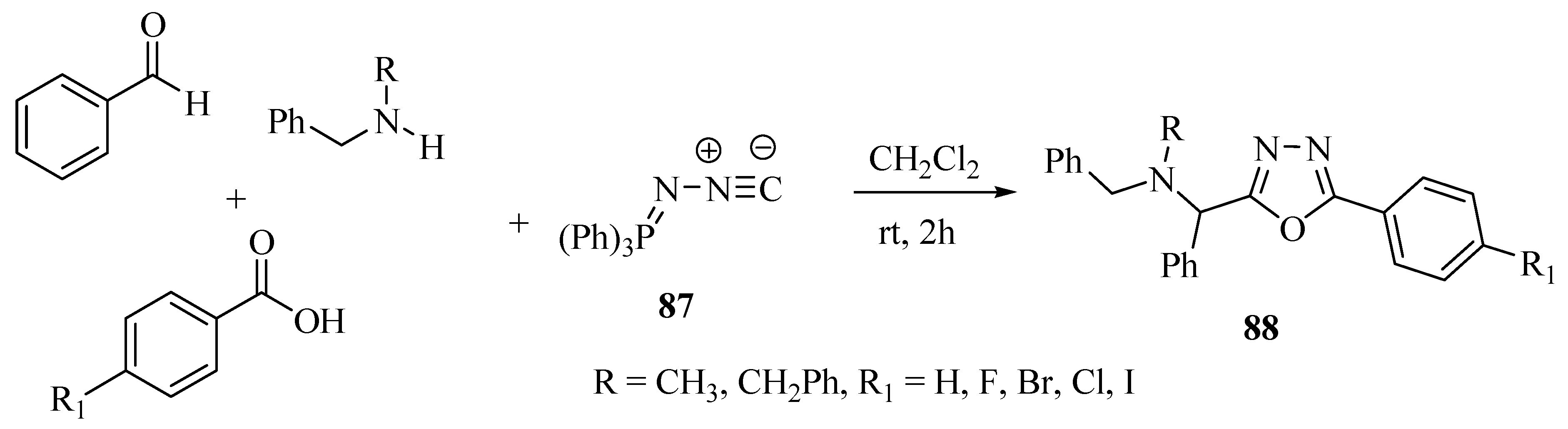
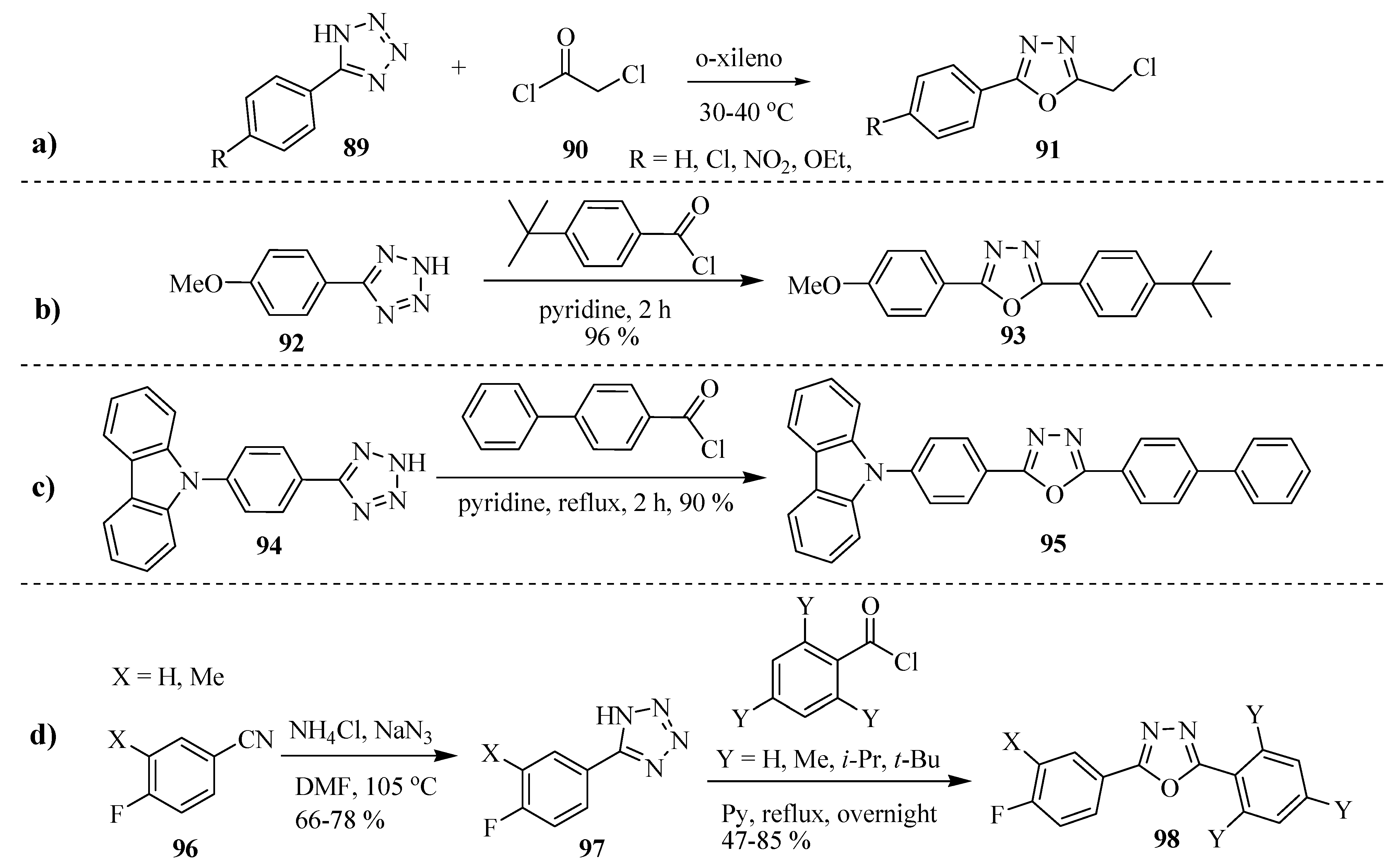
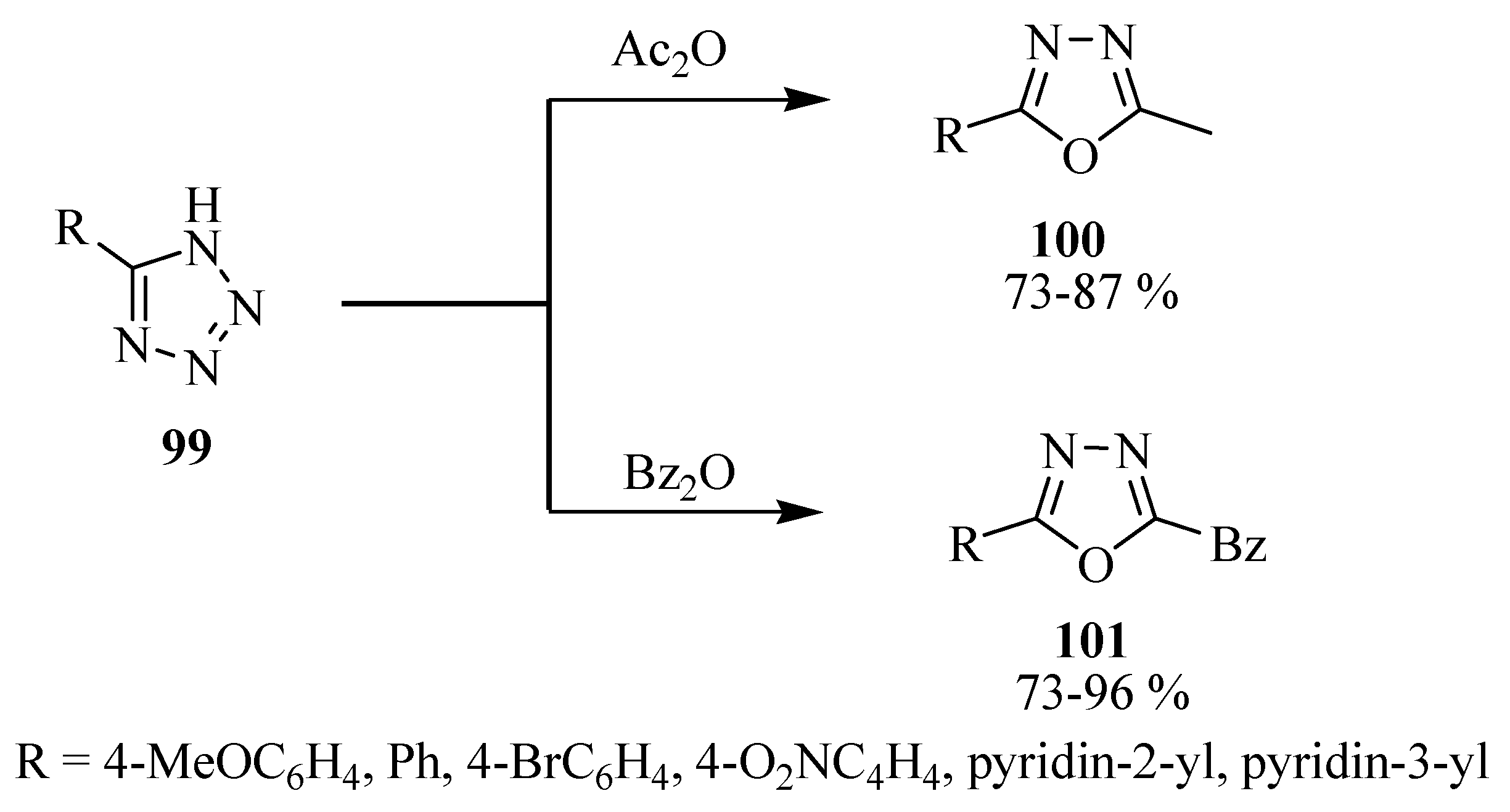
3. Pharmacological Activity of 1,3,4-Oxadiazoles
3.1. Antimicrobial Activity
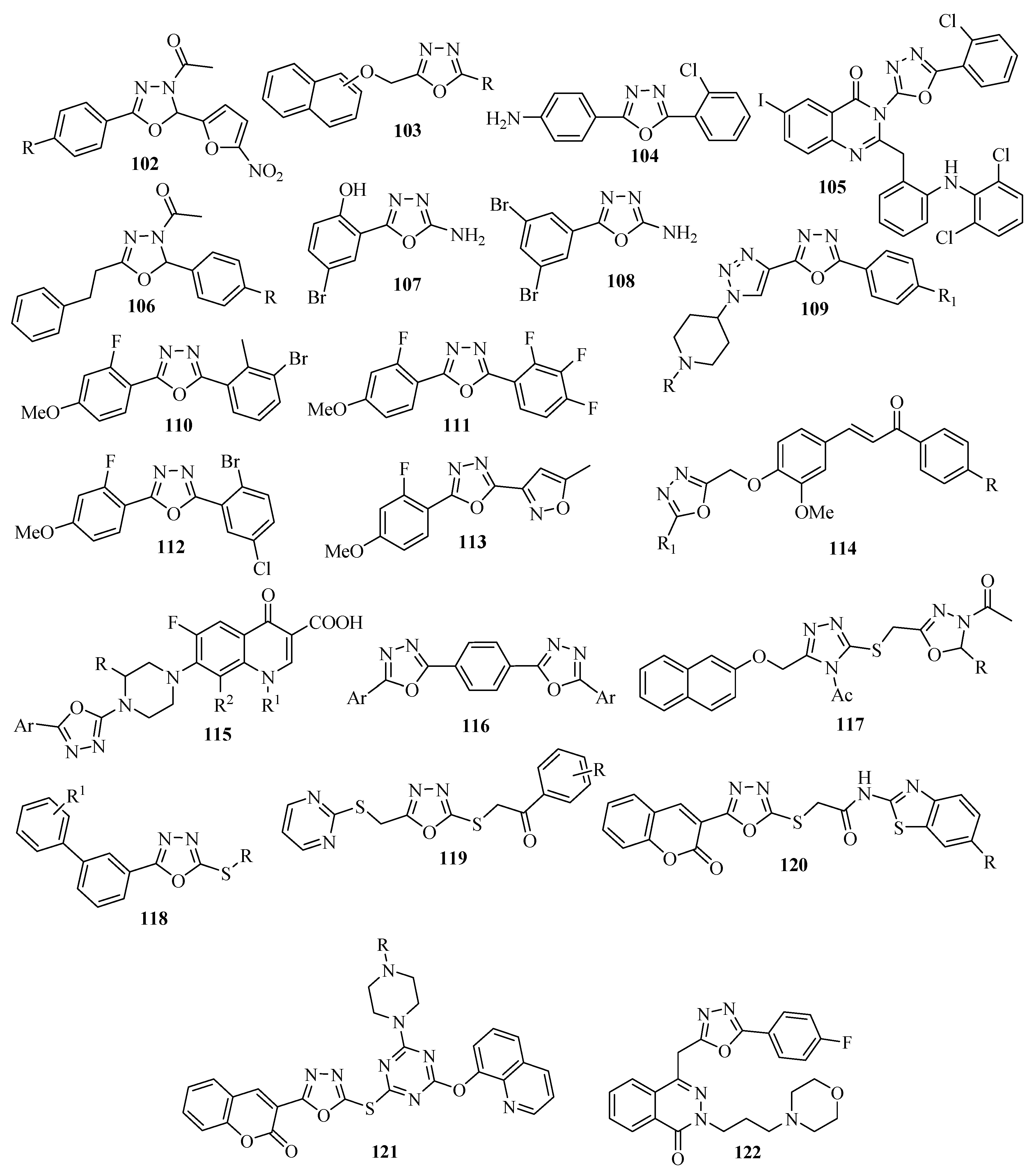
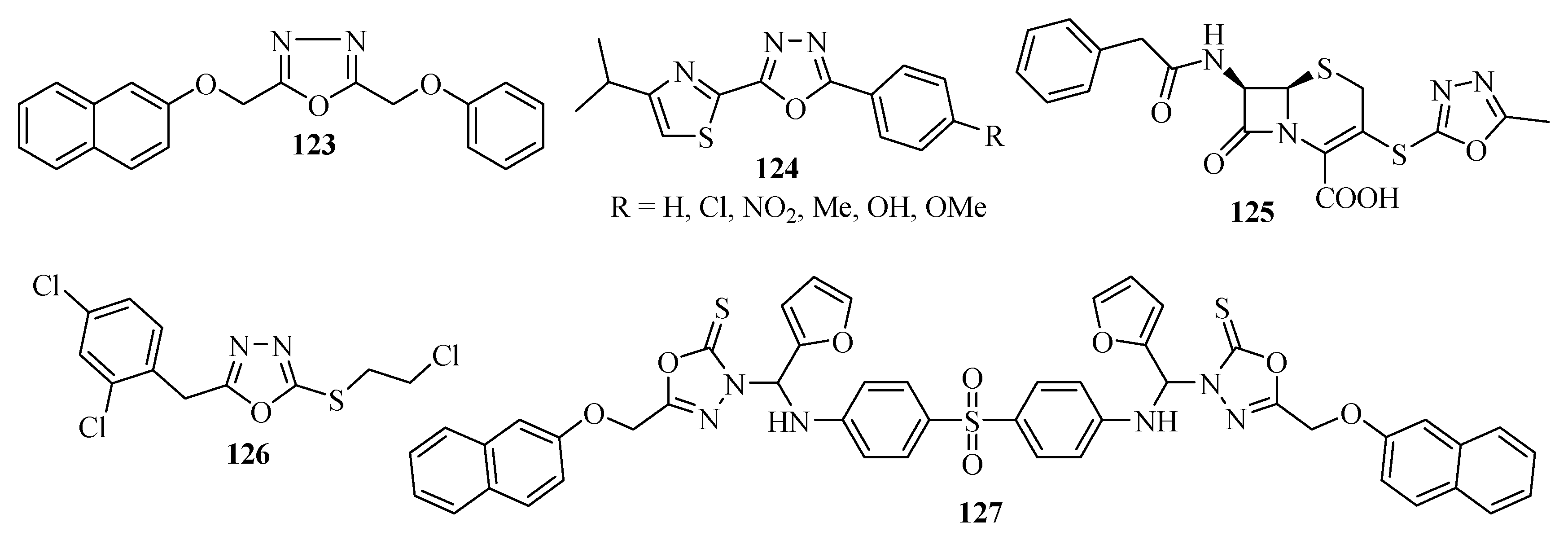
3.2. Anticonvulsant Activity
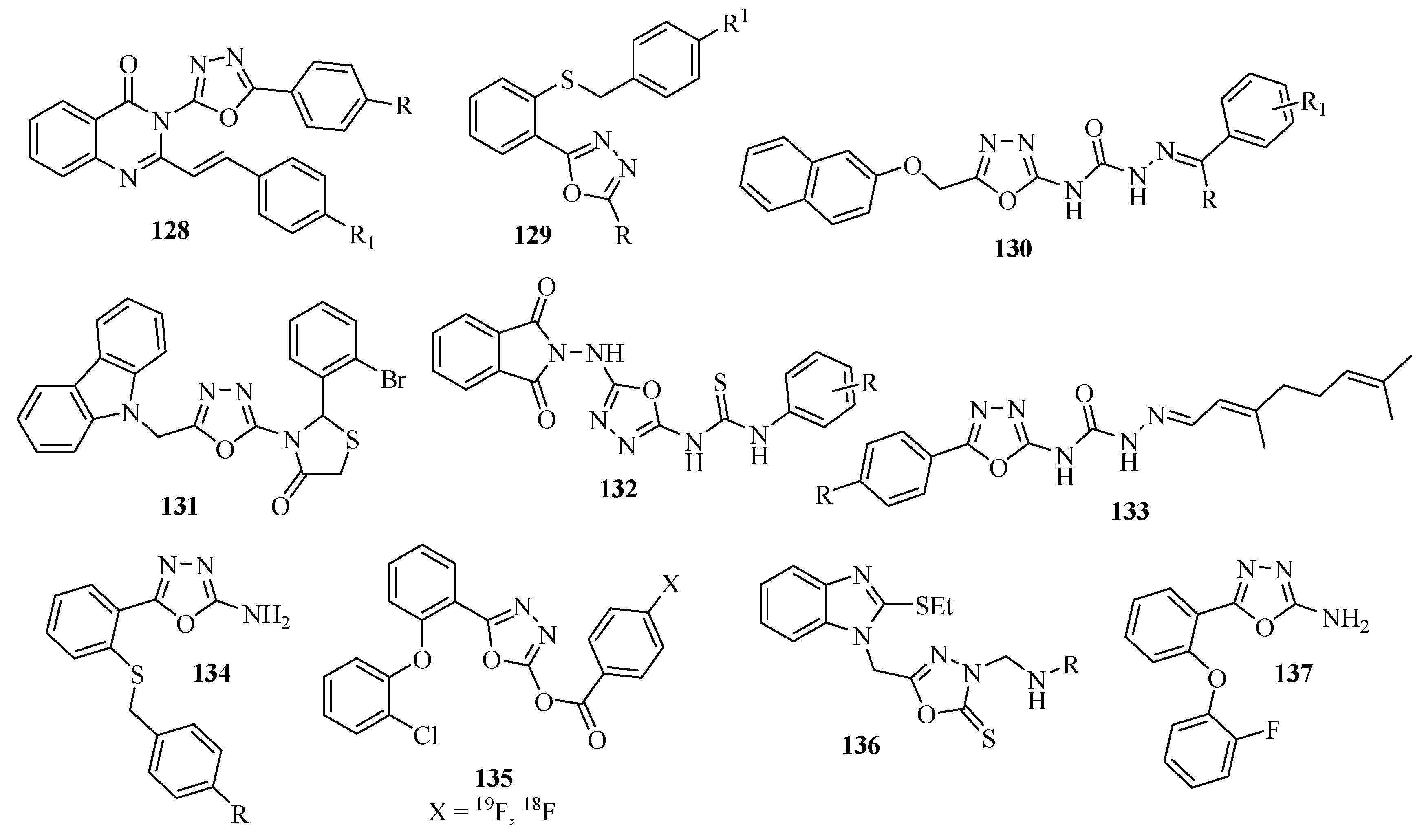
3.3. Anti-inflammatory Activity
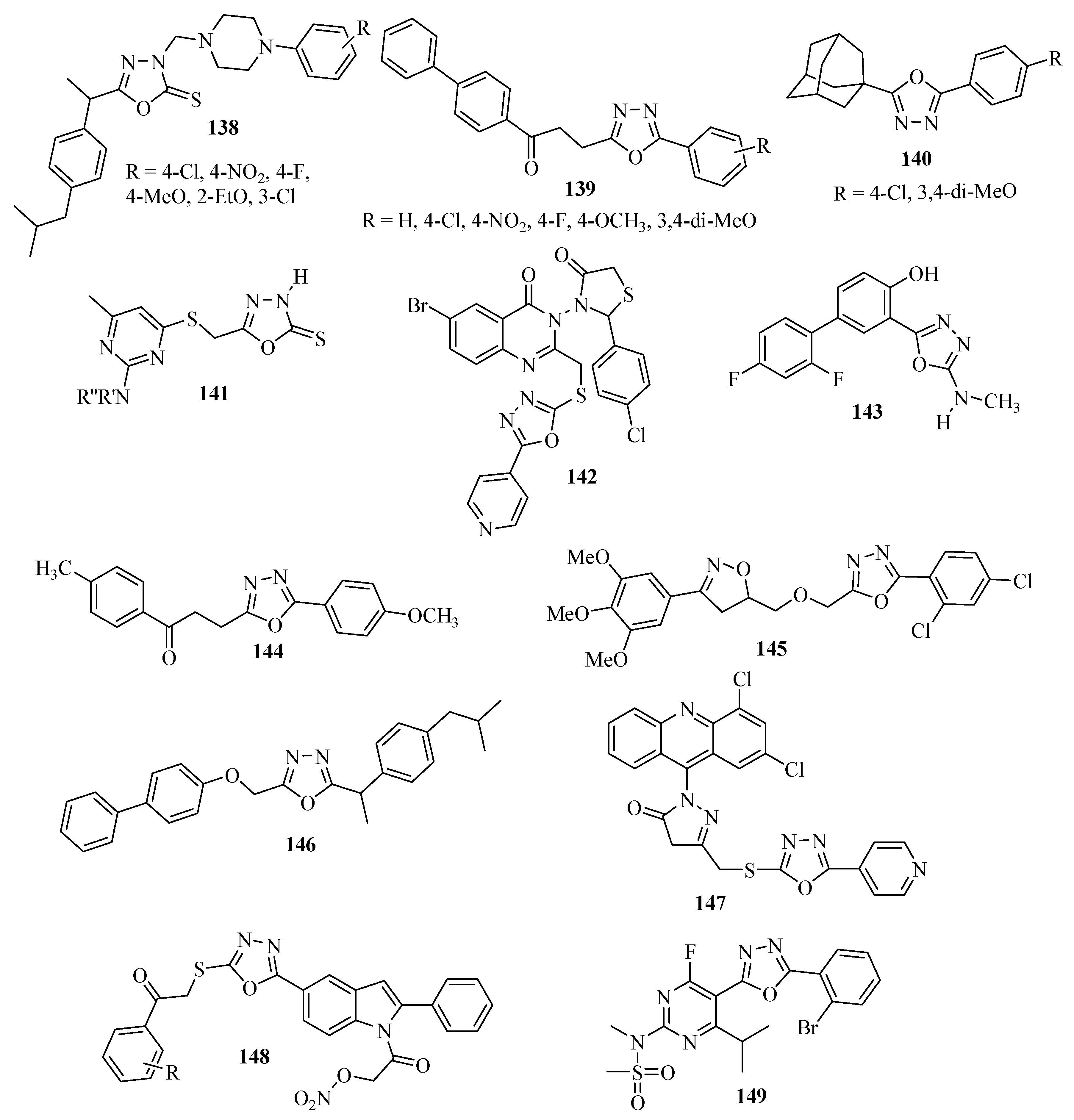
3.4. Analgesic Activity
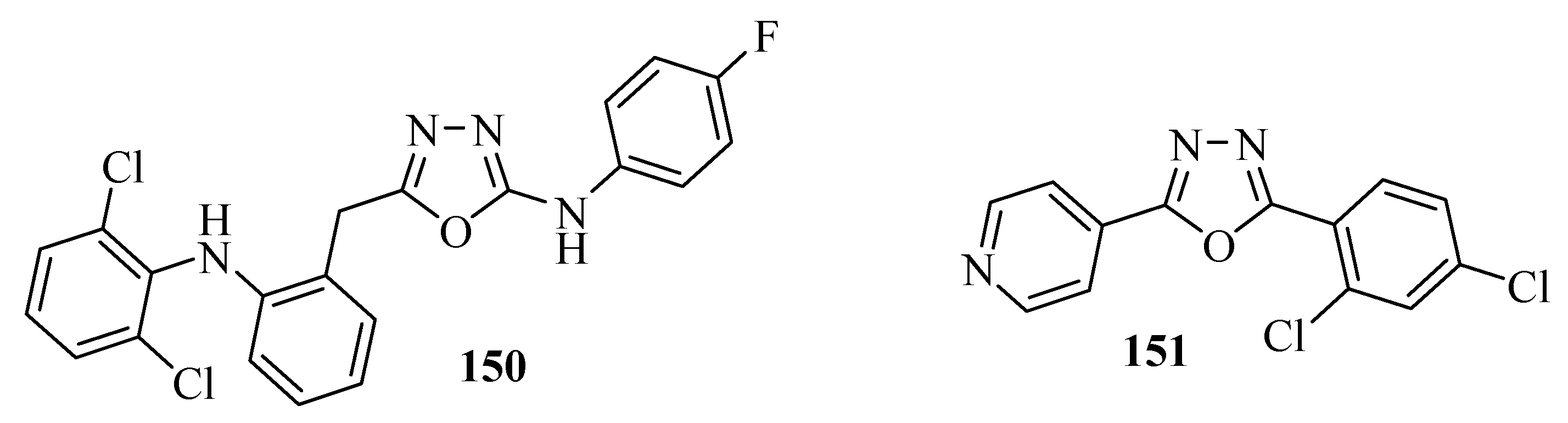
3.5. Antitumor Activity
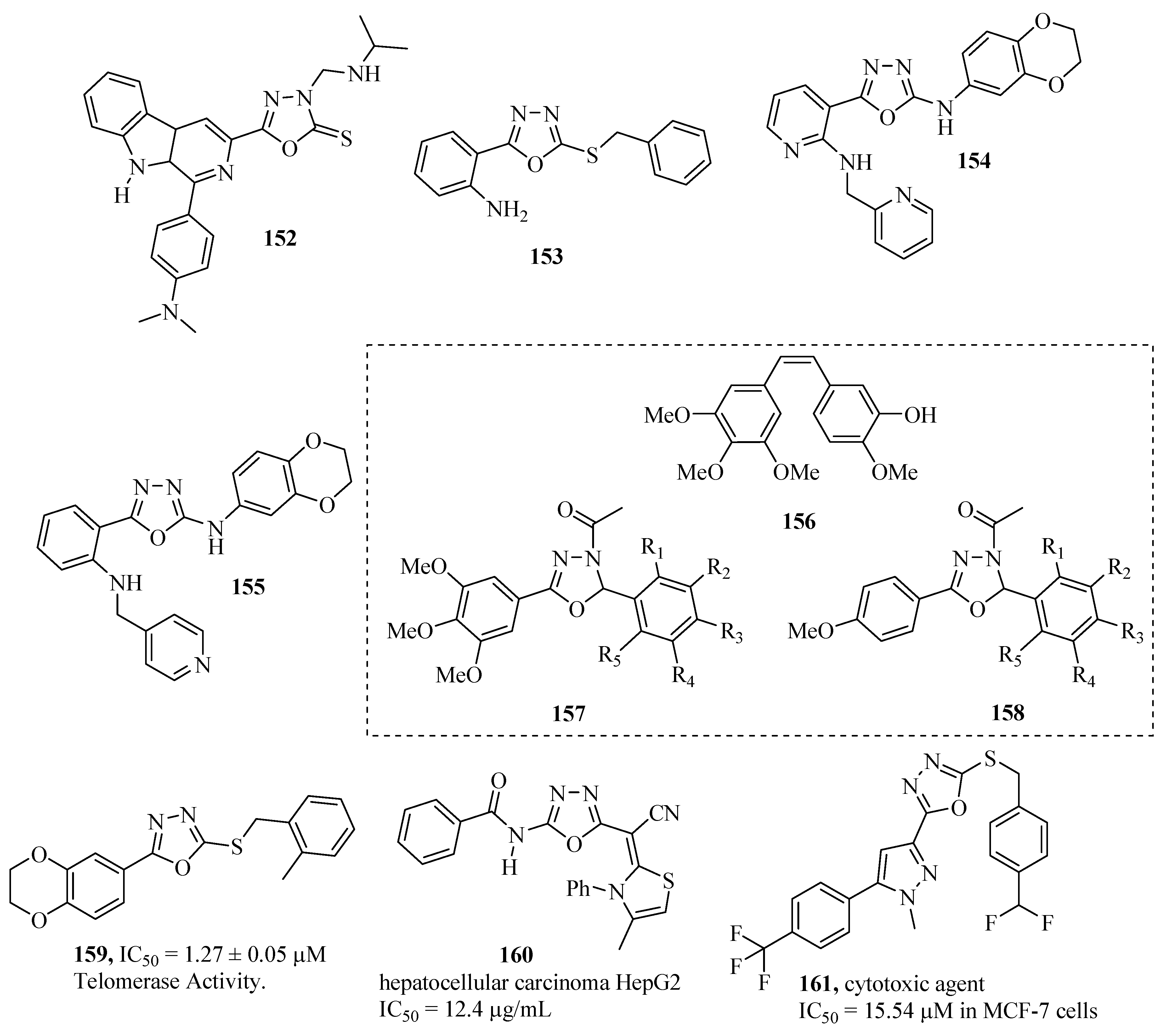
3.7. Antihypertensive Activity

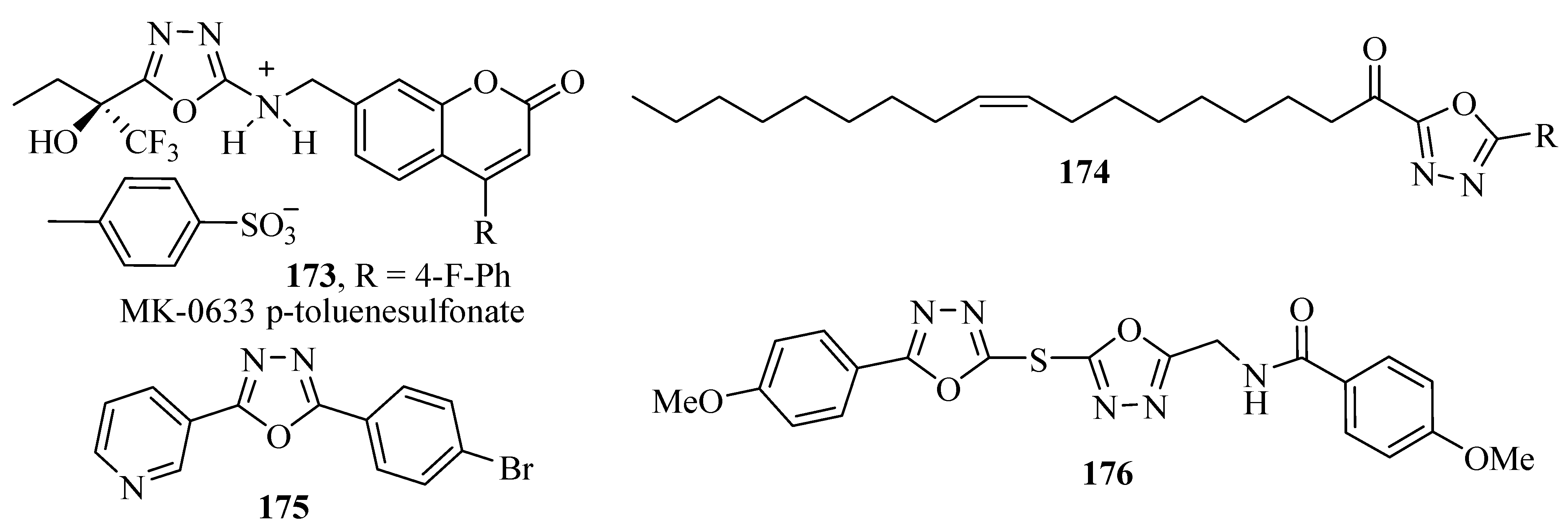
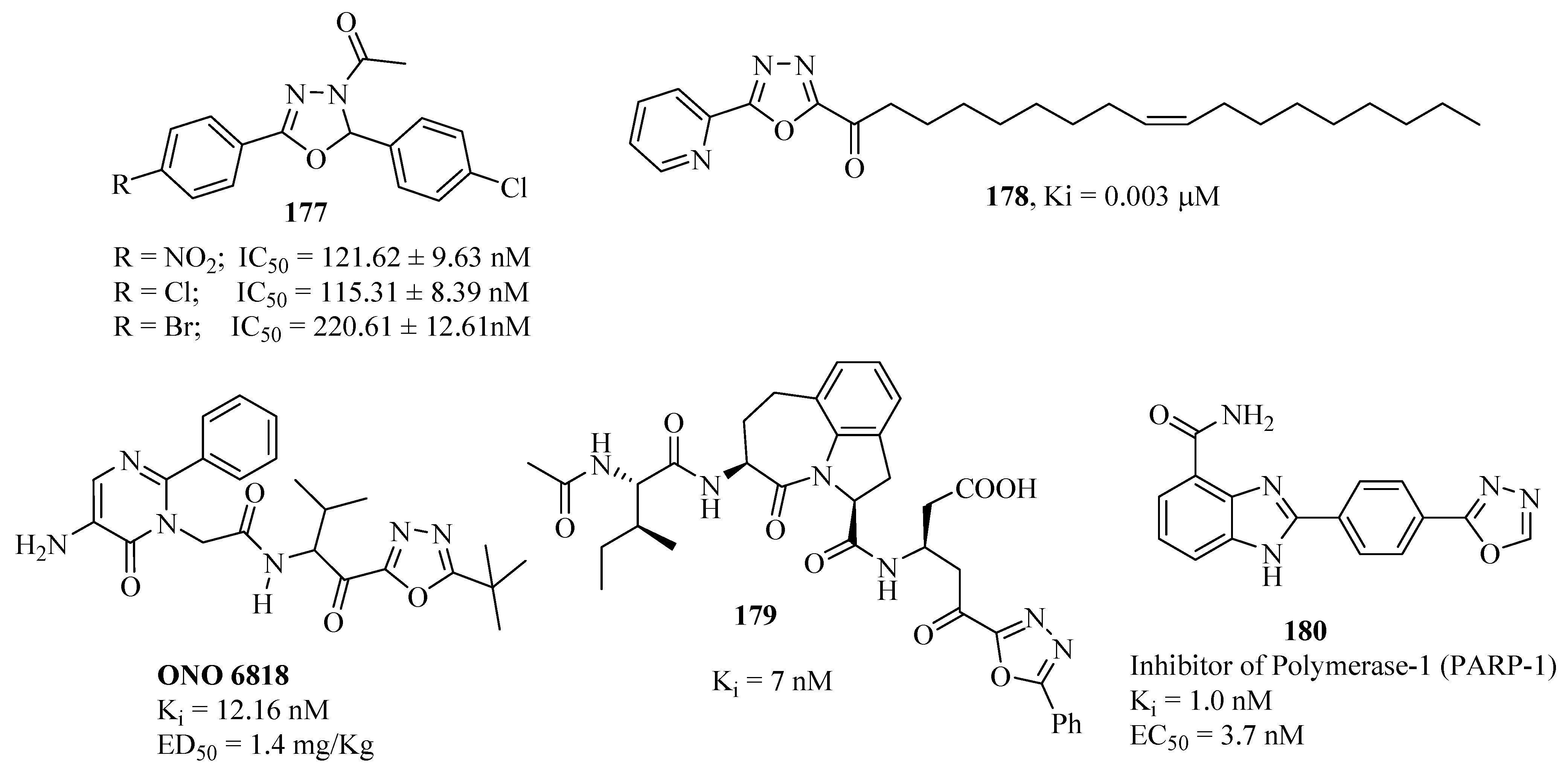
3.8. Enzyme Inhibitors

4. Conclusions
Acknowledgments
Conflict of Interest
References
- Nagaraj; Chaluvaraju, K.C.; Niranjan, M.S.; Kiran, S. 1,3,4-Oxadiazole: A potent drug candidate with various pharmacological activities. Int. J. Pharm. Pharm. Sci. 2011, 3, 9–16. [Google Scholar]
- Boström, J.; Hogner, A.; Llinàs, A.; Wellner, E.; Plowright, A.T. Oxadiazoles in medicinal chemistry. J. Med. Chem. 2012, 55, 1817–1830. [Google Scholar]
- Savarino, A. A historical sketch of the discovery and development of HIV-1 integrase inhibitors. Expert Opin. Investig. Drugs 2006, 15, 1507–1522. [Google Scholar] [CrossRef]
- James, N.D.; Growcott, J.W. Zibotentan. Drugs Future 2009, 34, 624–633. [Google Scholar]
- Scifinder Scholar. Criteria Used to Search: Research Topic: 1,3,4-Oxadiazole. Available online: http://www.cas.org/products/scifinder/ (accessed on 7 June 2012).
- Patel, N.B.; Patel, J.C. Synthesis and antimicrobial activity of 3-(1,3,4-oxadiazol-2-il)quinazolin-4(3H)-ones. Sci. Pharm. 2010, 78, 171–193. [Google Scholar] [CrossRef]
- Kerimov, I.; Ayhan-Kılcıgil, G.; Özdamar, E.D.; Can-Eke, B.; Çoban, T.; Özbey, S.; Kazak, C. Design and one-pot and microwave-assisted synthesis of 2-amino/5-aryl-1,3,4-oxadiazoles bearing a benzimidazole moiety as antioxidants. Arch. Pharm. Chem. Life Sci. 2012, 345, 349–356. [Google Scholar]
- Katritzky, A.R.; Vvedensky, V.; Cai, X.; Rogovoy, B.; Steel, P.J. Synthesis of 5-(2-arylazenyl)-1,2,4-triazoles and 2-amino-5-aryl-1,3,4-oxadiazoles. ARKIVOC 2002, 6, 82–90. [Google Scholar]
- Rajak, H.; Agarawal, A.; Parmar, P.; Thakur, B.S.; Veerasamy, R.; Sharma, P.C.; Kharya, M.D. 2,5-Disubstituted-1,3,4-oxadiazoles/thiadiazole as surface recognition moiety: Design and synthesis of novel hydroxamic acid based histone deacetylase inhibitors. Bioorg. Med. Chem. Lett. 2011, 21, 5735–5738. [Google Scholar]
- Gupta, V.; Kashaw, S.K.; Jatav, V.; Mishra, P. Synthesis and antimicrobial activity of some new 3-[5-(4-substituted) phenyl-1,3,4-oxadiazole-2yl]-2-styrylquinazoline-4(3H)-ones. Med. Chem. Res. 2008, 17, 205–211. [Google Scholar] [CrossRef]
- Sharma, L.K.; Kumar, S.; Singh, S.; Singh, R.K.P. Electrochemical synthesis of 5-substituted-2-amino-1,3,4-oxadiazoles at the llatinum electrode. Russ. J. Electrochem. 2010, 46, 34–40. [Google Scholar] [CrossRef]
- Lotfi, B.; Mustafa, B.; Leila, L.; Salima, M. Electrocyclization of semicarbazone; A novel route of green synthesis of 2,5-disubstituted-1,3,4-oxadiazoles. Int. J. Electrochem. Sci. 2011, 6, 1991–2000. [Google Scholar]
- El-Sayed, W.A.; Ali, O.M.; Hendy, H.A.; Abdel-Rahman, A.A.-H. Synthesis and antimicrobial activity of new 2,5-disubstituted 1,3,4-oxadiazoles and 1,2,4-triazoles and their sugar derivatives. Chin. J. Chem. 2012, 30, 77–83. [Google Scholar]
- Rivera, N.R.; Balsells, J.; Hansen, K.B. Synthesis of 2-amino-5-substituted-1,3,4-oxadiazoles using 1,3-dibromo-5,5-dimethylhydantoin as oxidant. Tetrahedron Lett. 2006, 47, 4889–4891. [Google Scholar] [CrossRef]
- Dolman, S.J.; Gosselin, F.; O’shea, P.D.; Davies, I.W. Superior reactivity of thiosemicarbazide in the synthesis of 2-amino-1,3,4-oxadizaoles. J. Org. Chem. 2006, 71, 9548–9551. [Google Scholar]
- Koparır, M.; Çetin, A.; Cansız, A. 5-Furan-2yl[1,3,4]oxadiazole-2-thiol, 5-furan-2yl-4H[1,2,4] triazole-3-thiol and their thiol-thione tautomerism. Molecules 2005, 10, 475–480. [Google Scholar] [CrossRef]
- Amir, M.; Shikha, K. Synthesis and anti-inflammatory, analgesic, ulcerogenic and lipid peroxidation activities of some new 2-[(2,6-dichloroanilino)phenyl]acetic acid derivatives. Eur. J. Med. Chem. 2004, 39, 535–545. [Google Scholar] [CrossRef]
- Vosooghi, M.; Akbarzadeh, T.; Fallah, A.; Fazeli, M.R.; Jamalifar, H.; Shafiee, A. Synthesis of substituted 1,3,4-oxadiazole, 1,3,4-thiadiazole and 1,2,4-triazole derivatives as potential antimicrobial agents. J. Sci. Islam. Repub. Iran 2005, 16, 145–151. [Google Scholar]
- Almasirad, A.; Vousooghi, N.; Tabatabai, S.A.; Kebriaeezadeh, A.; Shafiee, A. Synthesis, anticonvulsant and muscle relaxant activities of substituted 1,3,4-oxadiazole, 1,3,4-thiadiazole and 1,2,4-triazole. Acta Chim. Slov. 2007, 54, 317–324. [Google Scholar]
- Manjunatha, K.; Poojary, B.; Lobo, P.L.; Fernandes, J.; Kumari, N.S. Synthesis and biological evaluation of some 1,3,4-oxadiazole derivatives. Eur. J. Med. Chem. 2010, 45, 5225–5233. [Google Scholar] [CrossRef]
- Saitoh, M.; Kunitomo, J.; Kimura, E.; Hayase, Y.; Kobayashi, H.; Uchiyama, N.; Kawamoto, T.; Tanaka, T.; Mol, C.D.; Dougan, D.R.; et al. Design, synthesis and structure-activity relationships of 1,3,4-oxadiazole derivatives as novel inhibitors of glycogen synthase kinase-3β. Bioorg. Med. Chem. 2009, 17, 2017–2029. [Google Scholar]
- Almasirad, A.; Shafiee, A.; Abdollahi, M.; Noeparast, A.; Shahrokhinejad, N.; Vousooghi, N.; Tabatabai, S.A.; Khorasami, R. Synthesis and analgesic activity of new 1,3,4-oxadiazoles and 1,2,4-triazoles. Med. Chem. Res. 2011, 20, 435–442. [Google Scholar]
- Xu, W.; He, J.; He, M.; Han, F.; Chen, X.; Pan, Z.; Wang, J.; Tong, M. Synthesis and antifungal activity of novel sulfone derivatives containing 1,3,4-oxadiazole moieties. Molecules 2011, 16, 9129–9141. [Google Scholar]
- Kaplancikli, Z.A. Synthesis of some oxadiazole derivatives as new anticandidal agents. Molecules 2011, 16, 7662–7671. [Google Scholar] [CrossRef]
- Liu, Z.-M.; Chen, Q.; Chen, C.-N.; Tu, H.-Y.; Yang, G.-F. Syntheses of diheterocyclic compounds based on 2-thioacetohydrazide-5,7-dimethyl-1,2,4-triazolo[1,5-a]-pyrimidine. Molecules 2008, 13, 1353–1360. [Google Scholar]
- Zheng, X.; Li, Z.; Wang, Y.; Chen, W.; Huang, Q.; Liu, C.; Song, G. Synthesis and Insecticidal activities of novel 2,5-disubstituted 1,3,4-oxadiazoles. J. Fluorine Chem. 2003, 123, 163–169. [Google Scholar] [CrossRef]
- Amir, M.; Kumar, S. Synthesis and evaluation of anti-inflammatory, analgesic ulcerogenic and lipid peroxidation properties of ibuprofen derivatives. Acta Pharm. 2007, 57, 31–45. [Google Scholar] [CrossRef]
- Mickevičius, V.; Vaickelionienė, R.; Sapijanskaitė, B. Synthesis of substituted 1,3,4-oxadiazole derivatives. Chem. Heterocycl. Comp. 2009, 45, 215–218. [Google Scholar]
- Martin, P.J.; Bruce, D.B. Hydrogen-bonded oxadiazole mesogens. Liq. Crystals 2007, 34, 767–774. [Google Scholar] [CrossRef]
- Hernández-Ainsa, S.; Barberá, J.; Marcos, M.; Serrano, J.L. Liquid crystalline ionic dendrimers containing luminescent oxadiazole moieties. Macromolecules 2012, 45, 1006–1015. [Google Scholar]
- Nagendra, G.; Lamani, R.S.; Narendra, N.; Sureshbabu, V.V. A convenient synthesis of 1,3,4-thiadiazole and 1,3,4-oxadiazole based peptidomimetics employing diacylhydrazines derived from amino acids. Tetrahedron Lett. 2010, 51, 6338–6341. [Google Scholar]
- Li, Z.; Zhu, A.; Mao, X.; Sun, X.; Gong, X. Silica-supported dichlorophosphate: A recoverable cyclodehydrant for the eco-friendly synthesis of 2,5-disubstituted 1,3,4-oxadiazoles under solvent-free and microwave irradiation conditions. J. Braz. Chem. Soc. 2008, 19, 1622–1626. [Google Scholar] [CrossRef]
- Sharma, G.V.M.; Rakesh, B.A.; Krishna, P.R. Zirconium (IV) chloride mediated cyclodehydration of 1,2-diacylhydrazines: A convenient synthesis of 2,5-diaryl 1,3,4-oxadiazoles. Synth. Commun. 2004, 34, 2387–2391. [Google Scholar] [CrossRef]
- Yang, Y.-H.; Shi, M. Halogen effects in Robinson-Gabriel type reaction of cyclopropanecarboxylic acid N'-substituted-hydrazides with PPh3/CX4. Tetrahedron Lett. 2005, 46, 6285–6288. [Google Scholar] [CrossRef]
- Pouliot, M.-F.; Angers, L.; Hamel, J.D.; Paquin, J.F. Synthesis of 1,3,4-oxadiazoles from 1,2-diacylhydrazines using [Et2NSF2]BF4 as a practical cyclodehydration agent. Org. Biomol. Chem. 2012, 10, 988–993. [Google Scholar] [CrossRef]
- Li, C.; Dickson, H.D. A mild, one-pot preparation of 1,3,4-oxadiazoles. Tetrahedron Lett. 2009, 50, 6435–6439. [Google Scholar] [CrossRef]
- Rajapakse, H.A.; Zhu, H.; Young, M.B.; Mott, B.T. A mild and efficient one pot synthesis of 1,3,4-oxadiazoles from carboxylic acids and acyl hydrazides. Tetrahedron Lett. 2006, 47, 4827–4830. [Google Scholar] [CrossRef]
- Kangani, C.O.; Kelley, D.E.; Day, B.W. One pot direct synthesis of oxazolines, benzoxazoles and oxadiazoles from carboxylic acids using the deoxo-fluor reagent. Tetrahedron Lett. 2006, 47, 6497–6499. [Google Scholar]
- Mashraqui, S.H.; Ghadigaonkar, S.G.; Kenny, R.S. An expeditious and convenient one pot synthesis of 2,5-disubstituted-1,3,4-oxadiazoles. Synth. Commun. 2003, 33, 2541–2545. [Google Scholar]
- Kumar, G.V.S.; Rajendraprasad, Y.; Mallikarjuna, B.P.; Chandrashekar, S.M.; Kistayya, C. Synthesis of some novel 2-substituted-5-[isopropylthiazole] clubbed 1,2,4-triazole and 1,3,4-oxadiazoles as potential antimicrobial and antitubercular agents. Eur. J. Med. Chem. 2010, 45, 2063–2074. [Google Scholar] [CrossRef]
- Dabiri, M.; Salehi, P.; Baghbanzadeh, M.; Bahramnejad, M. A facile procedure for the one-pot synthesis of unsymmetrical 2,5-disubstituted 1,3,4-oxadiazoles. Tetrahedron Lett. 2006, 47, 6983–6986. [Google Scholar]
- Guin, S.; Ghosh, T.; Rout, S.K.; Banerjee, A.; Patel, B.K. Cu(II) Catalyzed imine C-H functionalization leading to synthesis of 2,5-substituted 1,3,4-oxadiazoles. Org. Lett. 2011, 13, 5976–5979. [Google Scholar]
- Li, X.; He, D. Synthesis and optical properties of novel anthracene-based stilbene derivatives containing an 1,3,4-oxadiazole unit. Dyes Pigm. 2012, 93, 1422–1427. [Google Scholar] [CrossRef]
- Gaonkar, S.L.; Nagashima, I.; Shimizu, H. Microwave-assisted solution phase synthesis of novel 2-{4-[2-(N-methyl-2-pyridylamino)ethoxy]phenyl}-5-substituted 1,3,4-oxadiazole library. Org. Chem. Int. 2011, 2011. [Google Scholar] [CrossRef]
- Pore, D.M.; Mahadik, S.M.; Desai, U.V. Trichloroisocyanuric acid-mediated one-pot synthesis of unsymmetrical 2,5-disubstituted 1,3,4-oxadiazoles at ambient temperature. Synth. Commun. 2008, 38, 3121–3128. [Google Scholar] [CrossRef]
- Pardeshi, S.P.; Patil, S.P.; Bobade, V.D. N-chlorosuccinimide/1,8-diazabicyclo[5.4.0]undec-7-ene (dbu)-mediated synthesis of 2,5-disubstituted 1,3,4-oxadiazoles. Synth. Commun. 2010, 40, 1601–1606. [Google Scholar]
- Dobrotã, C.; Paraschivescu, C.C.; Dumitru, I.; Matache, M.; Baciu, I.; Rutã, L.L. Convenient preparation of unsymmetrical 2,5-disubstituted 1,3,4-oxadiazoles promoted by Dess-Martin reagent. Tetrahedron Lett. 2009, 50, 1886–1888. [Google Scholar]
- Polshettiwar, V.; Varma, R.S. Greener and rapid access to bio-active heterocycles: One-pot solvent-free synthesis of 1,3,4-oxadiazoles and 1,3,4-thiadiazoles. Tetrahedron Lett. 2008, 49, 879–883. [Google Scholar]
- Kudelko, A.; Zieliǹski, W. Microwave-assisted synthesis of 2-styryl-1,3,4-oxadiazoles from cinnamic acid hydrazide and triethyl orthoesters. Tetrahedron Lett. 2012, 53, 76–77. [Google Scholar] [CrossRef]
- Cui, L.; Liu, Q.; Yu, J.; Ni, C.; Yu, H. A novel one-pot synthesis of a-keto-1,3,4-oxadiazole derivatives based on isocyanide-Nef reaction. Tetrahedron Lett. 2011, 52, 5530–5533. [Google Scholar]
- Ramazani, A.; Rezaei, A. Novel one-pot, four-component condensation reaction: An efficient approach for the synthesis of 2,5-disubstituted 1,3,4-oxadiazole derivatives by a Ugi-4CR/aza-wittig sequence. Org. Lett. 2010, 12, 2852–2855. [Google Scholar] [CrossRef]
- Ramazani, A.; Ahmadi, Y.; Mahyari, A. One-pot efficient synthesis of fully substituted 1,3,4-oxadiazole derivatives from (N-isocyanimino)triphenylphosphorane, carboxylic acids, and aromatic bis-aldehydes. Synth. Commun. 2011, 41, 2273–2282. [Google Scholar] [CrossRef]
- Ahankar, H.; Ramazani, A.; Amini, I.; Ahmadi, Y.; Souldozi, A. The reaction of (N-isocyanimino)triphenylphosphorane with (E)-3-aryl-2-propenoic acid derivatives: One-pot synthesis of 2-[(E)-2-aryl-1-ethenyl]-1,3,4-oxadiazoles via intramolecular aza-Wittig reaction. Heteroatom Chem. 2011, 22, 612–616. [Google Scholar] [CrossRef]
- Ramazani, A.; Shajari, N.; Mahyari, A.; Ahmadi, Y. A novel four-component reaction for the synthesis of disubstituted 1,3,4-oxadiazole derivatives. Mol. Divers. 2011, 15, 521–527. [Google Scholar] [CrossRef]
- Ramazani, A.; Ahmadi, Y.; Rouhani, M.; Shajari, N.; Souldozi, A. The reaction of (N-isocyanimino)triphenylphosphorane with an electron-poor α-haloketone in the presence of aromatic carboxylic acids: A novel three-component reaction for the synthesis of disubstituted 1,3,4-oxadiazole derivatives. Heteroatom Chem. 2010, 21, 368–372. [Google Scholar] [CrossRef]
- Ramazani, A.; Nasrabadia, F.Z.; Ahmadib, Y. One-pot, four-component synthesis of fully substituted 1,3,4-oxadiazole derivatives from (isocyanoimino)triphenylphosphorane, a primary amine, an aromatic carboxylic acid, and chloroacetone. Helv. Chim. Acta 2011, 94, 282–288. [Google Scholar] [CrossRef]
- Souldozi, A.; Ramazani, A. The reaction of (N-isocyanimino)triphenylphosphorane with benzoic acid derivatives: A novel synthesis of 2-aryl-1,3,4-oxadiazole derivatives. Tetrahedron Lett. 2007, 48, 1549–1551. [Google Scholar] [CrossRef]
- Baranov, A.B.; Tsypin, V.G.; Malin, A.S.; Laskin, B.M. Synthesis of 2-chloromethyl-5-(5-methyl-2-furyl)-, and 2-chloromethyl-5-(1,5-dimethyl-2-pyrrolyl)-1,3,4-oxadiazoles from tetrazole derivatives. Russ. J. Appl. Chem. 2005, 78, 773–775. [Google Scholar] [CrossRef]
- Jiang, X.; Register, R.A. Statistical copolymers with side-chain hole and electron transport groups for single-layer electroluminescent device applications. Chem. Mater. 2000, 12, 2542–2549. [Google Scholar] [CrossRef]
- Guan, M.; Bian, Z.Q.; Zhou, Y.F.; Li, F.Y.; Li, Z.J.; Huang, C.H. High-performance blue electroluminescent devices based on 2-(4-biphenylyl)-5-(4-carbazole-9-yl)phenyl-1,3,4-oxadiazole. Chem. Commun. 2003, 2003, 2708–2709. [Google Scholar]
- Zheng, Y.; Batsanov, A.S.; Jankus, V.; Dias, F.D.; Bryce, M.R.; Monkman, A.P. Bipolar molecules with high triplet energies: Synthesis, photophysical, and structural properties. J. Org. Chem. 2011, 76, 8300–8310. [Google Scholar]
- Efimova, Y.A.; Artamonova, T.V.; Koldobskii, G.I. Tetrazoles: LIII.* Microwave-activated acylation of 5-substituted tetrazoles. Russ. J. Org. Chem. 2008, 44, 1345–1347. [Google Scholar]
- Reichart, B.; Kappe, C.O. High-temperature continuous flow synthesis of 1,3,4-oxadiazoles via N-acylation of 5-substituted tetrazoles. Tetrahedron Lett. 2012, 53, 952–955. [Google Scholar] [CrossRef]
- Oliveira, C.S.; Lira, B.F.; Falcão-Silva, V.S.; Siqueira-Junior, J.P.; Barbosa-Filho, J.M.; Athayde-Filho, P.F. Synthesis, molecular properties prediction, and anti-staphylococcal activity of N-acylhydrazones and new 1,3,4-oxadiazole derivatives. Molecules 2012, 17, 5095–5107. [Google Scholar]
- Sahin, G.; Palaska, E.; Ekizoglu, M.; Ozalp, M. Synthesis and antimicrobial activity of some 1,3,4-oxadiazole derivatives. Il Farmaco 2002, 57, 539–542. [Google Scholar] [CrossRef]
- Fuloria, N.K.; Singh, V.; Shaharyar, M.; Ali, M. Synthesis and antimicrobial evaluation of some new oxadiazoles derived from phenylpropionohydrazides. Molecules 2009, 14, 1898–1903. [Google Scholar]
- Kumar, S. Anodic synthesis, spectral characterization and antimicrobial activity of novel 2-amino-5-substituted-1,3,4-oxadiazoles. J. Chil. Chem. Soc. 2010, 55, 126–129. [Google Scholar]
- Sangshetti, J.N.; Chabukswar, A.R.; Shinde, D.B. Microwave assisted one pot synthesis of some Novel 2,5-disubstituted 1,3,4-oxadiazoles as antifungal agents. Bioorg. Med. Chem. Lett. 2011, 21, 444–448. [Google Scholar] [CrossRef]
- Chandrakantha, B.; Shetty, P.; Nambiyar, V.; Isloor, N.; Isloor, A.M. Synthesis, characterization and biological activity of some new 1,3,4-oxadiazole bearing 2-flouro-4-methoxi phenyl moiety. Eur. J. Med. Chem. 2010, 45, 1206–1210. [Google Scholar] [CrossRef]
- Bakht, M.A.; Yar, M.S.; Abdel-Hamid, S.G.; Qasoumi, S.I.; Samad, A. Molecular properties prediction, synthesis and antimicrobial activity of some newer oxadiazole derivatives. Eur. J. Med. Chem. 2010, 45, 5862–5869. [Google Scholar]
- Kumar, R.; Kumar, A.; Jain, S.; Kaushik, D. Synthesis, antibacterial evaluation and QSAR studies of 7-[4-(5-aryl-1,3,4-oxadiazole-2-yl)piperazinyl] quinolone derivatives. Eur. J. Med. Chem. 2011, 46, 3543–3550. [Google Scholar] [CrossRef]
- Palekar, V.S.; Damle, A.J.; Shukla, S.R. Synthesis and antibacterial activity of some novel bis-1,2,4-triazolo[3,4-b]-1,3,4-thiadiazoles and bis-4-thiazolidinone derivatives from terephthalic dihydrazide. Eur. J. Med. Chem. 2009, 44, 5112–5116. [Google Scholar] [CrossRef]
- Ramaprasad, G.C.; Kalluraya, B.; Kumar, B.S.; Hunnur, R.K. Synthesis and biological property of some novel 1,3,4-oxadiazoles. Eur. J. Med. Chem. 2010, 45, 4587–4593. [Google Scholar] [CrossRef]
- Patel, R.V.; Kumari, P.; Rajani, D.P.; Chikhalia, K.H. Synthesis of coumarin-based 1,3,4-oxadiazol-2ylthio-N-phenyl/benzothiazolyl acetamides as antimicrobial and antituberculosis agents. Med. Chem. Res. 2012. [Google Scholar] [CrossRef]
- Patel, R.V.; Patel, A.B.; Kumari, P.; Chikhalia, K.H. Synthesis of novel 3-(5-sulfanyl-1,3,4-oxadiazol-2-yl)-2H-chromen-2-one condensed s-triazinyl piperazines and piperidines as antimicrobial agents. Med. Chem. Res. 2011. [Google Scholar] [CrossRef]
- Sridhara, A.M.; Reddy, K.R.V.; Keshavayya, J.; Goud, P.S.K.; Somashekar, B.C. Bose, P.; Peethambar, S.K.; Gaddam, S.K. Synthesis and antimicrobial activity of 2-substituted [4-(1,3,4-oxadiazol-2-yl methyl)] phthalazin-1(2H)-one derivatives. Eur. J. Med. Chem. 2010, 45, 4983–4989. [Google Scholar]
- Yar, M.S.; Siddiqui, A.A.; Ali, M.A. Synthesis and anti tuberculostatic activity of novel 1,3,4-oxadiazole derivatives. J. Chin. Soc. 2007, 54, 5–8. [Google Scholar]
- Yoshida, Y.; Matsuda, K.; Sasaki, H.; Matsumoto, Y.; Matsumoto, S.; Tawara, S.; Takasugi, H. Studies on anti-Helicobacter pylori agents. Part 2: New cephem derivatives. Bioorg. Med. Chem. 2000, 8, 2317–2335. [Google Scholar]
- Bakal, R.L.; Gattan, S.G. Identification and development of 2,5-disubstituted oxadiazole as potential candidate for treatment of XDR and MDR tuberculosis. Eur. J. Med. Chem. 2012, 47, 278–282. [Google Scholar] [CrossRef]
- Alia, M.A.; Shaharyar, M. Oxadiazole mannich bases: Synthesis and antimycobacterial activity. Bioorg. Med. Chem. Lett. 2007, 17, 3314–3316. [Google Scholar]
- Kashaw, S.K.; Gupta, V.; Kashaw, V.; Mishra, P.; Stables, J.P.; Jain, N.K. Anticonvulsant and sedative-hypnotic activity of some novel 3-[5-(4-substituted) phenyl-1,3,4-oxadiazole-2yl]-2-styrylquinazoline-4(3H)-ones. Med. Chem. Res. 2010, 19, 250–261. [Google Scholar] [CrossRef]
- Zarghi, A.; Tabatabai, S.A.; Faizi, M.; Ahadian, A.; Navabi, P.; Zanganeh, V.; Shafiee, A. Synthesis and anticonvulsant activity of new 2-substituted-5-(2-benzyloxyphenyl)-1,3,4-oxadiazoles. Bioorg. Chem. Lett. 2005, 15, 1863–1865. [Google Scholar]
- Rajak, H.; Deshmukh, R.; Veerasamy, R.; Sharma, A.K.; Mishra, P.; Kharya, M.D. Novel semicarbazones based 2,5-disubstituted-1,3,4-oxadiazoles: One more step towards establishing four binding site pharmacophoric model hypothesis for anticonvulsant activity. Bioorg. Med. Chem. Lett. 2010, 20, 4168–4172. [Google Scholar]
- Kaur, H.; Kumar, S.; Vishwakarma, P.; Sharma, M.; Saxena, K.K.; Kumar, A. Synthesis and antipsychotic and anticonvulsant activity of some new substituted oxa/thiadiazolylazetidinonyl/thiazolidinonylcarbazoles. Eur. J. Med. Chem. 2010, 45, 2777–2783. [Google Scholar]
- Bhat, M.A.; Al-Omar, M.A.; Siddiqui, N. Synthesis, anticonvulsant and neurotoxicity of some novel 1,3,4-oxadiazole derivatives of phthalimide. Pharm. Chem. 2010, 2, 1–10. [Google Scholar]
- Jain, N.; Kashaw, S.K.; Agrawal, R.K.; Gupta, A.; Soni, A. Synthesis, anticonvulsant and neurotoxic activity of some new 2,5-disubstituted-1,3,4-oxadiazoles. Med. Chem. Res. 2011, 20, 1696–1703. [Google Scholar]
- Jalilian, A.R.; Sheikhha, M.; Mirzaei, M.; Aslani, G.R.; Shafiee, A. Radiosynthesis of [18F]-5-[2-(2-chlorophenoxy)phenyl]-1,3,4-oxadiazole-2-yl-4-fluorobenzoate: A labeled ligand for benzodiazepine receptors. J. Radioanal. Nucl. Chem. 2004, 260, 373–377. [Google Scholar] [CrossRef]
- Somani, R.R.; Kadam, G.; Vohra, R.; Vijayaraghavan, S.; Shirodkar, P.Y. Studies of CNS activities of some mannich bases of 1,3,4-oxadiazole. Int. J. Pharmacol. 2010, 6, 696–704. [Google Scholar]
- Almasirad, A.; Tabatabai, S.A.; Faizi, M.; Kebriaeezadeh, A.; Mehrabi, N.; Dalvandia, A.; Shafiee, A. Synthesis and anticonvulsant activity of new 2-substituted-5-[2-(2-fluorophenoxy)phenyl]-1,3,4-oxadiazoles and 1,2,4-triazoles. Bioorg. Med. Chem. Lett. 2004, 14, 6057–6059. [Google Scholar]
- Husain, A.; Ahmad, A.; Alam, M.M.; Ajmal, M.; Ahuja, P. Fenbufen based 3-[5-(substituted aryl)-1,3,4-oxadiazol-2-yl)]-1-(biphenyl-4-yl)propan-1-ones as safer anti-inflammatory and analgesic agents. Eur. J. Med. Chem. 2009, 44, 3798–3804. [Google Scholar] [CrossRef]
- Kadi, A.A.; El-frollosy, N.R.; Al-deeb, O.A.; Habib, E.E.; Ibrahim, T.M.; El-emam, A.A. Synthesis, antimicrobial, and anti-inflammatory activities of novel 2-(1-adamantyl)-5-substituted-1,3,4-oxadiazoles and 2-(1-adamantylamino)-5-substituted-1,3,4-thiadiazoles. Eur. J. Med. Chem. 2007, 42, 235–242. [Google Scholar]
- Burbuliene, M.M.; Jakubkiene, V.; Mekuskiene, G.; Udrenaite, E.; Smicius, R.; Vainilavicius, P. Synthesis and anti-inflammatory activity of derivatives of 5-[(2-disubstitutedamino-6-methyl-pyrimidin-4-yl)-sulfanylmethyl]-3H-1,3,4-oxadiazole-2-thiones. Farmaco 2004, 59, 767–774. [Google Scholar] [CrossRef]
- Kumar, A.; Rajput, C.S. Synthesis and anti-inflammatory activity of newer quinazolin-4-one derivatives. Eur. J. Med. Chem. 2009, 44, 83–90. [Google Scholar] [CrossRef]
- Küçükgüzel, Ş.G.; Küçükgüzel, İ.; Tatar, E.; Rollas, S.; Şahin, F.; Güllüce, M.; Clercq, E.D.; Kabasakal, L. Synthesis of some novel heterocyclic compounds derived from diflunisal hydrazide as potential anti-infective and anti-inflammatory agents. Eur. J. Med. Chem. 2007, 42, 893–901. [Google Scholar]
- Akhter, M.; Husain, A.; Azad, B.; Ajmal, M. Aroylpropionic acid based 2,5-disubstituted-1,3,4-oxadiazoles: Synthesis and their anti-inflammatory and analgesic activities. Eur. J. Med. Chem. 2009, 44, 2372–2378. [Google Scholar]
- Jayashankar, B.; Rai, K.M.L.; Baskaran, N.; Sathish, H.S. Synthesis and pharmacological evaluation of 1,3,4-oxadiazole bearing bis(heterocycle) derivatives as anti-inflammatory and analgesic agents. Eur. J. Med. Chem. 2009, 44, 3898–3902. [Google Scholar]
- Kumar, H.; Javed, S.A.; Khan, S.A.; Amir, M. 1,3,4-Oxadiazole/thiadiazole and 1,2,4-triazole derivatives of biphenyl-4-yloxy acetic acid: Synthesis and preliminary evaluation of biological properties. Eur. J. Med. Chem. 2008, 43, 2688–2698. [Google Scholar] [CrossRef]
- Chandra, T.; Garg, N.; Lata, S.; Saxena, K.K.; Kumar, A. Synthesis of substituted acridinyl pyrazoline derivatives and their evaluation for anti-inflammatory activity. Eur. J. Med. Chem. 2010, 45, 1772–1776. [Google Scholar]
- Bhandari, S.V.; Parikh, J.K.; Bothara, K.G.; Chitre, T.S.; Lokwani, D.K.; Devale, T.L.; Modhave, N.S.; Pawar, V.S.; Panda, S. Design, synthesis, and evaluation of anti-inflammatory, analgesic, ulcerogenicity, and nitric oxide releasing studies of novel indomethacin analogs as non-ulcerogenic derivatives. J. Enzyme Inhib. Med. Chem. 2010, 25, 520–530. [Google Scholar] [CrossRef]
- Palusa, S.K.G.; Udupi, R.H.; Himabindu, V.; Sridhara, A.M. Synthesis and evaluation of a series of pyrimidine substituted 1,3,4-oxadiazole derivatives as antimicrobial and anti-inflammatory agents. Org. Commun. 2011, 4, 82–93. [Google Scholar]
- Gilani, S.J.; Khan, S.A.; Siddiqui, N. Synthesis and pharmacological evaluation of condensed heterocyclic 6-substituted 1,2,4-triazolo-[3,4-b]-1,3,4-thiadiazole and 1,3,4-oxadiazole derivatives of isoniazid. Bioorg. Med. Chem. Lett. 2010, 20, 4762–4765. [Google Scholar] [CrossRef]
- Savariz, F.C.; Formagio, A.S.N.; Barbosa, V.A.; Foglio, M.A. Carvalho, J.E.; Duarte, M.C.T.; Filho, B.P.D.; Sarragiotto, M.H. Synthesis, antitumor and antimicrobial activity of novel 1-substituted phenyl-3-[3-alkylamino(methyl)-2-thioxo-1,3,4-oxadiazol-5-yl]-b-carboline derivatives. J. Braz. Chem. Soc. 2010, 21, 288–298. [Google Scholar]
- Liu, K.; Lu, X.; Zhang, H.-J.; Sun, J.; Zhu, H.-L. Synthesis, molecular modeling and biological evaluation of 2-(benzylthio)-5-aryloxadiazole derivatives as anti-tumor agents. Eur. J. Med. Chem. 2012, 47, 473–478. [Google Scholar]
- Ouyang, X.; Piatnitski, E.L.; Pattaropong, V.; Chen, X.; He, H.Y.; Kiselyov, A.S.; Velankar, A.; Kawakami, J.; Labelle, M.; Smith, L.; et al. Oxadiazole derivatives as a novel class of antimitotic agents: Synthesis, inhibition of tubulin polymerization, and activity in tumor cell lines. Bioorg. Med. Chem. Lett. 2006, 16, 1191–1196. [Google Scholar]
- Tuma, M.C.; Malikzay, A.; Ouyang, X.; Surgulazde, D.; Fleming, J.; Mitelman, S.; Camara, M.; Finnerty, B.; Doody, J.; Chekler, E.L.P.; et al. Antitumor activity of IMC-038525, a novel oral tubulin polymerization inhibitor. Transl. Oncol. 2010, 3, 318–325. [Google Scholar]
- Lee, L.; Robb, L.M.; Davis, R.; Mackay, H.; Chavda, S.; Babu, B.; O’brien, E.L.; Risinger, A.L.; Mooberry, S.L.; Lee, M. Design, synthesis, and biological evaluations of 2,5-diaryl-2,3-dihydro-1,3,4-oxadiazoline analogs of combretastatin-A4. J. Med. Chem. 2010, 53, 325–334. [Google Scholar]
- Zhang, X.-M.; Qiu, M.; Sun, J.; Zhang, Y.-B.; Yang, Y.-S.; Wang, X.-L.; Tang, J.-F.; Zhu, H.-L. Synthesis, biological evaluation, and molecular docking studies of 1,3,4-oxadiazole derivatives possessing 1,4-benzodioxan moiety as potential anticancer agents. Bioog. Med. Chem. 2011, 19, 6518–6524. [Google Scholar]
- Bondock, S.; Adel, S.; Etman, H.A.; Badria, F.A. Synthesis and antitumor evaluation of some new 1,3,4-oxadiazole-based heterocycles. Eur. J. Med. Chem. 2012, 48, 192–199. [Google Scholar]
- Puthiyapurayil, P.; Poojary, B.; Chikkanna, C.; Buridipad, S.K. Design, synthesis and biological evaluation of a novel series of 1,3,4-oxadiazole bearing N-methyl-4-(trifluoromethyl)phenyl pyrazole moiety as cytotoxic agents. Eur. J. Med. Chem. 2012, 53, 203–210. [Google Scholar]
- Temesgen, Z.; Siraj, D.S. Raltegravir: first in class HIV integrase inhibitor. Ther. Clin. Risk Manage. 2008, 4, 493–500. [Google Scholar]
- Wang, Z.; Wang, M.; Yao, X.; Li, Y.; Qiao, W.; Geng, Y.; Liu, Y.; Wang, Q. Hydroxyl may not be indispensable for raltegravir: Design, synthesis and SAR studies of raltegravir derivatives as HIV-1 inhibitors. Eur. J. Med. Chem. 2012, 50, 361–369. [Google Scholar]
- El-Emam, A.A.; Al-Deeb, O.A.; Al-Omar, M.; Lehmann, J. Synthesis, antimicrobial, and anti-HIV-1 activity of certain 5-(1-adamantyl)-2-substituted thio-1,3,4-oxadiazoles and 5-(1-adamantyl)-3-substituted aminomethyl-1,3,4-oxadiazoline-2-thiones. Bioorg. Med. Chem. 2004, 12, 5107–5113. [Google Scholar]
- Iqbal, R.; Zareef, M.; Ahmed, S.; Zaidi, J.H.; Arfan, M.; Shafique, M.; Al-masoudi, N.A. Synthesis, antimicrobial and anti-HIV activity of some novel benzenesulfonamides bearing 2,5-disubstituted-1,3,4-oxadiazole moiety. J. Chin. Chem. Soc. 2006, 53, 689–696. [Google Scholar]
- Kim, R.M.; Rouse, E.A.; Chapman, K.T.; Schleif, W.A.; Olsen, D.B.; Stahlhurt, M.; Rutkowski, C.A.; Emini, E.A.; Tata, J.R. P1' oxadiazole protease inhibitors with excellent activity against native and protease inhibitor-resistant HIV-1. Bioorg. Med. Chem. Lett. 2004, 14, 4651–4654. [Google Scholar]
- Johns, B.; Weatherhead, J.G.; Allen, S.H.; Thompson, J.B.; Garvey, E.P.; Foster, S.A.; Jeffrey, J.L.; Miller, W.H. 1,3,4-Oxadiazole substituted naphthyridines as HIV-1 integrase inhibitors. Part 2: SAR of the C5 position. Bioorg. Med. Chem. Lett. 2009, 19, 1807–1810. [Google Scholar]
- Llinàs-Brunet, M.; Bailey, M.D.; Bolger, G.; Brochu, C.; Faucher, A.-M.; Ferland, J.M.; Garneau, M.; Ghiro, E.; Gorys, V.; Grand-Maître, C. Structure-activity study on a novel series of macrocyclic inhibitors of the hepatitis C virus NS3 protease leading to the discovery of BILN 2061. J. Med. Chem. 2004, 47, 1605–1608. [Google Scholar] [CrossRef]
- Bankar, G.R.; Nandakumar, K.; Nayak, P.G.; Thakur, A.; Chamallamudi, M.R.; Nampurath, G.K. Vasorelaxant effect in rat aortic rings through calcium channel blockage: A preliminary in vitro assessment of a 1,3,4-oxadiazole derivative. Chem. Biol. Interact. 2009, 181, 377–382. [Google Scholar] [CrossRef]
- Bankar, G.R.; Nampurath, G.K.; Nayak, P.G.; Bhattcharya, S. A possible correlation between the correction of endothelial dysfunction and normalization of high blood pressure levels by 1,3,4-oxadiazole derivative, an L-type Ca2+ channel blocker in deoxycorticosterone acetate and NG-nitro-l-arginine hypertensive rats. Chem. Biol. Interact. 2010, 183, 327–331. [Google Scholar]
- Xiang, Z.; Thompson, A.D.; Brogan, J.T.; Schulte, M.L.; Melancon, B.J.; Mi, D.; Lewis, L.M.; Zou, B.; Yang, L.; Morrison, R. The discovery and characterization of ML218: A novel, centrally active T-Type calcium channel inhibitor with robust effects in STN neurons and in a rodent model of Parkinson’s disease. Chem. Neurosci. 2011, 2, 730–742. [Google Scholar] [CrossRef]
- Ducharme, Y.; Blouin, M.; Brideau, C.; Châteauneuf, A.; Gareau, Y.; Grimm, E.L.; Juteau, H.; Laliberté, S.; MacKay, B.; Massé, F.; et al. The discovery of setileuton, a potent and selective 5-lipoxygenase inhibitor. Med. Chem. Lett. 2010, 1, 170–174. [Google Scholar] [CrossRef]
- Gosselin, F.; Britton, R.A.; Davies, I.W.; Dolman, S.J.; Gauvreau, D.; Hoerrner, R.S.; Hughes, G.; Janey, J.; Lau, S.; Molinaro, C.; et al. A practical synthesis of 5-lipoxygenase inhibitor MK-0633. J. Org. Chem. 2010, 75, 4154–4160. [Google Scholar]
- Leung, D.; Du, W.; Hardouin, C.; Cheng, H.; Hwang, I.; Cravatt, B.F.; Boger, D.L. Discovery of an exceptionally potent and selective class of fatty acid amide hydrolase inhibitors enlisting proteome-wide selectivity screening: Concurrent optimization of enzyme inhibitor potency and selectivity. Bioorg. Med. Chem. Lett. 2005, 15, 1423–1428. [Google Scholar]
- Khan, M.T.; Choudhary, M.I.; Khan, K.M.; Rani, M.; Atta-Ur, R. Structure-activity relationships of tyrosinase inhibitory combinatorial library of 2,5-disubstituted-1,3,4-oxadiazole analogues. Bioorg. Med. Chem. 2005, 16, 3385–3395. [Google Scholar]
- Tomi, I.H.R.; Al-Qaisi, A.H.J.; Al-Qaisi, Z.H.J. Synthesis, characterization and effect of bis-1,3,4-oxadiazole rings containing glycine moiety on the activity of some transferase enzymes. J. King Saud Univ. 2011, 23, 23–33. [Google Scholar] [CrossRef]
- Maccioni, E.; Alcaro, S.; Cirilli, R.; Vigo, S.; Cardia, M.C.; Sanna, M.L.; Meleddu, R.; Yanez, M.; Costa, G.; Casu, L.; et al. 3-Acetyl-2,5-diaryl-2,3-dihydro-1,3,4-oxadiazoles: A new scaffold for the selective inhibition of monoamine oxidase B. J. Med. Chem. 2011, 54, 6394–6398. [Google Scholar]
- Garfunkle, J.; Ezzili, C.; Rayl, T.J.; Hochstatter, D.G.; Hwang, I.; Boger, D.L. Optimization of the Central heterocycle of α-ketoheterocycle inhibitors of fatty acid amide hydrolase. J. Med. Chem. 2008, 51, 4392–4403. [Google Scholar] [CrossRef]
- Ohmoto, K.; Yamamoto, T.; Horiuchi, T.; Imanishi, H.; Odagaki, Y.; Kawabata, K.; Sekioka, T.; Hirota, Y.; Matsuoka, S.; Nakai, H.; et al. Design and synthesis of new orally active nonpeptidic inhibitors of human neutrophil elastase. J. Med. Chem. 2000, 43, 4927–4929. [Google Scholar] [CrossRef]
- Ohmoto, K.; Yamamoto, T.; Okuma, M.; Horiuchi, T.; Imanishi, H.; Odagaki, Y.; Kawabata, K.; Sekioka, T.; Hirota, Y.; Matsuoka, S.; et al. Development of orally active nonpeptidic inhibitors of human neutrophil elastase. J. Med. Chem. 2001, 44, 1268–1285. [Google Scholar]
- Willoughby, C.A.; Bull, H.G.; Garcia-Calvo, M.; Jiang, J.; Chapmana, K.T.; Thornberry, N.A. Discovery of potent, selective human granzyme B inhibitors that inhibit CTL mediated apoptosis. Bioorg. Med. Chem. Lett. 2002, 12, 2197–2200. [Google Scholar] [CrossRef]
- Zeng, H.; Zhang, H.; Jang, F.; Zhao, L.; Zhang, J. Molecular modeling studies on benzimidazole carboxamide derivatives as PARP-1 inhibitors using 3D-QSAR and docking. Chem. Biol. Drug Des. 2011, 78, 333–352. [Google Scholar] [CrossRef]
- Elzein, E.; Ibrahim, P.; Koltun, D.O.; Rehder, K.; Shenk, K.D.; Marquart, T.A.; Jiang, B.; Li, X.; Natero, R.; Li, Y.; et al. CVT-4325: A potent fatty acid oxidation inhibitor with favorable oral bioavailability. Bioorg. Med. Chem. Lett. 2004, 14, 6017–6021. [Google Scholar]
- Perni, R.B.; Pitlik, J.; Britt, S.D.; Court, J.J.; Courtney, L.F.; Deininger, D.D.; Farmer, L.J.; Gates, C.A.; Harbeson, S.L.; Levin, R.B.; et al. Inhibitors of hepatitis C virus NS3·4A protease 2. Warhead SAR and optimization. Bioorg. Med. Chem. Lett. 2004, 14, 1441–1446. [Google Scholar]
- Palanki, M.S.S.; Gayo-Fung, L.M.; Shevlin, G.I.; Erdman, P.; Sato, M.; Goldman, M.; Ransone, L.J.; Spooner, C. Structure-activity relationship studies of ethyl 2-[(3-methyl-2,5-dioxo(3-pyrrolinyl))amino]-4-(trifluoromethyl)pyrimidine-5-carboxylate: An inhibitor of AP-1 and NF-κB Mediated Gene Expression. Bioorg. Med. Chem. Lett. 2002, 12, 2573–2577. [Google Scholar] [CrossRef]
- Matsuno, K.; Masuda, Y.; Uehara, Y.; Sato, H.; Muroya, A.; Takahashi, O.; Yokotagawa, T.; Furuya, T.; Okawara, T.; Otsuka, M.; et al. Identification of a new series of STAT3 inhibitors by virtual screening. Med. Chem. Lett. 2010, 1, 371–375. [Google Scholar] [CrossRef]
- Rai, G.; Kenyon, V.; Jadhav, A.; Schultz, L.; Armstrong, M.; Jameson, J.B.; Hoobler, E.; Leister, W.; Simeonov, A.; Holman, T.R.; et al. Discovery of potent and selective inhibitors of human reticulocyte 15-lipoxygenase-1. J. Med. Chem. 2010, 53, 7392–7404. [Google Scholar]
- White, A.D.; Creswell, M.W.; Chucholowski, A.W.; Blankley, C.B.; Wilson, M.W.; Bousley, R.F.; Essenburg, A.D.; Hamelehle, K.L.; Krause, B.R.; Stanfield, R.L.; et al. Heterocyclic ureas: Inhibitors of acyl-CoA: Cholesterol O-acyltransferase as hypocholesterolemic agents. J. Med. Chem. 1996, 39, 4382–4395. [Google Scholar]
- Rydzewski, R.M.; Burrill, L.; Mendonca, R.; Palmer, J.T.; Rice, M.; Tahilramani, R.; Bass, K.E.; Leung, L.; Gjerstad, E.; Janc, J.W.; et al. Optimization of subsite binding to the β5 subunit of the human 20S proteasome using vinyl sulfones and 2-keto-1,3,4-oxadiazoles: Syntheses and cellular properties of potent, selective proteasome inhibitors. J. Med. Chem. 2006, 49, 2953–2968. [Google Scholar]
- Uto, Y.; Ueno, Y.; Kiyotsuka, Y.; Miyazawa, Y.; Kurata, H.; Ogata, T.; Yamada, M.; Deguchi, T.; Konishi, M.; Takagi, T.; et al. Synthesis and evaluation of novel stearoyl-CoA desaturase 1 inhibitors: 10-{6-[5-(pyridin-3-ylmethyl)-1,3,4-oxadiazol-2-yl]pyridazin-3-yl}-3,4-dihydrospiro[chromene-2,40-piperidine] analogs. Eur. J. Med. Chem. 2010, 45, 4788–4796. [Google Scholar]
- Hou, Z.; Nakanishi, I.; Kinoshita, T.; Takei, Y.; Yasue, M.; Misu, R.; Suzuki, Y.; Nakamura, S.; Kure, T.; Ohno, H.; et al. Structure-based design of novel potent protein kinase CK2 (CK2) inhibitors with phenyl-azole scaffolds. J. Med. Chem. 2012, 55, 2899–2903. [Google Scholar] [CrossRef]
- Uto, Y.; Kiyotsuka, Y.; Ueno, Y.; Miyazawa, Y.; Kurata, H.; Ogata, T.; Deguchi, T.; Yamada, M.; Watanabe, N.; Konishi, M.; et al. Novel spiropiperidine-based stearoyl-CoA desaturase-1 inhibitors: Identification of 10-{6-[5-(pyridin-3-ylmethyl)-1,3,4-oxadiazol-2-yl]-pyridazin-3-yl}-5-(trifluoromethyl)-3,4-dihydrospiro [chrome ne-2,40-piperidine]. Bioorg. Med. Chem. Lett. 2010, 20, 746–754. [Google Scholar]
- McCoull, W.; Addie, M.S.; Birch, A.M.; Birtles, S.; Buckett, L.K.; Butlin, R.J.; Bowker, S.S.; Boyd, S.; Chapman, S.; Davies, R.D.M.; et al. Identification, optimisation and in vivo evaluation of oxadiazole DGAT-1 inhibitors for the treatment of obesity and diabetes. Bioorg. Med. Chem. Lett. 2012, 22, 3873–3878. [Google Scholar]
- Iqbal, A.K.M.; Khan, A.Y.; Kalashetti, M.B.; Belavagi, N.S.; Gong, T.-D.; Khazi, I.A.M. Synthesis, hypoglycemic and hypolipidemic activities of novel thiazolidinedione derivatives containing thiazole/triazole/oxadiazole ring. Eur. J. Med. Chem. 2012, 53, 308–315. [Google Scholar]
- Khalilullah, H.; Ahsan, M.J.; Hedaitullah, M.; Khan, S.; Ahmed, B. 1,3,4-Oxadiazole: A biologically active scaffold. Mini Rev. Med. Chem. 2012, 12, 789–801. [Google Scholar] [CrossRef]
- Li, Z.; Zhan, P.; Liu, X. 1,3,4-Oxadiazole: A privileged structure in antiviral agents. Mini Rev. Med. Chem. 2011, 11, 1130–1142. [Google Scholar] [CrossRef]
- Suman, B.; Sunil, K.; Asho, K. Heterocyclic 1,3,4-oxadiazole compounds with diverse biological activities: A comprehensive review. J. Pharm. Res. 2010, 3, 2993–2997. [Google Scholar]
- Liao, Y.; Böttcher, H.; Harting, J.; Greiner, H.; Amsterdam, C.V.; Cremers, T.; Sundell, S.; März, J.; Rautenberg, W.; Wikström, H. New selective and potent 5-HT1B/1D antagonists: Chemistry and pharmacological evaluation of N-piperazinylphenyl biphenylcarboxamides and biphenylsulfonamides. J. Med. Chem. 2000, 43, 517–525. [Google Scholar] [CrossRef]
- Ali, K.A.; Ragab, E.A.; Farghaly, T.A.; Abdalla, M.M. Synthesis of new functionalized 3 subsitituted [1,2,4]triazolo [4,3-a]pyrimidine dreivatives: Potential antihypertensive agents. Acta Pol. Pharm. 2011, 68, 237–247. [Google Scholar]
- Khanfar, M.A.; Hill, R.A.; Kaddoumi, A.; El Sayed, K.A. Discovery of novel GSK-3β inhibitors with potent in vitro and in vivo activities and excellent brain permeability using combined ligand-and structure-based virtual screening. J. Med. Chem. 2010, 53, 8534–8545. [Google Scholar]
- Tan, T.M.C.; Chen, Y.; Kong, K.H.; Bai, J.; Li, Y.; Lim, S.G.; Ang, T.H.; Lam, Y. Synthesis and the biological evaluation of 2-benzenesulfonylalkyl-5-substituted-sulfanyl-[1,3,4]-oxadiazoles as potential anti-hepatitis B virus agents. Antivir. Res. 2006, 71, 7–14. [Google Scholar]
- Saitoh, M.; Kunitomo, J.; Kimura, E.; Iwashita, H.; Uno, Y.; Onishi, T.; Uchiyama, N.; Kawamoto, T.; Tanaka, T.; Mol, C.D.; et al. 2-{3-[4-(Alkylsulfinyl)phenyl]-1-benzofuran-5-yl}-5-methyl-1,3,4-oxadiazole derivatives as novel inhibitors of glycogen synthase kinase-3beta with good brain permeability. J. Med. Chem. 2009, 52, 6270–6286. [Google Scholar]
- Onishi, T.; Iwashita, H.; Uno, Y.; Kunitomo, J.; Saitoh, M.; Kimura, E.; Fujita, H.; Uchiyama, N.; Kori, M.; Takizawa, M. A novel glycogen synthase kinase-3 inhibitor 2-methyl-5-(3-{4-[(S)-methylsulfinyl]phenyl}-1-benzofuran-5-yl)-1,3,4-oxadiazole decrease tau phosphorylation and ameliorates cognitive deficits in a transgenic model of Alzheimer’s disease. J. Neurochem. 2011, 119, 1330–1340. [Google Scholar] [CrossRef]
- Kampmanna, T.; Yennamallia, R.; Campbella, P.; Stoermerc, M.J.; David, P.; Fairlie, D.P.; Kobea, B.; Young, P.R. In silico screening of small molecule libraries using the dengue virus envelope E protein has identified compounds with antiviral activity against multiple flaviviruses. Antivir. Res. 2009, 84, 234–241. [Google Scholar]
- Attaby, F.A.; Abdel-Fattah, A.M.; Shaif, L.M.; Elsayed, M.M. Anti-alzheimer and anti-cox-2 activities of the newly synthesized 2,3'-bipyridine derivatives (ii). Phosphorus Sulfur 2010, 185, 668–679. [Google Scholar] [CrossRef]
- Chrysina, E.D.; Kosmopoulou, M.N.; Tiraidis, C.; Kardakaris, R.; Bischler, N.; leonidas, D.D.; Hadady, Z.; Somsak, L.; Docsa, T.; Gergely, P.; et al. Kinetic and crystallographic studies on 2-(β-D-glucopyranosyl)-5-methyl-1, 3,4-oxadiazole, -benzothiazole, and -benzimidazole, inhibitors of muscle glycogen phosphorylase b. Evidence for a new binding site. Protein Sci. 2005, 14, 873–888. [Google Scholar] [CrossRef]
- Abdel-Aal, M.T.; El-Sayed, W.A.; El-Kosy, S.M.; El-Ashry, E.S.H. Synthesis and antiviral evaluation of novel 5-(N-arylaminomethyl-1,3,4-oxadiazol-2-yl)hydrazines and their sugars, 1,2,4-triazoles, tetrazoles and pyrazolyl derivatives. Arch. Pharm. Chem. Life Sci. 2008, 341, 307–313. [Google Scholar] [CrossRef]
- Shamroukh, A.H.; Ali, M.A. Anti-HIV activity of some newly synthesized triazolo[4,3-b]pyridazines. Arch. Pharm. Chem. Life Sci. 2008, 341, 223–230. [Google Scholar] [CrossRef]
- Maciolek, C.M.; Ma, B.; Menzel, K.; Laliberte, S.; Bateman, K.; Krolikowski, P.; Gibson, C.R. Novel cytochrome P450-mediated ring opening of the 1,3,4-oxadiazole in setileuton, a 5-lipoxygenase inhibitor. Drug Metab. Dispos. 2011, 39, 763–770. [Google Scholar] [CrossRef]
- Idrees, G.A.; Aly, O.M.; Abuo-Rahma, G.E.-D.A.A.; Radwan, M.F. Design, synthesis and hypolipidemic activity of novel 2-(naphthalen-2-yloxy)propionic acid derivatives as desmethyl fibrate analogs. Eur. J. Med. Chem. 2009, 44, 3973–3980. [Google Scholar]
- Zareef, M.; Iqbal, R.; Dominguez, N.G.; Rodrigues, J.; Zaidi, J.H.; Arfan, M.; Supuran, C.T. Synthesis and antimalarial activity of novel chiral and achiral benzenesulfonamides bearing 1,3,4-oxadiazole moieties. J. Enzym. Inhib. Med. Chem. 2007, 22, 301–308. [Google Scholar] [CrossRef]
- Maslat, A.O.; Abussaud, M.; Tashtoush, H.; Al-Talib, M. Synthesis, antibacterial, antifungal and genotoxic activity of bis-1,3,4-oxadiazole derivatives. Pol. J. Pharmacol. 2002, 54, 55–59. [Google Scholar]
- Tajik, H.; Dadras, A. Synthesis and herbicidal activity of novel 5-chloro-3-fluoro-2-phenoxypyridines with a 1,3,4-oxadiazole ring. J. Pestic. Sci. 2011, 36, 27–32. [Google Scholar] [CrossRef]
- Liu, Z.; Yang, G.; Qin, X. Syntheses and biological activities of novel diheterocyclic compounds containing 1,2,4-triazolo[1,5-a]pyrimidine and 1,3,4-oxadiazole. J. Chem. Technol. Biotechnol. 2001, 76, 1154–1158. [Google Scholar]
- Cao, S.; Qian, X.H.; Song, G.H.; Chai, B.; Jiang, Z.S. Synthesis and antifeedant activity of new oxadiazolyl 3(2H)-pyridazinones. J. Agric. Food Chem. 2003, 51, 152–155. [Google Scholar]
- Shi, W.; Qian, X.H.; Song, G.H.; Zhang, R.; Li, R.P. Syntheses and insecticidal activities of novel 2-fluorophenyl-5-aryl/cyclopropyl-1,3,4-oxadiazoles. J. Fluor. Chem. 2000, 106, 173–179. [Google Scholar] [CrossRef]
- Almajan, G.L.; Barbuceanu, S.F.; Saramet, I.; Dinu, M.; Doicin, C.V.; Draghici, C. Synthesis and biological evaluation of various new substituted 1,3,4-oxadiazole-2-thiols. Rev. Chim. 2008, 59, 395–399. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Oliveira, C.S.; Lira, B.F.; Barbosa-Filho, J.M.; Lorenzo, J.G.F.; De Athayde-Filho, P.F. Synthetic Approaches and Pharmacological Activity of 1,3,4-Oxadiazoles: A Review of the Literature from 2000–2012. Molecules 2012, 17, 10192-10231. https://doi.org/10.3390/molecules170910192
De Oliveira CS, Lira BF, Barbosa-Filho JM, Lorenzo JGF, De Athayde-Filho PF. Synthetic Approaches and Pharmacological Activity of 1,3,4-Oxadiazoles: A Review of the Literature from 2000–2012. Molecules. 2012; 17(9):10192-10231. https://doi.org/10.3390/molecules170910192
Chicago/Turabian StyleDe Oliveira, Cledualdo Soares, Bruno Freitas Lira, José Maria Barbosa-Filho, Jorge Gonçalo Fernandez Lorenzo, and Petrônio Filgueiras De Athayde-Filho. 2012. "Synthetic Approaches and Pharmacological Activity of 1,3,4-Oxadiazoles: A Review of the Literature from 2000–2012" Molecules 17, no. 9: 10192-10231. https://doi.org/10.3390/molecules170910192
APA StyleDe Oliveira, C. S., Lira, B. F., Barbosa-Filho, J. M., Lorenzo, J. G. F., & De Athayde-Filho, P. F. (2012). Synthetic Approaches and Pharmacological Activity of 1,3,4-Oxadiazoles: A Review of the Literature from 2000–2012. Molecules, 17(9), 10192-10231. https://doi.org/10.3390/molecules170910192