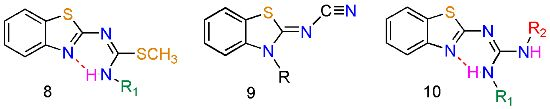
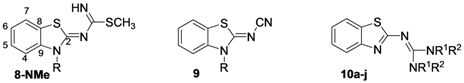A Synthetic Method to Access Symmetric and Non-Symmetric 2-(N,N'-disubstituted)guanidinebenzothiazoles
Abstract
:1. Introduction
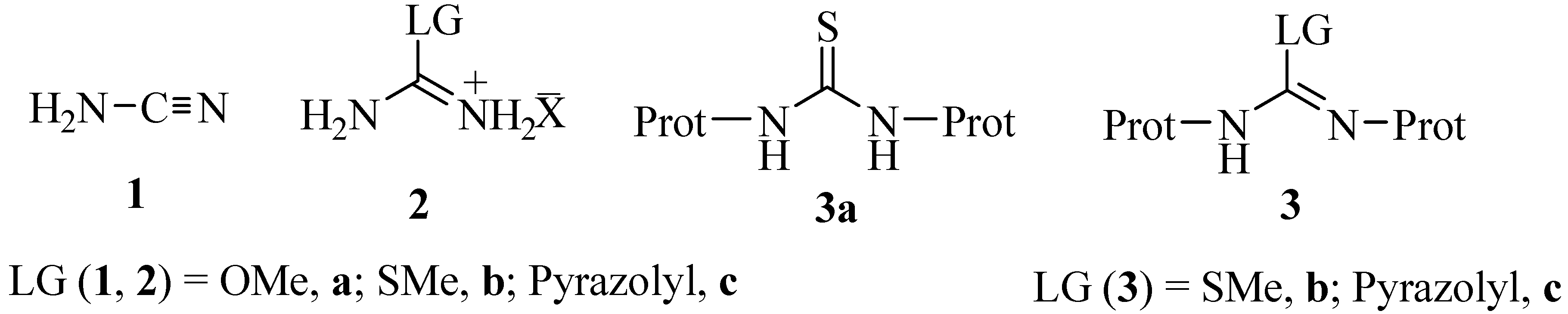
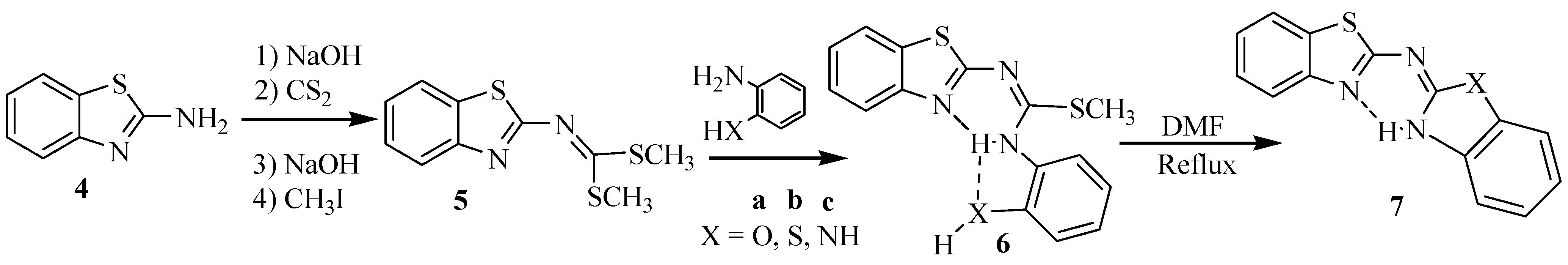
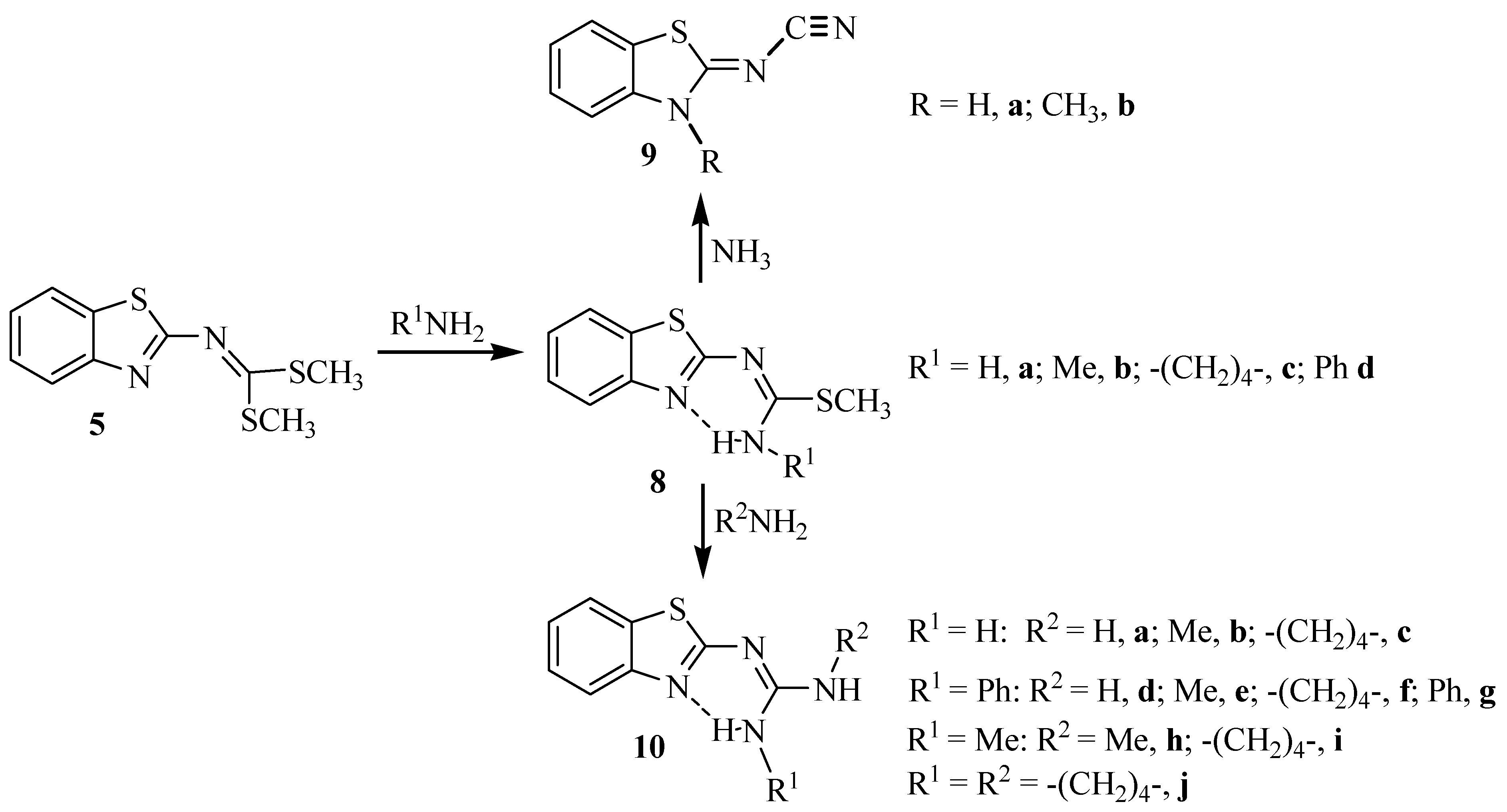
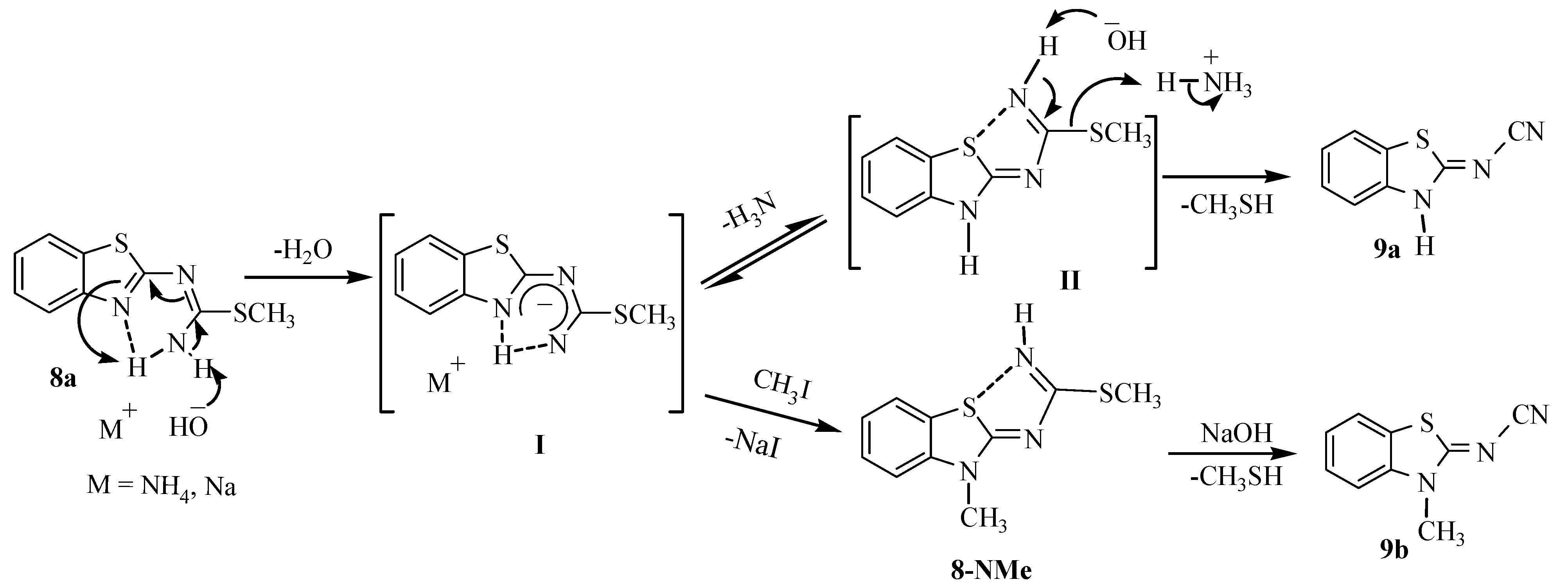
2. Results and Discussion
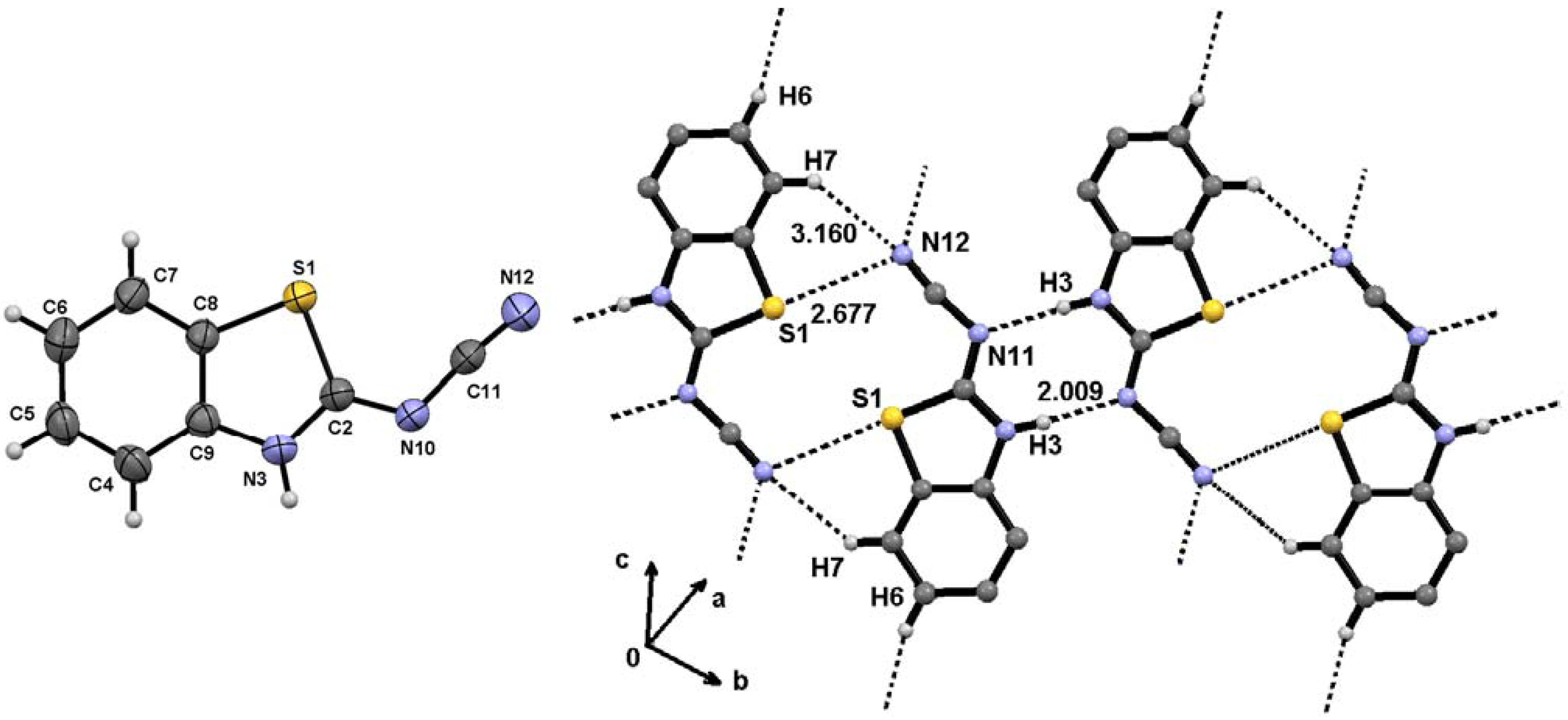
| Comp. | H4 | H5 | H6 | H7 | NH | NCH3, SCH3 | NPh | N(CH2CH2)2 |
|---|---|---|---|---|---|---|---|---|
| 8-NMea | 8.00 | 7.76 | 7.61 | 7.46 | 9.8 | 3.80, 2.66 | ||
| 9aa | 7.75 | 7.27 | 7.24 | 7.39 | ||||
| 9bb | 7.58 | 7.46 | 7.31 | 7.24 | 3.62 | |||
| 10ba | 7.62 | 7.21 | 7.03 | 7.42 | 7.7 | 2.75 | ||
| 10ca | 7.63 | 7.21 | 7.04 | 7.44 | 8.2 | 3.4, 1.8 | ||
| 10db | 7.61 | 7.30 | 7.14 | 7.59 | 7.4–7.3 | |||
| 10eb | 7.69 | 7.30 | 7.17 | 7.63 | 11.2 | 2.97 | 7.5–7.3 | |
| 10fb | 7.66 | 7.32 | 7.18 | 7.63 | 11.6 | 7.3–7.1 | 3.4, 1.8 | |
| 10ga | 7.73 | 7.31 | 11.9 | 7.4–7.3 | ||||
| 10hb | 7.61 | 7.24 | 7.10 | 7.55 | 9.6 | 2.92 | ||
| 10ib | 7.60 | 7.26 | 7.08 | 7.57 | 8.9 | 3.53 | 3.0, 1.9 | |
| 10jb | 7.51 | 7.20 | 7.00 | 7.49 | 3.4, 1.8 |
| Comp. | C2 | C4 | C5 | C6 | C7 | C8 | C9 | C11 | NCH3, SCH3 | Ph |
|---|---|---|---|---|---|---|---|---|---|---|
| 8-NMea | 166.0 | 114.6 | 124.1 | 124.6 | 128.7 | 126.2 | 138.5 | 175.1 | 33.6, 15.7 | |
| 9aa | 174.0 | 114.0 | 123.7 | 124.4 | 128.2 | 125.0 | 139.4 | 117.7 | ||
| 9bb | 171.5 | 112.2 | 122.9 | 124.8 | 127.9 | 123.3 | 139.4 | 116.9 | 31.3 | |
| 10ba | 158.4 | 125.9 | 122.3 | 121.4 | 118.9 | 130.9 | 152.5 | 174.3 | 28.3 | |
| 10ca | 155.4 | 125.9 | 122.2 | 121.4 | 119.0 | 130.9 | 152.6 | 174.4 | ||
| 10db | 156.0 | 125.7 | 122.7 | 122.1 | 119.6 | 131.3 | 151.8 | 173.7 | 136.8, 130.2, 125.7, 127.0 | |
| 10eb | 154.6 | 125.6 | 122.5 | 121.2 | 119.5 | 131.7 | 151.9 | 174.5 | 28.6 | 137.0, 130.2, 126.9, 126.0 |
| 10fb | 154.4 | 125.6 | 122.4 | 121.1 | 119.6 | 132.0 | 151.9 | 173.8 | 139.9, 129.5, 125.6, 123.3 | |
| 10ga | 151.5 | 125.8 | 121.5 | 121.3 | 119.9 | 132.0 | 151.0 | 173.6 | 137.3, 129.8, 123.6, 123.0 | |
| 10hb | 157.2 | 125.5 | 122.2 | 121.0 | 119.1 | 131.6 | 152.2 | 174.7 | 28.2 | |
| 10ib | 159.2 | 125.4 | 122.0 | 121.0 | 119.1 | 132.1 | 152.2 | 173.6 | 31.5 | |
| 10jb | 158.0 | 125.2 | 121.2 | 121.8 | 119.1 | 133.4 | 153.3 | 171.3 |
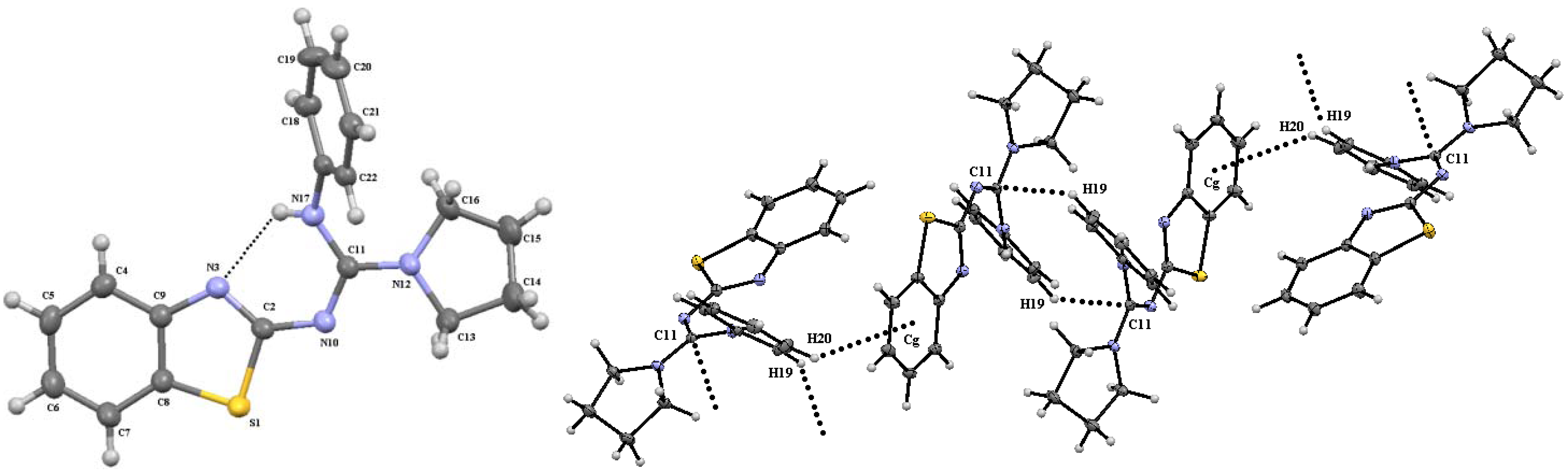
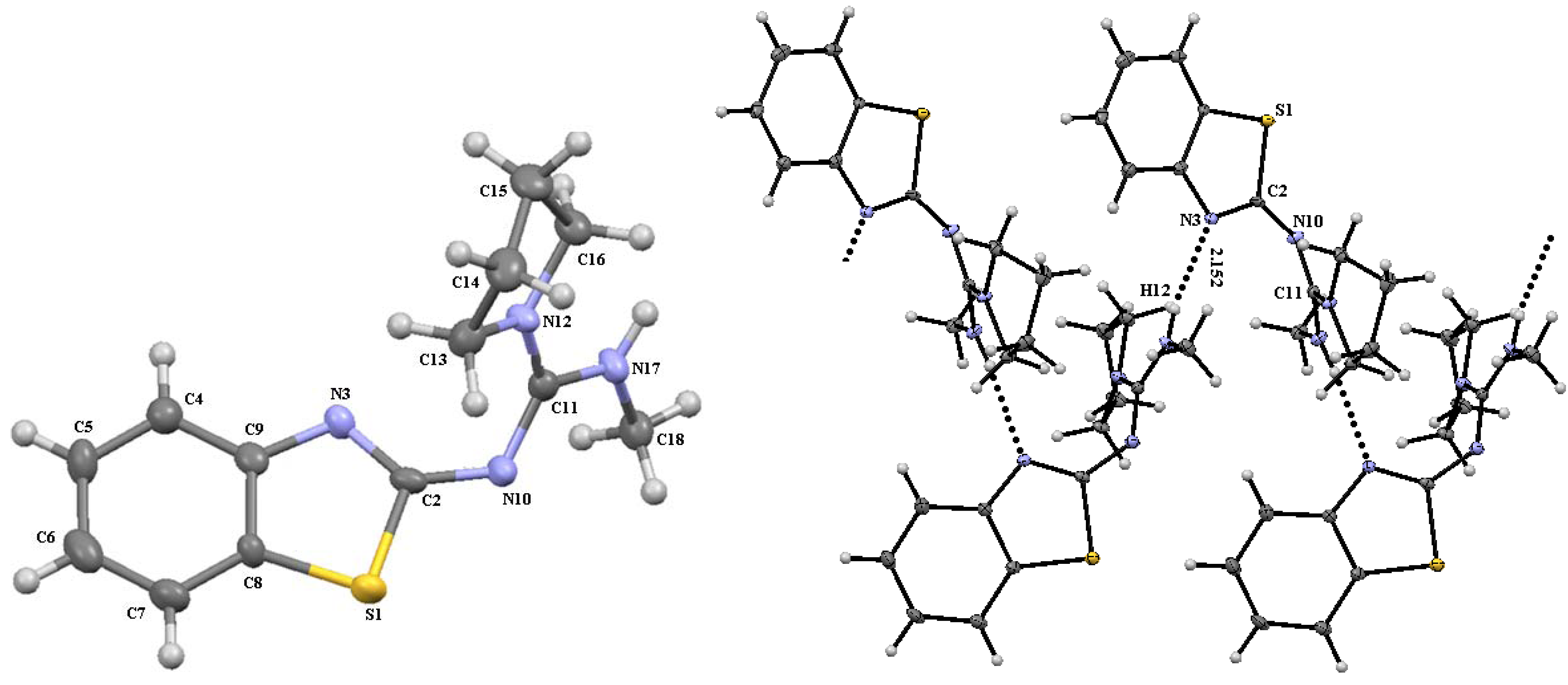
3. Experimental
3.1. General Procedures
| Comp. | Yield (%) | Physical appearance | M.p. (°C) | υ (cm−1) | m/z (%M+) | Elemental analysis Found (calculated) | ||
|---|---|---|---|---|---|---|---|---|
| C | H | N | ||||||
| 4 | SM | White solid | 126–129 | |||||
| 5 | 82 | Yellow powder | 72–73 | 509, 1464 | 254(20) | 47.05(47.24) | 3.95(3.94) | 11.13(11.02) |
| 9a | 62 | Colorless crystals | 198–199 | 2186, 1600, 1580 | 175(100) | 54.02(54.85) | 3.03(2.85) | 23.73(24.00) |
| 10b | 88 | White powder | 158–160 | 3406, 3260, 1624 | 206(100) | 52.14(52.42) | 4.88(4.85) | 27.20(27.18) |
| 10c | 92 | White powder | 242–244 | 3395, 3161, 1609, 1547 | 246(100) | 58.13(58.53) | 5.71(5.69) | 22.40(22.76) |
| 10d | 76 | White powder | 148–150 | 3436, 3198, 1613, 1568 | 57.42(62.68) | 4.57(4.48) | 19.05(20.89) | |
| 10e | 89 | White powder | 145–147 | 3418, 3200, 1597, 1560 | 63.14(63.83) | 4.98(4.96) | 19.95(19.86) | |
| 10f | 90 | Colorless crystals | 184–186 | 3395, 3161, 1609, 1547 | 66.99(67.08) | 5.70(5.59) | 17.72(17.39) | |
| 10g | 60 | White powder | 127–129 | 3400, 1613, 1580 | 344(19) | 68.19(69.76) | 4.72(4.65) | 16.17(16.28) |
| 10h | 90 | Brownish liquid | 1602, 1574 | 220(100) | 54.80(54.54) | 5.49(5.45) | 24.24(25.45) | |
| 10i | 92 | Colorless crystals | 136–137 | 3210, 3080, 1588, 1524 | 59.63(60.0) | 6.24(6.15) | 21.71(21.54) | |
| 10j | 89 | Brownish liquid | 300(100) | 63.2512(64.00) | 5.9812(6.66) | 19.12(18.66) | ||
| Compound | 10f | 10i | 9a |
|---|---|---|---|
| Unit cell information | |||
| Cell axes [Å]a | 11.3477[13] | 14.3400[20] | 5.6230[10] |
| b | 9.0463[11] | 7.8188[12] | 8.2300[9] |
| c | 16.5004[19] | 24.1730[40] | 17.2290[10] |
| Cell angles [deg]α | 90.000[0] | 90.000[0] | 90.000[0] |
| β | 101.858[2] | 101.858[2] | 90.000[0] |
| γ | 90.000[0] | 90.000[0] | 90.000[0] |
| Crystal system | Monoclinic | Orthorhombic | Monoclinic |
| Space group | P 2l/c | P na2l | P 2l/c |
| Molecular Formula | C18H18N4S | C13H16N4S | C8H5N3S |
| Density [g cm−1] | 1.29 | 1.28 | 1.46 |
| Formula weight | 322.4 | 520.7 | 175.2 |
| No. Form. Units Z | 4 | 4 | 4 |
| Reflection data | |||
| No. Meas. | 15334 | 22566 | 3406 |
| No. Uniq. | 2920 | 4245 | 1544 |
| No. Obs. | 2630 | 3049 | 1278 |
| Current refinement | |||
| No. Reflen. | 2920 | 4245 | 1544 |
| No. Param. | 208 | 325 | 110 |
| Delta-rho[eÅ−3]max, min | 0.242, −0.280 | 0.922, −0.272 | 0.274, −0.283 |
| R_all, R_obs | 0.054, 0.049 | 0.102, 0.073 | 0.054, 0.049 |
| wR2_all, wR2_aobs | 0.125, 0.121 | 0.189, 0.166 | 0.127, 0.116 |
3.2. General Procedure to Get Isothiourea Intermediates 8
3.3. General Procedure to Obtain Guanidines 10
4. Conclusions
Acknowledgments
References
- Lednicer, D.; Mitscher, L.A. The Organic Chemistry of Drugs Synthesis; Wiley: New York, NY, USA, 1980; Volume II. [Google Scholar]
- Mori, A.; Cohen, B.D.; Lowenthal, A. Historical, Biological, Biochemical and Clinical Aspects of the Naturally Occurring Guanidino Compounds; Plenium: New York, NY, USA, 1985. [Google Scholar]
- Berlinck, R.G.S. Some aspects of guanidine secondary metabolites. Fortschr. Chem. Org. Naturst. 1995, 66, 119–295. [Google Scholar] [CrossRef]
- Burgess, K. Solid-Phase Organic Synthesis; John Wiley Sons: New York, NY, USA, 2000. [Google Scholar]
- Xian, M.; Li, X.; Tang, X.; Chen, X.; Zheng, Z.; Galligan, J.J.; Kreulen, D.L.; Wang, P.G. N-Hydroxyl derivatives of guanidine based drugs as enzymatic NO donors. Bioorg. Med. Chem. Lett. 2001, 11, 2377–2380. [Google Scholar] [CrossRef]
- Durant, G.J. Guanidine derivatives acting at histaminergic receptors. Chem Soc. Rev. 1985, 14, 375–398. [Google Scholar] [CrossRef]
- Echavarren, A.; Galan, A.; Lehn, J.M.; De Mendoza, J. Chiral recognition of aromatic carboxylate anions by an optically active abiotic receptor containing a rigid guanidinium binding subunit. J. Am. Chem. Soc. 1989, 111, 4994–4995. [Google Scholar]
- Simoni, D.; Invidiata, F.P.; Manfredini, S.; Ferroni, R.; Lampronti, I.; Roberti, M.; Pollini, G.P. Facile synthesis of 2-nitroalkanols by tetramethylguanidine (TMG)-catalyzed addition of primary nitroalkanes to aldehydes and alicyclic ketones. Tetrahedron Lett. 1997, 38, 2749–2752. [Google Scholar] [CrossRef]
- Isobe, T.; Fukuda, K.; Ishikawa, T. Modified guanidines as potential chiral superbases. 1. Preparation of 1,3-disubstituted 2-iminoimidazolidines and the related guanidines through chloroamidine derivatives. J. Org. Chem. 2000, 65, 7770–7773. [Google Scholar]
- Isobe, T.; Fukuda, K.; Tokunaga, T.; Seki, H.; Yamaguchi, K.; Ishikawa, T. Modified guanidines as potential chiral superbases. 2. Preparation of 1,3-unsubstituted and 1-substituted 2-iminoimidazolidine derivatives and a related guanidine by the 2-chloro-1,3-dimethyl-imidazolinium chloride-induced cyclization of thioureas. J. Org. Chem. 2000, 65, 7774–7778. [Google Scholar]
- Isobe, T.; Fukuda, K.; Yamaguchi, K.; Seki, H.; Tokunaga, T.; Ishikawa, T. Modified guanidines as potential chiral superbases. 3. Preparation of 1,4,6-triazabicyclooctene systems and 1,4-disubstituted 2-iminoimidazolidines by the 2-chloro-1,3-dimethylimidazolinium chloride-induced cyclization of guanidines with a hydroxyethyl substituent. J. Org. Chem. 2000, 65, 7779–7785. [Google Scholar]
- Ryoda, A.; Yajima, N.; Haga, T.; Kumamoto, T.; Nakanishi, W.; Kawahata, M.; Yamaguchi, K.; Ishikawa, T. Optical resolution of (±)-1,2-bis(2-methylphenyl)ethylene-1,2-diamine as a chiral framework for 2-iminoimidazolidine with 2-methylphenyl pendant and the guanidine-catalyzed asymmetric Michael reaction of tert-butyl diphenyliminoacetate and ethyl acrylate. J. Org. Chem. 2008, 73, 133–141. [Google Scholar] [CrossRef]
- Saito, N.; Ryoda, A.; Nakanishi, W.; Kumamoto, T.; Ishikawa, T. Guanidine-catalyzed asymmetric synthesis of 2,2-disubstituted chromane skeletons by intramolecular oxa-Michael addition. Eur. J. Org. Chem. 2008, 2759–2766. [Google Scholar]
- Zhang, G.; Kumamoto, T.; Heima, T.; Ishikawa, T. Access to the nicotine system by application of a guanidine-catalyzed asymmetric Michael addition of diphenyliminoacetate with 3-pyridyl vinyl ketone. Tetrahedron Lett. 2010, 51, 3927–3930. [Google Scholar]
- Thai, K.; Gravel, M. Design, synthesis, and application of chiral electron-poor guanidines as hydrogen-bonding catalysts for the Michael reaction. Tetrahedron: Asymmetry 2010, 21, 751–755. [Google Scholar] [CrossRef]
- Isobe, T.; Fukuda, K.; Araki, Y.; Ishikawa, T. Modified guanidines as chiral superbases: The first example of asymmetric silylation of secondary alcohols. Chem. Commun. 2001, 243–244. [Google Scholar]
- Tang, Y.; Li, X.; Lian, C.; Zhu, J.; Deng, J. Synthesis of a water-soluble cationic chiral diamine ligand bearing a diguanidinium and application in asymmetric transfer hydrogenation. Tetrahedron: Asymmetry 2011, 22, 1530–1535. [Google Scholar] [CrossRef]
- Baker, T.J.; Luedke, N.W.; Tor, Y.; Goodman, M. Synthesis and Anti-HIV Activity of guanidinoglycosides. J. Org. Chem. 2000, 65, 9054–9058. [Google Scholar]
- Hui, Y.; Ptak, R.; Pallansch, M.; Chang, C.-W.W. Synthesis of novel guanidine incorporated aminoglycosides, guanidinopyranmycins. Tetrahedron Lett. 2002, 43, 9255–9257. [Google Scholar] [CrossRef]
- Izdebski, J.; Witkowska, E.; Kunce, D.; Orlowska, A.; Baranowska, B.; Radzikowska, M.; Smoluch, M. New potent hGH-RH analogues with increased resistance to enzymatic degradation. J. Peptide Sci. 2002, 8, 289–296. [Google Scholar] [CrossRef]
- Schow, S. Cyanamide. In Encyclopedia of Reagents for Organic Synthesis; Paquette, L.A., Ed.; Wiley: Sussex, UK, 1995; pp. 1408–1410. [Google Scholar]
- Palmer, D.C. O-Methylisourea. In Encyclopedia of Reagents for Organic Synthesis; Paquette, L.A., Ed.; Wiley: Sussex, UK, 1995; pp. 3525–3526. [Google Scholar]
- Bergeron, R.J.; Mcmanis, J.S. Total synthesis of (±)-15-deoxyspergualin. J. Org. Chem. 1987, 52, 1700–1703. [Google Scholar] [CrossRef]
- Dumas, D.J. Total synthesis of peramine. J. Org. Chem. 1988, 53, 4650–4653. [Google Scholar] [CrossRef]
- Moroni, M.; Kokschy, B.; Osipov, S.N.; Crucianelli, M.; Frigerio, M.; Bravo, P.; Burger, K. First synthesis of totally orthogonal protected α-(trifluoromethyl)- and α-(difluoromethyl)arginines. J. Org. Chem. 2001, 66, 130–133. [Google Scholar] [CrossRef]
- Bernatowics, M.S. 1H-Pyrazyle-1-carboxamidine Hydrochloride. In Encyclopedia of Reagents for Organic Synthesis; Paquette, L.D., Ed.; Wiley: Sussex, UK, 1995; pp. 4343–4344. [Google Scholar]
- Linton, B.R.; Carr, A.J.; Orner, B.P.; Hamilton, A.D. A Versatile one-pot synthesis of 1,3-substituted guanidines from carbamoyl isothiocyanates. J. Org. Chem. 2000, 65, 1566–1568. [Google Scholar] [CrossRef]
- Manimala, J.C.; Anslyn, E.B. Solid-phase synthesis of guanidinium derivatives from thiourea and isothiourea functionalities. Eur. J. Org. Chem. 2002, 2002, 3909–3922. [Google Scholar] [CrossRef]
- Bowser, A.M.; Madalengoitia, J.S. A 1,3-Diaza-Claisen rearrangement that affords guanidines. Org. Lett. 2004, 6, 3409–3412. [Google Scholar] [CrossRef]
- McAlpine, I.J.; Armstrong, R.W. Stereoselective synthesis of a tricyclic guanidinium model of cylindrospermopsin. Tetrahedron Lett. 2000, 41, 1849–1853. [Google Scholar] [CrossRef]
- Santagada, V.; Fiorino, F.; Severino, B.; Salvadori, S.; Lazarus, L.H.; Bryant, S.D.; Caliendo, G. A convenient synthesis of N-Fmoc-N,N′-bis-Boc-7-guanyl-1,2,3,4-tetrahydro-isoquinoline-3-carboxylic acid (Fmoc-N,N′-bis-Boc-7-guanyl-Tic-OH, GTIC). Tetrahedron Lett. 2001, 42, 3507–3509. [Google Scholar] [CrossRef]
- De Mong, D.E.; Williams, R.M. The asymmetric synthesis of (2S,3R)-capreomycidine. Tetrahedron Lett. 2001, 42, 3529–3532. [Google Scholar]
- Nagasawa, K.; Koshino, H.; Nakata, T. Stereoselective synthesis of tricyclic guanidine systems: confirmation of the stereochemistry of batzelladine F left-hand tricyclic guanidine portion. Tetrahedron Lett. 2001, 42, 4155–4158. [Google Scholar]
- Ghosh, A.K.; Hol, W.G.J.; Fan, E. Solid-phase synthesis of N-acyl-N'-alkyl/aryl disubstituted guanidines. J. Org. Chem. 2001, 66, 2161–2164. [Google Scholar] [CrossRef]
- Powell, D.A.; Phillip, D.; Ramsden, P.D.; Batey, R.A. Phase-transfer-catalyzed alkylation of guanidines by alkyl halides under biphasic conditions: A convenient protocol for the synthesis of highly functionalized guanidines. J. Org. Chem. 2003, 68, 2300–2309. [Google Scholar]
- Yong, Y.F.; Kowalski, J.A.; Lipton, M.A. A new reagent for solid and solution phase synthesis of protected guanidines from amines. Tetrahedron Lett. 1999, 40, 53–56. [Google Scholar] [CrossRef]
- Gers, T.; Kunce, D.; Markowski, P.; Izdebski, J. Reagents for efficient conversion of amines to protected guanidines. Synthesis 2004, 37–42. [Google Scholar]
- Cunha, S.; Rodriguez, M.T., Jr. The first bismuth(III)-catalyzed guanylation of thioureas. Tetrahedron Lett. 2006, 47, 6955–6956. [Google Scholar]
- Porcheddu, A.; Giacomelli, G.; Chinghine, A.; Masala, S. New cellulose-supported reagent: A sustainable approach to guanidines. Org. Lett. 2004, 6, 4925–4927. [Google Scholar]
- Deprez, P.; Vevert, J.P. Efficient two-step syntheses of sulfonylguanidines from sulfonamides. Synth. Commun. 1996, 26, 4299–4310. [Google Scholar] [CrossRef]
- Levallet, C.; Lerpiniere, J.; Ko, S.Y. The HgCl2-promoted guanylation reaction: The scope and limitations. Tetrahedron 1997, 53, 5291–5304. [Google Scholar]
- Atwal, K.S.; Ahmed, S.Z.; O’Reilly, B.C. A facile synthesis of cyanoguanidines from thioureas. Tetrahedron Lett. 1989, 30, 7313–7316. [Google Scholar]
- Wilson, L.J.; Klopfenstein, S.R.; Li, M. A traceless linker approach to the solid phase synthesis of substituted guanidines utilizing a novel acyl isothiocyanate resin. Tetrahedron Lett. 1999, 40, 3999–4002. [Google Scholar]
- Wang, H.; Ye, C.; Jin, H.; Liu, J.; Wu, J. An expeditious approach to 1-(isoquinolin-1-yl)guanidines via a three-component reaction of 2-alkynylbenzaldehyde, sulfonohydrazide, with carbodiimide. Tetrahedron 2011, 67, 5871–5877. [Google Scholar]
- Zhang, X.; Wang, C.; Qian, C.; Han, F.; Xu, F.; Shen, Q. Heterobimetallic dianionic guanidinate complexes of lanthanide and lithium: Highly efficient precatalysts for catalytic addition of amines to carbodiimides to synthesize guanidines. Tetrahedron 2011, 67, 8790–8799. [Google Scholar]
- Li, J.; Zhang, G.; Zhang, Z.; Fan, E. TFA-sensitive arylsulfonylthiourea-assisted synthesis of N,N'-substituted guanidines. J. Org. Chem. 2003, 68, 1611–1614. [Google Scholar]
- Kurser, F.; Sanderson, P.M. Thiadiazoles. Part X. The synthesis and isomerisation of 2-aryl-5-arylamino-3-arylimino-Δ4–1,2,4-thiadiazolines. J. Chem. Soc. 1960, 3240–3249. [Google Scholar]
- Weiss, V.S.; Kromer, H.; Prietzel, H. Uber 2-benzthiazolyl-guanidin: Biologische Wirksamkeit und verbessertes Dartellungsverfharen. Chemiker-Zeit. 1975, 99, 291, and references cited therein.. [Google Scholar]
- Téllez, F.; Cruz, A.; López-Sandoval, H.; Ramos-García, I.; Gayosso, M.; Castillo-Sierra, R.N.; Paz-Michel, B.; Nöth, H.; Flores-Parra, A.; Contreras, R. Dithiocarbamates, thiocarbamic esters, dithiocarboimidates, guanidines, thioureas, isothioureas, and tetraazathiapentalene derived from 2-aminobenzothiazole. Eur. J. Org. Chem. 2004, 4203–4214. [Google Scholar]
- Merchan, F.L.; Garín, J.; Meléndez, E. A facile synthesis of dimethyl N-(2-benzothiazolyl)-dithiocarbonimidates and methyl N-(2-benzothiazolyl)-dithiocarbamates. Synthesis 1982 1982, 590–591. [Google Scholar]
- Garín, J.; Meléndez, E.; Merchan, F.L.; Ortíz, D.; Tejero, T. 2-(2-Benzimidazolylamino-benzothiazoles and 2-(2-imidazolidinylidenamino)-benzothiazoles. Synthesis 1982, 1066–1067. [Google Scholar]
- Garín, J.; Meléndez, E.; Merchan, F.L.; Ortíz, D.; Tejero, T. A facile synthesis of 8-arylamino- and 8-hetarylaminopurines and their 1- and 3-deaza analogs. Synthesis 1985, 867–869. [Google Scholar]
- Merchan, F.L.; Garín, J.; Meléndez, E.; Tejero, T. A facile synthesis of 2-(2-benzothiazolylamino)-1,3-heterazoles. Synthesis 1987, 368–370. [Google Scholar]
- Cruz, A.; Padilla-Martínez, I.I.; García-Báez, E.V.; Juárez, M.J. S-Methyl-(-N-aryl and -N-alkyl)isothioureas derived from 2-aminobenzothiazole. ARKIVOC 2008, V, 200–209. [Google Scholar]
- Allen, F.H.; Kenard, O.; Watson, D.G.; Brammer, L.; Orpen, A.G.; Taylos, R. Tables of bond lengths determined by X-ray and neutron diffraction. Part 1. Bond lenghts in organic compounds. J. Chem. Soc. Perkin Trans. II 1987, S1–S19. [Google Scholar]
- Allen, F.H.; Kennard, O.; Watson, D.G.; Brammer, L.; Orpen, A.G.; Taylor, R. Typical Interatomic Distances: Organic Compounds. In International Tables for Crystallography; Wilson, A.J.C., Ed.; The International Union of Crystallography, Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992; Volume C, p. 685. [Google Scholar]
- Bruker, SMART and SAINT, Versions 6.02a; Bruker AXS Inc.: Madison, WI, USA, 2000.
- Sheldrick, G.M. SHELXS97 and SHELXL97; University of Göttingen: Göttingen, Germany, 1997. [Google Scholar]
- Spek, A.L. PLATON, Version of March 2002; University of Utrecht: Heidelberglaan, The Netherlands, 2002. [Google Scholar]
- Farrugia, L.J. WinGX suite for small molecule single crystal crystallography. J. Appl. Crystallogr. 1999, 32, 837–838. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 10f and 10i are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cruz, A.; Padilla-Martínez, I.I.; García-Báez, E.V. A Synthetic Method to Access Symmetric and Non-Symmetric 2-(N,N'-disubstituted)guanidinebenzothiazoles. Molecules 2012, 17, 10178-10191. https://doi.org/10.3390/molecules170910178
Cruz A, Padilla-Martínez II, García-Báez EV. A Synthetic Method to Access Symmetric and Non-Symmetric 2-(N,N'-disubstituted)guanidinebenzothiazoles. Molecules. 2012; 17(9):10178-10191. https://doi.org/10.3390/molecules170910178
Chicago/Turabian StyleCruz, Alejandro, Itzia I. Padilla-Martínez, and Efrén V. García-Báez. 2012. "A Synthetic Method to Access Symmetric and Non-Symmetric 2-(N,N'-disubstituted)guanidinebenzothiazoles" Molecules 17, no. 9: 10178-10191. https://doi.org/10.3390/molecules170910178
APA StyleCruz, A., Padilla-Martínez, I. I., & García-Báez, E. V. (2012). A Synthetic Method to Access Symmetric and Non-Symmetric 2-(N,N'-disubstituted)guanidinebenzothiazoles. Molecules, 17(9), 10178-10191. https://doi.org/10.3390/molecules170910178