Neuroprotective Activity of Lavender Oil on Transient Focal Cerebral Ischemia in Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Neurological Deficits and Infarct Area
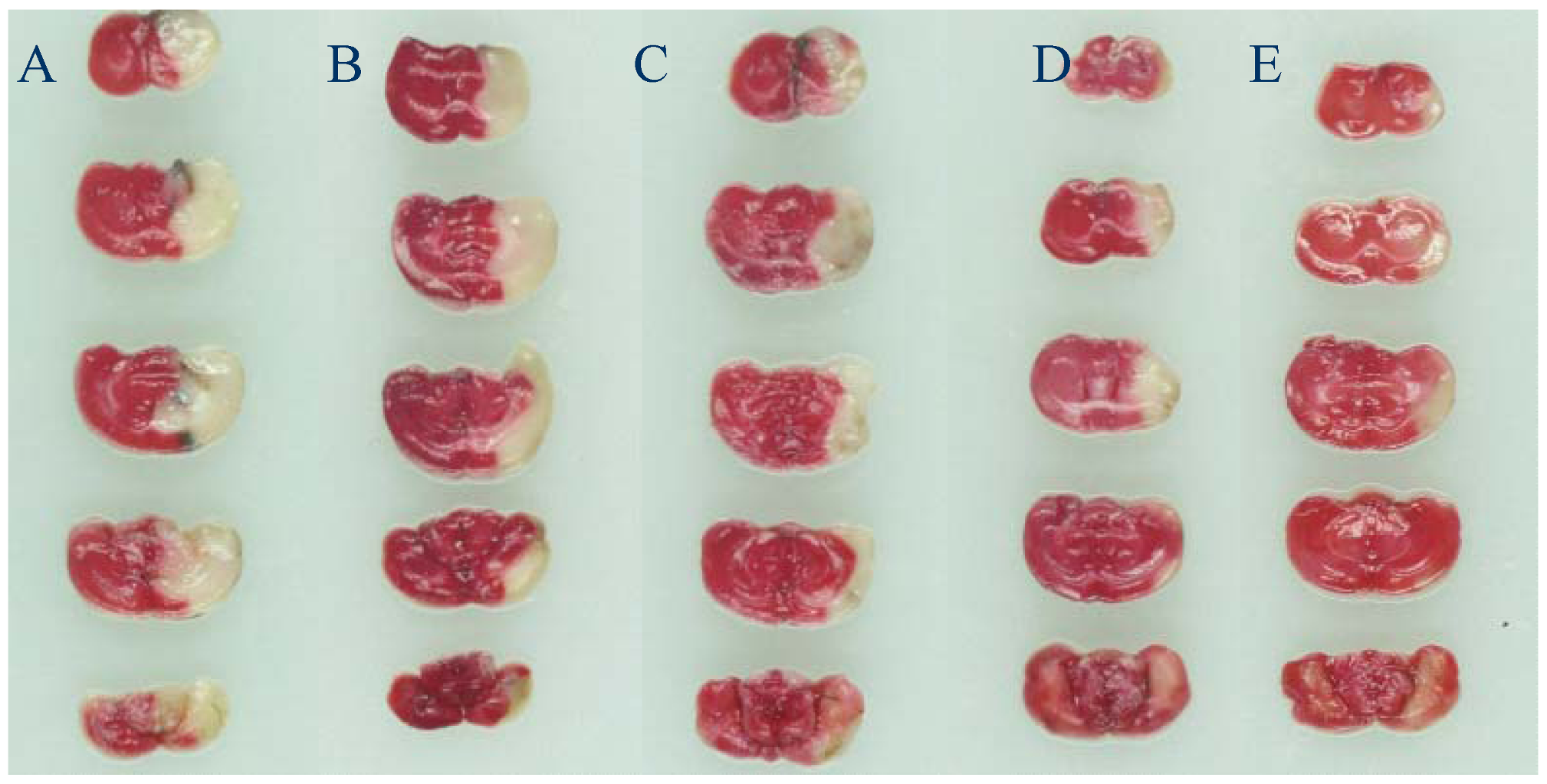
| Group | Dose (mg/kg) | Neurological deficit scores | Infarct area (%) |
|---|---|---|---|
| Vehicle | / | 2.20 ± 0.79 | 30.14 ± 3.83 |
| Sham | / | 0.00 ± 0.00 ** | 0.00 ± 0.00 ** |
| Lavender oil | 50 | 2.10 ± 0.74 | 28.11 ± 3.41 |
| Lavender oil | 100 | 2.00 ± 0.82 | 24.85 ± 2.30 * |
| Lavender oil | 200 | 1.50 ± 0.71 | 15.10 ± 2.93 ** |
| Edaravone | 3 | 1.40 ± 0.52 * | 11.01 ± 1.42 ** |
2.2. Histopathology (H&E) Staining
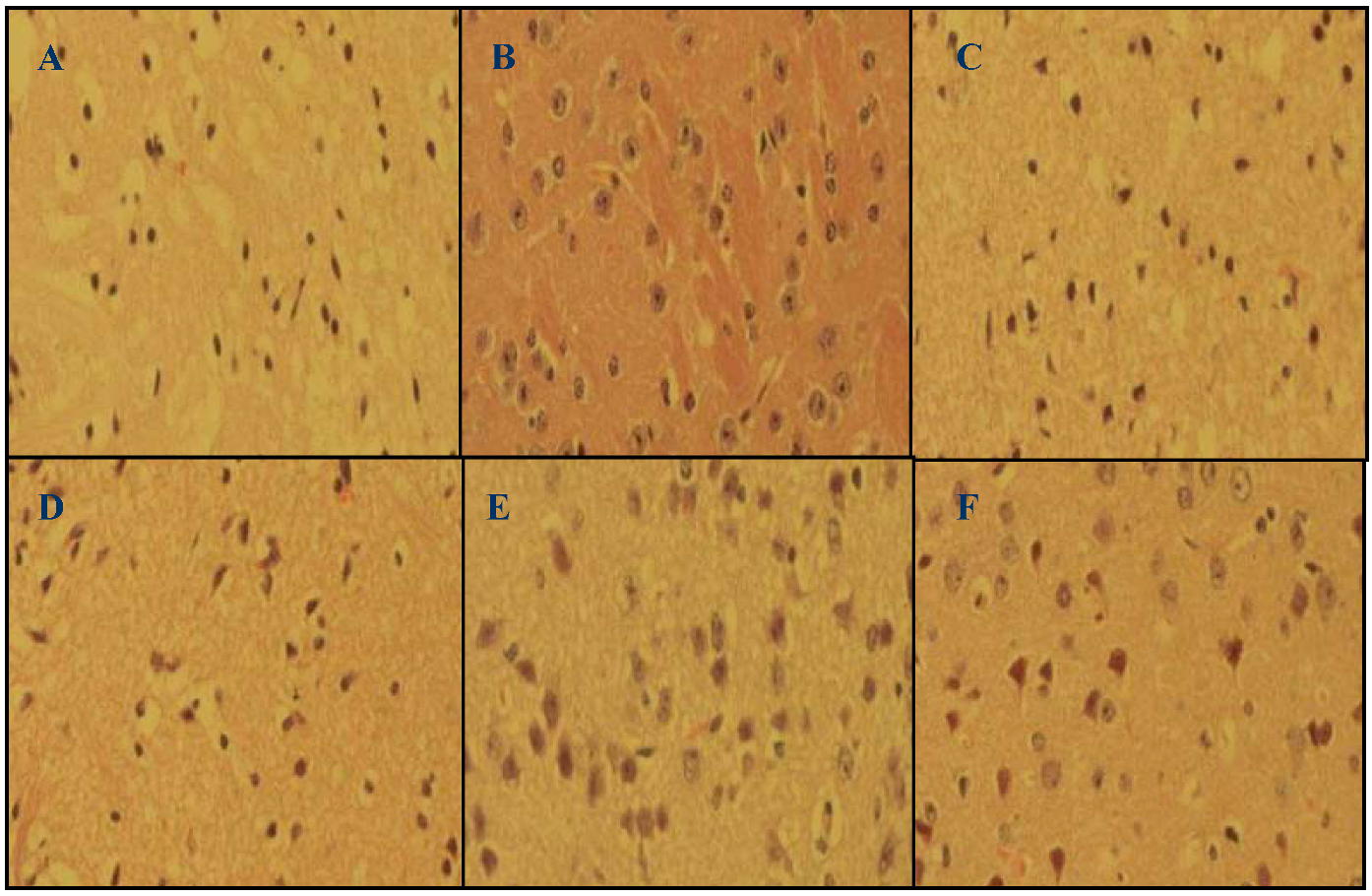
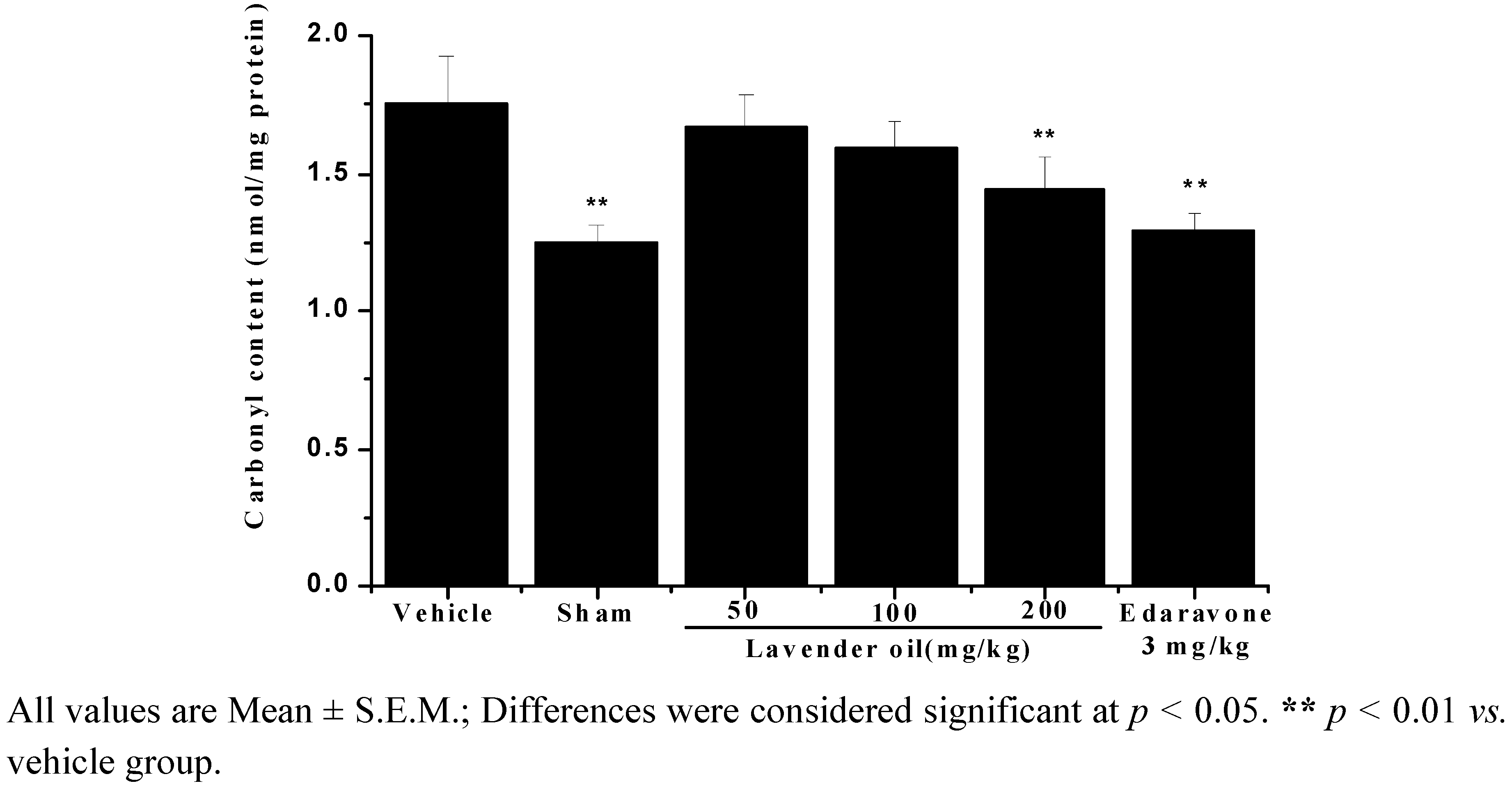
2.3. Protein Carbonyl Content (PCC) Decreased in Lavender Oil-Treated Groups
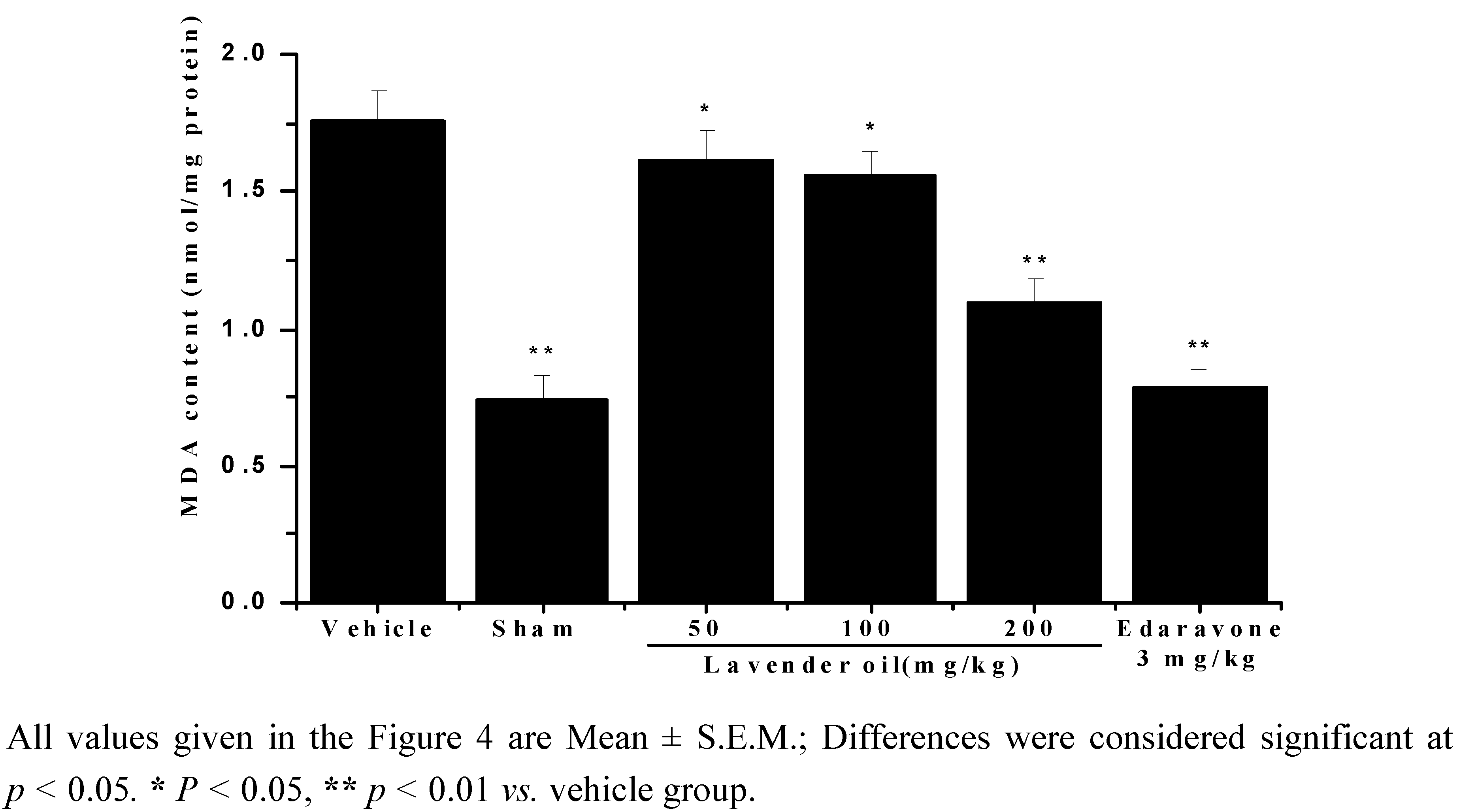
2.4. Lavender Oil-Treatment Decreased Malondialdehyde (MDA) Content
2.5. Lavender Oil-Treatment Alleviate the Decrease of Antioxidant Enzyme Activities Induced by IR
| Group | Dose (mg/kg) | CAT (U/mg protein) | SOD (U/mg protein) | GSH-Px (U/mg protein) |
|---|---|---|---|---|
| Vehicle | / | 28.38 ± 10.62 | 5.90 ± 1.19 | 27.04 ± 4.24 |
| Sham | / | 69.58 ± 13.25 ** | 12.64 ± 0.78 ** | 45.13 ± 3.87 ** |
| Lavender oil | 50 | 32.10 ± 8.36 | 6.22 ± 0.91 | 29.21 ± 4.15 |
| Lavender oil | 100 | 35.40 ± 5.78 | 6.73 ± 0.81 | 33.11 ± 3.77 * |
| Lavender oil | 200 | 39.92 ± 5.75 * | 7.68 ± 0.76 * | 34.02 ± 3.51 * |
| Edaravone | 3 | 56.54 ± 6.50 ** | 10.76 ± 0.54 ** | 37.99 ± 3.05 ** |
2.6. Lavender Oil-Treatment Alleviate the Decrease of GSH/GSSG Ratio Induced by IR

2.7. Lavender Oil-Treatment Decreased Reactive ROS Formation in Brain Mitochondria

2.8. Discussion
3. Experimental
3.1. Animals
3.2. Chemicals and Reagents
3.3. Drug Administration and Surgical Procedure
3.4. Measurement of Neurological Deficits
3.5. Measurement of Infarct Area
3.6. Measurement of Histopathological Changes
3.7. Biochemical Measurements
3.7.1. Tissue Preparation
3.7.2. Measurement of Protein Carbonyl Content
3.7.3. Measurement of Malondialdehyde (MDA) Content
3.7.4. Measurement of Superoxide Dismutase (SOD) Activity
3.7.5. Measurement of Catalase (CAT) Activity
3.7.6. Measurement of Glutathione Peroxidase (GSH-Px) Activity
3.7.7. Measurement of Protein Content
3.7.8. Measurement of GSH/GSSG Ratio
3.7.9. Measurement of Reactive Oxygen Species (ROS) in Brain Mitochondria
3.7.10. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Margaill, I.; Plotkine, M.; Lerouet, D. Antioxidant strategies in the treatment of stroke. Free Radic. Biol. Med. 2005, 39, 429–443. [Google Scholar] [CrossRef]
- Doyle, K.P.; Simon, R.P.; Stenzel-Poore, M.P. Mechanisms of ischemic brain damage. Neuropharmacology 2008, 55, 310–318. [Google Scholar] [CrossRef]
- Ritz, M.F.; Curin, Y.; Mendelowitsch, A.; Andriantsitohaina, R. Acute treatment with red wine polyphenols protects from ischemia-induced excitotoxicity, energy failure and oxidative stress in rats. Brain Res. 2008, 1239, 226–234. [Google Scholar] [CrossRef]
- Wang, Q.; Sun, A.Y.; Simonyi, A.; Kalogeris, T.J.; Miller, D.K.; Sun, G.Y.; Korthuis, R.J. Ethanol preconditioning protects against ischemia/reperfusion-induced brain damage: Role of NADPH oxidase-derived ROS. Free Radic. Biol. Med. 2007, 43, 1048–1060. [Google Scholar] [CrossRef]
- Ohtaki, H.; Takeda, T.; Dohi, K.; Yofu, S.; Nakamachi, T.; Satoh, K.; Hiraizumi, Y.; Miyaoka, H.; Matsunaga, M.; Shioda, S. Increased mitochondrial DNA oxidative damage after transient middle cerebral artery occlusion in mice. Neurosci. Res. 2007, 58, 349–355. [Google Scholar]
- Chan, P.H. Reactive oxygen radicals in signaling and damage in the ischemic brain. J. Cereb. Blood Flow Metab. 2001, 21, 2–14. [Google Scholar] [CrossRef]
- Lin, Z.H.; Zhu, D.; Yan, Y.Q.; Yu, B.Y. Herbal formula FBD extracts prevented brain injury and inflammation induced by cerebral ischemia-reperfusion. J. Ethnopharmacol. 2008, 118, 140–147. [Google Scholar] [CrossRef]
- Nakamura, T.; Kume, T.; Katsuki, H.; Niidome, T.; Sugimoto, H.; Akaike, A. Protective effect of serofendic acid on ischemic injury induced by occlusion of the middle cerebral artery in rats. Eur. J. Pharmacol. 2008, 586, 151–155. [Google Scholar] [CrossRef]
- Thiyagarajan, M.; Sharma, S.S. Neuroprotective effect of curcumin in middle cerebral artery occlusion induced focal cerebral ischemia in rats. Life Sci. 2004, 74, 969–985. [Google Scholar] [CrossRef]
- Sienkiewicz, M.; Kowalczyk, E.; Wasiela, M. Recent patents regarding essential oils and the significance of their constituents in human health and treatment. Recent Pat. Anti-Infect. Drug Discov. 2012, 7, 133–140. [Google Scholar] [CrossRef]
- Kivrak, İ.; Duru, M.E.; Öztürk, M.; Mercan, N.; H;rmandar, M.; Topçu, G. Antioxidant, anticholinesterase and antimicrobial constituents from the essential oil and ethanol extract of Salvia potentillifolia. Food Chem. 2009, 116, 470–479. [Google Scholar] [CrossRef]
- Yang, S.A.; Jeon, S.K.; Lee, E.J.; Shim, C.H.; Lee, I.S. Comparative study of the chemical composition and antioxidant activity of six essential oils and their components. Nat. Prod. Res. 2010, 24, 140–151. [Google Scholar] [CrossRef]
- Hai, P.; Shang, H.; Zhou, S.X. Initial approach on Dracocephalum Tanguticum Maxim against hypoxic-ischemic. J. Med. Pharm. Chin. Minorities 1999, 4, 32–33. [Google Scholar]
- Mazzanti, G.; Battinelli, L.; Salvatore, G. Antimicrobial properties of the linalol-rich essential oil of Hyssopus officinalis L. var decumbens (Lamiaceae). Flavour Frag. J. 1998, 13, 289–294. [Google Scholar] [CrossRef]
- Lu, M.; Battinelli, L.; Daniele, C.; Melchioni, C.; Salvatore, G.; Mazzanti, G. Muscle relaxing activity of Hyssopus officinalis essential oil on isolated intestinal preparations. Planta Med. 2002, 68, 213–216. [Google Scholar] [CrossRef]
- Reddy, K.K.; Grossman, L.; Rogers, G.S. Common complementary and alternative therapies with potential use in dermatologic surgery: Risks and benefits. J. Am. Acad. Dermatol. 2011. [Google Scholar] [CrossRef]
- Zhilyakova, E.T.; Novikov, O.O.; Naumenko, E.N.; Krichkovskaya, L.V.; Kiseleva, T.; Timoshenko, E.Y.; Novikova, M.Y.; Litvinov, S.A. Study of Monarda fistulosa essential oil as a prospective antiseborrheic agent. Bull. Exp. Biol. Med. 2009, 148, 612–614. [Google Scholar] [CrossRef]
- Sayorwan, W.; Siripornpanich, V.; Piriyapunyaporn, T.; Hongratanaworakit, T.; Kotchabhakdi, N.; Ruangrungsi, N. The effects of lavender oil inhalation on emotional states, autonomic nervous system, and brain electrical activity. J. Med. Assoc. Thai. 2012, 95, 598–606. [Google Scholar]
- Ballabeni, V.; Tognolini, M.; Chiavarini, M.; Impicciatore, M.; Bruni, R.; Bianchi, A.; Barocelli, E. Novel antiplatelet and antithrombotic activities of essential oil from Lavandula hybrida reverchon “grosso”. Phytomedicine 2004, 11, 596–601. [Google Scholar] [CrossRef]
- Evandri, M.G.; Battinelli, L.; Daniele, C.; Mastrangelo, S.; Bolle, P.; Mazzanti, G. The antimutagenic activity of Lavandula angustifolia (lavender) essential oil in the bacterial reverse mutation assay. Food Chem. Toxicol. 2005, 43, 1381–1387. [Google Scholar] [CrossRef]
- Holmes, C.; Hopkins, V.; Hensford, C.; MacLaughlin, V.; Wilkinson, D.; Rosenvinge, H. Lavender oil as a treatment for agitated behaviour in severe dementia: a placebo controlled study. Int. J. Geriatr. Psych. 2002, 17, 305–308. [Google Scholar] [CrossRef]
- Hajhashemi, V.; Ghannadi, A.; Sharif, B. Anti-inflammatory and analgesic properties of the leaf extracts and essential oil of Lavandula angustifolia Mill. J. Ethnopharmacol. 2003, 89, 67–71. [Google Scholar] [CrossRef]
- Hritcu, L.; Cioanca, O.; Hancianu, M. Effects of lavender oil inhalation on improving scopolamine-induced spatial memory impairment in laboratory rats. Phytomedicine 2012, 19, 529–534. [Google Scholar] [CrossRef]
- Durukan, A.; Tatlisumak, T. Acute ischemic stroke: Overview of major experimental rodent vehicle groups, pathophysiology, and therapy of focal cerebral ischemia. Pharmacol. Biochem. Behav. 2007, 87, 179–197. [Google Scholar] [CrossRef]
- Higashi, Y.; Jitsuiki, D.; Chayama, K.; Yoshizumi, M. Edaravone (3-Methyl-1-phenyl-2-pyrazolin- 5-one), a novel free radical scavenger, for treatment of cardiovascular diseases. Recent Pat. Cardiovasc. Drug Discov. 2006, 1, 85–93. [Google Scholar] [CrossRef]
- Hagvall, L.; Sköld, M.; Bråred-Christensson, J.; Börje, A.; Karlberg, A.T. Lavender oil lacks natural protection against autoxidation, forming strong contact allergens on air exposure. Contact Dermatitis 2008, 59, 143–145. [Google Scholar] [CrossRef]
- Di Sotto, A.; Mazzanti, G.; Carbone, F.; Hrelia, P.; Maffei, F. Genotoxicity of lavender oil, linalyl acetate, and linalool on human lymphocytes in vitro. Environ. Mol. Mutagen 2011, 52, 69–71. [Google Scholar] [CrossRef]
- Bhattacharya, S.; Mula, S.; Gamre, S.; Kamat, J.P.; Bandyopadhyay, S.K.; Chattopadhyay, S. Inhibitory property of Piper betel extract against photosensitization-induced damages to lipids and proteins. Food Chem. 2007, 100, 1474–1480. [Google Scholar] [CrossRef]
- Choi, Y.B.; Kim, Y.I.; Lee, K.S.; Kim, B.S.; Kim, D.J. Protective effect of epigallocatechin gallate on brain damage after transient middle cerebral artery occlusion in rats. Brain Res. 2004, 1019, 47–54. [Google Scholar] [CrossRef]
- Hou, H.; Li, B.F.; Zhao, X.; Zhuang, Y.L.; Ren, G.Y.; Yan, M.Y.; Cai, Y.P.; Zhang, X.K.; Chen, L. The effect of pacific cod (Gadus macrocephalus) shin gelatin polypeptides on UV radiation-induced skin photoaging in ICR mice. Food Chem. 2009, 115, 945–950. [Google Scholar] [CrossRef]
- Moro, M.A.; Almeida, A.; Bolaños, J.P.; Lizasoain, I. Mitochondrial respiratory chain and free radical generation in stroke. Free Radic. Biol. Med. 2005, 39, 1291–1304. [Google Scholar] [CrossRef]
- Chan, P.H. Mitochondria and neuronal death/survival signaling pathways in cerebral ischemia. Neurochem. Res. 2004, 29, 1943–1949. [Google Scholar] [CrossRef]
- Hua, F.; Ma, J.; Ha, T.Z.; Kelley, J.L.; Kao, R.L.; Schweitzer, J.B.; Kalbfleisch, J.H.; Williams, D.L.; Li, C.F. Differential roles of TLR2 and TLR4 in acute focal cerebral ischemia/reperfusion injury in mice. Brain Res. 2009, 1262, 100–108. [Google Scholar] [CrossRef]
- Longa, E.Z.; Weinstein, P.R.; Carlson, S.; Cummins, R. Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 1989, 20, 84–91. [Google Scholar] [CrossRef]
- Kuang, X.; Yao, Y.; Du, J.R.; Liu, Y.X.; Wang, C.Y.; Qian, Z.M. Neuroprotective role of Z-ligustilide against forebrain ischemic injury in ICR mice. Brain Res. 2006, 1102, 145–153. [Google Scholar] [CrossRef]
- Levine, R.L.; Garland, D.; Oliver, C.N.; Amici, A.; Climent, I.; Lenz, A.G.; Ahn, B.W.; Shaltiel, S.; Stadtman, E.R. Determination of carbonyl content in oxidatively modified proteins. Methods Enzymol. 1990, 186, 464–478. [Google Scholar]
- Cechetti, F.; Fochesatto, C.; Scopel, D.; Nardin, P.; Gonçalves, C.A.; Netto, C.A.; Siqueira, I.R. Effect of a neuroprotective exercise protocol on oxidative state and BDNF levels in the rat hippocampus. Brain Res. 2008, 1188, 182–188. [Google Scholar] [CrossRef]
- Jung, H.J.; Nam, J.H.; Choi, J.W.; Lee, K.T.; Park, H.J. Antiinflammatory effects of chiisanoside and chiisanogenin obtained from the leaves of Acanthopanax chiisanensis in the carrageenan- and Freund’s complete adjuvant-induced rats. J. Ethnopharmacol. 2005, 97, 359–367. [Google Scholar] [CrossRef]
- Campo, G.M.; Avenoso, A.; Campo, S.; Ferlazzo, A.M.; Micali, C.; Zanghi, L.; Calatroni, A. Hyaluronic acid and chondroitin-4-sulphate treatment reduces damage in carbon tetrachloride-induced acute rat liver injury. Life Sci. 2004, 74, 1289–1305. [Google Scholar] [CrossRef]
- Jagetia, G.C.; Rajanikant, G.K.; Rao, S.K.; Baliga, M.S. Alteration in the glutathione, glutathione peroxidase, superoxide dismutase and lipid peroxidation by ascorbic acid in the skin of mice exposed to fractionated γ radiation. Clin. Chim. Acta 2003, 332, 111–121. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye bingding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar]
- Zhu, Y.; Carvey, P.M.; Ling, Z. Age-related changes in glutathione and glutathione-related enzymes in rat brain. Brain Res. 2006, 1090, 35–44. [Google Scholar] [CrossRef]
- LeBel, C.P.; Ischiropoulos, H.; Bondy, S.C. Evaluation of the probe 2′,7′-dichlorofluorescin as an indicator of reactive oxygen species formation and oxidative stress. Chem. Res. Toxicol. 1992, 5, 227–231. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the Lavender oil is available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Wang, D.; Yuan, X.; Liu, T.; Liu, L.; Hu, Y.; Wang, Z.; Zheng, Q. Neuroprotective Activity of Lavender Oil on Transient Focal Cerebral Ischemia in Mice. Molecules 2012, 17, 9803-9817. https://doi.org/10.3390/molecules17089803
Wang D, Yuan X, Liu T, Liu L, Hu Y, Wang Z, Zheng Q. Neuroprotective Activity of Lavender Oil on Transient Focal Cerebral Ischemia in Mice. Molecules. 2012; 17(8):9803-9817. https://doi.org/10.3390/molecules17089803
Chicago/Turabian StyleWang, Dong, Xuan Yuan, Ting Liu, Liangliang Liu, Yanli Hu, Zhenhua Wang, and Qiusheng Zheng. 2012. "Neuroprotective Activity of Lavender Oil on Transient Focal Cerebral Ischemia in Mice" Molecules 17, no. 8: 9803-9817. https://doi.org/10.3390/molecules17089803
APA StyleWang, D., Yuan, X., Liu, T., Liu, L., Hu, Y., Wang, Z., & Zheng, Q. (2012). Neuroprotective Activity of Lavender Oil on Transient Focal Cerebral Ischemia in Mice. Molecules, 17(8), 9803-9817. https://doi.org/10.3390/molecules17089803




