Echinoclerodane A: A New Bioactive Clerodane-Type Diterpenoid from a Gorgonian Coral Echinomuricea sp.
Abstract
:1. Introduction
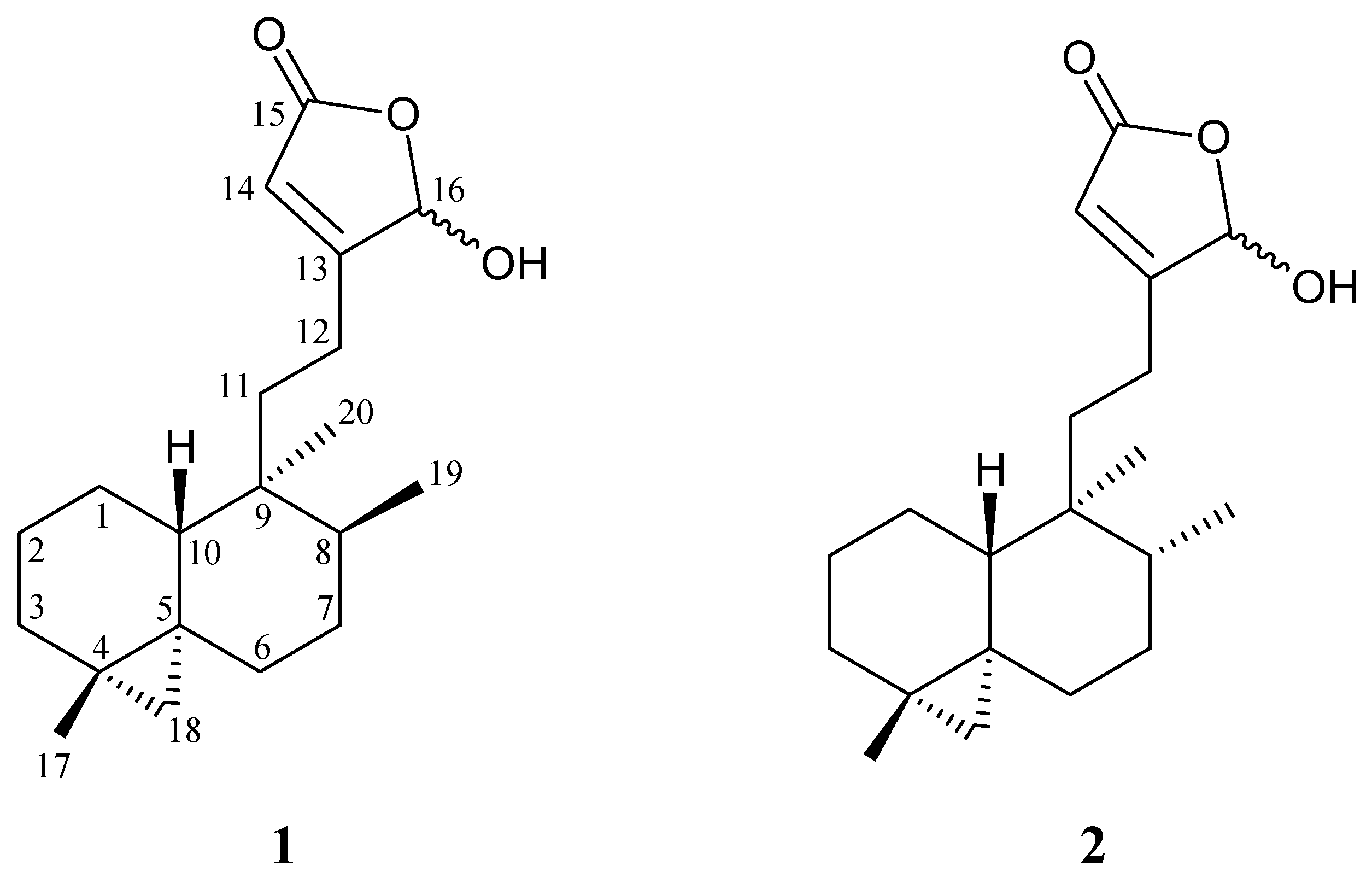
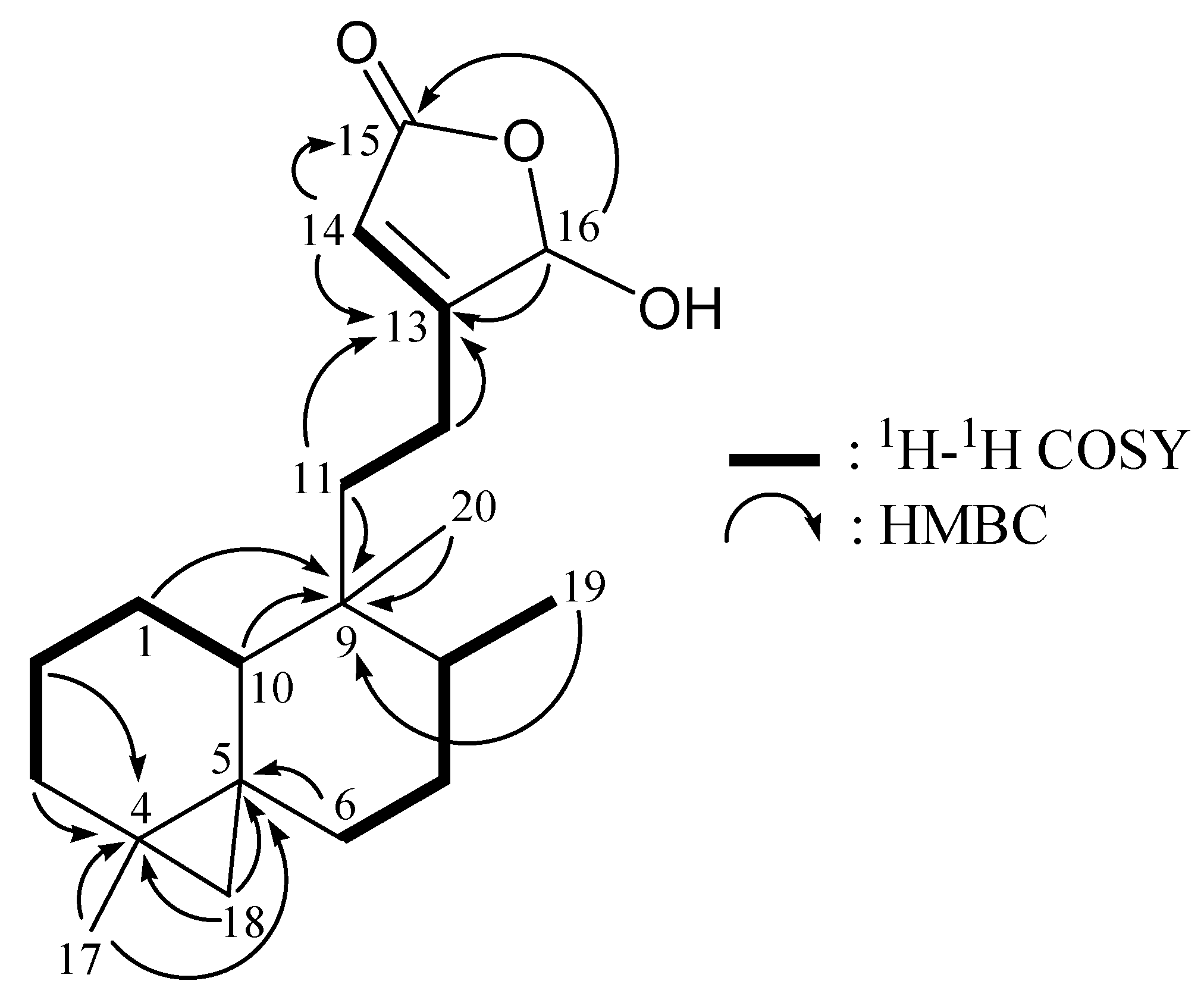
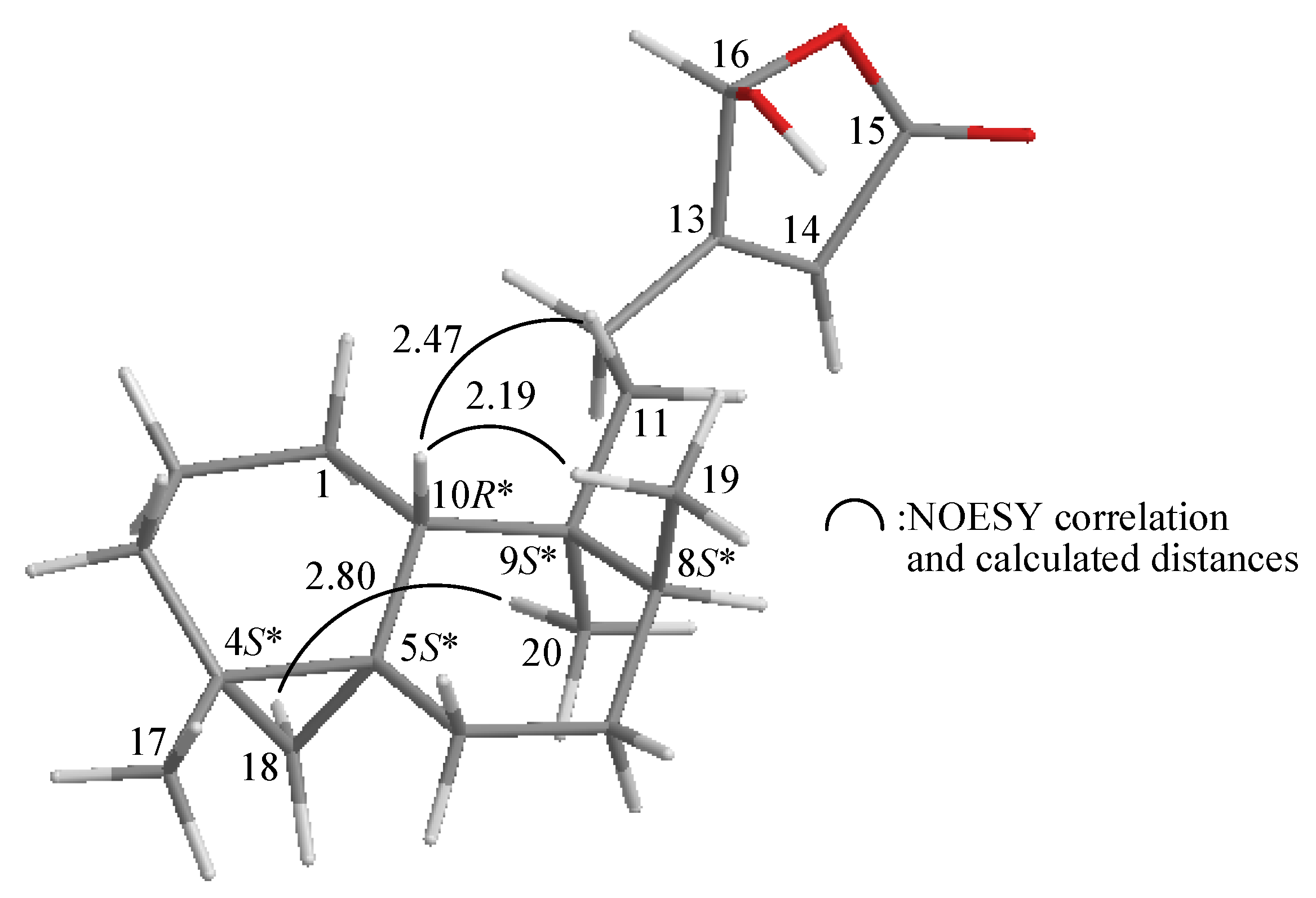
2. Results and Discussion
| Position | δH (J in Hz) | δC, Mult. | 1H–1H COSY | HMBC (H→C) |
|---|---|---|---|---|
| 1a/b | 0.75 dd (8.4, 2.8); 1.42 m | 19.9, CH2 | H2-2, H-10 | C-9, -10 |
| 2a/b | 1.19 m; 1.49 m | 23.2, CH2 | H2-1, H2-3 | C-1, -3, -4 |
| 3 | 1.58 m | 32.1, CH2 | H2-2 | C-4, -18 |
| 4 | 17.4, C | |||
| 5 | 26.3, C | |||
| 6a/b | 1.02 m; 1.80 td (14.0, 2.8) | 27.6, CH2 | H2-7 | C-5, -7, -8, -10, -18 |
| 7a/b | 1.35 m; 1.92 tt (14.0, 4.0) | 27.9, CH2 | H2-6, H-8 | C-6, -8, -19 |
| 8 | 1.68 m | 35.6, CH | H2-7, H3-19 | C-6, -7, -9, -19 |
| 9 | 39.1, C | |||
| 10 | 1.64 dd (12.4, 4.0) | 40.9, CH | H2-1 | C-8, -9, -10, -20 |
| 11 | 1.39 m; 1.59 m | 35.5, CH2 | H2-12 | C-8, -9, -12 |
| 12 | 2.35 dd (8.8, 7.2) | 21.5, CH2 | H2-11, H-14 | C-11, -13, -14, -16 |
| 13 | 171.2, C | |||
| 14 | 5.84 br s | 116.9, CH | H2-12 | C-12, -13,-15 |
| 15 | 171.8, C | |||
| 16 | 6.01 s | 99.2, CH | C-13, -15 | |
| 17 | 1.04 s | 22.4, CH3 | C-3, -4, -5 | |
| 18a/b | 0.13 d (4.4); 0.52 d (4.4) | 24.5, CH2 | C-3, -4, -5, -6, -10, -17 | |
| 19 | 0.97 d (7.2) | 14.2, CH3 | H-8 | C-7, -8, -9 |
| 20 | 1.00 s | 19.8, CH3 | C-8, -9 |
| Compounds | Cell lines IC50 (μM) | |||||
|---|---|---|---|---|---|---|
| K562 | MOLT-4 | HL-60 | DLD-1 | LoVo | DU-145 | |
| 1 | 37.05 | 13.18 | 14.89 | 23.44 | 21.69 | 53.93 |
| Doxorubicin a | 0.29 | 0.001 | 0.08 | 4.00 | 1.65 | 0.01 |
3. Experimental
3.1. General Experimental Procedures
3.2. Animal Material
3.3. Extraction and Isolation
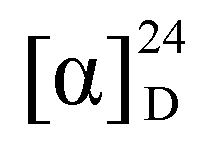 −43 (c 0.07, CHCl3); IR (neat) νmax 3,318, 1,741 cm–1; 1H- (CDCl3, 400 MHz) and 13C- (CDCl3, 100 MHz) NMR data, see Table 1; ESIMS m/z 341 [M+Na]+; HRESIMS: m/z 341.2095 (calcd. for C20H30O3Na, 341.2093).
−43 (c 0.07, CHCl3); IR (neat) νmax 3,318, 1,741 cm–1; 1H- (CDCl3, 400 MHz) and 13C- (CDCl3, 100 MHz) NMR data, see Table 1; ESIMS m/z 341 [M+Na]+; HRESIMS: m/z 341.2095 (calcd. for C20H30O3Na, 341.2093).3.4. Molecular Mechanics Calculations
3.5. Cytotoxicity Testing
3.6. Superoxide Anion Generation and Elastase Release by Human Neutrophils
4. Conclusions
- Sample Availability: Not Available.
Acknowledgments
References
- Blunt, J.W.; Copp, B.R.; Keyzers, R.A.; Munro, M.H.G.; Prinsep, M.R. Marine natural products. Nat. Prod. Rep. 2012, 29, 144–222. [Google Scholar] [CrossRef]
- Berrue, F.; Kerr, R.G. Diterpenes from gorgonian corals. Nat. Prod. Rep. 2009, 26, 681–710. [Google Scholar] [CrossRef]
- Chung, H.-M.; Hwang, T.-L.; Wang, W.-H.; Fang, L.-S.; Sung, P.-J. Curcuphenol derivatives from the gorgonian Echinomuricea sp. Heterocycles 2009, 78, 2595–2600. [Google Scholar] [CrossRef]
- Chung, H.-M.; Hong, P.-H.; Su, J.-H.; Hwang, T.-L.; Lu, M.-C.; Fang, L.-S.; Wu, Y.-C.; Li, J.-J.; Chen, J.-J.; Wang, W.-H.; et al. Bioactive compounds from a gorgonian coral Echinomuricea sp. (Plexauridae). 2012, 10, 1169–1179. [Google Scholar]
- Allinger, N.L. Conformation analysis. 130. MM2. A hydrocarbon force field utilizing V1 and V2 torsional terms. J. Am. Chem. Soc. 1977, 99, 8127–8134. [Google Scholar] [CrossRef]
- Shimbo, K.; Tsuda, M.; Fukushi, E.; Kawabata, J.; Kobayashi, J. Dytesinins A and B, new clerodane-type diterpenes with a cyclopropane ring from the tunicate Cystodytes sp. Tetrahedron 2000, 56, 7923–7926. [Google Scholar] [CrossRef]
- In the in vitro anti-inflammatory bioassay, the inhibitory effect on the generation of superoxide anion and the release of elastase by activated neutrophils were used as indicators. To indicate significant activity of pure compounds, an inhibition rate > 40% is required (inhibition rate < 10%, not active, 20% > inhibition rate > 10%, weakly anti-inflammatory; 40% > inhibition rate > 20%, modestly anti-inflammatory). Diphenyl indonium (DPI) and elastatinal were used as reference compounds in anti-inflammatory activity testing. DPI display an inhibitory effect on the generation of superoxide anion (IC50 = 0.9 μg/mL), and elastatinal exhibited an inhibitory effect on the release of elastase (IC50 = 30.1 μg/mL) by human neutrophils, respectively
- Bayer, F.M. Key to the genera of Octocorallia exclusive of Pennatulacea (Coelenterata: Anthozoa), with diagnoses of new taxa. Proc. Biol. Soc. Wash. 1981, 94, 902–947. [Google Scholar]
- Fabricius, K.; Alderslade, P. Soft Corals and Sea Fans—A Comprehensive Guide to the Tropical Shallow-Water Genera of the Central-West Pacific, the Indian Ocean and the Red Sea, 1st ed; Australian Institute of Marine Science: Queensland, Australia, 2001. [Google Scholar]
- Alley, M.C.; Scudiero, D.A.; Monks, A.; Hursey, M.L.; Czerwinski, M.J.; Fine, D.L.; Abbott, B.J.; Mayo, J.G.; Shoemaker, R.H.; Boyd, M.R. Feasibility of drug screening with panels of human tumor cell lines using a microculture tetrazolium assay. Cancer Res. 1988, 48, 589–601. [Google Scholar]
- Scudiero, D.A.; Shoemaker, R.H.; Paull, K.D.; Monks, A.; Tierney, S.; Nofziger, T.H.; Currens, M.J.; Seniff, D.; Boyd, M.R. Evaluation of a soluble tetrazolium/formazan assay for cell growth and drug sensitivity in culture using human and other tumor cell lines. Cancer Res. 1988, 48, 4827–4833. [Google Scholar]
- Yu, H.-P.; Hsieh, P.-W.; Chang, Y.-J.; Chung, P.-J.; Kuo, L.-M.; Hwang, T.-L. 2-(2-Fluorobenz-amido)benzoate ethyl ester (EFB-1) inhibits superoxide production by human neutrophils and attenuates hemorrhagic shock-induced organ dysfunction in rats. Free Radical Biol. Med. 2011, 50, 1737–1748. [Google Scholar] [CrossRef]
- Hwang, T.-L.; Wang, C.-C.; Kuo, Y.-H.; Huang, H.-C.; Wu, Y.-C.; Kuo, L.-M.; Wu, Y.-H. The hederagenin saponin SMG-1 is a natural FMLP receptor inhibitor that suppresses human neutrophil activation. Biochem. Pharmacol. 2010, 80, 1190–1200. [Google Scholar] [CrossRef]
- Hanson, J.R. Diterpenoids of terrestrial origin. Nat. Prod. Rep. 2011, 28, 1755–1772. [Google Scholar] [CrossRef]
- Harper, M.K.; Bugni, T.S.; Copp, B.R.; James, R.D.; Lindsay, B.S.; Richardson, A.D.; Schnabel, P.C.; Tasdemir, D.; van Wagoner, R.M.; Verbitski, S.M.; et al. Introduction to the Chemical Ecology of Marine Natural Products. In Marine Chemical Ecology; McClintock, J.B., Baker, B.J., Eds.; CRC Press: Washington, DC, USA, 2001; pp. 3–69. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Cheng, C.-H.; Chung, H.-M.; Hwang, T.-L.; Lu, M.-C.; Wen, Z.-H.; Kuo, Y.-H.; Wang, W.-H.; Sung, P.-J. Echinoclerodane A: A New Bioactive Clerodane-Type Diterpenoid from a Gorgonian Coral Echinomuricea sp. Molecules 2012, 17, 9443-9450. https://doi.org/10.3390/molecules17089443
Cheng C-H, Chung H-M, Hwang T-L, Lu M-C, Wen Z-H, Kuo Y-H, Wang W-H, Sung P-J. Echinoclerodane A: A New Bioactive Clerodane-Type Diterpenoid from a Gorgonian Coral Echinomuricea sp. Molecules. 2012; 17(8):9443-9450. https://doi.org/10.3390/molecules17089443
Chicago/Turabian StyleCheng, Ching-Hsiao, Hsu-Ming Chung, Tsong-Long Hwang, Mei-Chin Lu, Zhi-Hong Wen, Yueh-Hsiung Kuo, Wei-Hsien Wang, and Ping-Jyun Sung. 2012. "Echinoclerodane A: A New Bioactive Clerodane-Type Diterpenoid from a Gorgonian Coral Echinomuricea sp." Molecules 17, no. 8: 9443-9450. https://doi.org/10.3390/molecules17089443
APA StyleCheng, C.-H., Chung, H.-M., Hwang, T.-L., Lu, M.-C., Wen, Z.-H., Kuo, Y.-H., Wang, W.-H., & Sung, P.-J. (2012). Echinoclerodane A: A New Bioactive Clerodane-Type Diterpenoid from a Gorgonian Coral Echinomuricea sp. Molecules, 17(8), 9443-9450. https://doi.org/10.3390/molecules17089443






