A New Natural Lactone from Dimocarpus longan Lour. Seeds
Abstract
:1. Introduction
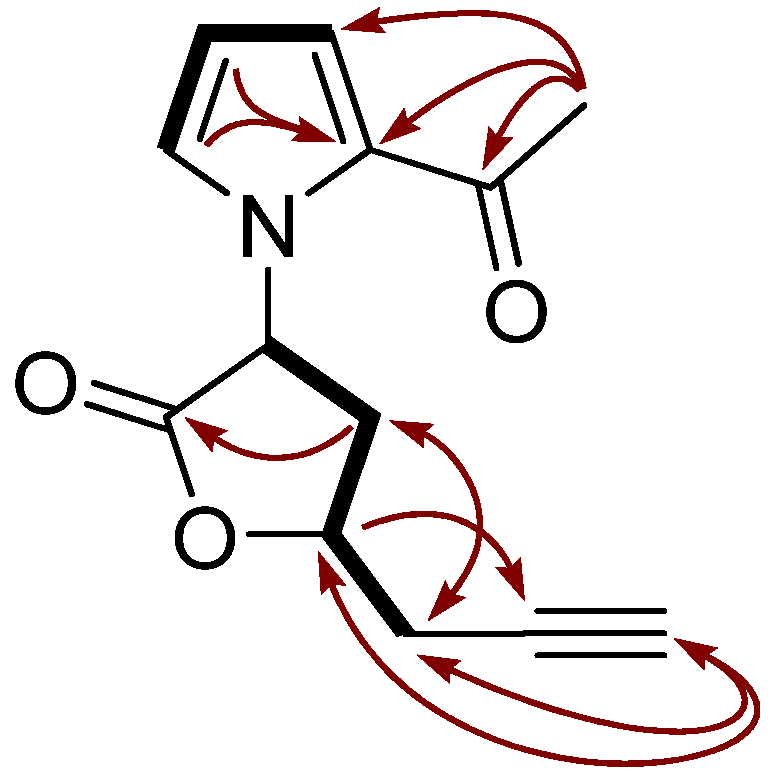
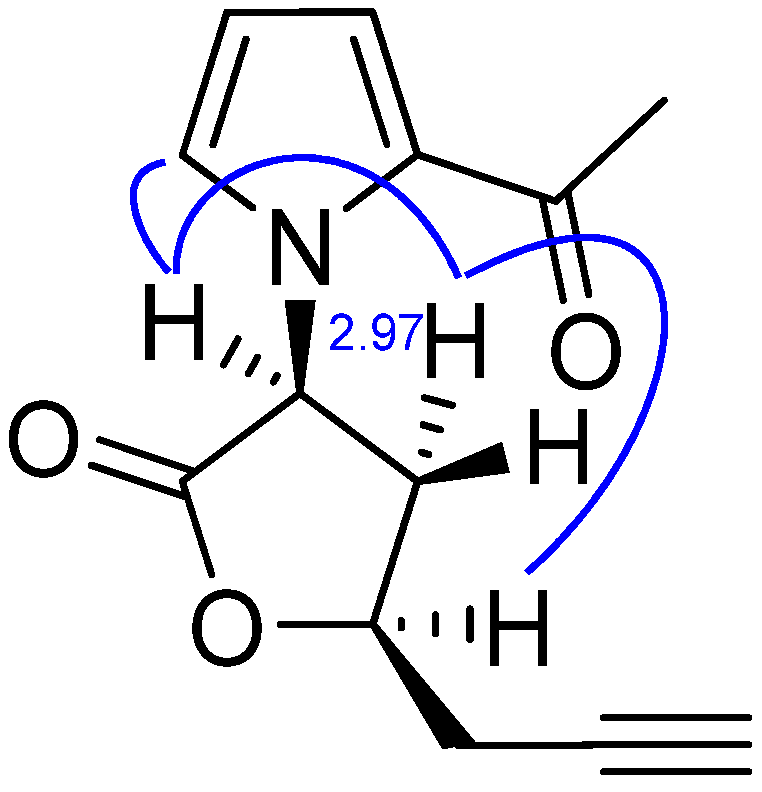
2. Results and Discussion
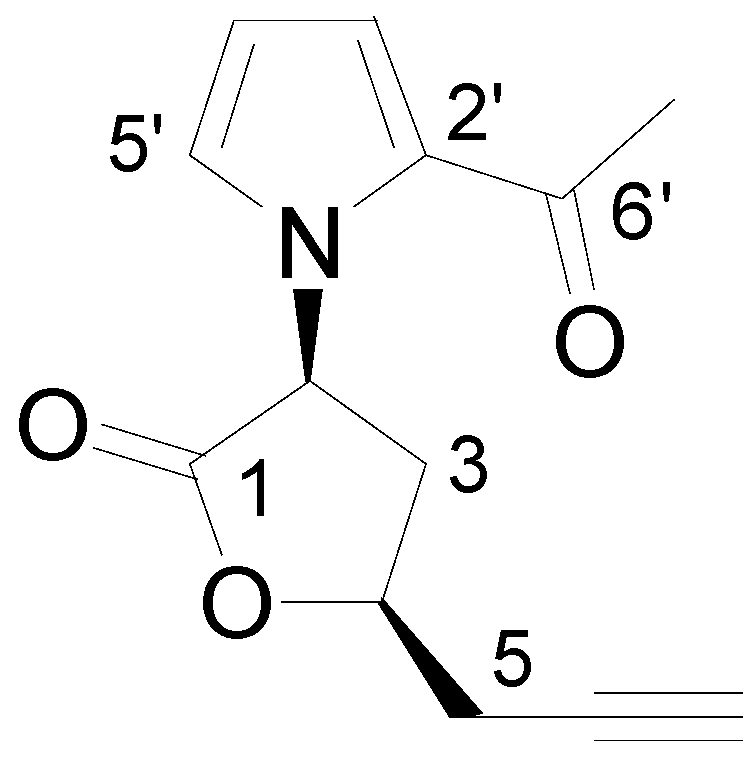
| Position | 1H | 13C | HMBC | NOESY |
|---|---|---|---|---|
| 1 | 171.8 s | |||
| 2 | 5.88 (br s) | 58.2 d | 3a, 3b, 4, 5' | |
| 3a | 2.97 (ddd, 12.4, 9.2, 6.1) | 35.5 t | 1, 2 | 2, 3b, 4 |
| 3b | 2.37 (ddd, 22.2, 12.4, 2.0) | 2, 4, 5 | 3a, 4, 5b, 5' | |
| 4 | 4.69(m) | 74.3 d | 6 | 2, 3a, 3b, 5a, 5b |
| 5a | 2.89 (ddd, 16.8, 5.3, 2.8) | 24.8 t | 3, 4, 6, 7 | 4 |
| 5b | 2.82 (ddd, 16.8, 7.1, 2.8) | 3, 4, 6, 7 | 3b, 4 | |
| 6 | 77.2 s | |||
| 7 | 2.07 (t, 2.6) | 71.6 d | 4, 5 | |
| 2' | 130.0 s | |||
| 3' | 7.07 (dd, 4.0, 1.6) | 121.4 d | 4', 5' | 4', 7' |
| 4' | 6.26 (dd, 4.0, 2.6) | 109.7 d | 2', 3' | 3', 5' |
| 5' | 6.97 (dd, 2.6, 1.6) | 129.7 d | 2', 3', 4' | 2, 3b, 4' |
| 6' | 188.7 s | |||
| 7' | 2.46 (s) | 27.0 q | 2', 3', 6' | 3' |
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
3.4. Spectral Data
4. Conclusions
Acknowledgments
References
- Jiang, Y.; Zhang, Z.; Joyce, D.C.; Ketsa, S. Postharvest biology and handling of longan fruit (Dimocarpus longan Lour.). Postharvest Biol. Technol. 2002, 26, 241–252. [Google Scholar] [CrossRef]
- Cai, C.; Tang, X.; Zhang, A.; Shu, Z.; Xiao, W.; Li, W. Importance and development of longan pulp as functional food and medication. Shi Pin Ke Xue 2002, 23, 328–330. [Google Scholar]
- Jiangsu New College of Medicine, Chinese Materia Medica Dictionary; Shanghai People’s Press: Shanghai, China, 1977; p. 637.
- Rangkadilok, N.; Sitthimonchai, S.; Worsuttayangkurn, L.; Mahidol, C.; Ruchirawat, M.; Satayavivad, J. Evaluation of free radical scavenging and antityrosinase activities of standardized longan fruit extract. Food Chem.Toxicol. 2007, 45, 328–336. [Google Scholar] [CrossRef]
- Zheng, S.Q.; Jiang, F.; Gao, H.Y.; Zheng, J.G. Preliminary observations on the antifatigue effects of longan (Dimocarpus longan Lour.) seed polysaccharides. Phytother. Res. 2010, 24, 622–624. [Google Scholar]
- Chung, Y.C.; Lin, C.C.; Chou, C.C.; Hsu, C.P. The effect of longan seed polyphenols on colorectal arcinoma cells. Eur. J. Clin. Invest. 2010, 40, 713–721. [Google Scholar] [CrossRef]
- Huang, R.Q.; Zou, X.Y.; Liu, X.M. Study on the hypoglycemic effect of longan seed extract. Nat. Prod. Res. Dev. 2006, 18, 991–992. [Google Scholar]
- Soong, Y.Y.; Barlow, P.J. Isolation and structure elucidation of phenolic compounds from longan (Dimocarpus longan Lour.) seed by high-performance liquid chromatography-electrospray ionization mass spectrometry. J. Chromatogr. A 2005, 1085, 270–277. [Google Scholar] [CrossRef]
- Sudjaroen, Y.; Hull, W.E.; Erben, G.; Würtele, G.; Changbumrung, S.; Ulrich, C.M.; Owen, R.W. Isolation and characterization of ellagitannins as the major polyphenolic components of Longan (Dimocarpus longan Lour.) seeds. Phytochemistry 2012, 77, 226–237. [Google Scholar]
- Zheng, G.; Xu, L.; Wu, P.; Xie, H.; Jiang, Y.; Chen, F.; Wei, X. Polyphenols from longan seeds and their radical scavenging activities. Food Chem. 2009, 116, 433–436. [Google Scholar] [CrossRef]
- Zheng, G.; Wei, X.; Xu, L.; Xie, H.; Wu, P. Chemical constituents from fruits of Dimocarpus longan. Zhong Cao Yao 2011, 42, 1053–1056. [Google Scholar]
- Sample Availability: Sample of the compound 3-(2-acetyl-1H-pyrrol-1-yl)-5-(prop-2-yn-1-yl)dihydrofuran-2(3H)-one is available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zheng, G.; Wei, X.; Xu, L.; Li, Z.; Liu, G.; Zhang, X. A New Natural Lactone from Dimocarpus longan Lour. Seeds. Molecules 2012, 17, 9421-9425. https://doi.org/10.3390/molecules17089421
Zheng G, Wei X, Xu L, Li Z, Liu G, Zhang X. A New Natural Lactone from Dimocarpus longan Lour. Seeds. Molecules. 2012; 17(8):9421-9425. https://doi.org/10.3390/molecules17089421
Chicago/Turabian StyleZheng, Gongming, Xiaoyi Wei, Liangxiong Xu, Zhongjun Li, Gangyong Liu, and Xiance Zhang. 2012. "A New Natural Lactone from Dimocarpus longan Lour. Seeds" Molecules 17, no. 8: 9421-9425. https://doi.org/10.3390/molecules17089421
APA StyleZheng, G., Wei, X., Xu, L., Li, Z., Liu, G., & Zhang, X. (2012). A New Natural Lactone from Dimocarpus longan Lour. Seeds. Molecules, 17(8), 9421-9425. https://doi.org/10.3390/molecules17089421




