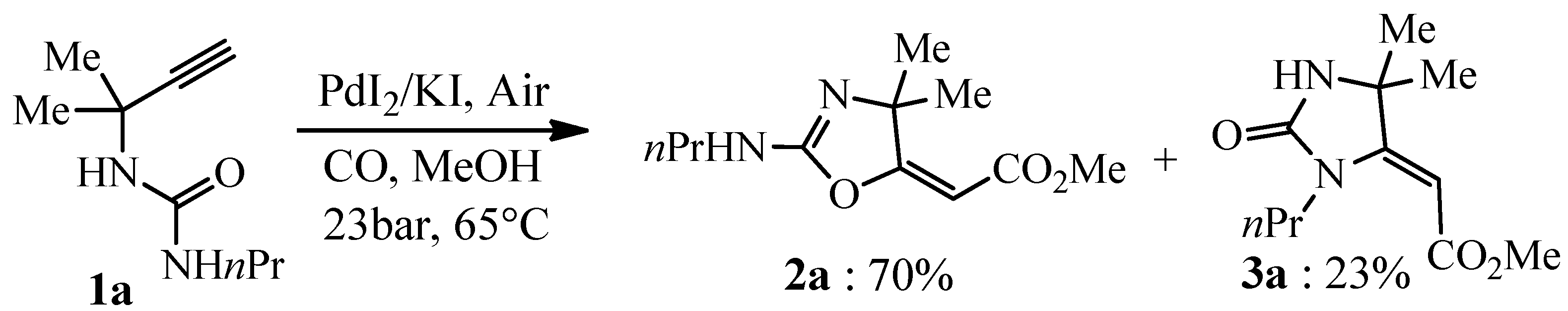
Cyclization-Carbonylation-Cyclization Coupling Reaction of Propargyl Ureas with Palladium(II)-Bisoxazoline Catalyst
Abstract
:1. Introduction
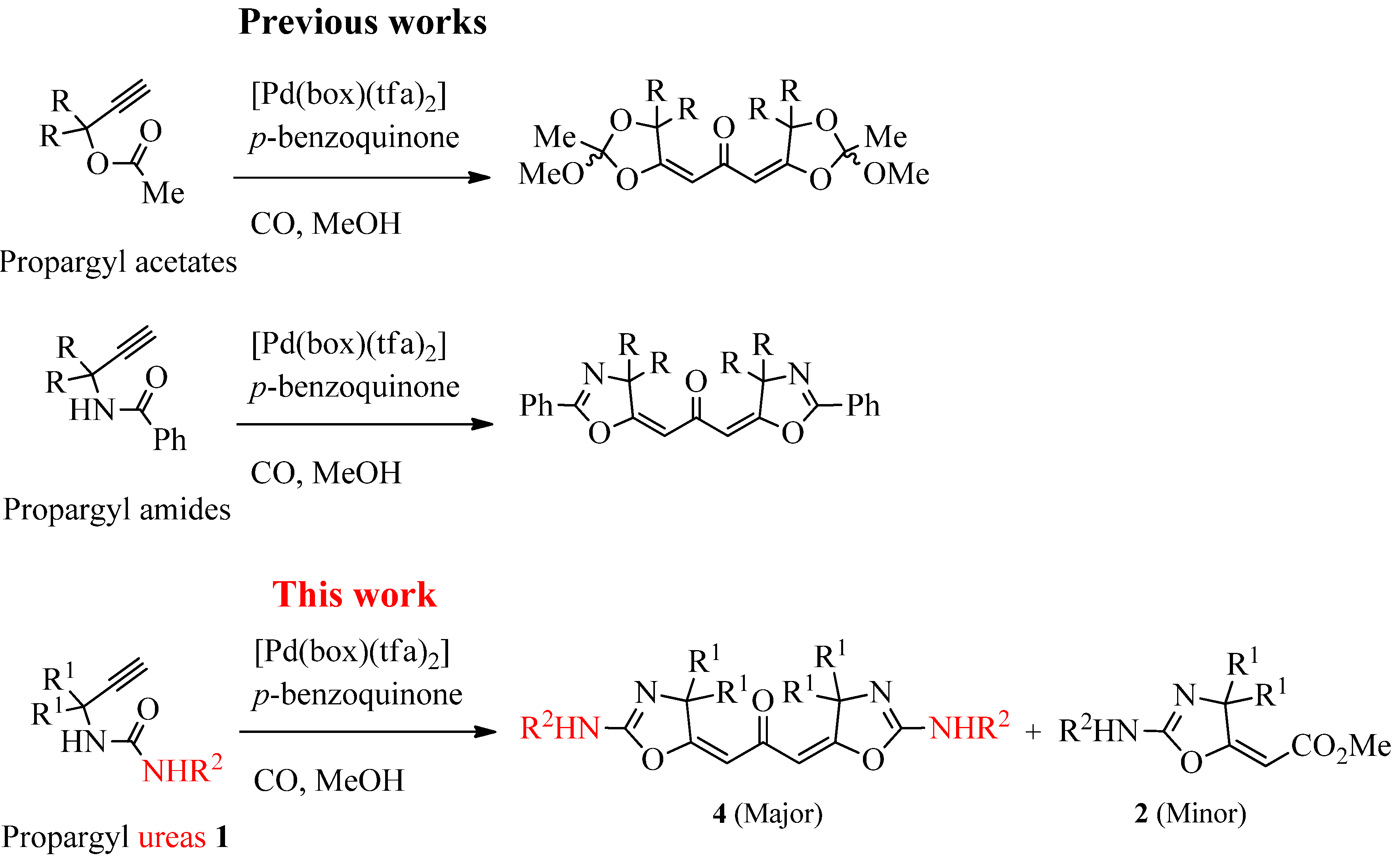
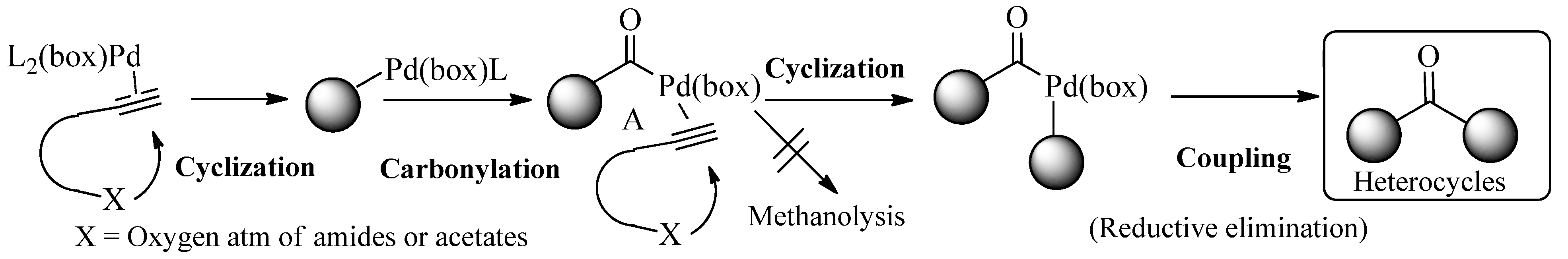
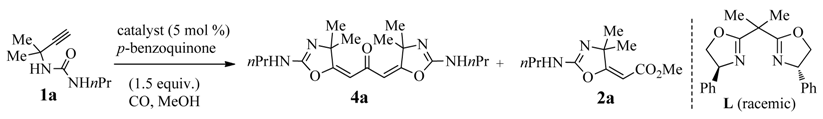
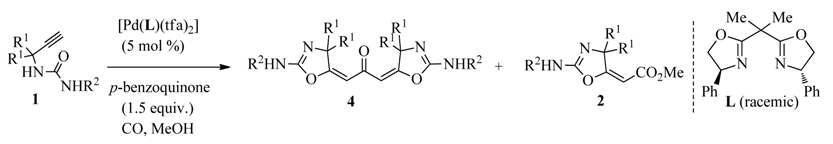
2. Results and Discussion
2.1. Optimization Studies
| Entry | Catalyst(5 mol %) | Conditions | Yield of4a (%) | Yield of2a (%) |
|---|---|---|---|---|
| 1 | (CH3CN)2PdCl2 | rt, 24 h | 4a: 46 | 2a: 36 |
| 2 | Pd(tfa)2 | −20 °C, 24 h | 4a: 24 | 2a: 20 |
| 3 | [Pd(bipy)Cl2] | rt, 24 h | 4a: 29 | 2a: 13 |
| 4 | (Ph3P)2PdCl2 | rt, 19 h | 4a: 16 | 2a: 61 |
| 5 | Pd(PPh3)4 | rt, 24 h | - | 2a: 44 |
| 6 | [Pd(L)(tfa)2] | rt, 24 h | 4a: 83 | 2a: 17 |
| 7 | [Pd(L)(tfa)2] | 50 °C, 3 h | 4a: 61 | 2a: 21 |
| 8 | [Pd(L)(tfa)2] | −20 °C, 24 h | 4a: 30 | 2a: 29 |
| 9 a | [Pd(L)(tfa)2] | rt, 24 h | N.R. | |
| 10 b | [Pd(L)(tfa)2] | rt, 24 h | N.R. | |
2.2. Substrate Scope and Limitations
| Entry | R1 | R2 | Conditions | Yield of4 (%) | Yield of2 (%) |
|---|---|---|---|---|---|
| 1 | Me | nPr | rt, 24 h | 4a: 83 | 2a: 17 |
| 2 | Me | Bn | rt, 18 h | 4b: 73 | 2b: 10 |
| 3 | Me | nBu | rt, 20 h | 4c: 61 | 2c: 25 |
| 4 | Me | Phenethyl | 7 °C, 48 h | 4d: 58 | 2d: 20 |
| 5 | Me | Cyclohexyl | rt, 24 h | 4e: 66 | 2e: 30 |
| 6 | -(CH2)5- | Phenethyl | rt, 24 h | 4f: 89 | 2f: 11 |
| 7 | -(CH2)5- | Cyclohexyl | rt, 24 h | 4g: 74 | 2g: 20 |
| 8 | -(CH2)5- | Ph | rt, 40 h | 4h: 24 | 2h: 23 |
| 9 a | -(CH2)5- | H | rt, 48 h | N.R. | |
| 10 | H | Phenethyl | rt, 24 h | N.R. | |
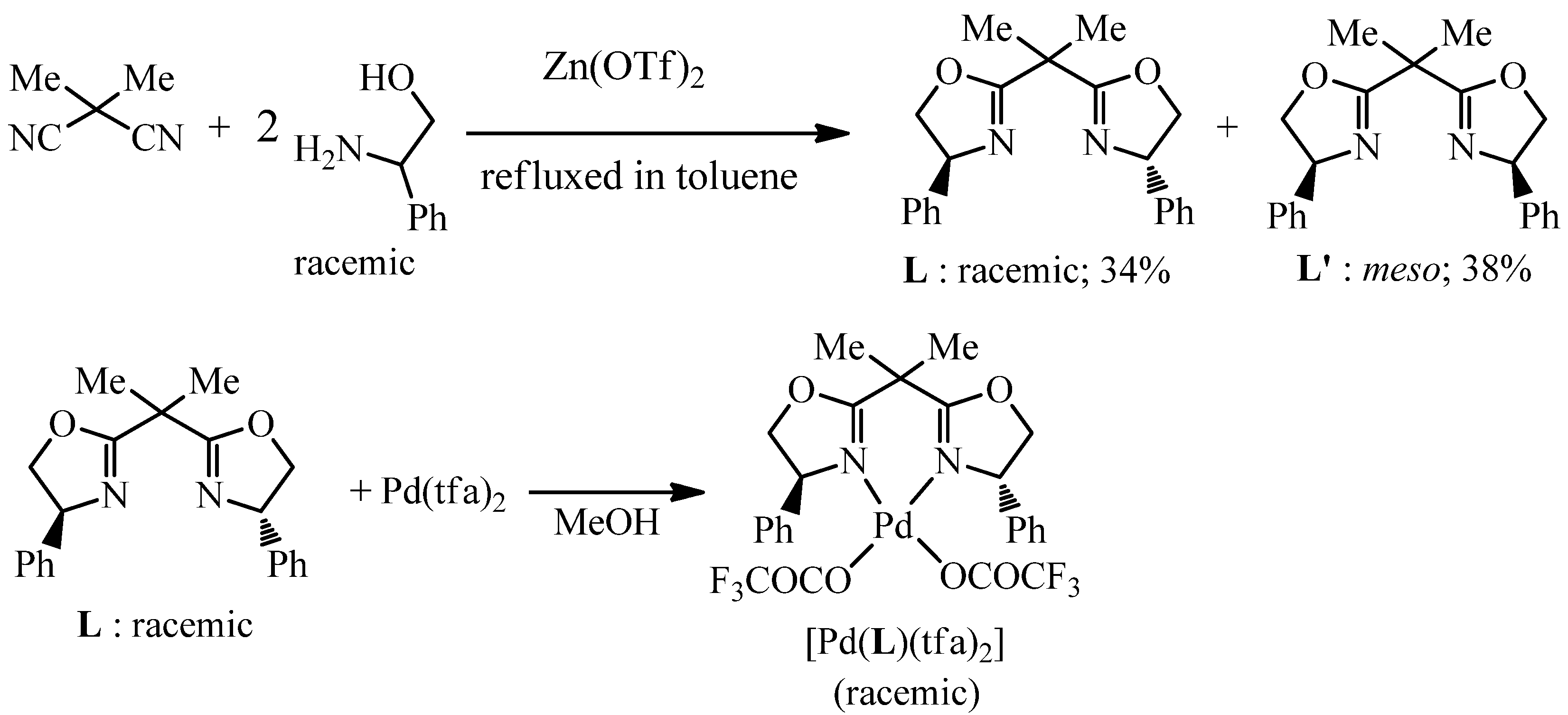
2.3. Preparation of [Pd(L)(tfa)2]
3. Experimental
3.1. General
3.2. Preparation of Substrates 1a–j
3.3. General Procedure for the CCC-Coupling Reaction of 1
3.3.1. Dimeric Ketone 4a
3.3.2. (2E)-Methyl 2-[4,4-dimethyl-2-(propylamino)-5(4H)-oxazolylidene]acetate (2a)
3.3.3. Dimeric Ketone 4b
3.3.4. (2E)-Methyl 2-[4,4-dimethyl-2-(phenylmethylamino)-5(4H)-oxazolylidene]acetate (2b)
3.3.5. Dimeric Ketone 4c
3.3.6. (2E)-Methyl 2-[4,4-dimethyl-2-(butylamino)-5(4H)-oxazolylidene]acetate (2c)
3.3.7. Dimeric Ketone 4d
3.3.8. (2E)-Methyl 2-[4,4-dimethyl-2-(phenylethylamino)-5(4H)-oxazolylidene]acetate (2d)
3.3.9. Dimeric Ketone 4e
3.3.10. (2E)-Methyl 2-[4,4-dimethyl-2-(cyclohexylethylamino)-5(4H)-oxazolylidene]acetate (2e)
3.3.11. Dimeric Ketone 4f
3.3.12. Monomeric Ester 2f
3.3.13. Dimeric Ketone 4g
3.3.14. Monomeric Estere 2g
3.3.15. Dimeric Ketone 4h
3.3.16. Dimeric Ketone 2h
3.4. Preparation of Ligands and Racemic-[Pd(L)(tfa)2]
3.4.1. Preparation of L and L′
3.4.2. Preparation of [Pd(L)(tfa)2]
4. Conclusions
Supplementary Materials
Acknowledgments
References
- Chaudhry, P.; Schoenen, F.; Neuenswander, B.; Lushington, G.H.; Aubé, J. One-Step Synthesis of Oxazoline and Dihydrooxazine Libraries. J. Comb. Chem. 2007, 9, 473–476. [Google Scholar] [CrossRef]
- Onishi, H.R.; Pelak, B.A.; Lynn, S.; Silver, L.L.; Kahan, F.M.; Chen, M.-H.; Patchett, A.A.; Galloway, S.M.; Hyland, S.A.; Anderson, M.S.; et al. Antibacterial Agents That Inhibit Lipid A Biosynthesis. Science 1996, 274, 980–982. [Google Scholar]
- Prinsep, M.R.; Moore, R.E.; Levine, I.A.; Patterson, G.M.L. Westiellamide, A Bistratamide-related Cyclic Peptide From The Blue-Green Alga Westiellopsis Prolifica. J. Nat. Prod. 1992, 55, 140–142. [Google Scholar] [CrossRef]
- Bruban, V.; Feldman, J.; Greney, H.; Dontenwill, M.; Schann, S.; Jarry, C.; Payard, M.; Boutin, J.; Scalbert, E.; Pfeiffer, B.; et al. Respective contributions of α-adrenergic and non-adrenergic mechanisms in the hypotensive effect of imidazoline-like drugs. Br. J. Pharmacol. 2001, 133, 261–266. [Google Scholar] [CrossRef]
- Bosc, J.J.; Forfar, I.; Jarry, C.; Laguerre, M.; Carpy, A. 2-Amino-2-oxazolines, VII: Influence of Structural Parameters on The Antidepressant Activity of 5-(1-Aryl-4-piperazino)methyl-2-amino-2-oxazolines. Arch. Pharm. 1994, 327, 187–192. [Google Scholar] [CrossRef]
- Kay, J.M.; Smith, P.; Heath, D. Aminorex and the Pulmonary Circulation. Thorax 1971, 26, 262–270. [Google Scholar] [CrossRef]
- Ueda, S.; Terauchi, H.; Yano, A.; Ido, M.; Matsumoto, M.; Kawasaki, M. 4,5-Disubstituted-1,3-oxazolidin-2-imine Derivatives: A New Class of Orally Bioavailable Nitric Oxide Synthase Inhibitor. Bioorg. Med. Chem. 2004, 14, 313–316. [Google Scholar] [CrossRef]
- Gally, G.; Norcross, R.; Polara, A. Preparation of amino oxazoline derivatives for use as TAAR1 ligands. U.S. Pat. Appl. Publ. 2011. US 20110112080 A1 20110512. [Google Scholar]
- Neumann, H.; Brennführer, A.; Beller, M. A General Synthesis of Diarylketones by Means of a Three-Component Cross-Coupling of Aryl and Heteroaryl Bromides, Carbon Monoxide, and Boronic acids. Chem. Eur. J. 2008, 14, 3645–3652, and references cited therein. [Google Scholar] [CrossRef]
- Lo Fiego, M.J.; Silbestri, G.F.; Chopa, A.B.; Lockhart, M.T. Selective Synthetic Routes to Sterically Hindered Unsymmetrical Diaryl Ketones via Arylstannanes. J. Org. Chem. 2011, 76, 1707–1714, and references cited therein. [Google Scholar] [CrossRef]
- Chao, E.Y.H.; Collins, J.L.; Gaillard, S.; Miller, A. B.; Wang, L.; Orband-Miller, L.A.; Nolte, R.T.; McDonnell, D.P.; Willson, T.M.; Zuercher, W.J. Structure-guided Synthesis of Tamoxifen Analogs with Improved Selectivity for The Orphan ERRγ. Bioorg. Med. Chem. Lett. 2006, 16, 821–824. [Google Scholar]
- Vlaar, T.; Ruijter, E.; Orru, R.V.A. Recent Advances in Palladium-Catalyzed Cascade Cyclizations. Adv. Synth. Catal. 2011, 353, 809–841. [Google Scholar] [CrossRef]
- Brennführer, A.; Neumann, H.; Beller, M. Palladium-catalyzed Carbonylation Reactions of Alkenes and Alkynes. ChemCatChem 2009, 1, 28–41. [Google Scholar] [CrossRef]
- Yasuhara, S.; Sasa, M.; Kusakabe, T.; Takayama, H.; Kimura, M.; Mochida, T.; Kato, K. Cyclization–Carbonylation–Cyclization Coupling Reactions of Propargyl Acetates and Amides with Palladium(II)–Bisoxazoline Catalysts. Angew. Chem. Int. Ed. 2011, 50, 3912–3915. [Google Scholar]
- Kusakabe, T.; Kawai, Y.; Shen, R.; Mochida, T.; Kato, K. Cyclization–carbonylation–cyclization coupling reactions of γ-propynyl-1,3-diketones with palladium(II)-bisoxazoline catalysts. Org. Biomol. Chem. 2012, 10, 3192–3194. [Google Scholar] [CrossRef]
- Kusakabe, T.; Sekiyama, E.; Motodate, S.; Kato, S.; Mochida, T.; Kato, K. Cyclization–Carbonylation–Cyclization Coupling Reactions of N-Propargylanilines and o-Alkynylphenols with Palladium(II)–Bisoxazoline Catalysts. Synthesis 2012, 44, 1825–1832. [Google Scholar] [CrossRef]
- Kato, K.; Teraguchi, R.; Yamamura, S.; Mochida, T.; Akita, H.; Peganova, T.A.; Vologdin, N.V.; Gusev, O.V. Ligand-controlled intramolecular carbonylative cyclization of 1,1-diethynyl acetates: New entry to the functionalized 4-cyclopentene-1,3-diones. Synlett 2007, 638–642. [Google Scholar]
- Kato, K.; Teraguchi, R.; Motodate, S.; Uchida, A.; Mochida, T.; Peganova, T.A.; Vologdin, N.V.; Akita, H. meso-Phbox-Pd(II) Catalyzed Tandem Carbonylative Cyclization of 1-Ethynyl-1-propargyl Acetate. Chem. Commun. 2008, 3687–3689. [Google Scholar]
- Kato, K.; Motodate, S.; Mochida, T.; Kobayashi, T.; Akita, H. Intermolecular Methoxy-carbonylation of Terminal Alkynes Catalyzed by Palladium(II) Bis(oxazoline) Complexes. Angew. Chem. Int. Ed. 2009, 48, 3326–3328. [Google Scholar] [CrossRef]
- Bacchi, A.; Chiusoli, G.P.; Costa, M.; Sani, C.; Gabriele, B.; Salerno, G. Nitrogen Heterocycles from palladium-catalysed oxidative cyclization-alkoxycarbonylation of acetylenic ureas. J. Organomet. Chem. 1998, 562, 35–43. [Google Scholar]
- Costa, M.; Della Cà, N.; Gabriele, B.; Massera, C.; Salerno, G.; Soliani, M. Synthesis of 4H-3,1-Benzoxazines, Quinazolin-2-ones, and Quinoline-4-ones by Palladium-Catalyzed Oxidative Carbonylation of 2-Ethynylaniline Derivatives. J. Org. Chem. 2004, 69, 2469–2477. [Google Scholar]
- See the electronic supporting information.
- Sammes, P.G.; Weller, D.J. Steric Promotion of Ring Formation. Synthesis 1995, 1205–1222. [Google Scholar]
- Cornejo, A.; Fraile, J.M.; García, J.I.; Gil, M.J.; Martínez-Merino, V.; Mayoral, J.A.; Pires, E.; Villalba, I. An Efficient and General One-Pot Method for the Synthesis of Chiral Bis(oxazoline) and Pyridine Bis(oxazoline) Ligands. Synlett 2005, 2321–2324. [Google Scholar]
- García, J.I.; Mayoral, J.A.; Pires, E.; Villalba, I. Synthesis of non-symmetric bisoxazoline compounds. An easy way to reach tailored chiral ligands. Tetrahedron: Asymmetry 2006, 17, 2270–2275. [Google Scholar] [CrossRef]
- Foltz, C.; Stecker, B.; Marconi, G.; Bellemin-Laponnaz, S.; Wadepohl, H.; Gade, L.H. Stereochemical Consequences of Threefold Symmetry in Asymmetric Catalysis: Distorting C3 Chiral 1,1,1-Tris(oxazolinyl)ethanes (“Trisox”) in CuII Lewis Acid Catalysts. Chem.Eur. J. 2007, 13, 9912–9923. [Google Scholar]
- Michael, F.E.; Sibbald, P.A.; Cochran, B.M. Palladium-catalyzed intramolecular chloroamination of alkenes. Org. Lett. 2008, 10, 793–796. [Google Scholar]
- Kato, K.; Matsuba, C.; Kusakabe, T.; Takayama, H.; Yamamura, S.; Mochida, T.; Akita, H.; Peganova, T.A.; Vologdin, N.V.; Gusev, O.V. 2,2′-Isopropylidenebis[(4S,5R)-4,5-di(2-naphthyl)-2-oxazoline] ligand for asymmetric cyclization-carbonylation of meso-2-alkyl-2-propargylcyclohexane-1,3-diols. Tetrahedron 2006, 62, 9988–9999. [Google Scholar]
- Sample Availability: Not available.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kusakabe, T.; Kawaguchi, K.; Kawamura, M.; Niimura, N.; Shen, R.; Takayama, H.; Kato, K. Cyclization-Carbonylation-Cyclization Coupling Reaction of Propargyl Ureas with Palladium(II)-Bisoxazoline Catalyst. Molecules 2012, 17, 9220-9230. https://doi.org/10.3390/molecules17089220
Kusakabe T, Kawaguchi K, Kawamura M, Niimura N, Shen R, Takayama H, Kato K. Cyclization-Carbonylation-Cyclization Coupling Reaction of Propargyl Ureas with Palladium(II)-Bisoxazoline Catalyst. Molecules. 2012; 17(8):9220-9230. https://doi.org/10.3390/molecules17089220
Chicago/Turabian StyleKusakabe, Taichi, Koujiro Kawaguchi, Miya Kawamura, Naohiko Niimura, Rong Shen, Hiroyuki Takayama, and Keisuke Kato. 2012. "Cyclization-Carbonylation-Cyclization Coupling Reaction of Propargyl Ureas with Palladium(II)-Bisoxazoline Catalyst" Molecules 17, no. 8: 9220-9230. https://doi.org/10.3390/molecules17089220
APA StyleKusakabe, T., Kawaguchi, K., Kawamura, M., Niimura, N., Shen, R., Takayama, H., & Kato, K. (2012). Cyclization-Carbonylation-Cyclization Coupling Reaction of Propargyl Ureas with Palladium(II)-Bisoxazoline Catalyst. Molecules, 17(8), 9220-9230. https://doi.org/10.3390/molecules17089220