Two New Compounds Isolated from Liriope muscari
Abstract
:1. Introduction
2. Results and Discussion
2.1. Structure Analysis and Characterization of Compounds 1–3
2.2. In Vitro Antioxidant Activity
3. Experimental
3.1. General
3.2. Plant Material
3.3. Compound Isolation
4. Conclusions
Acknowledgments
Conflict of Interest
References
- Yu, B.Y.; Xu, G.J.; Jin, R.L.; Xu, L.S. Drug resources and identification of commercial drugs on Radix Ophiopogonis (in Chinese with English abstract). J. Chin. Pharm. Univ. 1991, 22, 150–153. [Google Scholar]
- Tian, Y.Q.; Kou, J.P.; Li, L.Z.; Yu, B.Y. Anti-inflammatory effects of aqueous extract from Radix Liriope muscari and its major active fraction and component. Chin. J. Nat. Med. 2011, 9, 222–226. [Google Scholar]
- Wang, Z.J. Antioxidant activities of extracts from Ophiopogon japonicus (in Chinese with English abstract). Food Ferment. Ind. 2007, 33, 57–60. [Google Scholar]
- Yu, B.Y.; Hirai, Y.; Shoji, J.Z.; Xu, G.J. Comparative studies on the constituents of ophiopogonis tuber and its congeners. VI. Studies on the constituents of the subterranean part of Liriope spicata var. prolifera and L. muscari. Chem. Pharm. Bull. 1990, 38, 1931–1935. [Google Scholar] [CrossRef]
- Cheng, Z.H.; Wu, T.; Yu, B.Y.; Xu, L.S. Studies on Chemical constituents of Liriope muscari (in Chinese). Zhong Cao Yao 2005, 36, 823–826. [Google Scholar]
- Cheng, Z.H.; Wu, T.; Guo, Y.L.; Yu, B.Y.; Xu, L.S. Two new steroidal glycosides from Liriope muscari. Chin. Chem. Lett. 2006, 17, 31–34. [Google Scholar]
- Yu, B.Y.; Xu, G.J. Studies on resource utilization of Chinese drug Dwarf Lilyturf (Ophiopogon japonicus) (in Chinese with English abstract). Zhong Cao Yao 1995, 26, 205–210. [Google Scholar]
- Cheng, Z.H.; Tao, W.S. cis-Eudesmane Sesquiterpene Glycosides from Liriope muscari and Ophiopogon japonicus. J. Nat. Prod. 2004, 67, 1761–1763. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.M.; Wang, G.L.; Bai, C.Q. A new eudesmane sesquiterpene glucoside from Liriope muscari fibrous roots. Molecules 2011, 16, 9017–9024. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Liu, Z.H.; Li, L. A new eudesmane sesquiterpene glycosides from Liriope muscari. J. Asian Nat. Prod. Res. 2012, 14, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.F.; Chen, L.L.; Qi, J. Two new benzofuran derivatives with anti-inflammatory activity from Liriope spicata var. prolifera. Fitoterapia 2011, 82, 190–192. [Google Scholar] [CrossRef] [PubMed]
- Li, W.J.; Cheng, X.L.; Liu, J. Phenolic compounds and antioxidant activities of Liriope muscari. Molecules 2012, 17, 1797–1808. [Google Scholar] [CrossRef] [PubMed]
- Chang, W.L.; Lee, S.S. Norneolignan and phenols from Curculigo capitulata. Phytochemistry 1998, 7, 2133–2136. [Google Scholar] [CrossRef]
- Fang, J.M.; Hong, B.C.; Liao, L.F. Dichotomous regiochemistry of aldehyde and ketone in the reaction with dithio-substituted crotyllithium. J. Org. Chem. 1987, 52, 855–861. [Google Scholar] [CrossRef]
- Beecham, A.F. Optical activity and lactone ring configurations. Tetrahedron Lett. 1968, 9, 3591–3594. [Google Scholar] [CrossRef]
- Yi, B.; Hu, L.; Mei, W.; Zhou, K.; Wang, H.; Luo, Y.; Wei, X.; Dai, H. Antioxidant phenolic compounds of cassava (Manihot esculenta) from Hainan. Molecules 2011, 16, 10157–10167. [Google Scholar] [CrossRef] [PubMed]
- Marcos, M.; Jiménez, C.; Vlllaverde, M.C. Lignans and other constituents from south and central American Zanthoxylum Species. Planta Med. 1990, 56, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Tsukamoto, H.; Hisada, S.; Nishibe, S. Lignans from bark of Fraxinus mandshurica var. japonica and F. japonica. Chem. Pharm. Bull. 1984, 32, 4482–4489. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the crude extracts and pure compounds are available from the authors. |
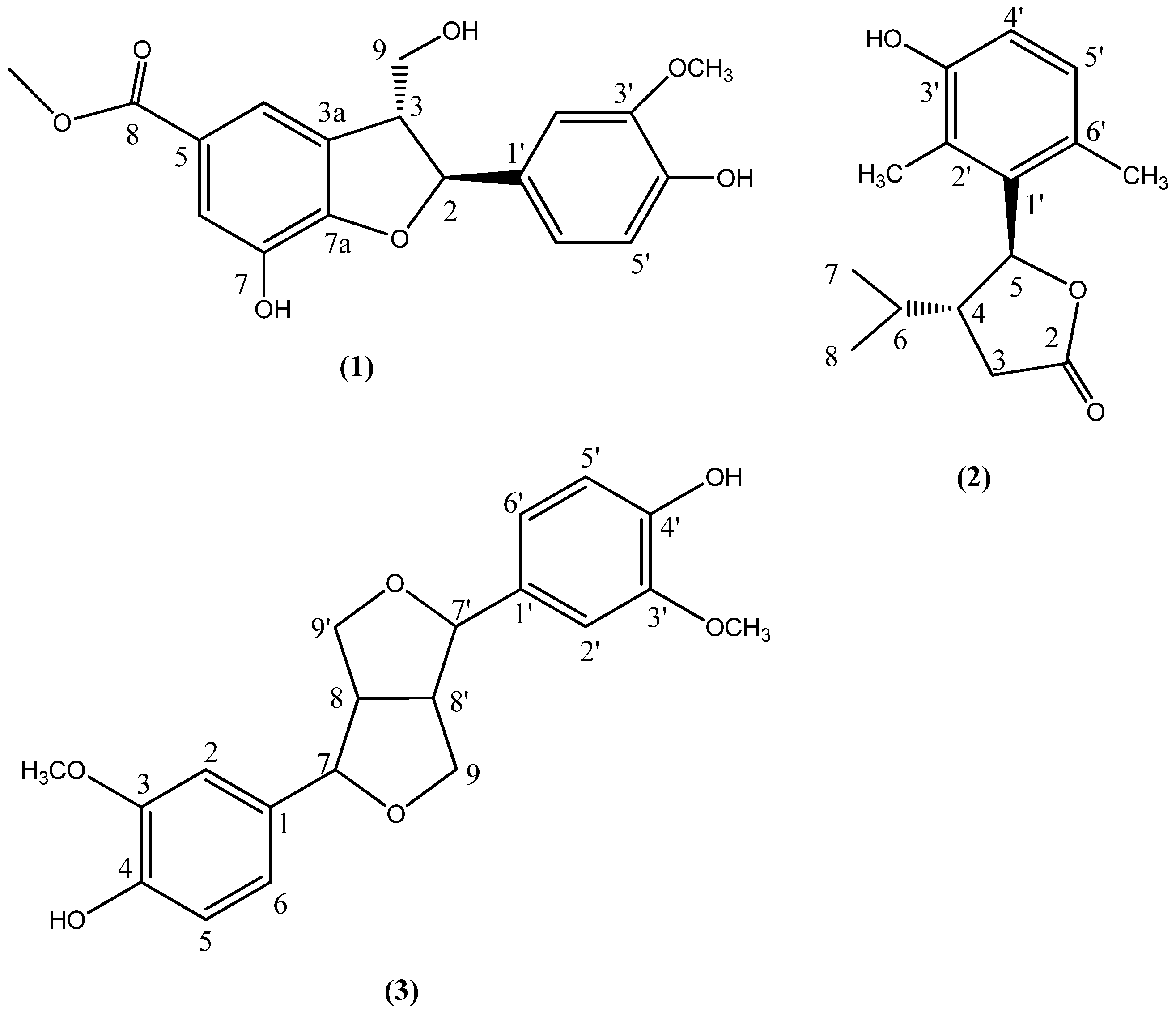
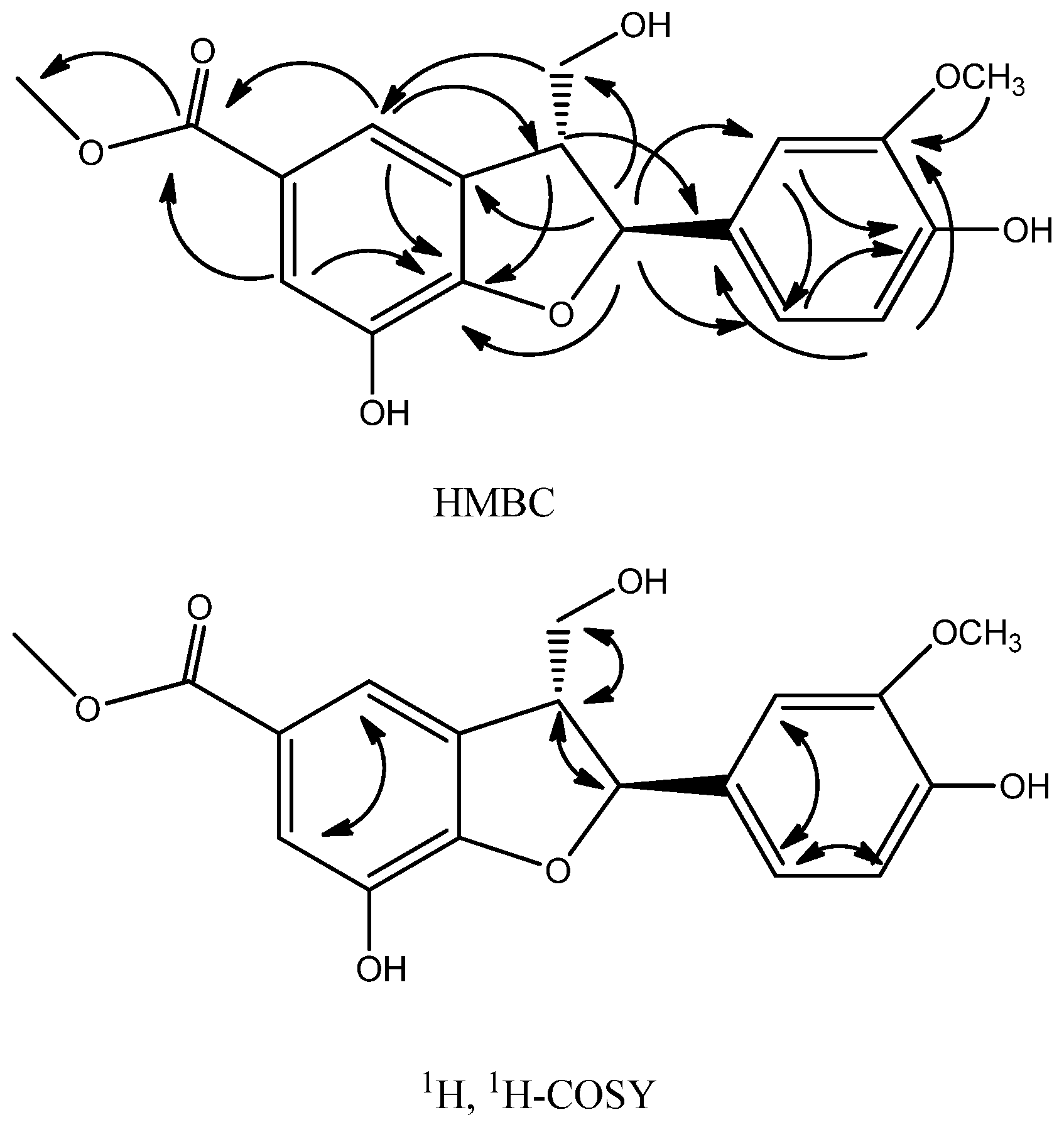
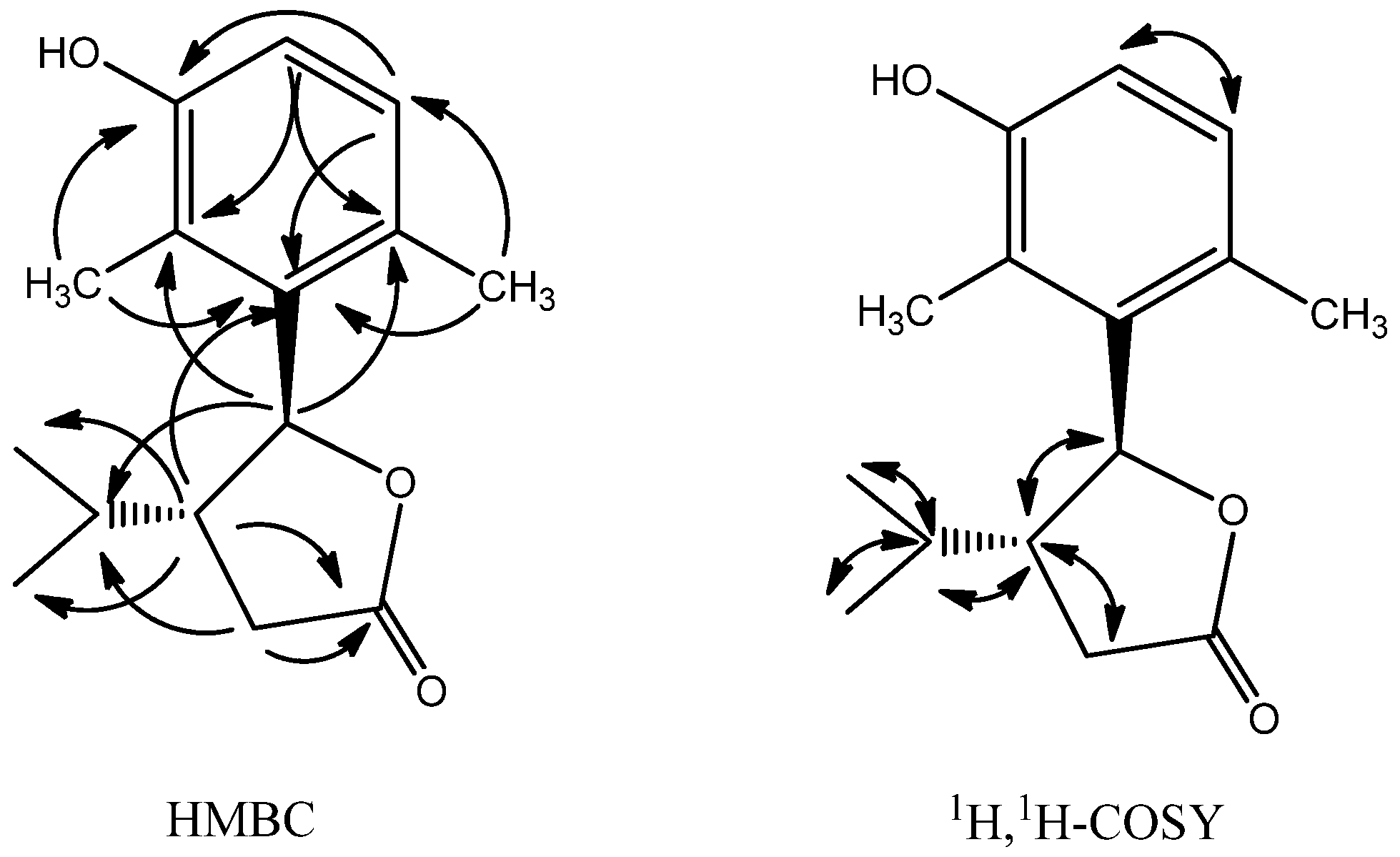
| Position | δC | δH |
|---|---|---|
| 2 | 88.2 | 5.52 (1H, d, 6.6 Hz) |
| 3 | 53.0 | 3.51 (1H, q, 6.6, 6.0 Hz) |
| 3a | 130.5 | |
| 4 | 117.9 | 7.40 (1H, d, 1.8 Hz) |
| 5 | 122.8 | |
| 6 | 117.6 | 7.33 (1H, d, 1.8 Hz) |
| 7 | 141.4 | |
| 7a | 151.7 | |
| 8 | 166.6 | |
| 9 | 63.1 | 3.68 (2H, dd, 6.0, 1.8 Hz) |
| 1′ | 132.2 | |
| 2′ | 110.9 | 6.93 (1H, d, 1.8 Hz) |
| 3′ | 148.1 | |
| 4′ | 147.0 | |
| 5′ | 115.8 | 6.75 (1H, d, 7.8 Hz) |
| 6′ | 119.3 | 6.78 (1H, dd, 7.8, 1.8 Hz) |
| 8-OCH3 | 52.2 | 3.77 (3H, s) |
| 3′-OCH3 | 56.1 | 3.74 (3H, s) |
| Position | δC | δH |
|---|---|---|
| 2 | 176.5 | |
| 3 | 32.1 | 2.70 (1H, m), 2.52 (1H, m) |
| 4 | 47.8 | 2.75 (1H, m) |
| 5 | 82.5 | 5.62 (1H, d, 9.0 Hz) |
| 6 | 29.3 | 1.75 (1H, m) |
| 7 | 18.6 | 0.98 (3H, d, 7.0 Hz) |
| 8 | 21.6 | 0.79 (3H, d, 6.5 Hz) |
| 1′ | 135.2 | |
| 2′ | 123.4 | |
| 3′ | 152.6 | |
| 4′ | 115.2 | 6.68 (1H, d, 8.5 Hz) |
| 5′ | 129.7 | 6.88 (1H, d, 8.5 Hz) |
| 6′ | 129 | |
| 2′-CH3 | 12.4 | 2.26 (3H, s) |
| 6′-CH3 | 20.5 | 2.32 (3H, s) |
| Compound | IC50-DPPH (µM) | IC50-ABTS (µM) |
|---|---|---|
| Compound 1 | 111.8 ± 9.0 | 18.5 ± 1.8 |
| Compound 2 | -- | 46.4 ± 3.7 |
| Compound 3 | 43.8 ± 3.7 | 23.1 ± 1.5 |
| VC | 17.3 ± 1.3 | 52.9 ± 4.2 |
| BHT | 188 ± 15.2 | 25 ± 2.4 |
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Li, W.-J.; Zhang, Z.-H.; Cheng, X.-L.; Liu, J.; He, Y.; Zhou, C.; Guo, Y.; Lin, R.-C.; Wang, G.-L. Two New Compounds Isolated from Liriope muscari. Molecules 2012, 17, 8773-8781. https://doi.org/10.3390/molecules17088773
Li W-J, Zhang Z-H, Cheng X-L, Liu J, He Y, Zhou C, Guo Y, Lin R-C, Wang G-L. Two New Compounds Isolated from Liriope muscari. Molecules. 2012; 17(8):8773-8781. https://doi.org/10.3390/molecules17088773
Chicago/Turabian StyleLi, Wen-Jie, Zhi-Hao Zhang, Xian-Long Cheng, Jing Liu, Yi He, Chao Zhou, Ying Guo, Rui-Chao Lin, and Gang-Li Wang. 2012. "Two New Compounds Isolated from Liriope muscari" Molecules 17, no. 8: 8773-8781. https://doi.org/10.3390/molecules17088773
APA StyleLi, W.-J., Zhang, Z.-H., Cheng, X.-L., Liu, J., He, Y., Zhou, C., Guo, Y., Lin, R.-C., & Wang, G.-L. (2012). Two New Compounds Isolated from Liriope muscari. Molecules, 17(8), 8773-8781. https://doi.org/10.3390/molecules17088773




