Synthesis, Tautomeric Structure and Antimicrobial Activity of 3-Arylhydrazono-4-phenyl-[1,2,4]-triazepino[2,3-a]quinazoline-2,7(1H)-diones
Abstract
:1. Introduction
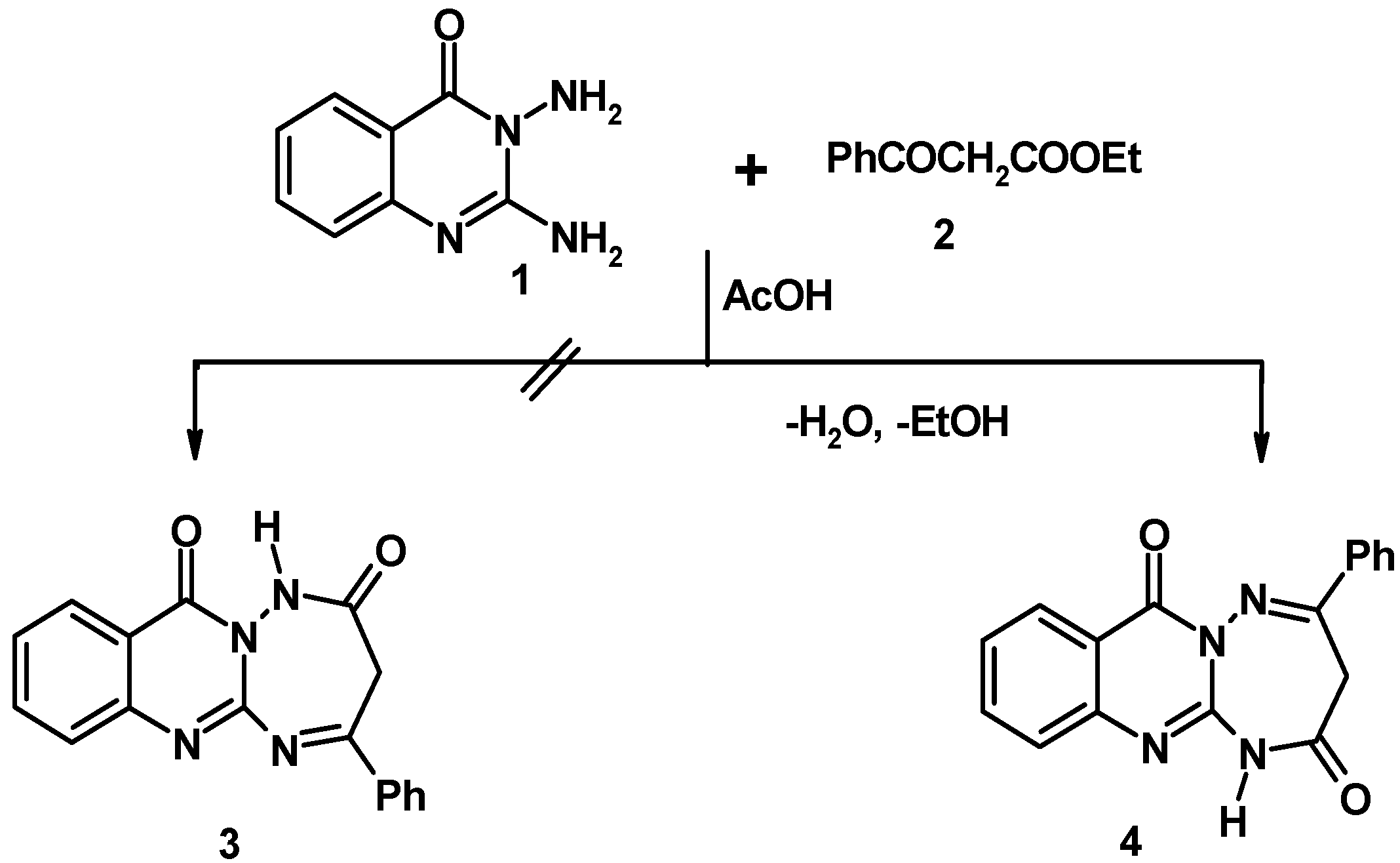
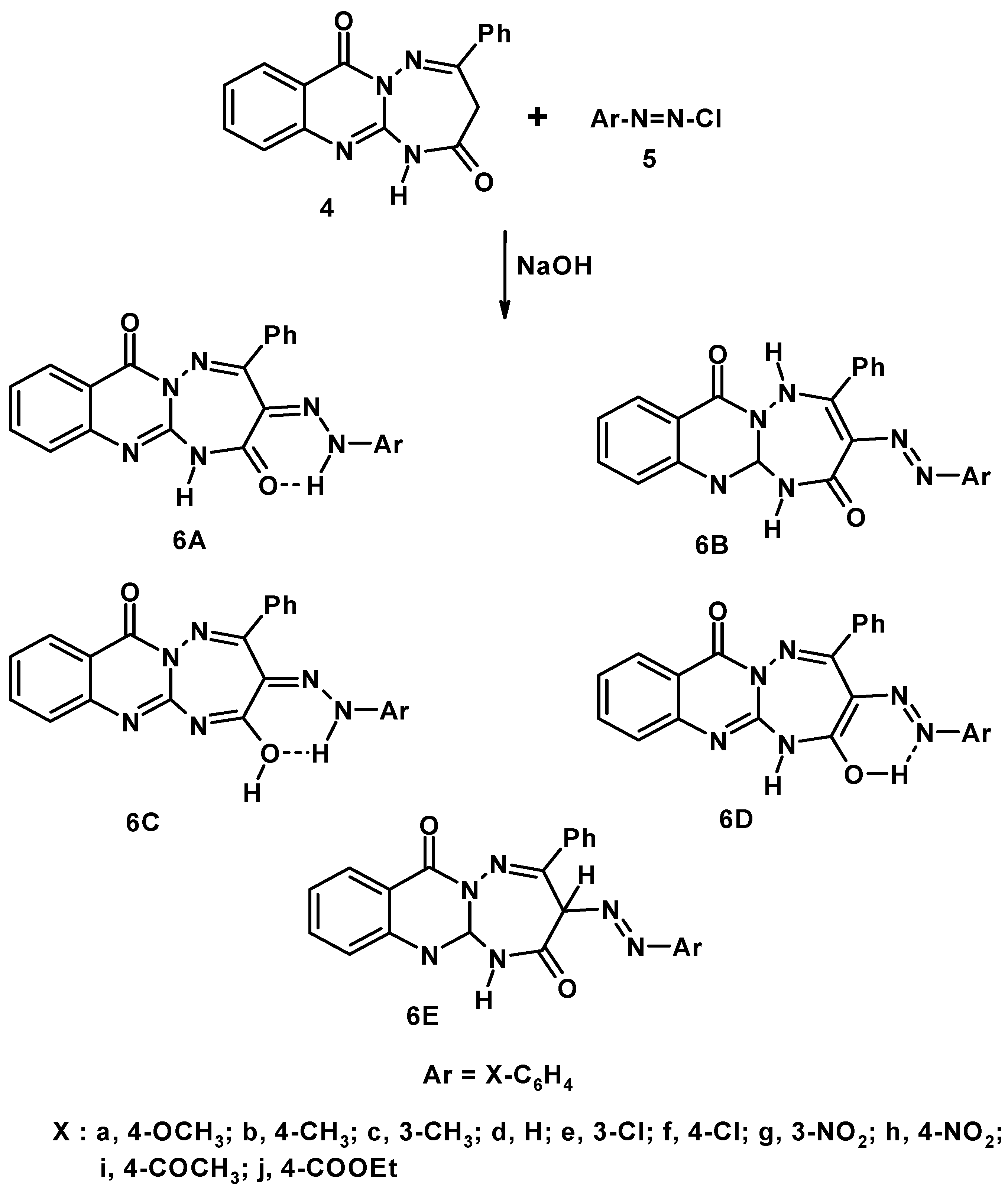

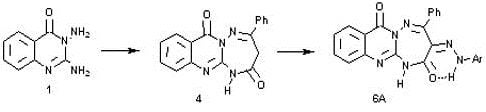
2. Results and Discussion
| Compd. No. | λmax (log ε) |
|---|---|
| 6a | 385 (3.72), 321 (4.22), 257 (4.63) |
| 6b | 381 (4.52), 260 (4.57), 231 (4.78) |
| 6c | 375 (4.02), 282 (4.82), 247 (4.81) |
| 6d a) | 370 (4.45), 248 (4.63) |
| 6e | 372 (4.25), 286 (4.26), 231 (4.55) |
| 6f | 372 (4.10), 328 (4.39), 233 (5.13) |
| 6g | 346 (3.62), 267 (4.78), 232 (5.01) |
| 6h | 383 (4.62), 260 (4.54), 234 (4.80) |
| 6i | 372 (3.94), 260 (3.68), 235 (4.01) |
| 6j | 381 (4.52), 260 (4.57), 231 (4.78) |
2.1. Anti-Microbial Activity
| Microorganism/IZD (mm/mg sample) * | ||||
|---|---|---|---|---|
| Cpd. no. | EC(G−) | SA (G+) | AF | CA |
| Control (DMSO) | 0.0 | 0.0 | 0.0 | 0.0 |
| 4 | 00 | 9 | 00 | 0.0 |
| 6a | 12 | 11 | 0.0 | 0.0 |
| 6b | 00 | 00 | 0.0 | 0.0 |
| 6c | 00 | 00 | 00 | 00 |
| 6d | 00 | 00 | 00 | 00 |
| 6e | 13 | 12 | 00 | 00 |
| 6f | 00 | 00 | 00 | 00 |
| 6g | 9 | 00 | 00 | 00 |
| 6h | 11 | 12 | 00 | 00 |
| 6i | 11 | 11 | 00 | 00 |
| 6j | 12 | 12 | 00 | 00 |
| Te ** | 31 | 29 | -- | -- |
| Am ** | -- | -- | 17 | 20 |
3. Experimental Section
3.1. General
3.2. General Procedure for Synthesis of 3-Arylhydrazono-4-phenyl-[1,2,4]triazepino[2,3-a]quinazoline-2,7(1H)-diones (6a–j).
3.2. Agar Diffusion Well Method to Determine the Antimicrobial Activity
4. Conclusions
References
- Maillard-Boyer, M.; Castera-Ducros, C.; Verhaeghe, P.; Sifredi, F.; Rathelot, P.; Vanelle, P. Access to original vinylic chlorides in the quinazoline series via a monoelectronic transfer reaction approach. Molecules 2010, 15, 2719–2729. [Google Scholar]
- Laddha, S.S.; Bhatnagar, S.P. Novel fused quinazolinones: Further studies on the anticonvulsant activity of 1,2,9,11-tetrasubstituted-7H-thieno[2′,3′:4,5]pyrimido[6,1-b]-quinazolin-7-one and 1,3,10,12-tetrasubstituted-8H-pyrido[2′,3′:4,5]pyrimido[6,1-b]quinazolin-8-one. Future Med. Chem. 2010, 2, 565–573. [Google Scholar] [CrossRef]
- Rohini, R.; Shanker, K.; Reddy, M.P.; Ravinder, V. Synthesis and antimicrobial activities of a new class of 6-arylbenzimidazo[1,2-c]quinazolines. J. Braz. Chem. Soc. 2010, 21, 49–57. [Google Scholar] [CrossRef]
- Saeed, A.; Shaheen, U.; Bolte, M. Synthesis, characterization and crystal structure of some novel 1-aryl-2-thioxo-2,3-dihydro-1H-quinazolin-4-ones. J. Chin. Chem. Soc. 2010, 57, 82–88. [Google Scholar]
- Shakhadoyatov, K.M.; Yangibaev, S.; Yun, L.M.; Kadyrov, C.S. Synthesis and alkylation of 2-mercapto-4-quinazolone and the fungicidal activities of the compounds obtained. Chem. Nat. Comp. 1982, 18, 106–109. [Google Scholar]
- El-Tombary, A.A.; Ismail, K.A.; Aboulwafa, O.M.; Omar, A.M.; El-Azzouni, M.Z.; El-Mansoury, S.T. Novel triazolo[4,3-a]quinazolinone and bis-triazolo[4,3-a:4,3'-c]quinazolines: synthesis and antitoxoplasmosis effect. Il Farmaco 1999, 54, 486–488. [Google Scholar]
- Gürsoy, A.; Karali, N. Synthesis and primary cytotoxicity evaluation of 3-[[(3-phenyl-4(3H)-quinazolinone-2-yl)mercaptoacetyl]hydrazono]-1H-2-indolinones. Eur. J. Med. Chem. 2003, 38, 633–643. [Google Scholar]
- Hamel, E.; Lin, C.M.; Plowman, J.; Wang, H.K.; Lee, K.H.; Paull, K.D. Antitumor 2,3-dihydro-2-(aryl)-4(1H)-quinazolinone derivatives: Interactions with tubulin. Biochem. Pharmacol. 1996, 51, 53–59. [Google Scholar]
- Xia, Y.; Yang, Z.Y.; Hour, M.-J.; Kuo, S.C.; Xia, P.; Bastow, K.F.; Nakanishi, Y.; Nampoothiri, P.; Hackl, T.; Hamel, E.; Lee, K. H. Antitumor agents. Part 204: Synthesis and biological evaluation of substituted 2-aryl quinazolinones. Bioorg. Med. Chem. Lett. 2001, 11, 1193–1196. [Google Scholar] [CrossRef]
- Peet, N.P.; Baugh, L.E.; Sundler, S.; Lewis, J.E.; Matthews, E.H.; Olberding, E.L.; Shah, D.N. 3-(1H-Tetrazol-5-yl)-4(3H)-quinazolinone sodium salt (MDL 427): A new antiallergic agent. J. Med. Chem. 1986, 29, 2403–2409. [Google Scholar]
- Cross, P.E.; Dickison, R.P.; Parry, M.J.; Randall, M.J. Selective thromboxane synthetase inhibitors. 1. 1-[(aryloxy)alkyl]-1H-imidazoles. J. Med. Chem. 1985, 28, 1427–1432. [Google Scholar]
- Hankovszky, H.O.; Hideg, K.; Bódi, I.; Frank, L. New antiarrhythmic agents. 2,2,5,5-Tetramethyl-3-pyrroline-3-carboxamides and 2,2,5,5-tetramethyl-pyrrolidine-3-carboxamides. J. Med. Chem. 1986, 29, 1138–1152. [Google Scholar] [CrossRef]
- Van Dyck, C.H.; Tan, P.-Z.; Baldwin, R.M.; Amici, L.A.; Garg, P.K.; Ng, C.K.; Soufer, R.; Charney, D.S.; Innis, R.B. PET quantification of 5-HT2A receptors in the human brain: a constant infusion paradigm with [18F]altanserin. J. Nucl. Med. 2000, 41, 234–241. [Google Scholar]
- Du, X.; Jiang, Y.; Qian, W.; Lu, X.; Walsh, J.P. Fatty acids inhibit growth-factor-induced diacylglycerol kinase alpha activation in vascular smooth-muscle cells. Biochem. J. 2001, 1, 275–282. [Google Scholar]
- Santagati, A.; Modica, M.; Monsu Scolaro, L.; Santagati, M. New synthetic approaches to fused heterocyclo-quinazolines. J. Chem. Res. (s) 1999, 86–87. [Google Scholar]
- Barghash, A.E.M.; El-Kerdawy, M.M.; Shehata, I.A.; Abdel-Aziz, N.I. Synthesis and antiviral testing of certain derivatives of thiazolo[4,3-b]benzothiazoles, thiazolo[3′,4′:2,3][1,2,4]-triazolo[5,1-b]-, {triazino [3,2-b]- and triazepino[3,2-b]}-quinazolines. Alex. J. Pharm. Sci. 2004, 18, 109–113. [Google Scholar]
- Tiwari, V.K.; Kale, R.R.; Mishra, B.B.; Singh, A. A facile one-pot MW approach for N3-(heteroaryl-2’-yl)-2-thioxo-2,3-dihydro-1H-quinazolin-4-one. ARKIVOC 2008, 14, 27–36. [Google Scholar]
- Mosselhi, M.A.; Pfleiderer, W. Syntheses, properties, and reactions of 8-aminoxanthines, purines. Part XVI: Syntheses, properties, and reactions of 8-aminoxanthines. Helv. Chim. Acta 2010, 93, 2115–2134. [Google Scholar] [CrossRef]
- Shawali, A.S.; Mosselhi, M.A.; Altablawy, F.M.A.; Farghaly, T.A.; Tawfik, N.M. Synthesis and tautomeric structure of 3,7-bis(arylazo)-6-methyl-2-phenyl-1H-imidazo[1,2-b]pyrazoles in ground and excited states. Tetrahedron 2008, 64, 5524–5530. [Google Scholar] [CrossRef]
- Shawali, A.S.; Mosselhi, M.A.; Farghaly, T.A.; Shehata, M.R.; Tawfik, N.M. Synthesis and tautomeric structure of 3,6-bis(arylazo)pyrazolo[1,5-a]pyrimidine-5,7(4H,6H)-diones. J. Chem. Res. 2008, 452–456. [Google Scholar]
- Shawali, A.S.; Mosselhi, M.A.; Farghaly, T.A. Synthesis and tautomeric structure of 2-arylazo-4H-imidazo[2,1-b][1,3,4]-thiadiazines. J. Chem. Res. 2007, 479–483. [Google Scholar]
- Shawali, A.S.; Abdallah, M.A.; Mosselhi, M.A.N.; Mohamed, Y.F. Synthesis and tautomeric structure of 1,2-bis(7-arylhydrazono-7H-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazin-3-yl)ethanes. Z. Naturforsch. B: Chem. Sci. 2002, 552–556. [Google Scholar]
- Mosselhi, M.N.; Abdallah, M.A.; Mohamed, Y.F.; Shawali, A.S. Synthesis and tautomeric structure of 7-arylhydrazono-7H-[1,2,4]triazolo[3,4-b][1,3,4]thiadiazines. Phosphorus Sulfur Silicon 2002, 177, 487–496. [Google Scholar] [CrossRef]
- Mosselhi, M.A.N.; Abdallah, M.A.; Riyadh, S.M.; Harhash, A.E.; Shawali, A.S. Tautomerism and reactions of 1H-1,2,4-thiazole-5-thiones with hydrazonoyl halides. J. Prakt. Chem. 1998, 340, 160–164. [Google Scholar]
- Hlavka, J.J.; Bitha, P.; Lin, Y.; Strohmeyer, T. Synthesis and reactions of 2,3-diamino-4(3H)-pyrimidinones and 3-amino-2-hydrazino-4(3H)-pyrimidinones. J. Heterocycl. Chem. 1984, 21, 1537–1541. [Google Scholar] [CrossRef]
- Essassi, E.M.; Lavergne, J.P.; Viallefont, P.; Daunis, J. Recherches en série triazepine-1,2,4: 1-détermination de la structure de la triazolotriazépinone obtenue par action de l'acétylacétate d'éthyle sur le diamino-3,4 triazole-1,2,4. J. Heterocycl. Chem. 1975, 12, 661–663. [Google Scholar]
- Hassan, K.M.; Ahmed, R.A.; Abdel-Hafez, S.H.; Abdel-Azim, M.A. Condensed thioxocyclopentapyridine (isoquinoline)-1,2,4-azines. Phosphorus Sulfur Silicon 2006, 181, 481–496. [Google Scholar]
- Romano, C.; De La Cuesta, E.; Avendano, C.; Florencio, F.; Sainz-Aparicio, J. Reactions of 1,2-diaminobenzimidazoles with β-dicarbonyl compounds. Tetrahedron 1988, 44, 7185–7192. [Google Scholar]
- Shawali, A.S.; Sherif, S.M.; Farghaly, T.A.; Shehata, M.R.; Darwish, M.A.A. Synthesis and tautomeric structure of the azo-coupling products of 2-methyl-7-phenylpyrimido[1,2-b][1,2,4]triazepine-4,9(3H,5H)-dione. J. Chem. Res. 2007, 44–47. [Google Scholar]
- Jones, R.; Ryan, A.J.; Sternhell, S.; Wright, S.E. The structures of some 5-pyrazolones and derived 4-arylazo-5-pyrazolones. Tetrahedron 1963, 19, 1497–1507. [Google Scholar]
- Smania, J.A.; Monache, F.D.; Smania, E.F.A.; Cuneo, R.S. Antibacterial activity of steroidal compounds isolated from Ganoderma applanatum (Pers.) Pat. (Aphyllophoromycetideae) fruit body. Int. J. Med. Mushrooms 1999, 1, 325–330. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 4 and 6 are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Farghaly, T.A.R.; Edrees, M.M.; Abdelnabi Mosselhi, M. Synthesis, Tautomeric Structure and Antimicrobial Activity of 3-Arylhydrazono-4-phenyl-[1,2,4]-triazepino[2,3-a]quinazoline-2,7(1H)-diones. Molecules 2012, 17, 8483-8493. https://doi.org/10.3390/molecules17078483
Farghaly TAR, Edrees MM, Abdelnabi Mosselhi M. Synthesis, Tautomeric Structure and Antimicrobial Activity of 3-Arylhydrazono-4-phenyl-[1,2,4]-triazepino[2,3-a]quinazoline-2,7(1H)-diones. Molecules. 2012; 17(7):8483-8493. https://doi.org/10.3390/molecules17078483
Chicago/Turabian StyleFarghaly, Thoraya Abdel Reheem, Mastoura Mohamed Edrees, and Mosselhi Abdelnabi Mosselhi. 2012. "Synthesis, Tautomeric Structure and Antimicrobial Activity of 3-Arylhydrazono-4-phenyl-[1,2,4]-triazepino[2,3-a]quinazoline-2,7(1H)-diones" Molecules 17, no. 7: 8483-8493. https://doi.org/10.3390/molecules17078483
APA StyleFarghaly, T. A. R., Edrees, M. M., & Abdelnabi Mosselhi, M. (2012). Synthesis, Tautomeric Structure and Antimicrobial Activity of 3-Arylhydrazono-4-phenyl-[1,2,4]-triazepino[2,3-a]quinazoline-2,7(1H)-diones. Molecules, 17(7), 8483-8493. https://doi.org/10.3390/molecules17078483