Terpenes as Green Solvents for Extraction of Oil from Microalgae
Abstract
:1. Introduction
| Solvent | n-Hexane | d-Limonene | α-Pinene | ρ-Cymene |
|---|---|---|---|---|
| Chemical Structure | 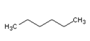 | 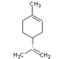 | 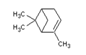 | 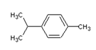 |
| Molecular formula | C6H14 | C10H16 | C10H16 | C10H14 |
| Molecular weight (g·mol−1) | 86.18 | 136.23 | 136.24 | 134.22 |
| Density 25 °C (g·cm−3) | 0.675 | 0.834 | 0.879 | 0.861 |
| Flash point (°C) | −23 | 48.3 | 32 | 47.2 |
| Boiling point (°C) | 69 | 176 | 155 | 176 |
| Viscosity, 25 °C (Cp) | 0.31 | 0.83 | 1.32 | 0.83 |
| Enthalpy of vaporization (kJ·mol−1) | 29.74 | 39.49 | 37.83 | 39.34 |
| Surface tension, 25 °C (dyne·cm−1) | 20.3 | 25.8 | 25.3 | 28.5 |
| Dielectric constant, 20 °C | 1.87 | 2.44 | 2.58 | 2.34 |
| Hansen Solubility Parameters δa (cal1/2·cm−3/2) | 7.3 | 8 | 7.9 | 8.1 |
| δd | 7.3 | 8 | 7.6 | 8.1 |
| δp | 0 | 0.1 | 2.1 | 0.3 |
| δh | 0 | 0.1 | 0 | 0 |

2. Results and Discussion
2.1. Extraction Kinetics
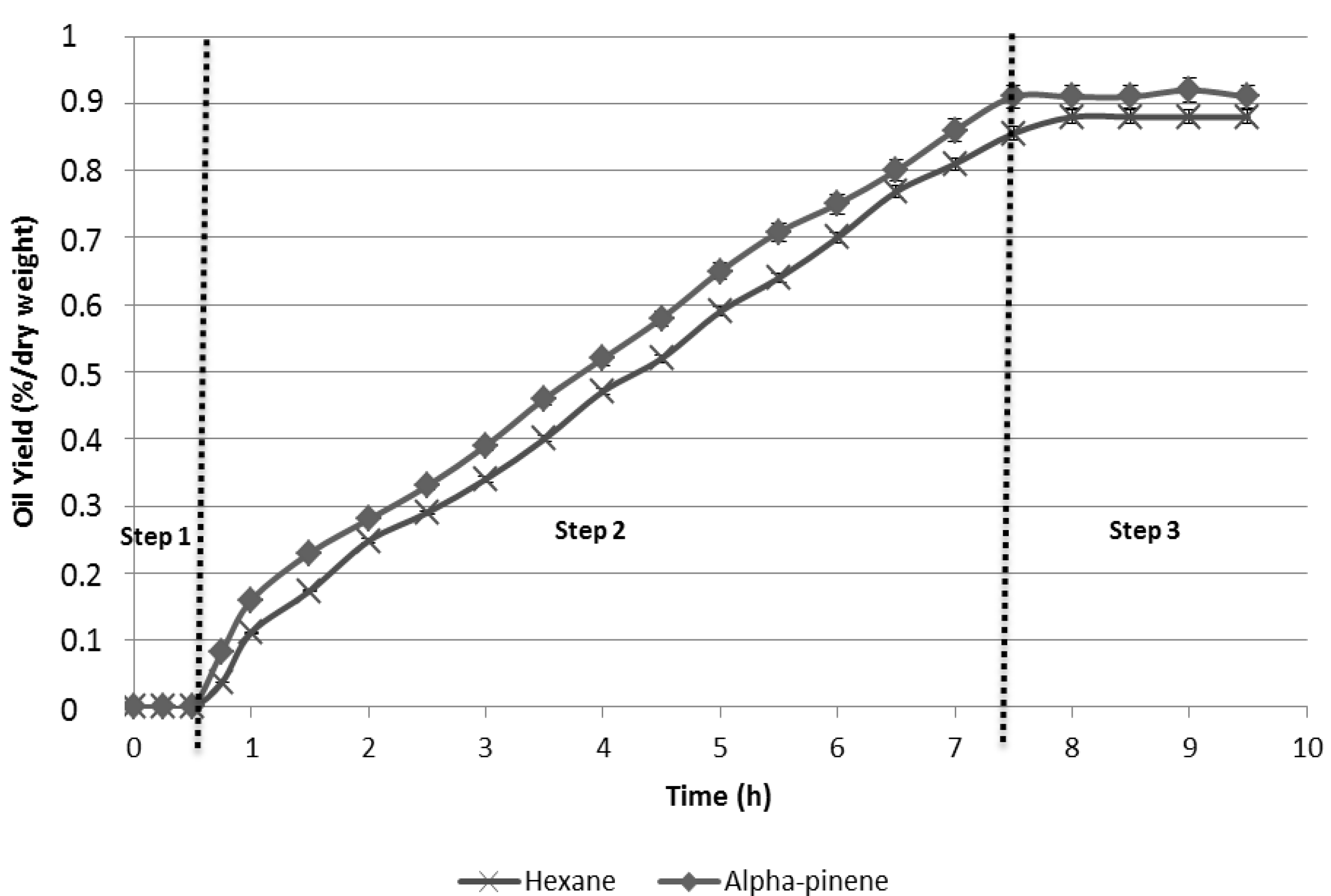
2.2. Oil Extraction Yields from the Different Solvents
| Fatty Acids | Extracted amounts of fatty acids (%) | ||||
|---|---|---|---|---|---|
| Bligh and Dyer | n-hexane | d-limonene | α-pinene | ρ-cymene | |
| C14 | 1.29 ± 0.04 | 1.83 ± 0.11 | 0.98 ± 0.31 | 1.38 ± 0.06 | 1.43 ± 0.08 |
| C16:0 | 26.63 ± 0.23 | 25.99 ± 0.52 | 27.62 ± 0.97 | 27.53 ± 0.32 | 28.91 ± 1.10 |
| C16:1 | 4.93 ± 0.32 | 2.03 ± 0.09 | 4.95 ± 0.66 | 3.02 ± 0.21 | 4.47 ± 0.06 |
| C16:2 | 1.37 ± 0.10 | 0.70 ± 0.03 | 2.83 ± 0.30 | 1.49 ± 0.17 | 0.92 ± 0.13 |
| C16:3 | 3.27 ± 0.18 | 1.65 ± 0.48 | 2.35 ± 1.67 | 1.26 ± 0.18 | 1.39 ± 0.13 |
| C16:4 | 9.89 ± 0.14 | 4.95 ± 1.78 | 6.64 ± 1.44 | 5.08 ± 0.16 | 3.36 ± 0.13 |
| C18:0 | 1.67 ± 0.31 | 4.44 ± 0.08 | 6.37 ± 1.00 | 4.58 ± 0.11 | 3.92 ± 0.21 |
| C18:1 | 17.79 ± 0.20 | 35.91 ± 0.07 | 23.97 ± 0.50 | 33.04 ± 0.10 | 36.70 ± 0.59 |
| C18:2 | 6.99 ± 0.36 | 7.32 ± 0.24 | 5.87 ± 0.61 | 7.37 ± 0.31 | 5.49 ± 0.35 |
| C18:3 | 23.48 ± 0.09 | 12.67 ± 0.10 | 14.55 ± 0.20 | 13.54 ± 0.07 | 12.33 ± 0.24 |
| C18:4 | 2.50 ± 0.10 | 2.13 ± 0.09 | 3.64 ± 0.29 | 1.36 ± 0.08 | 0.66 ± 0.10 |
| C20:0 | 0.20 ± 0.06 | 0.40 ± 0.04 | 0.23 ± 0.09 | 0.35 ± 0.08 | 0.41 ± 0.06 |
| ∑SFAs | 29.7922.7247.49 | 32.6537.9429.41 | 35.228.935.9 | 33.8336.0630.11 | 34.6841.1724.15 |
| ∑MUFAs | |||||
| ∑PUFAs | |||||
| Oil Yield [%] | 3.36 ± 0.02 | 0.88 ± 0.01 | 1.29 ± 0.01 | 0.91 ± 0.05 | 1.52 ± 0.03 |
2.3. Oil Quality Comparison Results
2.4. Microscopic Observations

3. Experimental
3.1. Chemical and Reagents
3.2. Microalgae Cultivation and Harvesting
3.3. Determination of the Total Lipid Content
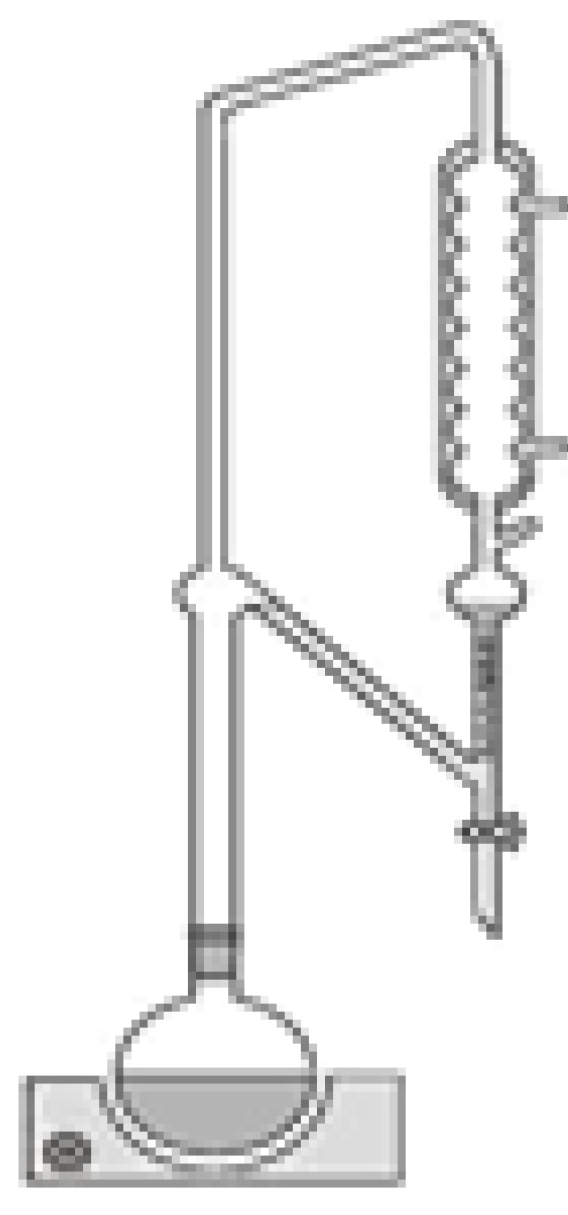
3.4. Soxhlet Extraction
3.5. Preparation of Fatty Acid Methyl Ester Derivatives
3.6. Chromatographic Analysis of Fatty Acids
3.7. Microscopic Observations
4. Conclusions
Acknowledgments
References
- Sheehan, J.; Dunahay, T.; Benemann, J.; Roessler, P. A. A Look Back at the US Department of Energy’s Aquatic Species Program: Biodiesel from Algae. NREL/TP-580-24190. U.S Department of Energy’s Office of Fuels Development, Golden, CO, USA. 1998; 1–325. [Google Scholar]
- Wan, P.J.; Hron, R.J.; Dowd, M.K.; Kuk, M.S.; Conkerton, E.J. Alternative hydrocarbon solvents for cottonseed extraction: Plant trials. J. Am. Oil Chem. Soc. 1995, 72, 661–664. [Google Scholar] [CrossRef]
- Hanmoungjai, P.; Pyle, L.; Niranjan, K. Extraction of rice bran oil using aqueous media. J. Chem. Technol. Biotechnol. 2000, 75, 348–352. [Google Scholar] [CrossRef]
- Anastas, P.; Warner, J. Theory and Practice; Oxford University Press: New York, NY, USA, 1998. [Google Scholar]
- Toplisek, T.; Gustafson, R. Cleaning with D-Limonenes: A substitute for chlorinated solvents. Precis. Clean. 1995, 3, 17–20. [Google Scholar]
- Mamidipally, P.K.; Liu, S.X. First approach on rice bran oil extraction using limonene. Eur. J. Lipid Sci. Technol. 2004, 106, 122–125. [Google Scholar] [CrossRef]
- Liu, S.X.; Mamidipally, P.K. Quality Comparison of rice bran oil extracted with D-limonene and hexane. Cereal. Chem. 2005, 82, 209–215. [Google Scholar] [CrossRef]
- Virot, M.; Tomao, V.; Ginies, C.; Visinoni, F.; Chemat, F. Green procedure with a green solvent for fats and oils’ determination Microwave-integrated Soxhlet using limonene followed by microwave Clevenger distillation. J. Chromatogr. A 2008, 1196-1197, 147–152. [Google Scholar] [CrossRef]
- Yatagai, M.; Sato, T.; Takahashi, T. Terpenes of leaf oils from Cupressaceae. Biochem. Syst. Ecol. 1984, 13, 377–385. [Google Scholar]
- Hansen, C.M. The three dimensional solubility parameter—Key to paint component affinities II. Dyes, emulsifiers, mutual solubility and compatibility, and pigments. J. Paint Technol. 1967, 39, 505–510. [Google Scholar]
- Albuquerque, M.R.J.R.; Lemos, T.L.G.; Pessoa, O.D.L.; Nunes, E.P.; Nascimento, R.F.; Silveira, E.R. Chemical composition of the essential oil from Vernonia scorpioides (Asteraceae). Flavour Fragr. J. 2007, 22, 249–250. [Google Scholar] [CrossRef]
- Bligh, E.G.; et Dyer, W.J. A rapid method of total lipid extraction and purification. Biochem. Cell Biol. 1959, 37-38, 911–917. [Google Scholar]
- Petkov, G.; Garcia, G. Which are fatty acids of the green alga Chlorella? Biochem. Syst. Ecol. 2007, 35, 281–285. [Google Scholar] [CrossRef]
- Plaza, M.; Santoyo, S.; Jaime, L.; Avalo, B.; Cifuentes, A.; Reglero, G.; García-Blairsy, G.; Señoráns, F.J.; Ibáñez, E. Comprehensive characterization of the functional activities of pressurized liquid and ultrasound-assisted extracts from Chlorella vulgaris. LWT Food Sci. Technol. 2011, 46, 245–253. [Google Scholar]
- Rashid, U.; Anwar, F.; Moser, B.R.; Knothe, G. Moringa oleifera oil: A possible source of biodiesel. Bioresour. Technol. 2008, 99, 8175–8179. [Google Scholar] [CrossRef]
- Demirbas, A. Progress and recent trends in biodiesel fuels. Energy Convers. Manag. 2009, 50, 14–34. [Google Scholar] [CrossRef]
- Soxhlet, F. Die gewichtsanalytische bestimmung des milchfettes. Dinglers’ Polyt. J. 1879, 232, 461–465. [Google Scholar]
- Morrison, W.R.; et Smith, L.M. Preparation of fatty acid methyl esters and dimethylacetals from lipids with boron fluoride-methanol. J. Lipid Res. 1964, 5, 600–608. [Google Scholar]
- Sample Availability: Samples of the different experiments are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Dejoye Tanzi, C.; Abert Vian, M.; Ginies, C.; Elmaataoui, M.; Chemat, F. Terpenes as Green Solvents for Extraction of Oil from Microalgae. Molecules 2012, 17, 8196-8205. https://doi.org/10.3390/molecules17078196
Dejoye Tanzi C, Abert Vian M, Ginies C, Elmaataoui M, Chemat F. Terpenes as Green Solvents for Extraction of Oil from Microalgae. Molecules. 2012; 17(7):8196-8205. https://doi.org/10.3390/molecules17078196
Chicago/Turabian StyleDejoye Tanzi, Celine, Maryline Abert Vian, Christian Ginies, Mohamed Elmaataoui, and Farid Chemat. 2012. "Terpenes as Green Solvents for Extraction of Oil from Microalgae" Molecules 17, no. 7: 8196-8205. https://doi.org/10.3390/molecules17078196
APA StyleDejoye Tanzi, C., Abert Vian, M., Ginies, C., Elmaataoui, M., & Chemat, F. (2012). Terpenes as Green Solvents for Extraction of Oil from Microalgae. Molecules, 17(7), 8196-8205. https://doi.org/10.3390/molecules17078196







