Short and Long-Term Effects of Baccharis articulata on Glucose Homeostasis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Phytochemical Characterization
| Extract/fraction | Total phenolic | Total flavonoids |
|---|---|---|
| CE | 151.8 ± 0.93 a | 38.9 ± 0.66 a |
| BF | 154.1 ± 1.08 a | 44.9 ± 0.71 b |
| ARF | 135.6 ± 0.95 b | 25.3 ± 0.42 c |
2.2. Effect of Crude Extract and n-Butanol and Aqueous Residual Fractions of B. Articulata on Oral Glucose Tolerance Curve
2.3. Effect of Crude Extract and n-Butanol and Aqueous Residual Fractions of B. Articulata on Insulin Secretion and Glycogen Content
| Time (min) | Group I Hyper Glucose (4 g/kg) | Group II Hyper + glipizide (10 mg/kg) | Group III Hyper + CE | Group IV Hyper + BF | Group V Hyper + ARF | ||||
|---|---|---|---|---|---|---|---|---|---|
| 50 mg/kg | 100 mg/kg | 200 mg/kg | 50 mg/kg | 100 mg/kg | 50 mg/kg | 100 mg/kg | |||
| 0 | 112 ± 4 # | 104 ± 3 | 110 ± 6 | 116 ± 5 | 103 ± 3 | 116 ± 2 | 113 ± 3 | 116 ± 3 | 103 ± 2 |
| 15 | 162 ± 8 | 121 ± 4 *** | 128 ± 5 ** | 123 ± 2 *** | 134 ± 6 ** | 124 ± 6 *** | 187 ± 4 | 138 ± 4 ** | 136 ± 10 * |
| 30 | 185 ± 6 | 148 ± 7 *** | 144 ± 4 *** | 137 ± 1 *** | 146 ± 3 *** | 143 ± 2 *** | 178 ± 6 | 148 ± 3 *** | 159 ± 6 * |
| 60 | 164 ± 4 | 122 ± 4 *** | 145 ± 5 | 130 ± 8 ** | 140 ± 6 * | 134 ± 9 ** | 186 ± 8 | 154 ± 4 | 161 ± 10 |
| 180 | 135 ± 4 | 116 ± 5 ** | 130 ± 3 | 129 ± 3 | 136 ± 4 | 145 ± 5 | 126 ± 4 | 142 ± 5 | 133 ± 5 |
| Time (min) | Hyper Glucose (4 g/kg) | Hyper + glipizide (10 mg/kg) | Hyper + B. articulata | ||
|---|---|---|---|---|---|
| CE 100 mg/kg | BF 50 mg/kg | ARF 50 mg/kg | |||
| 0 | 0.57 ± 0.03 | - | - | - | - |
| 15 | 0.77 ± 0.06 # | 2.27 ± 0.20 *** | 1.29 ± 0.27 ** | 1.25 ± 0.10 * | 0.82 ± 0.04 |
| 30 | 0.90 ± 0.10 | 1.37 ± 0.02 * | 1.30 ± 0.14 * | 1.74 ± 0.06 *** | 0.79 ± 0.09 |
| 60 | 0.54 ± 0.04 | 1.03 ± 0.15 * | 1.45 ± 0.24 *** | 1.32 ± 0.20 *** | 0.63 ± 0.10 |
| II | 0.44 | 1.09 | 0.96 | 1.03 | 0.49 |
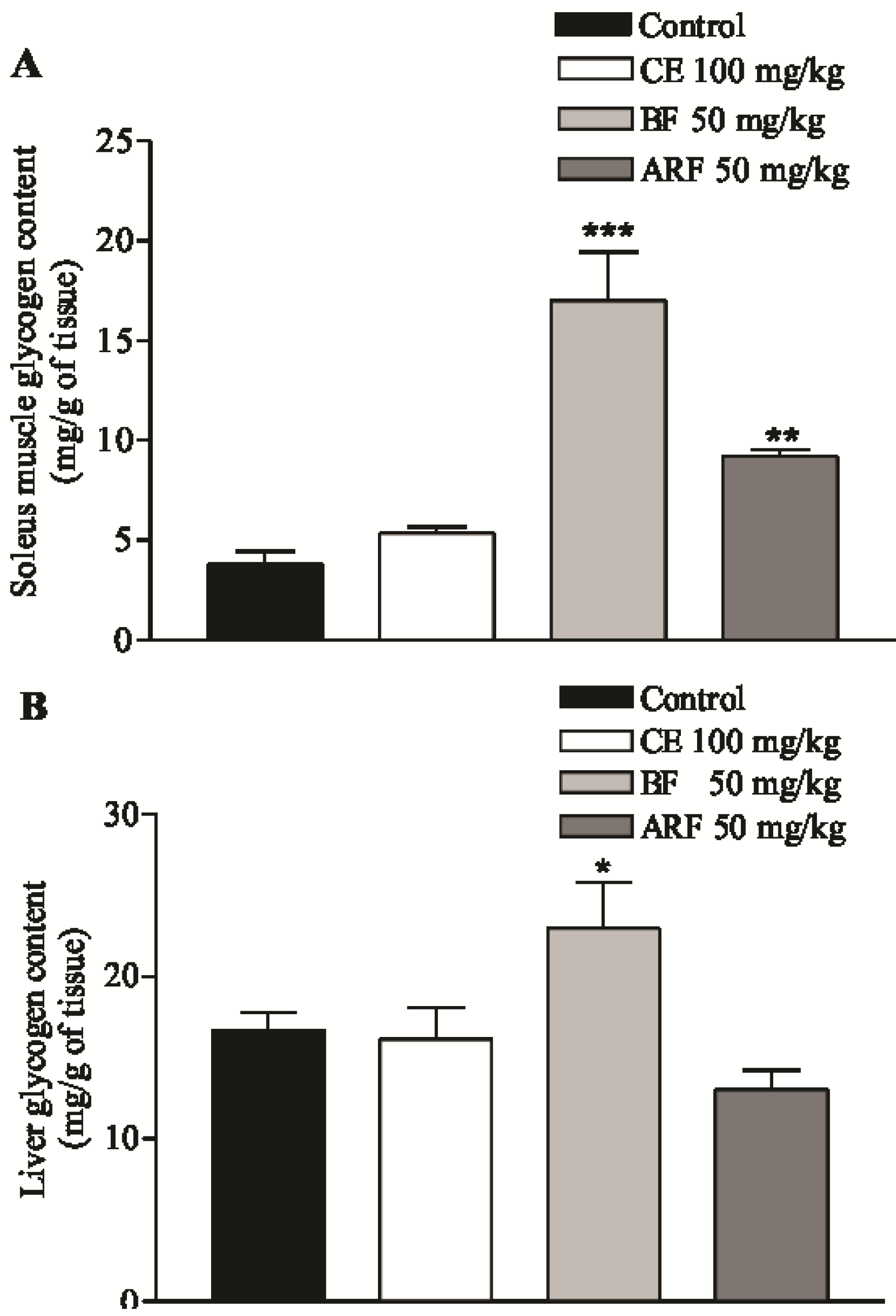
2.4. Effect of Crude Extract and n-Butanol and Aqueous Residual Fractions of B. Articulata on the Disaccharidases

2.5. Effect of Crude Extract and n-Butanol and Aqueous Residual Fractions of B. Articulata on in vitro Albumin Glycation

3. Experimental
3.1. Materials
3.2. Plant Material
3.3. Preparation of Extract and Fractions of B. Articulata
3.4. Thin-Layer Chromatographic Analysis
3.5. Determination of Total Phenolic Content
3.6. Determination of Total Flavonoid Content
3.7. Animals
3.8. Oral Glucose Tolerance Curve (OGTC)
3.9. Determination of the Plasma Glucose Concentration
3.10. Insulin Serum Measurements
3.11. Glycogen Content Measurements
3.12. Disaccharidase Extraction and Assays
3.13. Formation of Advanced Glycation End-Products (AGEs) in Bovine Serum Albumin/Glucose and Fructose Systems
3.14. Data and Statistical Analysis
4. Conclusions
Conflict of Interest
Acknowledgements
- Sample Availability: Samples of the extracts are available from the authors.
References and Notes
- American Diabetes Association. Diagnosis and Classification of Diabetes Mellitus. In Diabetes Care; 2011; 34 (Suppl. 1), pp. 62–69. [Google Scholar]
- Souto, S.B.; Souto, E.B.; Braga, D.C.; Medina, J.L. Prevention and current onset delay approaches of type 2 diabetes mellitus (T2DM). Eur. J. Clin. Pharmacol. 2011, 67, 653–661. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural products as sources of new drugs over the last 25 years. J. Nat. Prod. 2007, 70, 461–477. [Google Scholar] [CrossRef]
- Lamba, S.S.; Buch, K.Y.; Lewis, H.; Lamba, J. Phytochemicals as potencial hypoglicemic agents. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier Science B.V.: Amsterdam, The Netherlands, 2000; pp. 457–496. [Google Scholar]
- Abad, M.J.; Bermejo, P. Baccharis (Compositae): A review update. ARKIVOC 2007, 7, 76–96. [Google Scholar]
- De Oliveira, S.Q.; Barbon, G.; Gosmann, G.; Bordignon, S. Differentiation of South Brazilian Baccharis species by TLC. J. Liq. Chromatogr. Relat. Technol. 2006, 29, 2603–2609. [Google Scholar] [CrossRef]
- De Oliveira, S.Q.; Dal-Pizzol, F.; Gosmann, G.; Guillaume, D.; Moreira, J.C.F.; Schenkel, E.P. Antioxidant activity of Baccharis articulata extracts: Isolation of a new compound with antioxidant activity. Free Radic. Res. 2003, 37, 555–559. [Google Scholar] [CrossRef]
- Verdi, L.G.; Brighente, I.M.C.; Pizzolatti, M.G. Gênero Baccharis (ASTERACEAE): Aspectos químicos, econômicos e biológicos. Quim. Nova 2005, 28, 85–94. [Google Scholar] [CrossRef]
- Scalbert, A.; Manach, C.; Morand, C.; Rémésy, C.; Jiménez, L. Dietary Polyphenols and the Prevention of Diseases. Crit. Rev. Food. Sci. Nutr. 2007, 5, 287–306. [Google Scholar]
- Jung, M.; Park, M.; Lee, H.C.; Kang, Y.; Kang, E.S.; Kim, S.K. Antidiabetic agents from medicinal plants. Curr. Med. Chem. 2006, 13, 1203–1218. [Google Scholar] [CrossRef]
- Oliveira, A.C.P.; Endringer, D.C.; Amorim, L.A.S.; Brandão, M.G.L.; Coelho, M.M. Effect of the extracts and fractions of Baccharis trimera and Syzygium cumini on glycaemia of diabetic and non-diabetic mice. J. Ethnopharmacol. 2005, 102, 465–469. [Google Scholar] [CrossRef]
- Beardsall, K.; Yuen, K.; Williams, R.; Dunger, D. Applied physiology of glucose control. Curr. Pediatr. 2003, 13, 543–548. [Google Scholar] [CrossRef]
- Folador, P.; Cazarolli, L.H.; Gazola, A.C.; Reginatto, F.H.; Schenkel, E.P.; Silva, F.R.M.B. Potential insulin secretagogue effects of isovitexin and swertisin isolated from Wilbrandia ebracteata roots in non-diabetic rats. Fitoterapia 2010, 81, 1180–1187. [Google Scholar] [CrossRef]
- Ferrer, J.C.; Favre, C.; Gomis, R.R.; Fernández-Novell, J.M.; Garcia-Rocha, M.; De la Iglesia, N.; Cid, E.; Guinovart, J.J. Control of glycogen deposition. FEBS Lett. 2003, 546, 127–132. [Google Scholar] [CrossRef]
- Park, S.A.; Choi, M.S.; Cho, S.Y.; Seo, J.S.; Jung, U.J.; Kim, M.J.; Sung, M.K.; Park, Y.B.; Lee, M.K. Genistein and daidzein modulate hepatic glucose and lipid regulating enzyme activities in C57BL/KsJ-db/db mice. Life Sci. 2006, 79, 1207–1213. [Google Scholar] [CrossRef]
- De Melo, E.B.; Carvalho, I. a e b-Glucosidases como alvos moleculares para desenvolvimento de fármacos. Quim. Nova 2006, 29, 840–843. [Google Scholar] [CrossRef]
- Kumar, S.K.; Smita, N.; Vipin, K.; Om, P. α-glucosidase inhibitors from plants: A natural approach to treat diabetes. Pharmacogn. Rev. 2011, 5, 19–29. [Google Scholar] [CrossRef]
- De Souza, S.P.; Pereira, L.L.S.; Souza, A.A.; Dos Santos, C.D. Inhibition of pancreatic lipase by extracts of Baccharis trimera (Less.) DC., Asteraceae: Evaluation of antinutrients and effect on glycosidases. Braz. J. Pharmacogn. 2011, 21, 450–455. [Google Scholar]
- Goh, S.Y.; Cooper, M.E. The Role of Advanced Glycation End Products in progression and Complications. J. Clin. Endocrinol. Metab. 2008, 93, 1143–1152. [Google Scholar] [CrossRef]
- Wu, C.H.; Yen, G.C. Inhibitory Effect of Naturally Occurring Flavonoids on the Formation of Advanced Glycation Endproducts. J. Agric. Food. Chem. 2005, 53, 3167–3173. [Google Scholar] [CrossRef]
- Kim, H.Y.; Kim, K. Effect of flavonoids on formation of advanced glycation endproducts in vitro. Int. Congr. Ser. 1245, 1245, 511. [Google Scholar]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of Folin-Ciocalteu reagent. Meth. Enzymol. 1999, 299, 152–178. [Google Scholar]
- Miliauskas, G.; Venskutonis, P.R.; van Beek, T.A. Screening of radical scavenging activity of some medicinal and aromatic plant extracts. Food Chem. 2004, 85, 231–237. [Google Scholar] [CrossRef]
- Varley, H.; Gowenlock, A.H.; Bell, M. Practical Biochemistry, 5th ed; I.W. Heinemann: London, UK, 1976. [Google Scholar]
- Damazio, R.G.; Zanatta, A.P.; Cazarolli, L.H.; Mascarello, A.; Chiaradia, L.D.; Nunes, R.J.; Yunes, R.A.; Silva, F.R.M.B. Nitrochalcones: Potential in vivo insulin secretagogues. Biochimie 2009, 91, 1493–1498. [Google Scholar] [CrossRef]
- Krisman, C.R. A method for the colorimetric estimation of glycogen with iodine. Anal. Biochem. 1962, 4, 17–23. [Google Scholar] [CrossRef]
- Zanatta, L.; Rosso, A.; Folador, P.; Figueiredo, M.S.R.B.; Pizzolatti, M.G.; Leite, L.D.; Silva, F.R.M.B. Insulinomimetic effect of kaempferol 3-neohesperidoside on the rat soleus muscle. J. Nat. Prod. 2008, 71, 532–535. [Google Scholar] [CrossRef]
- Dahlqvist, A. Assay of intestinal disaccharidases. Scand. J. Clin. Lab. Invest. 1984, 44, 169–172. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Kiho, T.; Usui, S.; Hirano, K.; Aizawa, K.; Inakuma, T. Tomato paste fraction inhibiting the formation of advanced glycation end-products. Biosci. Biotechnol. Biochem. 2004, 68, 200–205. [Google Scholar] [CrossRef]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kappel, V.D.; Pereira, D.F.; Cazarolli, L.H.; Guesser, S.M.; Da Silva, C.H.B.; Schenkel, E.P.; Reginatto, F.H.; Silva, F.R.M.B. Short and Long-Term Effects of Baccharis articulata on Glucose Homeostasis. Molecules 2012, 17, 6754-6768. https://doi.org/10.3390/molecules17066754
Kappel VD, Pereira DF, Cazarolli LH, Guesser SM, Da Silva CHB, Schenkel EP, Reginatto FH, Silva FRMB. Short and Long-Term Effects of Baccharis articulata on Glucose Homeostasis. Molecules. 2012; 17(6):6754-6768. https://doi.org/10.3390/molecules17066754
Chicago/Turabian StyleKappel, Virginia D., Danielle F. Pereira, Luisa H. Cazarolli, Samantha M. Guesser, Carlos H. Blum Da Silva, Eloir P. Schenkel, Flávio H. Reginatto, and Fátima R. M. B. Silva. 2012. "Short and Long-Term Effects of Baccharis articulata on Glucose Homeostasis" Molecules 17, no. 6: 6754-6768. https://doi.org/10.3390/molecules17066754
APA StyleKappel, V. D., Pereira, D. F., Cazarolli, L. H., Guesser, S. M., Da Silva, C. H. B., Schenkel, E. P., Reginatto, F. H., & Silva, F. R. M. B. (2012). Short and Long-Term Effects of Baccharis articulata on Glucose Homeostasis. Molecules, 17(6), 6754-6768. https://doi.org/10.3390/molecules17066754




