Antiatherogenic Properties of Acetone Extract of Alpinia zerumbet Seeds
Abstract
:1. Introduction
2. Results and Discussion
2.1. GC-MS Analysis
| Compounds | RT | Peak area (%) | |||||
|---|---|---|---|---|---|---|---|
| Rhizome | Stem | Leaf | Flower | Pericarp | Seed | ||
| Cholestane | 13.342 | - | 0.33 | - | - | 0.20 | - |
| 3α,7β-Dihydroxy-5β,6β-epoxycholestane | 17.442 | - | - | - | - | 0.17 | 0.65 |
| Cholest-4-ene-3,6-dione | 21.067 | - | - | - | - | - | 1.84 |
| Cholest-5-en-3-ol | 21.125 | - | - | - | 0.12 | - | - |
| Ergost-5-en-3-ol | 21.133 | - | - | - | - | 0.29 | - |
| Sitosterol | 21.333 | 8.25 | 14.49 | 2.86 | - | 0.11 | - |
| Cholestenone | 21.567 | - | - | - | - | 0.96 | - |
| 5α-Ergost-8(14)-ene | 21.758 | - | - | 0.49 | - | - | - |
| 9,19-Cyclolanostan-3-ol | 21.983 | - | - | 0.38 | - | - | - |
| Cholest-4-en-3-ol | 22.075 | - | - | - | - | 0.27 | - |
| Stigmasterol | 22.225 | - | 2.70 | - | 1.46 | 1.72 | - |
| Cholest-8-ene-3,6-diol | 22.725 | 0.28 | 0.16 | 0.16 | - | - | - |
| 4,22-Stigmastadiene-3-one | 22.750 | - | - | - | - | 0.25 | - |
| Campesterol | 24.067 | - | 1.55 | - | 0.35 | - | - |
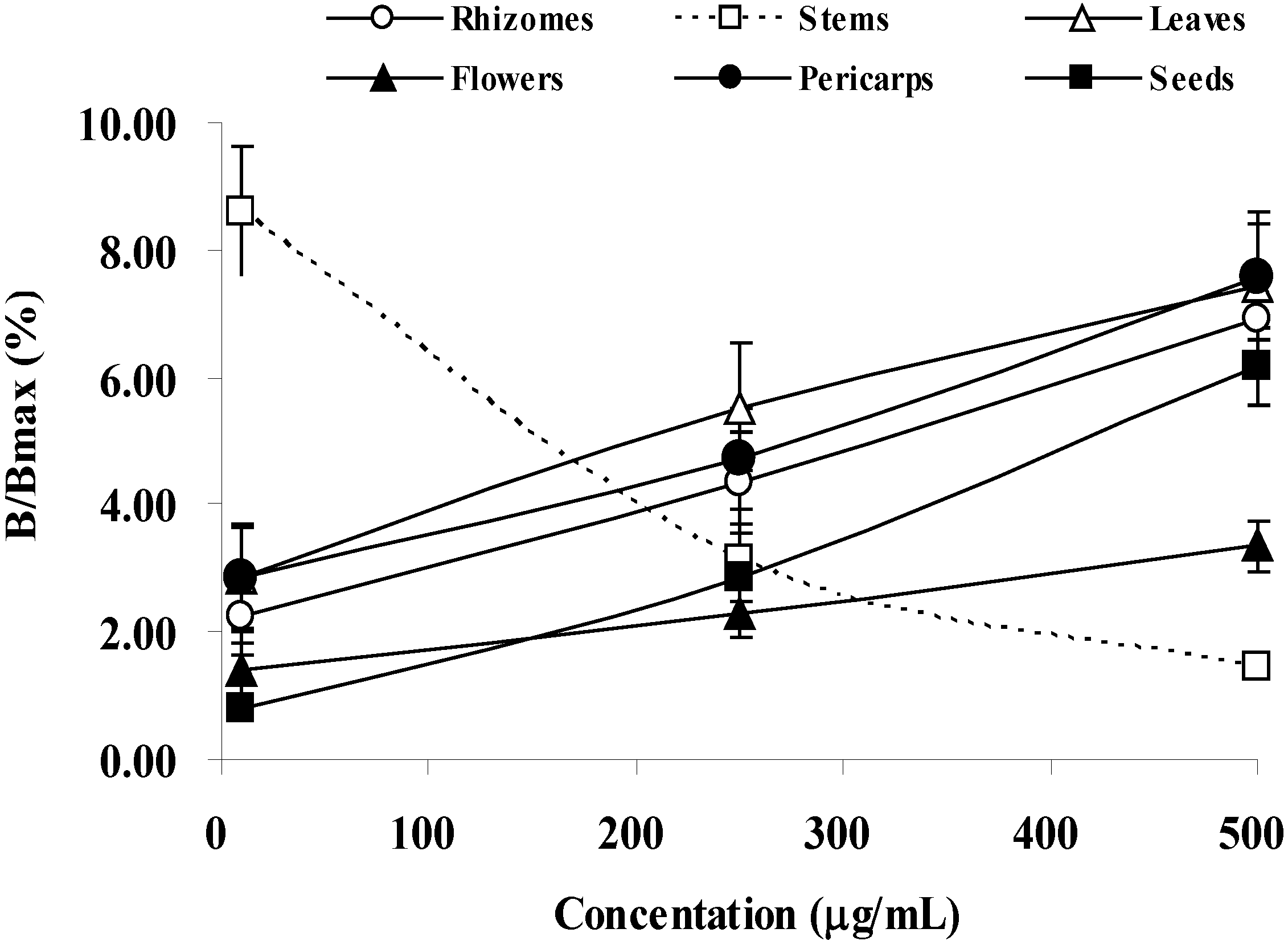
2.2. Anti-Tyrosinase Activity
| Sample | 50% of Inhibition (μg/mL) | |||
|---|---|---|---|---|
| Tyrosinase | Pancreatic lipase | 15-Lipoxygenase | LDL oxidation | |
| Rhizomes | 224.53 ± 4.34 d 1 | 277.75 ± 1.60 e | 142.15 ± 1.44 c | 164.90 ± 0.71 b |
| Stems | 312.53 ± 1.43 e | 747.09 ± 2.34 f | 1866.75 ± 9.12 f | 443.97 ± 3.64 d |
| Leaves | 152.23 ± 1.66 b | 58.88 ± 1.55 b | 759.59 ± 3.13 d | 507.59 ± 3.73 e |
| Flowers | 210.33 ± 1.76 c | 76.73 ± 0.82 c | 1356.57 ± 0.52 e | 423.43 ± 3.47 c |
| Pericarps | 151.31 ± 0.26 b | 256.30 ± 3.12 d | 87.90 ± 1.60 b | 515.48 ± 2.14 e |
| Seeds | 2.30 ± 0.02 a | 5.00 ± 0.70 a | 1.26 ± 0.07 a | 15.40 ± 0.86 a |
2.2. Pancreatic Lipase Inhibition
2.3. 15-Lipoxygenase Inhibition

2.4. LDL Oxidation Inhibition
2.5. Estrogenic Activity
2.6. Discussion
3. Experimental
3.1. General
3.2. Plant Material and Preparation of Extracts
3.3. GS-MS Analysis
3.4. Anti-Tyrosinase Activity Assay
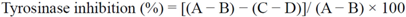
3.5. Pancreatic Lipase Inhibition Assay
3.6. 15-Lipoxygenase Inhibition Assay
3.7. LDL Oxidation Inhibition Assay
3.8. Estrogenic Activity Assay
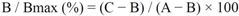
3.9. Statistical Analysis
4. Conclusion
References and Notes
- Ross, R. Atherosclerosis—An inflammatory disease. N. Engl. J. Med. 1999, 340, 115–126. [Google Scholar] [CrossRef]
- Shepherd, J.; Cobbe, S.M.; Ford, I.; Isles, C.G.; Lorimer, A.R.; Macfarlane, P.W.; Mckillop, J.H.; Packard, C.J. Prevention of coronary heart disease with pravastatin in men with hypercholesterolemia. N. Engl. J. Med. 1995, 333, 1301–1307. [Google Scholar] [CrossRef]
- Fox, P.L.; Mazumder, B.; Ehrenwald, E.; Mukhopadhyay, C.K. Forum: Role of oxidation in atherosclerosis. Ceruloplasmin and cardiovascular disease. Free Radic. Biol. Med. 2000, 28, 1735–1744. [Google Scholar] [CrossRef]
- Gholamhoseinian, A.; Shahouzehi, B.; Sharifi-far, F. Inhibitory effect of some plant extracts on pancreatic lipase. Int. J. Pharmacol. 2010, 6, 18–24. [Google Scholar] [CrossRef]
- Barter, P. The role of HDL-cholesterol in preventing atherosclerotic disease. Eur. Heart J. Suppl. 2005, 7F, F4–F8. [Google Scholar] [CrossRef]
- Harats, D.; Shaish, A.; George, J.; Mulkins, M.; Kurihara, H.; Levkovitz, H.; Sigal, E. Overexpression of 15-lipoxygenase in vascular endothelium accelerates early atherosclerosis in LDL receptor-deficient mice. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 2100–2105. [Google Scholar] [CrossRef]
- Zoghbi, M.G.B.; Andrade, E.H.A.; Maia, J.G.S. Volatile constituents from leaves and flowers of Alpiniaspeciosa K. Schum. and A. purpurata (Viell.) Schum. Flavour Fragr. J. 1999, 14, 411–414. [Google Scholar] [CrossRef]
- Bezerra, M.A.C.; Leal-Cardoso, J.H.; Coelho-de-Souza, A.N.; Criddle, D.N.; Fonteles, M.C. Myorelaxant and antispasmodic effect of the essential oil of Alpipiaspeciosa on rat ileum. Phytother. Res. 2000, 14, 549–551. [Google Scholar] [CrossRef]
- Elzaawely, A.A.; Xuan, T.D.; Koyama, H.; Tawata, S. Antioxidant activity and contents of essential oil and phenolic compounds in flowers and seeds of Alpiniazerumbet (Pers.) B.L. Burtt. & R.M. Sm. Food Chem. 2007, 104, 1648–1653. [Google Scholar] [CrossRef]
- Upadhyay, A.; Chompoo, J.; Kishimoto, W.; Makise, T.; Tawata, S. HIV-1 integrase and neuraminidase inhibitors from Alpiniazerumbet. J. Agric. Food Chem. 2011, 59, 2857–2862. [Google Scholar] [CrossRef]
- Chompoo, J.; Upadhyay, A.; Kishimoto, W.; Makise, T.; Tawata, S. Advanced glycation end products inhibitors from Alpiniazerumbet rhizomes. Food Chem. 2011, 129, 709–715. [Google Scholar] [CrossRef]
- Carpenter, K.L.H.; Van der Veen, C.; Hied, R.; Dennis, I.F.; Ding, T.; Mitchinson, M.J. The carotenoids beta-carotene, canthaxanthin and zeaxanthin inhibit macrophage-mediated LDL oxidation. FEBS Lett. 1997, 401, 262–266. [Google Scholar] [CrossRef]
- Calpe-Berdiel, L.; Escolà-Gil, J.C.; Blanco-Vaca, F. New insights into the molecular actions of plant sterols and stanols in cholesterol metabolism. Atherosclerosis 2009, 203, 18–31. [Google Scholar] [CrossRef]
- Chang, T.S. An updated review of tyrosinase inhibitors. Int. J. Mol. Sci. 2009, 10, 2440–2475. [Google Scholar] [CrossRef]
- Bocan, T.M.A.; Rosebury, W.S.; Mueller, S.B.; Kuchera, S.; Welch, K.; Daugherty, A.; Cornicelli, J.A. A specific 15-lipoxygenase inhibitor limits the progression and monocyte-macrophage enrichment of hypercholesterolemia-induced atherosclerosis in the rabbit. Atherosclerosis 1998, 136, 203–216. [Google Scholar] [CrossRef]
- Christiansen, C. Sex steroids and the cardiovascular system. Osteoporosis Int. Suppl. 1997, 1, S8–S11. [Google Scholar] [CrossRef]
- Fischer, G.M.; Cherian, K.; Swain, M.L. Increased synthesis of aortic collagen and elastin in experiment atherosclerosis: Inhibition by contraceptive steroids. Atherosclerosis 1981, 39, 463–467. [Google Scholar] [CrossRef]
- Lee, S.E.; Shin, H.T.; Hwang, H.J.; Kim, J.H. Antioxidant activity of extracts from Alpiniazerumbetkatsumadai seed. Phytother. Res. 2003, 17, 1041–1047. [Google Scholar] [CrossRef]
- Li, H.; Park, J.H.; Yan, B.; Yoo, K.Y.; Lee, C.H.; Choi, J.H.; Hwang, I.K.; Won, M.H. Neuroprotection of Alpiniakatsumadai seed extract against neuronal damage in the ischemic gerbil hippocampus in linked to altered brain-derived neurotrophic factor. Lab. Anim. Res. 2011, 27, 67–71. [Google Scholar] [CrossRef]
- Lee, M.Y.; Lee, N.H.; Seo, C.S.; Lee, J.A.; Jung, D.; Kim, J.H.; Shin, H.K. Alpiniakatsumadai seed extract attenuate oxidative stress and asthmatic activity in a mouse model of allergic asthma. Food Chem. Toxicol. 2010, 48, 1746–1752. [Google Scholar] [CrossRef]
- Mitsui, S.; Kobayashi, S.; Nagahori, H.; Ogiso, A. Constituents from seeds of Alpiniagalangal Willd. And their anti-ucler activities. Chem. Pharm. Bull. 1976, 24, 2377–2382. [Google Scholar] [CrossRef]
- Fernández, C.; Suárez, Y.; Ferruelo, A.J.; Gómez-Coronado, D.; Lasunción, A. Inhibition of cholesterol biosynthesis by Δ22-unsaturated phytosterols via competitive inhibition of sterol Δ24-reductase in mammalian cells. Biochem. J. 2002, 366, 109–119. [Google Scholar] [CrossRef]
- Nestler, J.E.; Barlascini, C.O.; Clore, J.N.; Blackard, W.G. Dehydroepiandrosteronereduces serum low density lipoprotein levels and body fat but does not alter insulin sensitivity in normal men. J. Clin. Endocrinol. Metab. 1988, 135, 1519–1528. [Google Scholar]
- Eich, D.M.; Nestler, J.E.; Johnson, D.E.; Dworkin, G.H.; Ko, D.; Wechsler, A.S.; Hess, M.L. Inhibition of accelerated coronary atherosis with dehydroepiandrosterone in the heterotopic rabbit model of cardiac transplantation. Circulation 1993, 87, 261–269. [Google Scholar] [CrossRef]
- Badria, F.A.; Dawidar, A.A.A.; Houssen, W.E.; Shier, W.T. In vitro study of flavonoids, fatty acids, and sterois on proliferatin of rat hepatic stellate cells. Z. Naturforsch. 2005, 60, 139–142. [Google Scholar]
- Han, L.; Shi, D.; Xu, F.; Yuan, Z.; Sun, J.; Shi, J. Bioactivity sterols from red alga Acanthophora spicifera boergesen. China J. Chin. Mat. Med. 2009, 34, 60–63. [Google Scholar]
- Zhu, X.; Bonet, B.; Gillenwater, H.; Knopp, R.H. Opposing effects of estrogen and progestins on LDL oxidation and vascular wall cytotoxicity: Implications for atherogenesis. Exp. Biol. Med. 1999, 222, 214–221. [Google Scholar]
- Phuruengrat, A.; Phaisansuthichol, S. Preliminary study of steroids in Sericocalyxschomburgkii (Craib) Bremek by GC-MS. Songklanakarin J. Sci. Technol. 2006, 28, 39–44. [Google Scholar]
- Tadtong, S.; Viriyaroj, A.; Vorarat, S.; Nimkultat, S.; Suksamrarn, S. Antityrosinase and antibacterial activities of mangosteen pericarp extract. J. Health Res. 2009, 23, 99–120. [Google Scholar]
- Kim, J.H.; Kim, H.J.; Park, H.W.; Youn, S.H.; Choi, D.Y.; Shin, C.S. Development of inhibitors against lipase and α-glucosidase from derivatives of monascus pigment. FEMS Microbiol. Lett. 2007, 276, 93–98. [Google Scholar] [CrossRef]
- Lyckander, I.M.; Malterud, K.E. Lipophilic flavonoids from Orthosiphon spicatus prevent oxidative inactivation of 15-lipoxygenase. Prostaglandins Leukot. Essent. Fatty Acids 1996, 54, 239–246. [Google Scholar] [CrossRef]
- Rattan, A.K.; Arad, Y. Inhibition of LDL oxidation by new estradiol receptor modulator compound LY-139478, comparative effect with other steroids. Atherosclerosis 1998, 136, 305–314. [Google Scholar] [CrossRef]
- Steinbrecher, U.P.; Parthasarathy, S.; Leake, D.S.; Witztum, J.L.; Steinberg, D. Modification of low density lipoprotein by endothelial cells involves lipid peroxidation and degradation of low density lipoprotein phospholipids. Proc. Natl. Acad. Sci. USA 1984, 81, 3883–3887. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chompoo, J.; Upadhyay, A.; Gima, S.; Fukuta, M.; Tawata, S. Antiatherogenic Properties of Acetone Extract of Alpinia zerumbet Seeds. Molecules 2012, 17, 6237-6248. https://doi.org/10.3390/molecules17066237
Chompoo J, Upadhyay A, Gima S, Fukuta M, Tawata S. Antiatherogenic Properties of Acetone Extract of Alpinia zerumbet Seeds. Molecules. 2012; 17(6):6237-6248. https://doi.org/10.3390/molecules17066237
Chicago/Turabian StyleChompoo, Jamnian, Atul Upadhyay, Shinichi Gima, Masakazu Fukuta, and Shinkichi Tawata. 2012. "Antiatherogenic Properties of Acetone Extract of Alpinia zerumbet Seeds" Molecules 17, no. 6: 6237-6248. https://doi.org/10.3390/molecules17066237
APA StyleChompoo, J., Upadhyay, A., Gima, S., Fukuta, M., & Tawata, S. (2012). Antiatherogenic Properties of Acetone Extract of Alpinia zerumbet Seeds. Molecules, 17(6), 6237-6248. https://doi.org/10.3390/molecules17066237




