Antibacterial Evaluation of Some Schiff Bases Derived from 2-Acetylpyridine and Their Metal Complexes
Abstract
:1. Introduction
2. Results and Discussion
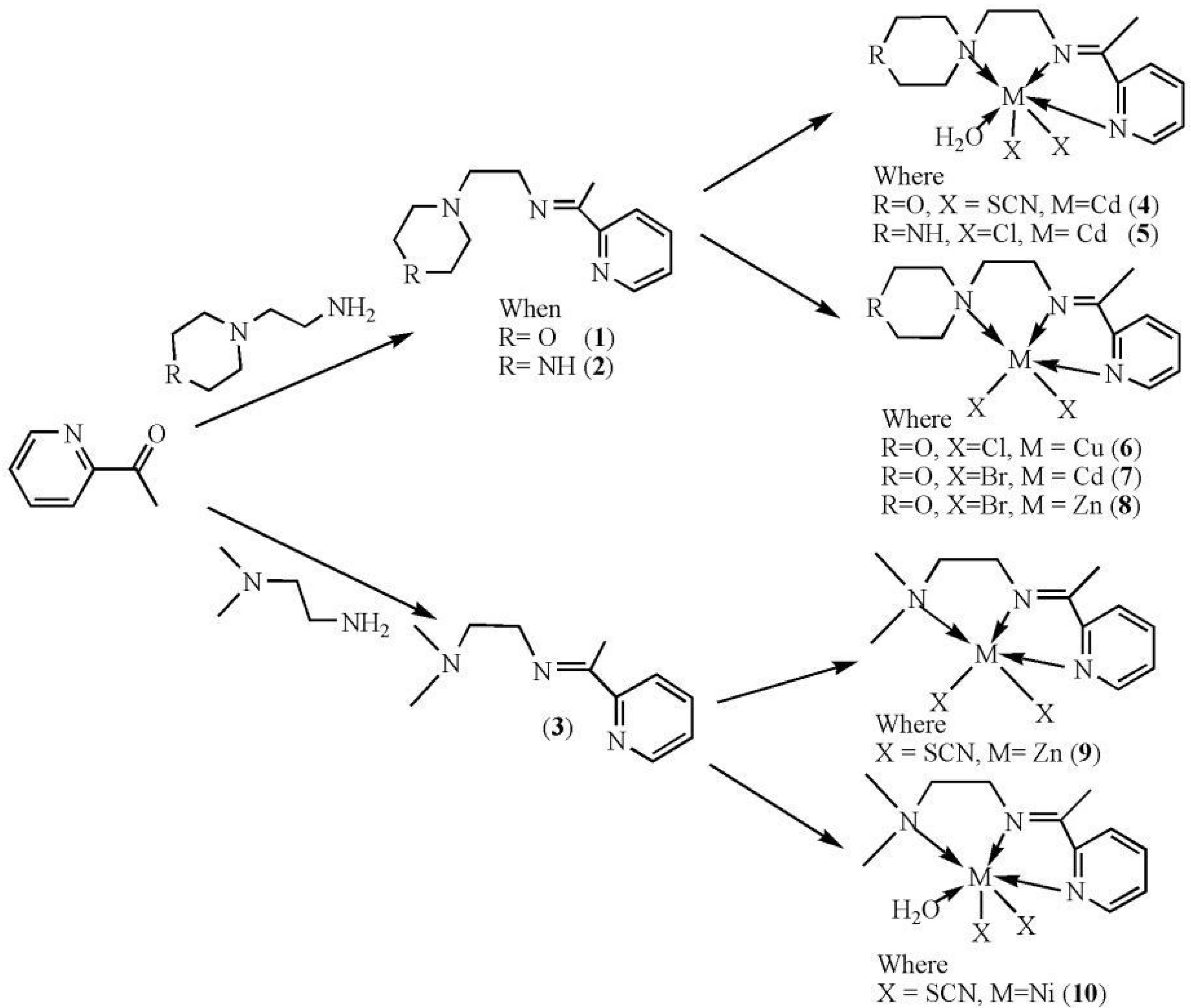
2.1. Infrared Spectra
2.2. Electronic Spectra
2.3. 1H- and 13C-NMR-NMR Spectra
2.4. X-Ray Crystal and Molecular Structure
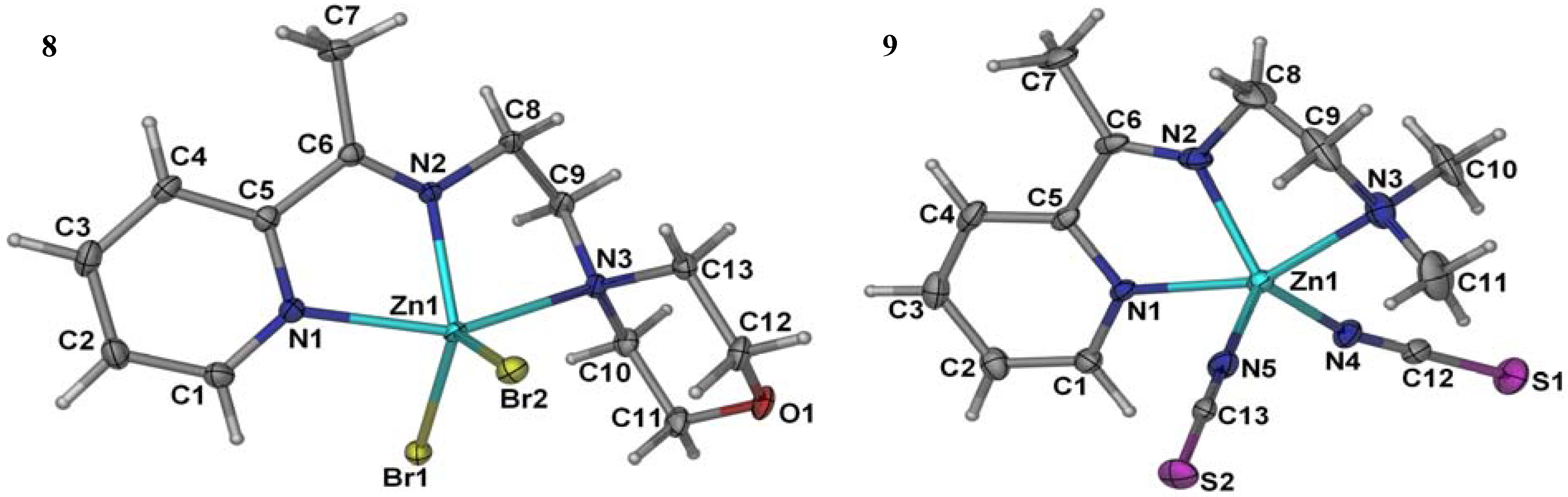
| 8 | 9 | ||||||
|---|---|---|---|---|---|---|---|
| Zn1 | Br1 | 2.4036(4) | Zn1 | N1 | 2.1752(18) | ||
| Zn1 | Br2 | 2.4172(4) | Zn1 | N2 | 2.0752(18) | ||
| Zn1 | N1 | 2.243(3) | Zn1 | N3 | 2.217(2) | ||
| Zn1 | N2 | 2.076(2) | Zn1 | N4 | 1.9681(18) | ||
| Zn1 | N3 | 2.331(2) | Zn1 | N5 | 1.9698(19) | ||
| Br1 | Zn1 | Br2 | 116.905(15) | N1 | Zn1 | N3 | 154.18(7) |
| N1 | Zn1 | Br1 | 92.48(6) | N2 | Zn1 | N1 | 75.30(7) |
| N1 | Zn1 | Br2 | 95.00(6) | N2 | Zn1 | N3 | 79.64(8) |
| N1 | Zn1 | N3 | 151.50(8) | N4 | Zn1 | N1 | 97.48(7) |
| N2 | Zn1 | Br1 | 133.79(6) | N4 | Zn1 | N2 | 133.47(8) |
| N2 | Zn1 | Br2 | 108.28(6) | N4 | Zn1 | N3 | 95.22(7) |
| N2 | Zn1 | N1 | 73.92(9) | N4 | Zn1 | N5 | 113.13(8) |
| N2 | Zn1 | N3 | 79.37(9) | N5 | Zn1 | N1 | 96.56(7) |
| N3 | Zn1 | Br1 | 99.01(6) | N5 | Zn1 | N2 | 113.35(8) |
| N3 | Zn1 | Br2 | 102.71(5) | N5 | Zn1 | N3 | 98.97(8) |
| C1 | N1 | Zn1 | 128.0(2) | C1 | N1 | Zn1 | 126.71(14) |
| C5 | N1 | Zn1 | 113.25(18) | C5 | N1 | Zn1 | 114.26(14) |
| C6 | N2 | Zn1 | 120.85(19) | C6 | N2 | Zn1 | 120.19(15) |
| C8 | N2 | Zn1 | 115.86(19) | C8 | N2 | Zn1 | 114.51(17) |
| C9 | N3 | Zn1 | 100.28(17) | C9 | N3 | Zn1 | 100.2(2) |
| C13 | N3 | Zn1 | 115.48(17) | C10 | N3 | Zn1 | 112.0(2) |
| C6 | N2 | C8 | 122.7(2) | C11 | N3 | Zn1 | 107.3(3) |
| C8 | N2 | Zn1 | 115.86(19) | C9' | N3 | Zn1 | 104.0(2) |
| C9 | N3 | C13 | 109.5(2) | C11 | N3 | Zn1 | 112.3(2) |
| C1 | N1 | C5 | 118.7(3) | C12 | N4 | Zn1 | 162.3(2) |
| C10 | N3 | C13 | 107.5(2) | C13 | N5 | Zn1 | 175.5(2) |
| 8 | 9 | |
|---|---|---|
| Empirical formula | C13H19Br2N3OZn | C13H17N5S2Zn |
| Formula weight | 458.50 | 372.81 |
| Temperature/K | 100(2) | 100(2) |
| Crystal system | monoclinic | monoclinic |
| Space group | P21/n | P21/n |
| a/Å | 9.8290(2) | 13.7663(2) |
| b/Å | 14.0218(2) | 9.4949(2) |
| c/Å | 12.1371(2) | 13.8089(2) |
| β/° | 106.9180(10) | 109.9460(10) |
| Volume/Å3 | 1600.35(5) | 1696.68(5) |
| Z | 4 | 4 |
| ρcalcmg/mm3 | 1.903 | 1.459 |
| m/mm−1 | 6.527 | 1.693 |
| F(000) | 904 | 768 |
| Crystal size/mm3 | 0.33 × 0.25 × 0.11 | 0.35 × 0.31 × 0.19 |
| 2Θ range for data collection | 4.56 to 54° | 5.14 to 54° |
| Reflections collected | 12535 | 15059 |
| Independent reflections | 3496[R(int) = 0.0351] | 3707[R(int) = 0.0232] |
| Data/restraints/parameters | 3496/0/182 | 3707/14/225 |
| Goodness-of-fit on F2 | 1.028 | 1.015 |
| Final R indexes [I >= 2σ (I)] | R1 = 0.0271, wR2 = 0.0595 | R1 = 0.0296, wR2 = 0.0714 |
| Final R indexes [all data] | R1 = 0.0360, wR2 = 0.0624 | R1 = 0.0402, wR2 = 0.0772 |
| Largest diff. peak/hole/e Å−3 | 0.679/−0.466 | 0.455/−0.428 |
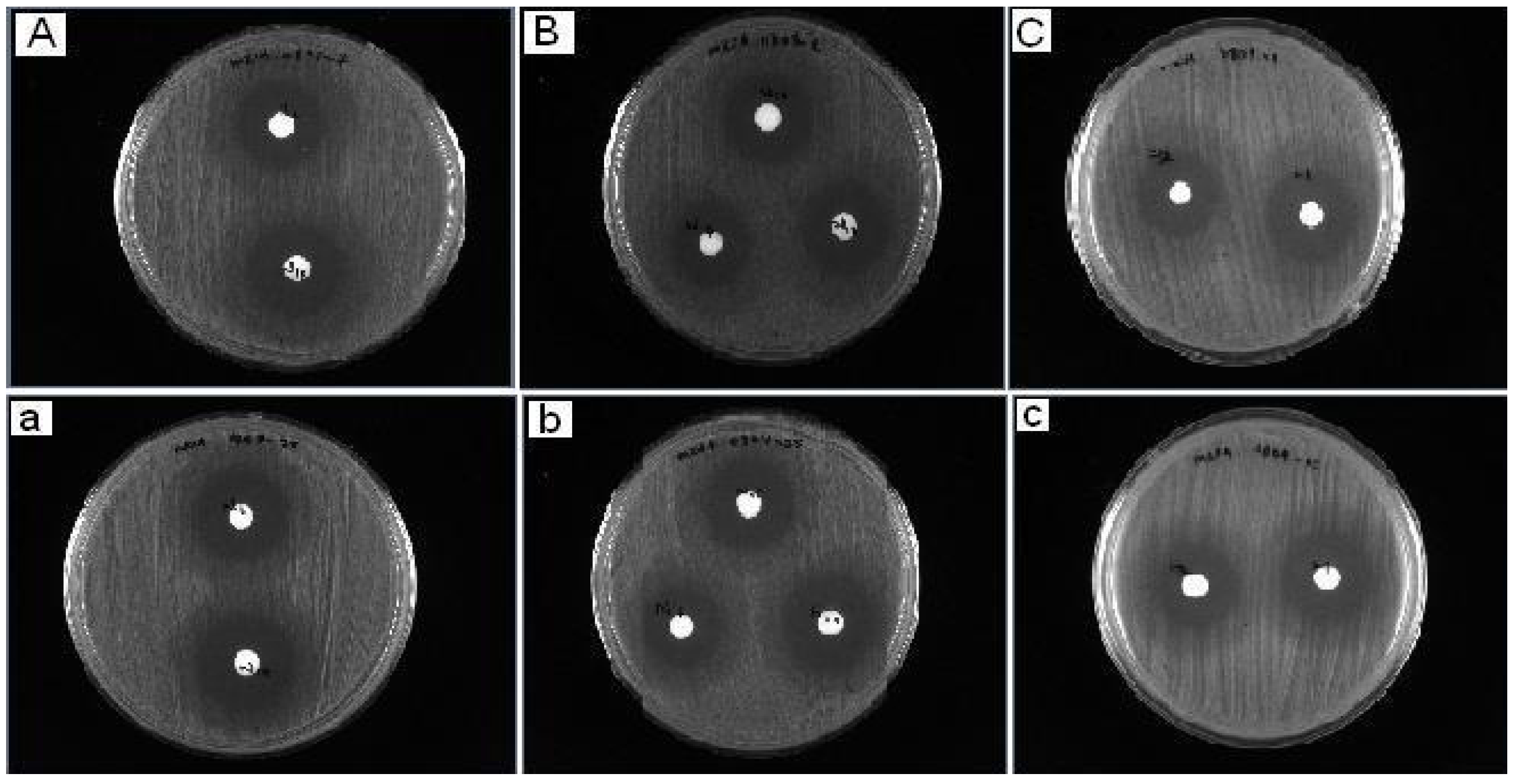
2.5. Antibacterial Activity Results
| Compound | MRSA 0804-25 | MRSA 0807-7 | AC 0612-7 | AC 0903-21 | KB 71 | KB 83 | PA 45 | PA 104 |
|---|---|---|---|---|---|---|---|---|
| 1 | 6 | 6 | 0 | 0 | 0 | 0 | 5 | 6 |
| 2 | 5 | 5 | 0 | 0 | 0 | 0 | 5 | 5 |
| 3 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 4 | 21 | 22 | 7 | 7 | 0 | 0 | 8 | 8 |
| 5 | 24 | 23 | 9 | 8 | 0 | 0 | 11 | 19 |
| 6 | 22 | 23 | 9 | 8 | 0 | 0 | 10 | 10 |
| 7 | 23 | 22 | 10 | 10 | 0 | 0 | 8 | 8 |
| 8 | 21 | 20 | 10 | 9 | 0 | 0 | 9 | 9 |
| 9 | 23 | 23 | 9 | 8 | 0 | 0 | 10 | 9 |
| 10 | 22 | 22 | 7 | 7 | 0 | 0 | 10 | 9 |
| Control (DMSO) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Minimum inhibitory concentration (MIC; µg/mL)/minimum bactericidal concentration (MBC; µg/mL) | ||||||||
|---|---|---|---|---|---|---|---|---|
| MRSA 0804-25 | MRSA 0807-7 | AC 0612-7 | AC 0903-21 | KB 71 | KB 83 | PA 45 | PA 104 | |
| 1-3 | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. | n.d. |
| 4 | 0.7/2.9 | 1.5/2.9 | 93.8/750 | 93.8/750 | 375/750 | 375/750 | 187.5/750 | 187.5/750 |
| 5 | 0.7/2.9 | 0.7/2.9 | 93.8/750 | 93.8/750 | 187.5/750 | 187.5/750 | 187.5/750 | 187.5/750 |
| 6 | 1.5/5.9 | 1.5/5.9 | 93.8/375 | 93.8/375 | 375/750 | 375/750 | 187.5/750 | 187.5/750 |
| 7 | 1.5/5.9 | 1.5/5.9 | 93.8/375 | 93.8/375 | 187.5/750 | 187.5/750 | 187.5/750 | 187.5/750 |
| 8 | 1.5/5.9 | 1.5/2.9 | 93.8/750 | 93.8/750 | 375/750 | 375/750 | 187.5/750 | 187.5/750 |
| 9 | 1.5/5.9 | 1.5/23.4 | 187.5/750 | 187.5/750 | 375/750 | 375/750 | 187.5/750 | 187.5/750 |
| 10 | 2.9/46.9 | 2.9/46.9 | 187.5/750 | 187.5/750 | 375/750 | 375/750 | 187.5/750 | 187.5/750 |
| DMSO | 375/>375 | 375/>375 | 375/>375 | 375/>375 | 375/>375 | 375/>375 | 375/>375 | 375/>375 |
| Compounds | MRSA 0805-21 | MRSA 0806-1 | MRSA 0807-1 | MRSA 0807-19 | MRSA 0808-35 | MRSA 0809-15 | MRSA 0809-25 | MRSA 0809-38 |
|---|---|---|---|---|---|---|---|---|
| 4 | 23 | 24 | 0 | 23 | 0 | 22 | 22 | 21 |
| 5 | 23 | 24 | 0 | 25 | 0 | 23 | 23 | 23 |
| 6 | 24 | 22 | 0 | 22 | 0 | 21 | 21 | 22 |
| 7 | 24 | 20 | 0 | 21 | 0 | 19 | 20 | 20 |
| 8 | 25 | 23 | 0 | 24 | 0 | 21 | 21 | 22 |
| 9 | 23 | 23 | 0 | 22 | 0 | 23 | 24 | 23 |
| 10 | 22 | 22 | 0 | 22 | 0 | 22 | 22 | 22 |
| (DMSO) | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| Strain No | E | CN | LZD | MUP | RD | DA | TEC | CIP | NET | TE | VA | FD | OX | SXT |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MRSA 0805-21 | R | R | S | S | S | S | S | R | R | R | S | S | R | R |
| MRSA 0806-1 | R | R | S | S | S | S | S | R | R | R | S | S | R | R |
| MRSA 0807-1 | R | R | S | S | S | S | S | R | R | R | S | S | R | R |
| MRSA 0807-19 | R | R | S | S | S | S | S | R | I | S | S | S | R | S |
| MRSA 0808-35 | R | R | S | S | S | S | S | I | I | R | S | S | R | R |
| MRSA 0809-15 | R | R | S | S | S | S | S | R | R | R | S | S | R | R |
| MRSA 0809-24 | R | R | S | S | S | S | S | R | R | R | S | S | R | R |
| MRSA 0809-38 | R | R | S | S | S | S | S | R | R | R | S | S | R | S |
| MRSA 0807-7 | R | R | S | S | S | S | S | R | I | R | S | S | R | R |
| MRSA 0804-25 | R | R | S | S | S | S | S | R | S | S | S | S | R | S |
3. Experimental
General










3.1. X-Ray Structure Analyses
4. Pharmacology
4.1. Bacterial Strains
4.1.1. Antimicrobial Testing
4.1.2. MIC Determination
4.1.3. MBC Determination
5. Conclusions
Supplementary Data
Acknowledgments
Conflict of Interest
References and Notes
- Prakash, A.; Singh, B.K.; Bhojak, N.; Adhikari, D. Synthesis and characterization of bioactive zinc(II) and cadmium(II) complexes with new Schiff bases derived from 4-nitrobenzaldehyde and acetophenone with ethylenediamine. Spectrochim. Acta 2010, 76, 356–362. [Google Scholar] [CrossRef]
- Sridhar, S.K.; Pandeya, S.N.; Stables, J.P.; Ramesh, A. Anticonvulsant activity of hydrazones, Schiff and Mannich bases of Isatin derivatives. Eur. J. Pharm. Sci. 2002, 16, 129–132. [Google Scholar] [CrossRef]
- Bharti, S.K.; Nath, G.; Tilak, R.; Singh, S.K. Synthesis, anti-bacterial and anti-fungal activities of some novel Schiff bases containing 2,4-disubstituted thiazole ring. Eur. J. Med. Chem. 2010, 45, 651–660. [Google Scholar] [CrossRef]
- Kalagouda, B.G.; Manjula, S.P.; Ramesh, S.V.; Rashmi, V.S.; Siddappa, A.P. X-ray crystal structure of the N-(2-hydroxy-1-naphthalidene)phenylglycineSchiff base. Synthesis and characterization of its transition metal complexes. Trans. Met. Chem. 2006, 31, 580–585. [Google Scholar] [CrossRef]
- Cukurovali, A.; Yilmaz, İ.; Kirbag, S. Spectroscopic characterization and biological activity of salicylaldehyde thiazolyl hydrazone ligands and their metal complexes. Trans. Met. Chem. 2006, 31, 207–213. [Google Scholar] [CrossRef]
- Tansir, A.; Nahid, N.; Shadma, P. Synthesis, characterization and antimicrobial studies of newly developed polymeric schiff base and its metal-polychelates. J. Coord. Chem. 2008, 61, 1963–1972. [Google Scholar] [CrossRef]
- Manabu, F.; Hisanobu, W.; Takayuki, M.; Toshiyuki, S. Preparation of 14-, 18-, and 22-membered tetraaza macrocycles and their complexing ability for copper(ii) and nickel(II) ions. Bull. Chem. Soc. Jpn. 1990, 63, 3443–3449. [Google Scholar] [CrossRef]
- Pandeya, S.N.; Sriram, D.; Nath, G.; DeClercq, E. Synthesis, antibacterial, antifungal and anti HIV activities of Schiff and Mannich bases derived from isatin derivatives and N-[4-(4-chlorophenyl) thiazol-2-yl] thiosemicarbazide. Eur. J. Pharm. Sci. 1999, 9, 25–31. [Google Scholar] [CrossRef]
- Zhang, J.A.; Pan, M.; Zhang, J.Y.; Kang, B.S.; Su, C.Y. Syntheses, structures and bioactivities of cadmium(II) complexes with a tridentate heterocyclic N- and S-ligand. Inorg. Chim. Acta 2009, 362, 3519–3525. [Google Scholar] [CrossRef]
- Mandal, S.; Karmakar, T.K.; Ghosh, A.; Fleck, M.; Bandyopadhyay, D. Synthesis, crystal structure and antibacterial activity of a group of mononuclear manganese(II) Schiff base complexes. Polyhedron 2011, 30, 790–795. [Google Scholar] [CrossRef]
- Yusnita, J.; Puvaneswary, S.; Ali, H.M.; Robinson, W.T.; Lin, T.K. Synthesis, structural characterization and antibacterial activity of 2,6-diacetylpyridine bis(benzenesulfonohydrazide) Schiff bases and their copper(II) complexes. Polyhedron 2009, 28, 3050–3054. [Google Scholar] [CrossRef]
- Pignatello, R.; Panico, A.; Mazzone, P.; Pinizzotto, M.R.; Garozzo, A.; Fumeri, P.M. Schiff bases of N-hydroxy-N′-aminoguanidines as antiviral, antibacterial and anticancer agents. Eur. J. Med. Chem. 1994, 29, 781–785. [Google Scholar] [CrossRef]
- Ceyhan, G.; Çelik, C. Antioxidant, electrochemical, thermal, antimicrobial and alkane oxidation properties of tridentate Schiff base ligands and their metal complexes. Spectrochim. Acta 2011, 81, 184–198. [Google Scholar] [CrossRef]
- Tajudeen, S.S.; Radha, E. Synthesis, characterization and antimicrobial activity of transition metal complexes of schiff base derivatives from isonicotinic acid hydrazide. Asian J. Chem. 2009, 21, 313–316. [Google Scholar]
- Bagihalli, G.B.; Avaji, P.G. Synthesis, spectral characterization, in vitro antibacterial, antifungal and cytotoxic activities of Co(II), Ni(II) and Cu(II) complexes with 1,2,4-triazole Schiff bases. Eur. J. Med. Chem. 2008, 43, 2639–2649. [Google Scholar] [CrossRef]
- Ispir, E.; Toroglu, S.; Kayraldrz, A. Syntheses, characterization, antimicrobial and genotoxic activities of new Schiff bases and their complexes. Transit. Met Chem. 2008, 33, 53–960. [Google Scholar]
- Guidos, R.J. The 10×'20 Initiative: Pursuing a Global Commitment to Develop 10 New Antibacterial Drugs by 2020. Am. Clin. Infect. Dis. 2010, 50, 1081–1083. [Google Scholar] [CrossRef]
- Esposito, S.; Leone, S. Antimicrobial treatment for Intensive Care Unit (ICU) infections including the role of the infectious disease specialist. Int. J. Antimicrob. Agents 2007, 29, 494–500. [Google Scholar] [CrossRef]
- Noskin, G.A.; Siddiqui, F.; Stosor, V.; Hacek, D.; Peterson, L.R. In vitro activity of linezolid against important Gram-positive bacteria pathogens including vancomycin-resistant enterococci. Antimicrob. Agent Chemother. 1999, 43, 2059–2062. [Google Scholar]
- Prystowsky, J.; Siddiqui, F.; Chosay, J.; Shinabarger, D.L.; Millichap, J.; Peterson, L.R.; Noskin, G.A. Resistance to linezolid: Characterization of mutations in rRNA and comparison of their occurrences in vancomycin-resistant enterococci. Antimicrob. Agent Chemother. 2001, 45, 2154–2156. [Google Scholar] [CrossRef]
- Raparti, V.; Chitre, T.; Bothara, K.G.; Kumar, V.; Dangre, S.; Khachane, C.; Gore, S.; Deshmane, B. Novel 4-(morpholin-4-yl)-N′-(arylidene)benzohydrazides: Synthesis, antimycobacterial activity and QSAR invest- tiga-tions. Eur. J. Med. Chem. 2009, 44, 3954–3960. [Google Scholar] [CrossRef]
- Laskar, I.R.; Maji, T.K.; Das, D.; Lu, T.H.; Wong, W.-T.; Okamoto, K.-i.; Chaudhuri, N.R. Syntheses, characterisation and solid state thermal studies of 1-(2-aminoethyl)piperidine (L), 1-(2-aminoethyl)pyrrolidine (L) and 4-(2-aminoethyl)morpholine (L.) complexes of nickel(II): X-ray single crystal structure analyses of trans-[NiL2(CH3CN)2](ClO4)2, trans-[NiL2(NCS)2] and trans-[NiL-2(NCS)2]. Polyhedron 2001, 20, 2073–2082. [Google Scholar] [CrossRef]
- Raman, N.; Selvan, A.; Sudharsan, S. Metallation of ethylenediamine based Schiff base with biologically active Cu(II), Ni(II) and Zn(II) ions: Synthesis, spectroscopic characterization, electrochemical behaviour, DNA binding, photonuclease activity and in vitro antimicrobial efficacy. Spectrochim. Acta A 2011, 79, 873–883. [Google Scholar] [CrossRef]
- Khan, N.U.; Pandya, N.; Prathap, K.J.; Kureshy, R.I.; Abdi, S.H.; Mishra, S.; Bajaj, H.C. Chiral discrimination asserted by enantiomers of Ni (II), Cu (II) and Zn (II) Schiff base complexes in DNA binding, antioxidant and antibacterial activities. Spectrochim. Acta 2011, 81, 199–208. [Google Scholar] [CrossRef]
- Nakamoto, K. Infrared and Raman Spectra of Inorganic and Coordination Compounds; Wiley: New York, NY, USA, 1978. [Google Scholar]
- Bhowmik, P.; Chattopadhyyay, S.; Drew, M.G.B.; Diaz, C.; Ghosh, A. Synthesis, structure and magnetic properties of mono- and di-nuclear nickel(II) thiocyanate complexes with tridentate N3 donor Schiff bases. Polyhedron 2010, 29, 2637–2642. [Google Scholar] [CrossRef]
- Banerjee, S.; Wu, B.; Lassahn, P.G.; Janiak, C.; Ghosh, A. Synthesis, structure and bonding of cadmium(II) thiocyanate systems involving nitrogen containing ligands of different denticity. Inorg. Chim. Acta 2005, 358, 535–544. [Google Scholar] [CrossRef]
- Chattopadhyay, S.; Drew, M.G.B.; Ghosh, A. Binuclear complexes of M(II) thiocyanate (M = Ni and Cu) containing a tridentate Schiff base ligand: synthesis, structural diversity and magnetic properties. Eur. J. Inorg. Chem. 2005, 12, 2376–2383. [Google Scholar]
- Mohamed, M.; Hapipah, M.A.; Mahmood, A.A.; Robinson, T.W. Synthesis, structural characterization, and anti-ulcerogenic activity of Schiff base ligands derived from tryptamine and 5-chloro, 5-nitro, 3,5-ditertiarybutyl salicylaldehyde and their nickel(II), copper(II), and zinc(II) complexes. Polyhedron 2009, 28, 3993–3998. [Google Scholar] [CrossRef]
- Lakshmi, B.; Avaji, P.G.; Shivananda, K.N.; Naggella, P.; Manohar, S.H.; Mahendra, K.N. Synthesis, spectral characterization and in vitro microbiological evaluation of novel glyoxal, biacetyl and benzil bis-hydrazone macrocyclic Schiff bases and their Co(II), Ni(II) and Cu(II) complexs. Polyhedron 2011, 30, 1507–1515. [Google Scholar] [CrossRef]
- Deoghoria, S.; Mostafa, G.; Lu, T.H.; Chandra, S.K. Synthesis, characterisation and properties of manganese(II) complexes having pseudohalide coordination: X-ray crystal structure of an unusually distorted hexacoordinated [MnL(NCS)](ClO4) species (L = pentadentate Schiff base ligand). Ind. J. Chem. 2004, 43A, 329–332. [Google Scholar]
- Shahabadi, N.; Kashanian, S. DNA binding and DNA cleavage studies of a water soluble cobalt(II) complex containing dinitrogen Schiff base ligand: The effect of metal on the mode of binding. Eur. J. Med. Chem. 2010, 45, 4239–4245. [Google Scholar] [CrossRef]
- Chen, W.; Li, Y.; Cui, Y.; Zhang, X. Synthesis, molecular docking and biological evaluation of Schiff base transition metal complexes as potential urease inhibitors. Eur. J. Med. Chem. 2010, 45, 4473–4478. [Google Scholar] [CrossRef]
- Creaven, B.S.; Duff, B.; Egan, D.A.; Kavanagh, K.; Rosair, G.; Thangella, V.R.; Walsh, M. Anticancer and antifungal activity of copper(II) complexes of quinolin-2(1H)-one-derived Schiff bases. Inorg. Chim. Acta 2010, 363, 4048–4058. [Google Scholar] [CrossRef]
- Raman, N.; Jeyamurugan, R.; Senthilkumar, R.; Rajkapoor, B.; Franzblau, S.G. In vivo and in vitro evaluation of highly specific thiolate carrier group copper(II) and zinc(II) complexes on Ehrlich ascites carcinoma tumor model. Eur. J. Med. Chem. 2010, 45, 5438–5451. [Google Scholar] [CrossRef]
- Ali, M.A.; Mirza, A.H.; Tan, A.L.; Bujang, F.H.; Hamid, M.H.S.A.; Bernhardt, P.V. Preparation and structural characterization of nickel(II), cobalt(II), zinc(II) and tin(IV) complexes of the isatin Schiff bases of S-methyl and S-benzyldithiocarbazates. Polyhedron 2008, 25, 977. [Google Scholar]
- Ceyhan, G.; Çelik, C.; Uruş, S.; Demirtaş, İ.; Elmastaş, M.; Tümer, M. Antioxidant, electrochemical, thermal, antimicrobial and alkane oxidation properties of tridentate Schiff base ligands and their metal complexes. Spectrochim. Acta A Mol. Biomol. Spectr. 2011, 81, 184–198. [Google Scholar] [CrossRef]
- Hisham, N.A.I.; Gwaram, N.S.; Khaledi, H.; Ali, H.M. Dichlorido{2-morpholino-N-[1-(2-pyridyl)ethylidene] ethanamine- 3N,N',N''}zinc(II). Acta Crystallogr. 2011, 67, 55. [Google Scholar]
- Addison, A.W.; Rao, T.N.; Reedijk, J.; Rijn, V.J.; Verschoor, G.C. Synthesis, structure, and spectroscopic properties of copper(II) compounds containing nitrogen-sulphur donor ligands; the crystal and molecular structure of aqua[1,7-bis(N-methylbenzimidazol-2′-yl)-2,6-dithiaheptane]copper(II) perchlorate. J. Chem. Soc. Dalton Trans. 1984, 7, 1349–1356. [Google Scholar]
- You, Z.L.; Chi, J.Y. Synthesis, crystal structures and antibacterial activities of two Schiff base zinc(II) complexes. Synth. React. Inorg. Met.-Org., Nano-Met. Chem. 2006, 36, 713–717. [Google Scholar] [CrossRef]
- Zakrzewski, G.; Lingafelter, E.C. The crystal and molecular structure of dibromo-1- (2-pyridyl)-2,5-diaza-5-methyl-hexa-1-enezinc(II). Inorg. Chim. Acta 1970, 4, 251–257. [Google Scholar] [CrossRef]
- Cai, B.H. (2-Morpholinoeth-yl)(2-pyridylmethyl-ene)amine]dithio-cyanato-zinc(II). Acta Crystallgr. 2009, 65, 142. [Google Scholar]
- Chen, G.; Bai, Z.P.; Qu, S.J. N,N'-Dimethyl-N''-(2-pyridylmethylene)ethane-1,2-diamine] dithiocyanato zinc(II). Acta Crystallogr. 2005, 61, 2483. [Google Scholar]
- Gwaram, N.S.; Khaledi, H.; Ali, H.M. catena-Poly-N,N-dimethyl-N'-[1-(pyridin-2-yl)ethylidene]ethane-1,2-diamine-κ3N,N',N''}(thiocyanato-κN)cadmium]-μ-thiocyanato-κ2S:N. Acta Crystallogr. 2011, 67, 480. [Google Scholar]
- Chohan, Z.H.; Arif, M.; Akhtar, M.A.; Supuran, C.T. Metal-based antibacterial and antifungal agents: Synthesis, characterization, and in vitro biological evaluation of Co(II), Cu(II), Ni(II), and Zn(II) complexes with amino acid-derived compounds. Bioinorg. Chem. Appl. 2006, 83131. [Google Scholar]
- Chohan, Z.H.; Scozzafava, A.; Supuran, C.T. Zinc complexes of benzothiazole-derived Schiff bases with antibacterial activity. J. Enzyme Inhib. Med. Chem. 2003, 18, 259–263. [Google Scholar] [CrossRef]
- Bruker APEX2 and SAINT. Bruker AXS Inc.: Madison, WI, USA, 2007.
- Sheldrick, G.M. A short history of SHELX. Acta Crystallogr. A 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Barbour, L.J. X-Seed-A software tool for supramolecular crystallography. J. Supramol. Chem. 2001, 1, 189–191. [Google Scholar] [CrossRef]
- Clinical and Laboratory Standards Institute (CLSI), Performance Standard for Antimicrobial Susceptibility Testing; Twenty-First Information Supplement; CLSI document M100-S21; CLSI: Wayne, PA, USA, 2011.
- Sample Availability: Samples of the compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gwaram, N.S.; Ali, H.M.; Khaledi, H.; Abdulla, M.A.; Hadi, A.H.A.; Lin, T.K.; Ching, C.L.; Ooi, C.L. Antibacterial Evaluation of Some Schiff Bases Derived from 2-Acetylpyridine and Their Metal Complexes. Molecules 2012, 17, 5952-5971. https://doi.org/10.3390/molecules17055952
Gwaram NS, Ali HM, Khaledi H, Abdulla MA, Hadi AHA, Lin TK, Ching CL, Ooi CL. Antibacterial Evaluation of Some Schiff Bases Derived from 2-Acetylpyridine and Their Metal Complexes. Molecules. 2012; 17(5):5952-5971. https://doi.org/10.3390/molecules17055952
Chicago/Turabian StyleGwaram, Nura Suleiman, Hapipah Mohd Ali, Hamid Khaledi, Mahmood Ameen Abdulla, A. Hamid A. Hadi, Thong Kwai Lin, Chai Lay Ching, and Cher Lin Ooi. 2012. "Antibacterial Evaluation of Some Schiff Bases Derived from 2-Acetylpyridine and Their Metal Complexes" Molecules 17, no. 5: 5952-5971. https://doi.org/10.3390/molecules17055952
APA StyleGwaram, N. S., Ali, H. M., Khaledi, H., Abdulla, M. A., Hadi, A. H. A., Lin, T. K., Ching, C. L., & Ooi, C. L. (2012). Antibacterial Evaluation of Some Schiff Bases Derived from 2-Acetylpyridine and Their Metal Complexes. Molecules, 17(5), 5952-5971. https://doi.org/10.3390/molecules17055952