Antimicrobial and Antioxidant Activities of Essential Oils of Satureja thymbra Growing Wild in Libya
Abstract
:1. Introduction
2. Results and Discussion
| Constituents | a KIE | b KIL | % | |
|---|---|---|---|---|
| α-thujene | 917.7 | 924 | 1.55 ± 0.02 | |
| α-pinene | 923.4 | 932 | 1.48 ± 0.03 | |
| camphene | 937.4 | 946 | 0.69 ± 0.01 | |
| sabinene | 963.6 | 969 | 0.14 ± 0.00 | |
| β-pinene | 965.7 | 974 | 0.70 ± 0.01 | |
| myrcene | 983.2 | 988 | 1.76 ± 0.01 | |
| α-phellandrene | 995.4 | 1002 | 0.28 ± 0.00 | |
| δ3-carene | 1001.2 | 1008 | 0.10 ± 0.01 | |
| α-terpinene | 1007.8 | 1014 | 3.26 ± 0.02 | |
| p-cymene | 1016.1 | 1020 | 7.17 ± 0.05 | |
| sylvestrene | 1019.5 | 1025 | 0.78 ± 0.01 | |
| cis-β-ocimene | 1030.6 | 1032 | 0.52 ± 0.00 | |
| trans-β-ocimene | 1040.6 | 1044 | 0.81 ± 0.01 | |
| γ-terpinene | 1051.5 | 1054 | 39.23 ± 0.27 | |
| cis-sabinene hydrate | 1060.6 | 1065 | 0.23 ± 0.01 | |
| terpinolene | 1079.1 | 1086 | 0.11 ± 0.00 | |
| linalool | 1051.5 | 1095.6 | 0.34 ± 0.00 | |
| borneol | 1157.2 | 1165 | 0.82 ± 0.00 | |
| terpinen-4-ol | 1169.4 | 1174 | 0.36 ± 0.01 | |
| α-terpineol | 1185.3 | 1186 | 0.05 ± 0.01 | |
| neo-dihydro carveol | n/a | 1193 | 0.06 ± 0.04 | |
| carvacrol methyl ether | 1235.6 | 1241 | 3.33 ± 1.88 | |
| thymol | 1291.0 | 1289 | 25.16 ± 1.95 | |
| carvacrol | 1297.9 | 1298 | 4.18 ± 0.23 | |
| thymyl acetate | 1346.9 | 1349 | 0.21 ± 0.21 | |
| β-caryophyllene | 1407.1 | 1417 | 2.76 ± 0.03 | |
| aromadendrene | 1426.5 | 1439 | 0.36 ± 0.01 | |
| α-humulene | 1441.3 | 1452 | 0.15 ± 0.00 | |
| alloaromadendrene | 1448.4 | 1458 | 0.22 ± 0.22 | |
| cis-β-guaiene | 1484.6 | 1492 | 1.56 ± 0.02 | |
| spathulenol | 1565.9 | 1577 | 0.95 ± 0.03 | |
| caryophyllene oxide | 1570.0 | 1582 | 0.32 ± 0.01 | |
| cis-α-bergamotene | c n/a | 0.17 ± 0.16 | ||
| Total | 99.81% | |||
| Yield (v/w) % | 4.9 | |||
| Number of constituents | 33 | |||
| Monoterpene hydrocarbons | 58.57% | |||
| Oxygenated monoterpenes | 31.21% | |||
| Sesquiterpene hydrocarbons | 5.23% | |||
| Oxygenated hydrocarbons | 1.27% | |||
| Other | 3.53% | |||
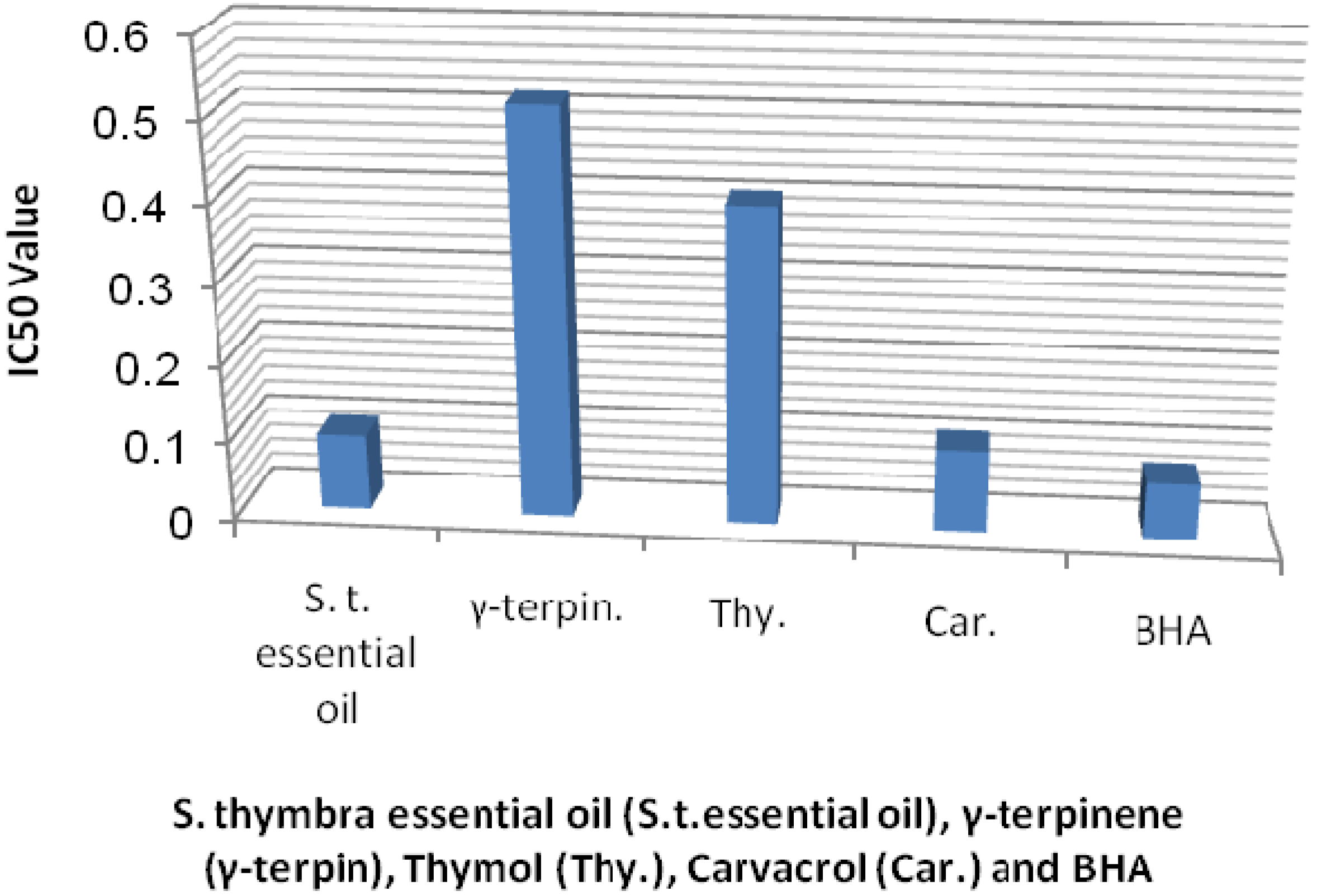
| Bacteria | S. thymbra | γ-terpenene | thymol | carvacrol | streptomycin | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | MIC | MBC | |
| Gram (+) bacteria | ||||||||||
| Bacillus cereus | 0.05 | 0.1 | 0.05 | 0.07 | 0.025 | 0.05 | 0.0125 | 0.025 | 0.0005 | 0.0005 |
| Micrococcus flavus | 0.001 | 0.002 | 0.05 | 0.07 | 0.025 | 0.05 | 0.0025 | 0.005 | 0.0005 | 0.001 |
| Staphylococcus aureus | 0.05 | 0.1 | 0.05 | 0.1 | 0.025 | 0.05 | 0.025 | 0.05 | 0.001 | 0.001 |
| Listeria monocytogenes | 0.1 | 0.2 | 0.1 | 0.2 | 0.1 | 0.1 | 0.05 | 0.05 | 0.001 | 0.002 |
| Gram (−) bacteria | ||||||||||
| Escherichia coli | 0.05 | 0.1 | 0.15 | 0.2 | 0.1 | 0.15 | 0.05 | 0.05 | 0.0005 | 0.001 |
| Pseudomonas aeruginosa | 0.05 | 0.1 | 0.15 | 0.3 | 0.1 | 0.15 | 0.05 | 0.1 | 0.001 | 0.002 |
| Proteus mirabilis | 0.05 | 0.1 | 0.2 | 0.3 | 0.01 | 0.15 | 0.05 | 0.1 | 0.001 | 0.002 |
| Salmonella typhimurium | 0.05 | 0.1 | 0.1 | 0.2 | 0.05 | 0.1 | 0.05 | 0.05 | 0.001 | 0.001 |
| Fungi | S. thymbra | γ-terpenene | thymol | carvacrol | Bifonazole | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | MIC | MFC | |
| Penicillium funiculosum | 0.025 | 0.05 | 0.025 | 0.07 | 0.0125 | 0.025 | 0.0125 | 0.0125 | 2.0 | 2.5 |
| Penicillium ochrochloron | 0.001 | 0.001 | 0.025 | 0.07 | 0.025 | 0.025 | 0.0025 | 0.005 | 1.5 | 2.0 |
| Aspergillus fumigatus | 0.025 | 0.05 | 0.05 | 0.07 | 0.025 | 0.05 | 0.025 | 0.025 | 1.5 | 2.0 |
| Aspergillus niger | 0.025 | 0.05 | 0.02 | 0.03 | 0.01 | 0.02 | 0.025 | 0.025 | 1.5 | 2.0 |
| Aspergillus flavus | 0.025 | 0.05 | 0.02 | 0.03 | 0.01 | 0.01 | 0.005 | 0.01 | 1.5 | 2.0 |
| Aspergillus ochraceus | 0.025 | 0.05 | 0.015 | 0.02 | 0.01 | 0.015 | 0.005 | 0.01 | 1.5 | 2.0 |
| Candida albicans | 0.025 | 0.1 | 0.05 | 0.1 | 0.05 | 0.05 | 0.025 | 0.05 | 1.5 | 2.0 |
| Trichoderma viride | 0.025 | 0.05 | 0.05 | 0.07 | 0.01 | 0.01 | 0.005 | 0.01 | 2.0 | 2.5 |
3. Experimental
3.1. Plant Material
3.2. Isolation of the Essential Oil
3.3. Gas Chromatography and Gas Chromatography-Mass Spectrometry Analysis
3.4. Antioxidant Activity (DPPH Assay)
3.5. Antifungal Activity
3.6. Antibacterial Activity
4. Conclusions
Acknowledgments
- Sample Availability: Samples of the essential oils are available from Abdulhamid Giweli.
References and Notes
- Jafri, S.M.H.; El-Gadi, A. Flora of Libya; Al Faateh University, Faculty of Science, Department of Botany: Tripoli, Libya, 1985; Volume 118, pp. 88–92. [Google Scholar]
- Ball, P.W.; Getliffe, F.M. Satureja L. In Flora Europaea; Tutin, T.G., Heywood, V.H., Burges, N.A., Moore, D.M., Valentine, D.H., Walters, S.M., Webb, D.A., Eds.; Cambridge University Press: London, UK, 1972; Volume 3, pp. 163–165. [Google Scholar]
- Rios, J.L.; Recico, M.C. Medicinal plants and antimicrobial activity. J. Ethnopharmacol. 2005, 100, 80–84. [Google Scholar] [CrossRef]
- Capone, W.; Mascia, C.; Spanedda, L.; Chiappini, M. Chemical composition and antibacterial activity of the essential oil from sardinian Satureja thymbra. Fitoterapia 1989, 60, 90–92. [Google Scholar]
- Gulluce, M.; Sokmen, M.; Daferera, D.; Agar, G.; Ozkan, H.; Kartal, N. In vitro antibacterial, antifungal and antioxidant activities of the essential oil and methanol extracts of herbal parts and callus cultures of Satureja hortensis L. J. Agric. Food Chem. 2003, 51, 3958–3965. [Google Scholar] [CrossRef]
- Zargari, A. Medicinal Plants; Tehran University Press: Tehran, Iran, 1990; Volume IV, pp. 325–328. [Google Scholar]
- Gören, A.C.; Topçu, G.; Bilsel, G.; Bilsel, M.; Wilkinson, J.M.; Cavanagh, H.M.A. Analysis of essential oil of Satureja thymbra by hydrodistillation, thermal desorber, and headspace GC/MS techniques and its antimicrobial activity. Nat. Prod. Res. 2004, 18, 189–195. [Google Scholar] [CrossRef]
- Sarac, N.; Ugur, A. Antimicrobial activities of the essential oils of Origanum onites L., Origanum vulgare L. subspecies hirtum (Link) Ietswaart, Satureja thymbra L., and Thymus cilicicus Boiss. & Bal. growing wild in Turkey. J. Med. Food 2008, 11, 568–573. [Google Scholar] [CrossRef]
- Azaz, A.D.; Kürkcüoglu, M.; Satil, F.; Baser, K.H.C.; Tümen, G. In vitro antimicrobial activity and chemical composition of some Satureja essential oils. Flavour Fragr. J. 2005, 20, 587–591. [Google Scholar] [CrossRef]
- Loizzo, M.R.; Saab, A.M.; Tundis, R.; Statti, G.A.; Menichini, F.; Lampronti, I.; Gambari, R.; Cinatl, J.; Doerr, H.W. Phytochemical analysis and in vitro antiviral activities of the essential oils of seven Lebanon species. Chem. Biodivers. 2008, 5, 461–470. [Google Scholar] [CrossRef]
- Chorianopoulos, N.G.; Giaouris, E.D.; Skandamis, P.N.; Haroutounian, S.A.; Nychas, G.-J.E. Disinfectant test against monoculture and mixed-culture biofilms composed of technological, spoilage and pathogenic bacteria: Bactericidal effect of essential oil and hydrosol of Satureja thymbra and comparison with standard acid-base sanitizers. J. Appl. Microbiol. 2008, 104, 1586–1596. [Google Scholar] [CrossRef]
- Cetin, H.; Cilek, J.E.; Oz, E.; Aydin, L.; Deveci, O.; Yanikoglu, A. Acaricidal activity of Satureja thymbra L. essential oil and its major components, carvacrol and gamma-terpinene against adult Hyalomma marginatum (Acari: Ixodidae). Vet. Parasitol. 2010, 170, 287–290. [Google Scholar] [CrossRef]
- Ayvaz, A.; Sagdic, O.; Karaborklu, S.; Ozturk, I. Insecticidal activity of the essential oils from different plants against three stored-product insects. J. Insect Sci. 2010, 10, 1–13. [Google Scholar]
- Shafie, M.S.B.; Hasan, Z.; Shah, R.M. Study of genetic variability of Wormwood capillary (Artemisia capillaris) using inter simple sequence repeat (ISSR) in Pahang region, Malaysia. Plant Omics J. 2009, 2, 127–134. [Google Scholar]
- Pourohit, S.S.; Vyas, S.P. Medicinal Plants Cultivation; Agrobios Press: Jodhpur, India, 2004. [Google Scholar]
- Rahimmalek, M.; Tabatabaei, B.E.S.; Etemadi, N.; Goli, S.A.H.; Arzani, A.; Zeinali, H. Essential oil variation among and within six Achillea species transferred from different ecological regions in Iran to the field conditions. Ind. Crops Prod. 2009, 29, 348–355. [Google Scholar] [CrossRef]
- Basu, S.K.; Acharya, S.N.; Bandara, M.S.; Friebel, D.; Thomas, J.E. Effects of genotype and environment on seed and forage yield in fenugreek (Trigonella foenum-graecum L.) grown in western Canada. Aust. J. Crop Sci. 2009, 3, 305–314. [Google Scholar]
- Mockute, D.; Bernotiene, G.; Judzentiene, A. The essential oil of Origanum vulgare L. ssp. Vulgare growing wild in Vilnius district (Lithuania). Phytochemistry 2001, 57, 65–69. [Google Scholar]
- Russell, M.F.; Southwell, I.A. Monoterpenoid accumulation in 1,8-cineol, terpinolene and terpinen-4-ol chemotypes of Melaleuca alternifolia seedlings. Phytochemistry 2003, 62, 683–689. [Google Scholar]
- Curado, M.A.; Oliveira, C.B.A.; Jesus, J.G.; Santos, S.C.; Searphin, J.C.; Ferri, P.H. Environmental factors influence on chemical polymorphism of the essential oils of Lychnophora ericoides. Phytochemistry 2006, 67, 2363–2369. [Google Scholar] [CrossRef]
- Letchamo, W.; Xu, H.L.; Gosselin, A. Variations in photosynthesis and essential oil in thyme. J. Plant Physiol. 1995, 147, 29–37. [Google Scholar] [CrossRef]
- Karousou, R.; Koureas, D.N.; Kokkini, S. Essential oil composition is related to the natural habitats: Coridothymus capitatus and Satureja thymbra in NATURA 2000 sites of Crete. Phytochemistry 2005, 66, 2668–2673. [Google Scholar] [CrossRef]
- Sokovic, M.; Tzakou, O.; Pitarakoli, D.; Couladis, M. Antifungal activities of selected aromatic plants growing wild in Greece. Food / Nahrung 2002, 46, 317–320. [Google Scholar] [CrossRef]
- Skoula, M.; Grayer, R.J. Volatile oils of Coridothymus capitatus, Satureja thymbra, Satureja spinosa and Thymbra calostachya (Lamiaceae) from Crete. Flavour Fragr. J. 2005, 20, 573–576. [Google Scholar] [CrossRef]
- Schulz, H.; Ozkan, G.; Baranska, M.; Krüger, H.; Özcan, M. Characterisation of essential oil plants from Turkey by IR and Raman spectroscopy. Vib. Spectrosc. 2005, 39, 249–256. [Google Scholar] [CrossRef]
- Chorianopoulos, N.; Evergetis, E.; Mallouchos, A.; Kalpoutzakis, E.; Nychas, G.-J.; Haroutounian, S.A. Characterization of the essential oil volatiles of Satureja thymbra and Satureja parnassica: Influence of harvesting time and antimicrobial activity. J. Agric. Food Chem. 2006, 54, 3139–3145. [Google Scholar]
- Michaelakis, A.; Theotokatofs, S.A.; Koliopoulos, G.; Chorianopoulos, N.G. Essential oils of Satureja species: Insecticidal effect on Culex pipiens larvae (Diptera: Culicidae). Molecules 2007, 12, 2567–2578. [Google Scholar] [CrossRef]
- Economou, G.; Panagopoulos, G.; Tarantilis, P.; Kalivas, D.; Kotoulas, V. Variability in essential oil content and composition of Origanum hirtum L., Origanum onites L., Coridothymus capitatus (L.) and Satureja thymbra L. populations from the Greek island Ikaria. Ind. Crops Prod. 2011, 33, 236–241. [Google Scholar] [CrossRef]
- Piras, A.; Cocco, V.; Falconieri, D. Isolation of the volatile oil from Satureja thymbra by supercritical carbon dioxide extraction: Chemical composition and biological activity. Nat. Prod. Commun. 2011 6, 1523–1526.
- Karpouhtsis, I.; Pardali, E.; Feggou, E.; Kokkini, S.; Scouras, Z.G.; MavraganiI-Tsipidou, P. Insecticidal and genotoxic activities of oregano essential oils. J. Agric. Food Chem. 1998, 46, 1111–1115. [Google Scholar] [CrossRef]
- Ravid, U.; Putievsky, E. Constituentsof the essentail oils from Majorana syriaca, Coridothymus capitatus and Satureja thymbra. Planta Med. 1983, 49, 248–249. [Google Scholar] [CrossRef]
- Galego, L.; Almeida, V.; Gonçalves, V.; Costa, M.; Monteiro, I.; Matos, F.; Miguel, G. Antioxidant activity of the essential oils of Thymbra capitata, Origanum vulgare, Thymus mastichina and Calamintha baetica. Acta Hortic. 2008, 765, 325–334. [Google Scholar]
- Albano, S.M.; Lima, A.S.; Miguel, M.G.; Pedro, L.G.; Barroso, J.G.; Figueiredo, A.C. Antioxidant, anti-5-lipoxygenase and antiacetylcholinesterase activities of essential oils and decoction waters of some aromatic plants. Rec. Nat. Prod. 2012, 6, 35–48. [Google Scholar]
- Ruberto, G.; Baratta, M.T. Antioxidant activity of selected essential oil components in two lipid model systems. Food Chem. 2000, 69, 167–174. [Google Scholar] [CrossRef]
- Glamočlija, J.; Soković, M.; Vukojević, J.; Milenković, I.; van Griensven, L.J.L.D. Chemical composition and antifungal activities of essential oils of Satureja thymbra L. and Salvia pomifera ssp. calycina (Sm.) Hayek. J. Essent. Oil Res. 2006, 18, 115–117. [Google Scholar] [CrossRef]
- Marković, T.; Chatzopoulou, P.; Šiljegović, J.; Nikolić, M.; Glamočlija, J.; Ćirić, A.; Soković, M. Chemical analysis and antimicrobial activities of the essential oils of Satureja thymbra L. and Thymbra spicata L. and their main components. Arch. Biol. Sci. 2011, 63, 457–464. [Google Scholar] [CrossRef]
- Adams, S.; Kunz, B.; Weidenbörner, M. Mycelial deformations of Cladosporium herbarum due to the application of eugenol and carvacrol. J. Essent. Oil Res. 1996, 8, 535–540. [Google Scholar] [CrossRef]
- Adam, K.; Sivropoulu, A.; Kokkini, S.; Lanaras, T.; Arsenakis, M. Antifungal activities of Origanum vulgare subsp. hirtum, Mentha spicata, Lavandula angustifolia and Salvia fruticosa essential oils against human pathogenic fungi. J. Agric. Food Chem. 1998, 46, 1739–1745. [Google Scholar]
- Skandamis, P.; Koutsoumanis, K.; Fasseas, K.; Nychas, G.-J.E. Inhibition of oregano essential oil and EDTA on Escherichia coli O157:H7. Ital. J. Food Sci. 2001, 13, 65–75. [Google Scholar]
- Carson, C.F.; Mee, B.J.; Riley, T.V. Mechanism of action of Melaleuca alternifolia (tea tree) oil on Staphylococcus aureus determined by time-kill, lysis, leakage and salt tolerance assays and electron microscopy. Antimicrob. Agents Chemother. 2002, 46, 1914–1920. [Google Scholar] [CrossRef]
- Knobloch, K.; Weigand, H.; Weis, N.; Schwarm, H.M.; Vigenschow, H. Action of Terpenoids on Energy Metabolism. In Progress in Essential Oil Research; Brunke, E.J., Ed.; Walter de Gruyter: Berlin, Germany, 1986; pp. 429–445. [Google Scholar]
- Sikkema, J.; de Bont, J.A.M.; Poolman, B. Interactions of cyclic hydrocarbons with biological membranes. J. Biol. Chem. 1994, 269, 8022–8028. [Google Scholar]
- Burt, S. Essential oils: Their antibacterial properties and potential applications in foods—A review. Int. J. Food Microbiol. 2004, 94, 223–253. [Google Scholar] [CrossRef]
- Denyer, S.P.; Hugo, W.B. Biocide-Induced Damage to the Bacterial Cytoplasmic Membrane. In Mechanisms of Action of Chemical Biocides; Denyer, S.P., Hugo, W.B., Eds.; The Society for Applied Bacteriology, Technical Series No 27. Oxford Blackwell Scientific Publication: Oxford, UK, 1991; pp. 171–188. [Google Scholar]
- Soković, M.; Glamočlija, J.; Marin, D.P.; Brkić, D.; van Griensven, J.L.D.L. Antibacterial effects of the essential oils of commonly consumed medicinal herbs using an in vitro model. Molecules 2010, 15, 7532–7546. [Google Scholar] [CrossRef]
- Cosentino, S.; Tuberoso, C.I.G.; Pisano, B.; Satta, M.; Mascia, V.; Arzedi, E.; Palmas1, F. In-vitro antimicrobial activity and chemical composition of Sardinian Thymus essential oils. Lett. Appl. Microbiol. 1999, 29, 130–135. [Google Scholar] [CrossRef]
- Kalemba, D.; Kunicka, A. Antibacterial and antifungal properties of essential oils. Curr. Med. Chem. 2003, 10, 813–829. [Google Scholar] [CrossRef]
- Nikaido, H.; Vaara, M. Molecular basis of bacterial outer membrane permeability. Microbiol. Rev. 1985, 49, 1–32. [Google Scholar]
- Nikaido, H. Outer membrane barrier as a mechanism of antimicrobial resistance. Antimicrob. Agents Chemother. 1989, 33, 1831–1836. [Google Scholar] [CrossRef]
- Gao, Y.; Van Belkum, M.J.; Stiles, M.E. The outer membrane of gram negative bacteria inhibits antibacterial activity of brochocin-C. Appl. Environ. Microbiol. 1999, 65, 4329–4333. [Google Scholar]
- Nikaido, H. Prevention of drug access to bacterial targets: Permeability barriers and active efflux. Science 1994, 264, 382–388. [Google Scholar]
- Delamare, A.P.L.; Moschen-Pistorello, I.T.; Artico, L.; Echeverrigaray, L.A.S. Antibacterial activity of the essential oils of Salvia officinalis L. and Salvia triloba L. cultivated in South Brazil. Food Chem. 2007, 100, 603–608. [Google Scholar] [CrossRef]
- Marino, M.; Bersani, C.; Comi, G. Impedance measurementsto study the antimicrobial activity of essential oils from Lamiaceae and Compositae. Int. J. Food Microbiol. 2001, 67, 187–195. [Google Scholar] [CrossRef]
- Cowan, M.M. Plant products as antimicrobial agents. Clin. Microbiol. Rev. 1999, 12, 564–582. [Google Scholar]
- Wendakoon, C.N.; Sakaguchi, M. Inhibition of amino acid decaboxylase activity of Enterobacter aerogenes by active components of spices. J. Food Prot. 1995, 58, 280–283. [Google Scholar]
- Farag, R.S.; Daw, Z.Y.; Hewedi, F.M.; El-Baroty, G.S.A. Antimicrobial activity of some Egyptian spice essential oils. J. Food Prot. 1989, 52, 665–667. [Google Scholar]
- Lemos, T.L.G.; Matos, F.J.A.; Alencar, J.W.; Craveiro, A.A.; Clark, A.M.; McChesney, J.D. Antimicrobial activity of essential oils of Brazilian plants. Phytoter. Res. 1990, 4, 82–84. [Google Scholar] [CrossRef]
- Smith-Palmer, A.; Stewart, J.; Fyfe, L. Antimicrobial properties of plant essential oils and essences against five importantfood-borne pathogens. Lett. Appl. Microbiol. 1998, 26, 118–122. [Google Scholar]
- British Pharmacopoeia II. P 109. H.M. Stationary Office: London, UK.
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed; Allured Publ. Corp.: Carol Stream, IL, USA, 2007; pp. 10–87. [Google Scholar]
- Blois, M.S. Antioxidant determinations by the use of a stable free radical. Nature 1958, 181, 1199–1200. [Google Scholar] [CrossRef]
- Booth, C. Fungal Culture Media. In Methods in Microbiology; Norris, J.R., Ribbons, D.W., Eds.; Academic Press: New York, NY, USA, 1971; Volume IV, pp. 49–94. [Google Scholar]
- Daouk, K.D.; Dagher, M.S.; Sattout, J.E. Antifungal activity of the essential oil of Origanum syriacum L. J. Food Prot. 1995, 58, 1147–1149. [Google Scholar]
- Hanel, H.; Raether, W. A more sophisticated method of determining the fungicidal effect of water-insoluble preparations with a cell harvester, using miconazole as an example. Mycoses 1988, 31, 148–154. [Google Scholar] [CrossRef]
- Espinel-Ingroff, A. Comparasion of the E-test with the NCCLS M38-P method for antifungal susceptibility testing of common and emerging pathogenic filamentous fungi. J. Clin. Microbiol. 2001, 39, 1360–1367. [Google Scholar] [CrossRef]
- Kabara, J.J. Phenols and Chelators. In Food Preservatives; Russell, N.J., Gould, G.W., Eds.; Blackie: London, UK, 1991; pp. 200–214. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Giweli, A.; Džamić, A.M.; Soković, M.; Ristić, M.S.; Marin, P.D. Antimicrobial and Antioxidant Activities of Essential Oils of Satureja thymbra Growing Wild in Libya. Molecules 2012, 17, 4836-4850. https://doi.org/10.3390/molecules17054836
Giweli A, Džamić AM, Soković M, Ristić MS, Marin PD. Antimicrobial and Antioxidant Activities of Essential Oils of Satureja thymbra Growing Wild in Libya. Molecules. 2012; 17(5):4836-4850. https://doi.org/10.3390/molecules17054836
Chicago/Turabian StyleGiweli, Abdulhmid, Ana M. Džamić, Marina Soković, Mihailo S. Ristić, and Petar D. Marin. 2012. "Antimicrobial and Antioxidant Activities of Essential Oils of Satureja thymbra Growing Wild in Libya" Molecules 17, no. 5: 4836-4850. https://doi.org/10.3390/molecules17054836
APA StyleGiweli, A., Džamić, A. M., Soković, M., Ristić, M. S., & Marin, P. D. (2012). Antimicrobial and Antioxidant Activities of Essential Oils of Satureja thymbra Growing Wild in Libya. Molecules, 17(5), 4836-4850. https://doi.org/10.3390/molecules17054836






