Ionic Liquid Applications in Peptide Chemistry: Synthesis, Purification and Analytical Characterization Processes
Abstract
:| [BF4]− = tetrafluoroborate | [C1C1IM]+ = 1,3-dimethylimidazolium |
| [C1mim]+ = 1-methyl-3-methylimidazolium | [C2mim]+ = 1-ethyl-3-methylimidazolium |
| [C4C1pyrr]+ = 1-butyl-1-methylpyrrolidinium | [C4mim]+ = 1-butyl-3-methylimidazolium |
| [C6mim]+ = 1-hexyl-3-methylimidazolium | [C8mim]+ = 1-octyl-3-methylimidazolium |
| [Ci,j,k,lN]+ = tetraalkylammonium | [DEP]− = diethyl phosphate |
| ESI = electrospray ionization | [Et3NH]+ = triethylammonium |
| [EtOSO3]− = ethylsulfate | [guan]+ = guanidinium |
| [Me2PO4]− = dimethylphosphate | [MeOSO3]− = methylsulfate |
| [MOEMIm]+ = 3-(2-methoxyethyl)-1-methyl-imidazolium | |
| [N(CN)2]− = dicyanamide | [OAc]− = acetate |
| [OTf]− = triflate | [OTs]− = tosylate |
| [PF6]− = hexafluorophoshate | [SbF6]− = hexafluoroantimonate |
| [SCN]− = thiocyanate | [NTf2]− = bis(trifluoromethanesulfonyl)imide |
| BOP = benzotriazole-1-yl-oxy-tris-(dimethylamino)-phosphonium hexafluorophosphate | |
| CCA = α-cyano-4-hydroxycinnamic acid | DCC = N,N'-dicyclohexylcarbodiimide |
| DHB = 2,5-dihydroxybenzoic acid | DIEA = N,N-diisopropylethylamine, or Hünig’s base |
| DMAP = 4-dimethylaminopyridine | DMED = N,N-dimethyl-ethylenediamine |
| GSH = glutathione | |
| GTHAP = [1,1,3,3,-tetramethylguanidinium-2,4,6,-trihydroxyacetophenone] | |
| H2O2 = hydrogen peroxide | H3PO4 = phosphoric acid |
| HATU = O-(7-azabenzotriazol-1-yl)-N,N,N',N'-tetramethyluronium hexafluorophosphate | |
| HPLC = high-performance liquid chromatography (high-pressure liquid chromatography) | |
| IL = ionic liquid | ILM = ionic liquid matrices |
| kDa = kilodalton | LSER = linear solvation free energy relationship |
| MPG = 2-methyl-2-(p-tolyl)-glycine | MALDI = matrix-assisted laser desorption/ ionization |
| MS = mass spectrometry | MOPS = 3-[N-morpholino]propanesulfonic acid |
| NCL = native chemical ligation | NMR = nuclear magnetic resonance |
| OGp = chymotrypsin and trypsin | RP = reversed phase |
| PyBOP = (benzotriazol-1-yl-oxytripyrrolidinophosphonium hexafluorophosphate) | |
| SCIL = surface-confined ionic liquids | SCm = V8 protease |
| SPPS = solid phase peptide synthesis | TFA = trifluoroacetic acid |
| THF = tetrahydofurane | TLC = thin layer chromatography |
| TOF = time-of-flight | TRH = thyroliberin (thyrotropin-releasing hormone) |
1. Introduction
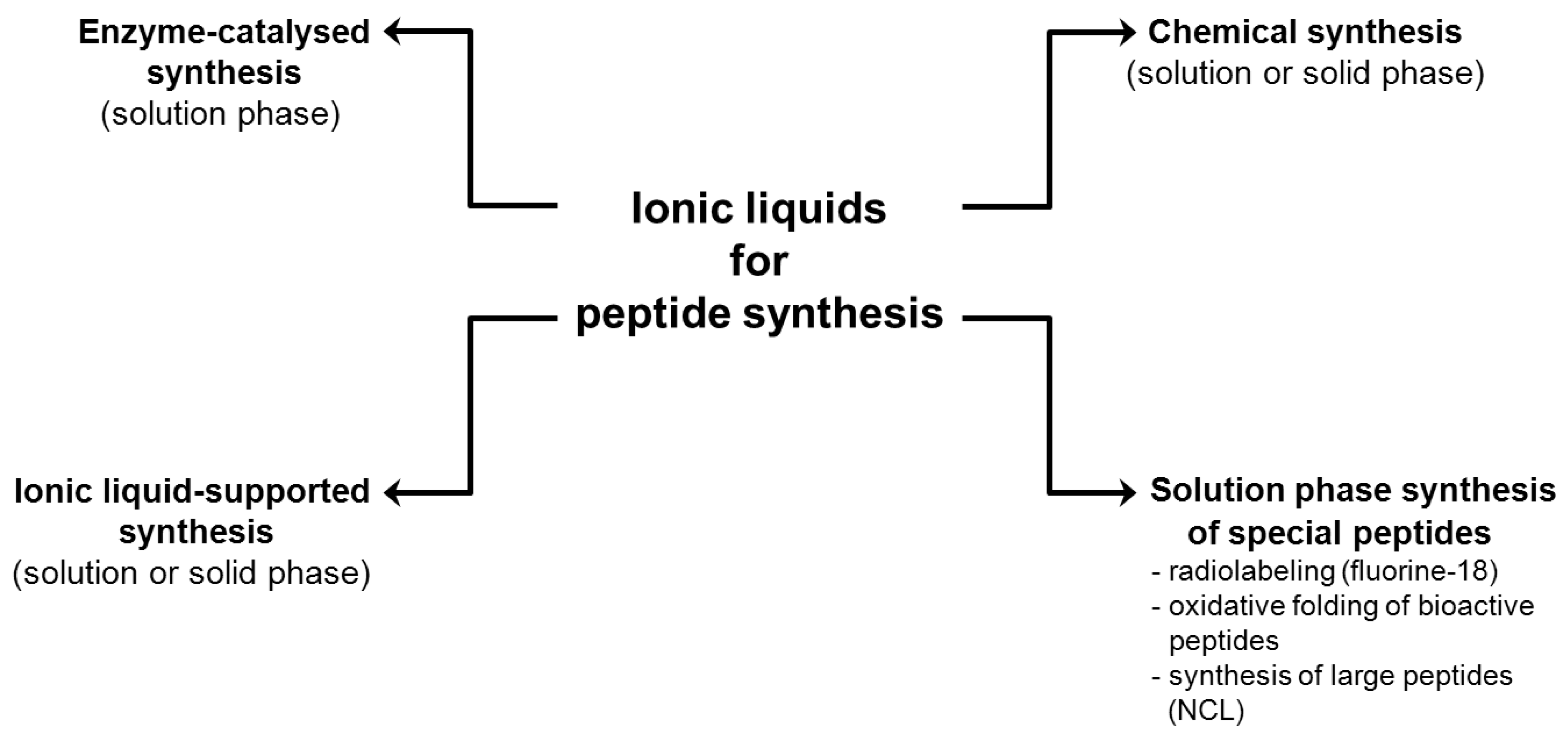
2. Ionic Liquids in Peptide Chemistry
2.1. Peptide Assembly
| Ionic liquid | Abbreviation | Application | Ref. |
|---|---|---|---|
| 1-methoxyethyl-3-methyl-imidazolium hexafluoro-phosphate or tetrafluoroborate | [MOEMIM][PF6] [MOEMIM][BF4] | Enzymatic peptide synthesis | [30] |
| 1-butyl-3-methylimidazolium hexafluorophosphate | [C4mim][PF6] | Chemical peptide synthesis | [15,31,32,33,34] |
| 1-hydroxyethyl-1-methyl-imidazolium tetrafluoroborate 3-(2-hydroxyethyl)-1-methyl-imidazolium tetrafluoroborate | - | IL supported peptide synthesis | [35,36] |
| 1-butyl-3-methylimidazolium X | [C4mim][X] (X = BF4, PF6, SbF6, OTf, NTf2) | Radiolabeling of peptides | [37,38] |
| 1-ethyl-3-methylimidazolium X 1-butyl-3-methylimidazolium acetate | [C2mim][X], (X = OAc, DEP, OTs, N(CN2)2) [C4mim][OAc] | Oxidative folding and native chemical ligation of cysteine-containing peptides | [14,39] |
2.1.1. Enzymatic Peptide Synthesis
2.1.2. Chemical Synthesis of Peptides
2.1.2.1. Condensation of Free Amino Acids in Solution
2.1.2.2. Ionic Liquid-Supported Amino Acid Condensation
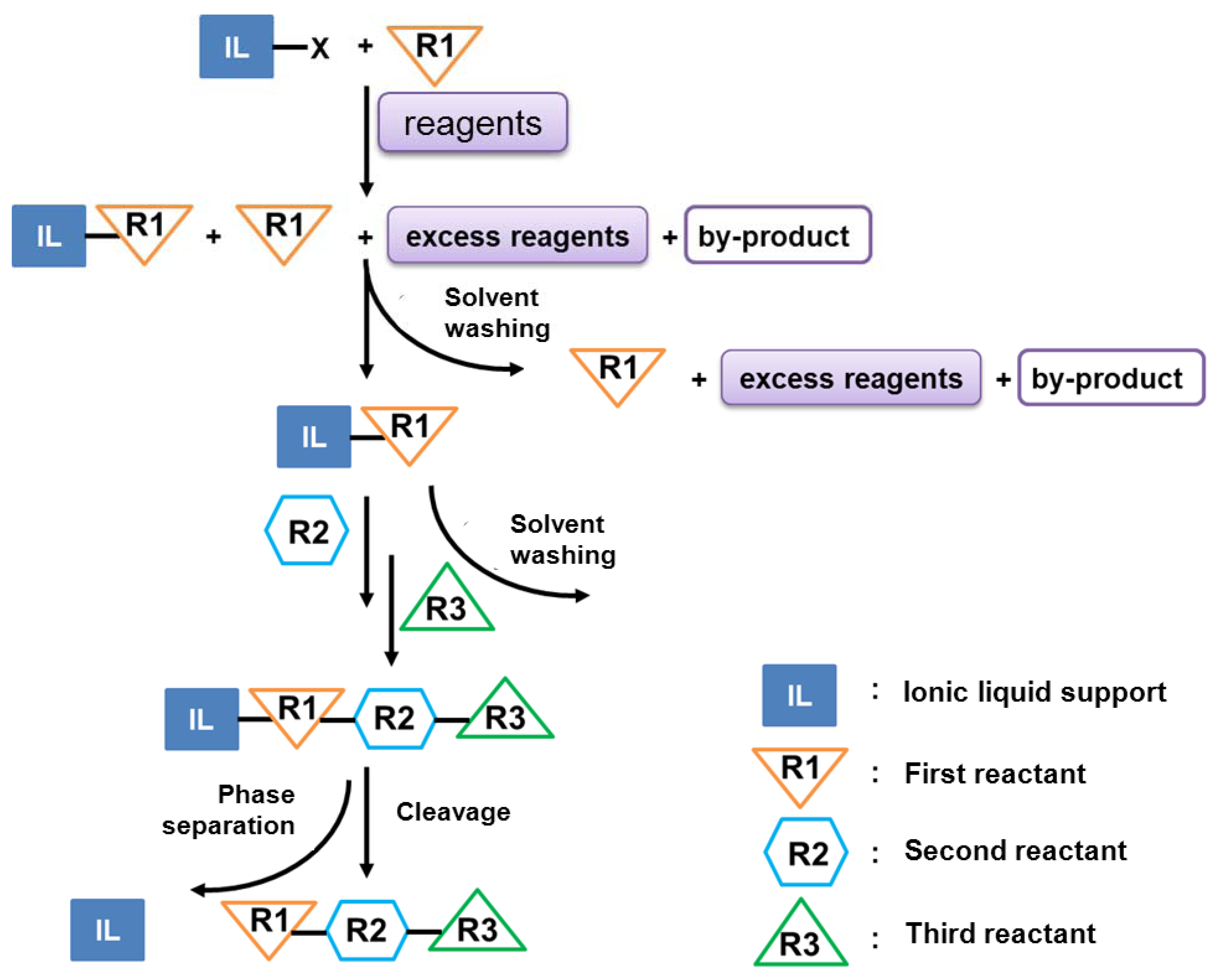
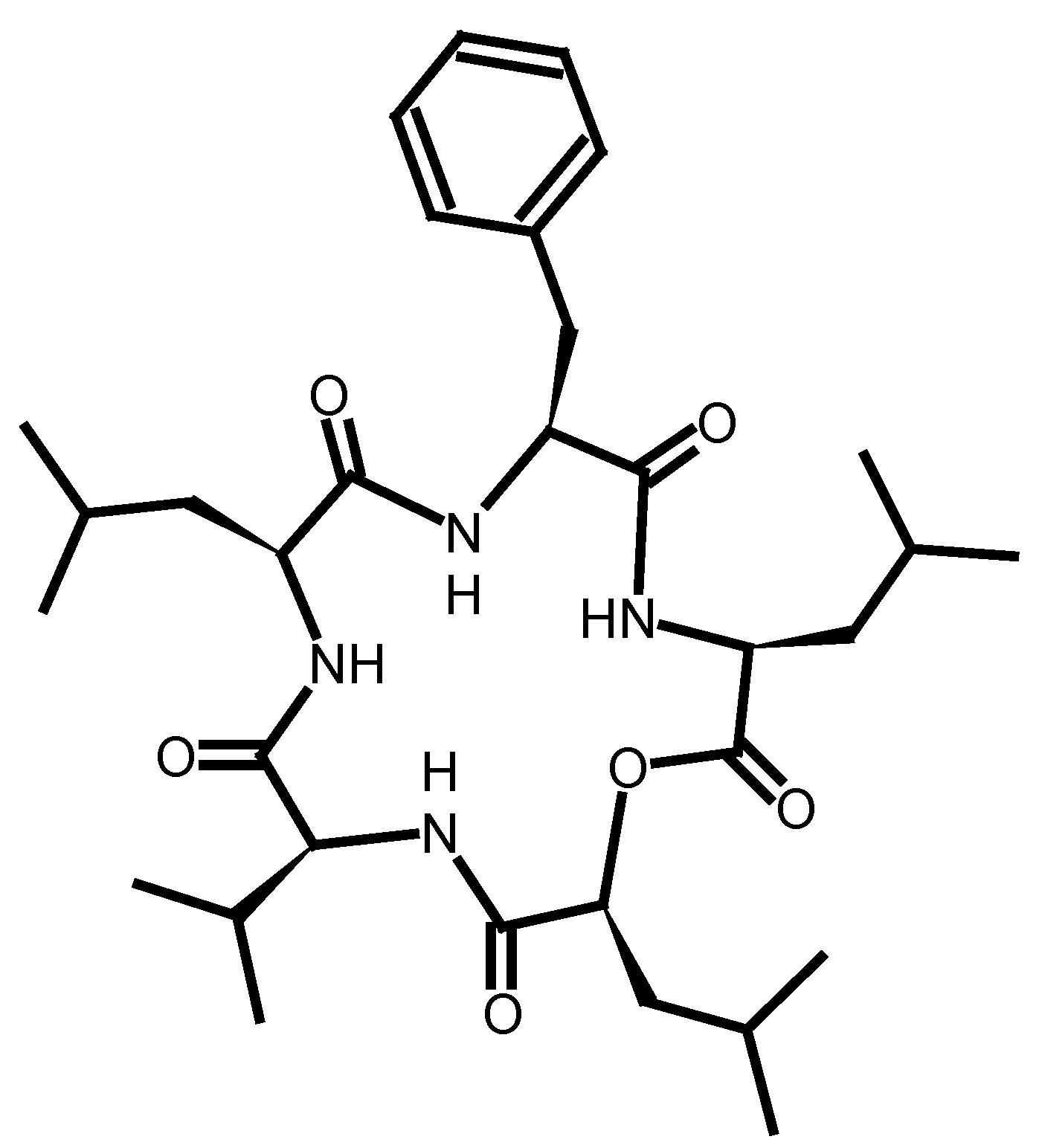
2.2. Peptide Modifications
2.2.1. Radiolabeling of Peptides

2.2.2. Disulfide Bond Formation and Peptide Folding
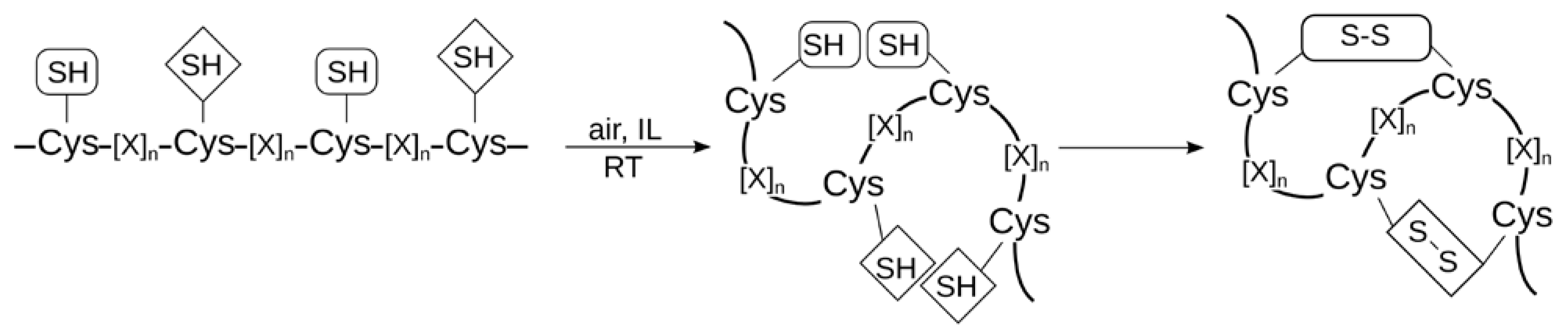
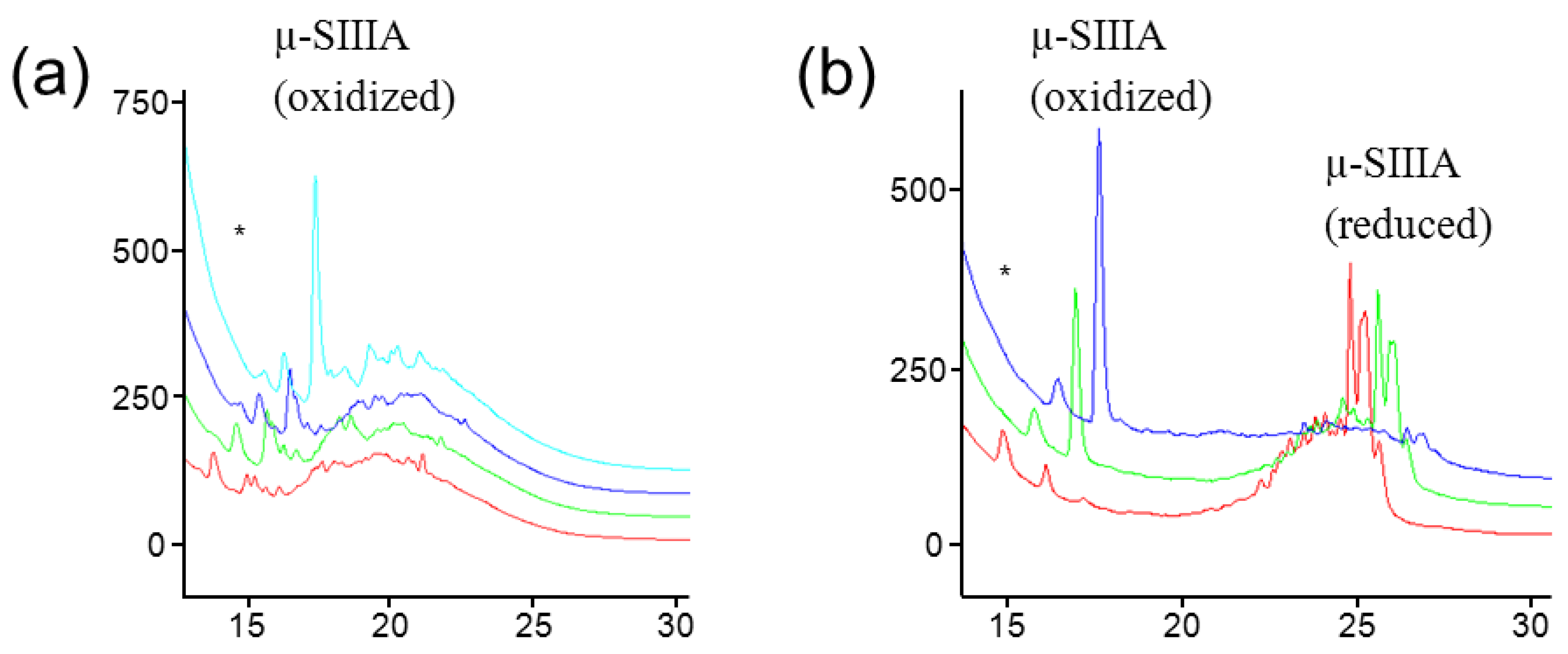
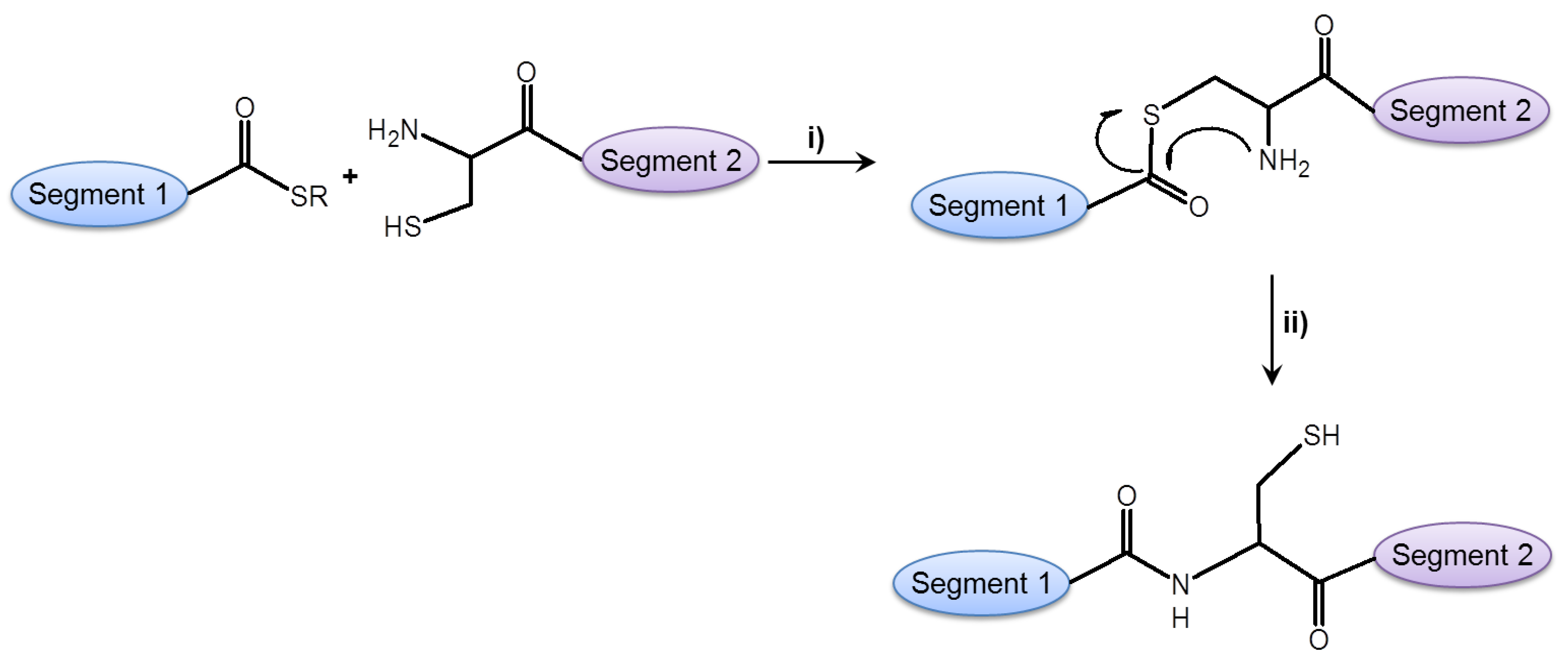
2.2.3. Ligation Reactions in Peptide Synthesis
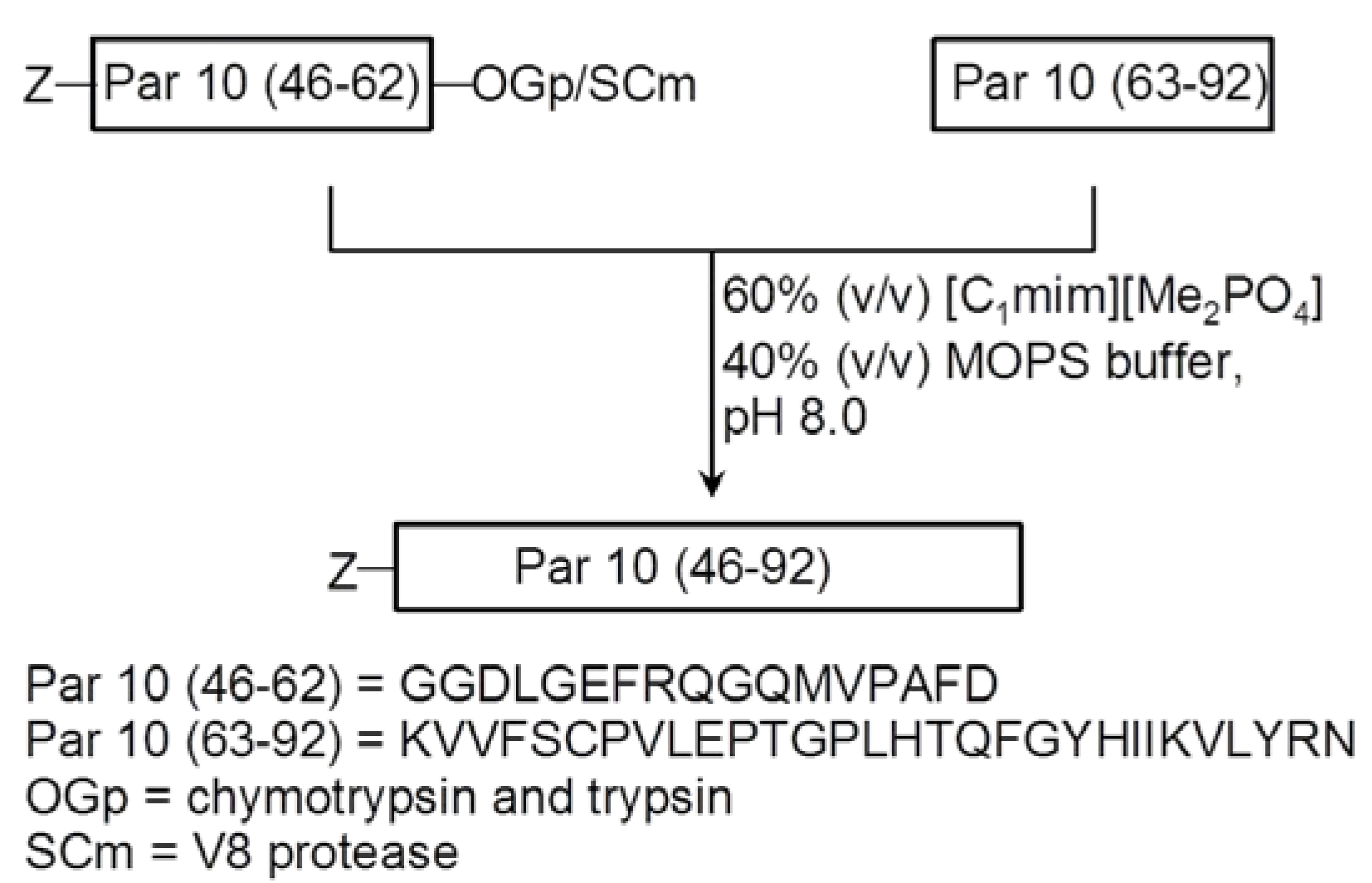
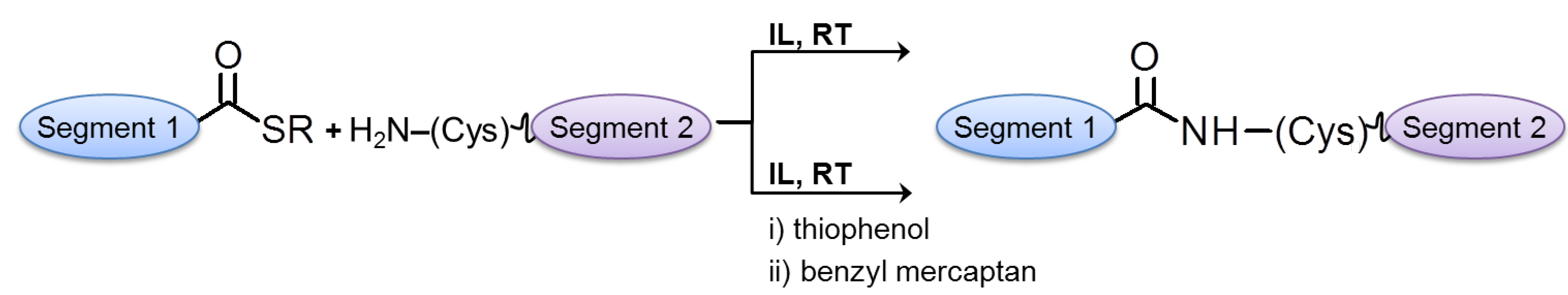
3. Ionic Liquids for Peptide Purification and Characterization
3.1. Peptide Separation by Liquid Chromatography Methods
3.1.1. Reversed-Phase HPLC
3.1.1.1. ILs as Additives for RP HPLC Mobile Phase
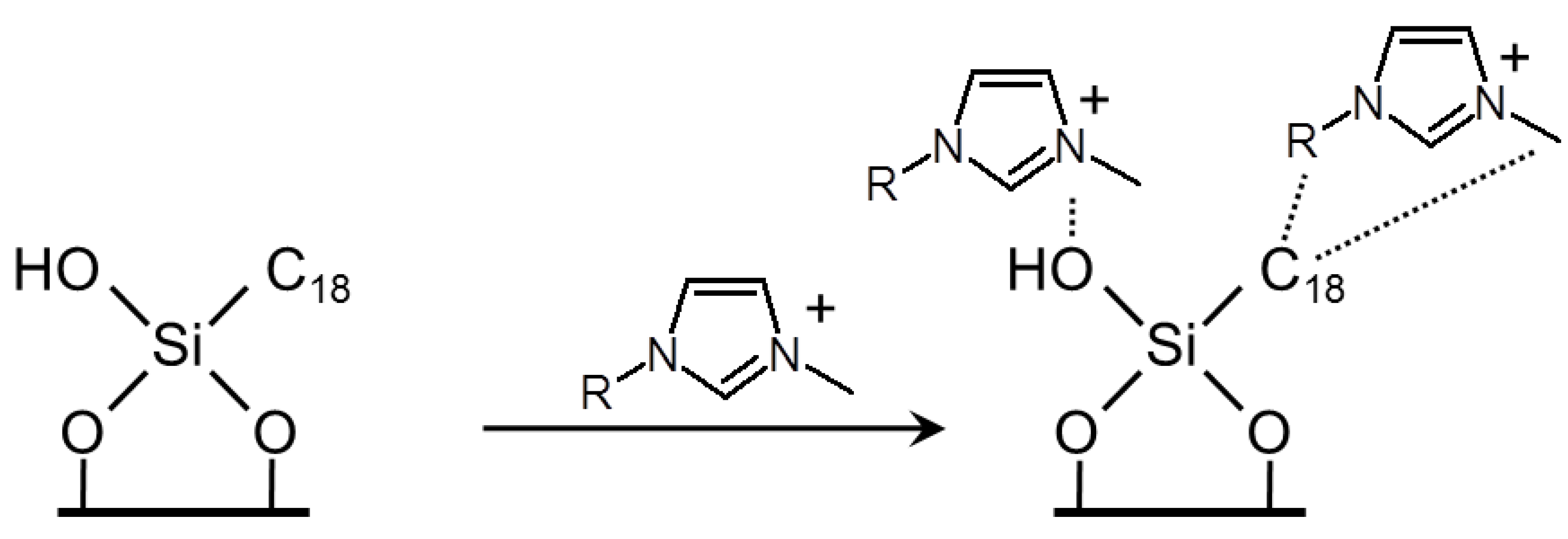
3.1.1.2. ILs as Material for RP HPLC Stationary Phase
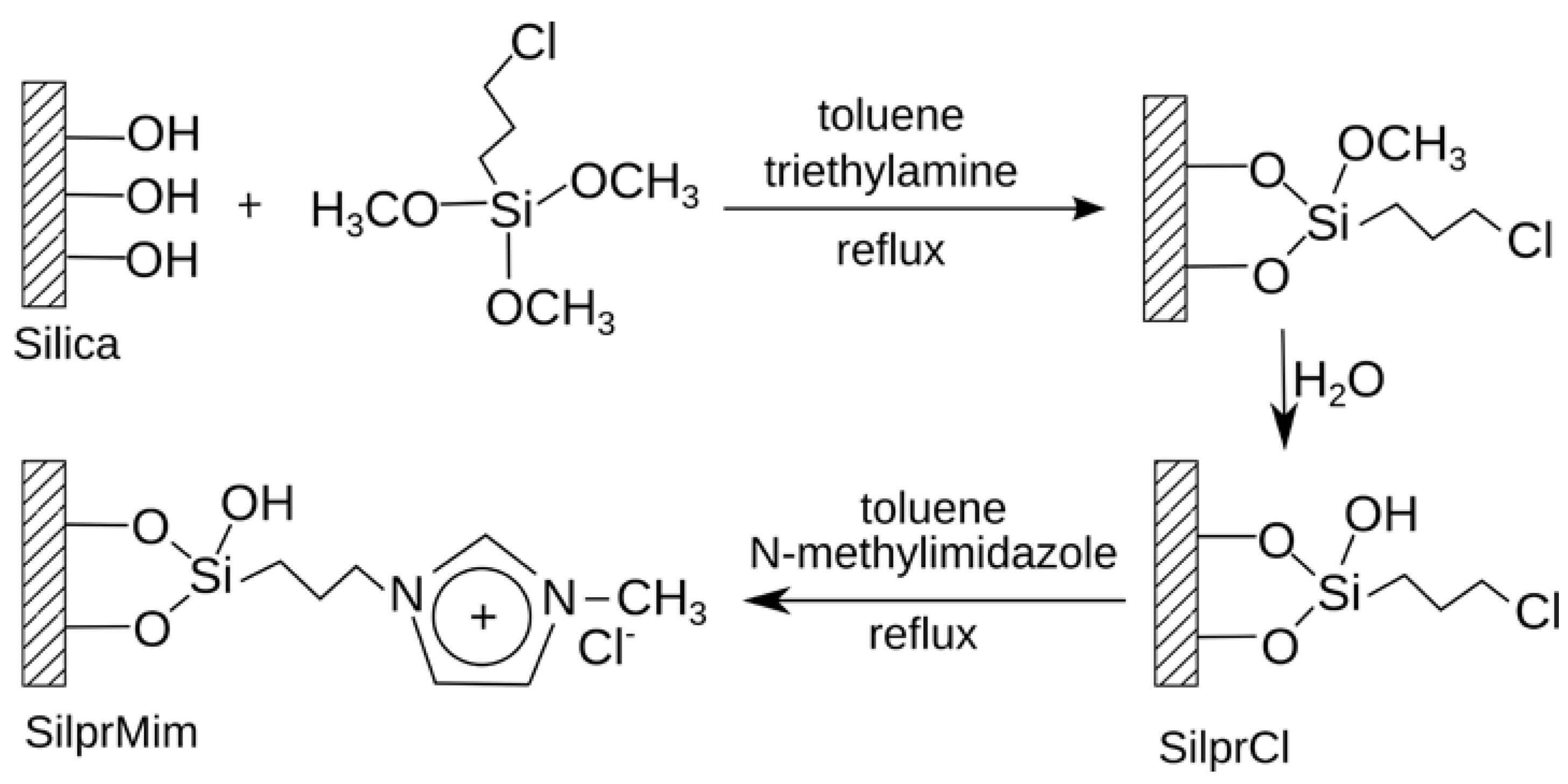
3.1.2. Thin Layer Chromatography
3.2. Mass Spectrometry
3. Conclusions
Acknowledgments
References
- Sheldon, R. Catalytic reactions in ionic liquids. Chem. Commun. 2001, 2399–2407. [Google Scholar]
- Welton, T. Room-temperature ionic liquids. Solvents for synthesis and catalysis. Chem. Rev. 1999, 99, 2071–2083. [Google Scholar] [CrossRef]
- Wasserscheid, P.; Keim, W. Ionic liquids—New “solutions” for transition metal catalysis. Angew.Chem. Int. Ed. 2000, 39, 3772–3789. [Google Scholar]
- Lange, C.; Patil, G.; Rudolph, R. Ionic liquids as refolding additives: N'-alkyl and N'-(omega-hydroxyalkyl) N-methylimidazolium chlorides. Protein Sci. 2005, 14, 2693–2701. [Google Scholar] [CrossRef]
- Moon, Y.H.; Lee, S.M.; Ha, S.H.; Koo, Y.M. Enzyme-catalyzed reactions in ionic liquids. Korean J. Chem. Eng. 2006, 23, 247–263. [Google Scholar]
- Hekmat, D.; Hebel, D.; Joswig, S.; Schmidt, M.; Weuster-Botz, D. Advanced protein crystallization using water-soluble ionic liquids as crystallization additives. Biotechnol.Lett. 2007, 29, 1703–1711. [Google Scholar]
- Baker, S.N.; Brauns, E.B.; McCleskey, T.M.; Burrell, A.K.; Baker, G.A. Fluorescence quenching immunoassay performed in an ionic liquid. Chem. Commun. 2006, 2851–2853. [Google Scholar]
- Kragl, U.; Eckstein, M.; Kaftzik, N. Enzyme catalysis in ionic liquids. Curr.Opin. Biotechnol. 2002, 13, 565–571. [Google Scholar]
- Stark, A. Ionic liquid structure-induced effects on organic reactions. Top.Curr. Chem. 2009, 290, 41–81. [Google Scholar]
- Fujita, K.; MacFarlane, D.R.; Forsyth, M. Protein solubilising and stabilising ionic liquids. Chem. Commun. 2005, 4804–4806. [Google Scholar]
- Baker, S.N.; McCleskey, T.M.; Pandey, S.; Baker, G.A. Fluorescence studies of protein thermostability in ionic liquids. Chem. Commun. 2004, 940–941. [Google Scholar]
- Feher, E.; Major, B.; Belafi-Bako, K.; Gubicza, L. On the background of enhanced stability and reusability of enzymes in ionic liquids. Biochem. Soc. Trans. 2007, 35, 1624–1627. [Google Scholar]
- Dreyer, S.; Kragl, U. Ionic liquids for aqueous two-phase extraction and stabilization of enzymes. Biotechnol.Bioeng. 2008, 99, 1416–1424. [Google Scholar]
- Miloslavina, A.A.; Leipold, E.; Kijas, M.; Stark, A.; Heinemann, S.H.; Imhof, D. A room temperature ionic liquid as convenient solvent for the oxidative folding of conopeptides. J. Pept. Sci. 2009, 15, 72–77. [Google Scholar]
- Plaquevent, J.C.; Levillain, J.; Guillen, F.; Malhiac, C.; Gaumont, A.C. Ionic liquids: New targets and media for alpha-amino acid and peptide chemistry. Chem. Rev. 2008, 108, 5035–5060. [Google Scholar]
- Brown, R.; Carr, B.; Lennon, J. Factors that influence the observed fast fragmentation of peptides in matrix-assisted laser desorption. J. Am. Soc. Mass Spectrom. 1996, 7, 225–232. [Google Scholar]
- McDonnell, L.A.; Heeren, R.M. Imaging mass spectrometry. Mass Spectrom.Rev. 2007, 26, 606–643. [Google Scholar]
- Tietze, D.; Breitzke, H.; Imhof, D.; Kothe, E.; Weston, J.; Buntkowsky, G. New insight into the mode of action of nickel superoxide dismutase by investigating metallopeptide substrate models 1. Chem. Eur. J. 2009, 15, 517–523. [Google Scholar]
- Becker, S.; Terlau, H. Toxins from cone snails: Properties, applications and biotechnological production. Appl. Microbiol. Biotechnol. 2008, 79, 1–9. [Google Scholar]
- Escoubas, P.; Bernard, C.; Lambeau, G.; Lazdunski, M.; Darbon, H. Recombinant production and solution structure of PcTx1, the specific peptide inhibitor of ASIC1a proton-gated cation channels. Protein Sci. 2003, 12, 1332–1343. [Google Scholar]
- Anangi, R.; Chen, C.Y.; Cheng, C.H.; Chen, Y.C.; Chen, C.C.; Chu, Y.P.; Chang, C.H.; Jeng, W.Y.; Shiu, J.H.; Chuang, W.J. Expression of snake venom toxins in Pichia pastoris. Toxin Rev. 2007, 26, 169–187. [Google Scholar] [CrossRef]
- Olivera, B.M.; Gray, W.R.; Zeikus, R.; McIntosh, J.M.; Varga, J.; Rivier, J.; de Santos, V.; Cruz, L.J. Peptide neurotoxins from fish-hunting cone snails. Science 1985, 230, 1338–1343. [Google Scholar]
- Guzman, F.; Barberis, S.; Illanes, A. Peptide synthesis: Chemical or enzymatic. J. Biotechnol. 2007, 10, 279–314. [Google Scholar]
- Marsh, K.N.; Deev, A.; Wu, A.C.T.; Tran, E.; Klamt, A. Room temperature ionic liquids as replacements for conventional solvents—A review. Korean J. Chem. Eng. 2002, 19, 357–362. [Google Scholar]
- Kumar, D.; Bhalla, T.C. Microbial proteases in peptide synthesis: Approaches and applications. Appl. Microbiol. Biotechnol. 2005, 68, 726–736. [Google Scholar]
- Merrifield, R.B. Solid phase peptide synthesis 1. Synthesis of a tetrapeptide. J. Am. Chem. Soc. 1963, 85, 2149–2154. [Google Scholar] [CrossRef]
- Mutter, M.; Hagenmai, H.; Bayer, E. New method of polypeptide synthesis. Angew.Chem. Int. Ed. 1971, 10, 811–812. [Google Scholar]
- Horvath, I.T. Fluorousbiphase chemistry. Acc. Chem. Res. 1998, 31, 641–650. [Google Scholar]
- Mizuno, M.; Goto, K.; Miura, T.; Hosaka, D.; Inazu, T. A novel peptide synthesis using fluorous chemistry. Chem. Commun. 2003, 972–973. [Google Scholar]
- Xing, G.W.; Li, F.Y.; Ming, C.; Ran, L.N. Peptide bond formation catalyzed by alpha-chymotrypsin in ionic liquids. Tetrahedron Lett. 2007, 48, 4271–4274. [Google Scholar]
- Erbeldinger, M.; Mesiano, A.J.; Russell, A.J. Enzymatic catalysis of formation of Z-aspartame in ionic liquid—An alternative to enzymatic catalysis in organic solvents. Biotechnol. Prog. 2000, 16, 1129–1131. [Google Scholar] [CrossRef]
- Smith, V.F.; de Long, H.C.; Sutto, T.E.; Trulove, P.C. Nonenzymatic Synthesis of Peptides in Ionic Liquids. In Proceedings of the Electrochemical Society, Molten Salts XIII, Annapolis, MD, USA; pp. 268–275, PV 2002-19.
- Vallette, H.; Ferron, L.; Coquerel, G.; Gaumont, A.C.; Plaquevent, J.C. Peptide synthesis in room temperature ionic liquids. Tetrahedron Lett. 2004, 45, 1617–1619. [Google Scholar]
- Vallette, H.; Ferron, L.; Coquerel, G.; Guillen, F.; Plaquevent, J.C. Room temperature ionic liquids (RTIL’s) are convenient solvents for peptide synthesis. ARKIVOC 2006, iv, 200–211. [Google Scholar]
- Chan, T.-H.; He, X. Imidazolium-type Ionic Oligomers. U.S. Patent 2010/0093975 A1, 15 April 2010. [Google Scholar]
- Miao, W.; Chan, T.H. Ionic-liquid-supported peptide synthesis demonstrated by the synthesis of Leu(5)-enkephalin. J. Org. Chem. 2005, 70, 3251–3255. [Google Scholar]
- Kim, D.W.; Choe, Y.S.; Chi, D.Y. A new nucleophilic fluorine-18 labeling method for aliphatic mesylates: Reaction in ionic liquids shows tolerance for water. Nucl. Med. Biol. 2003, 30, 345–350. [Google Scholar]
- Schirrmacher, R.; Wängler, C.; Schirrmacher, E. Recent developments and trends in 18F-radiochemistry: Syntheses and applications. Mini-Rev. Org. Chem. 2007, 4, 317–329. [Google Scholar] [CrossRef]
- Miloslavina, A.; Ebert, C.; Tietze, D.; Ohlenschlager, O.; Englert, C.; Gorlach, M.; Imhof, D. An unusual peptide from Conus villepinii: Synthesis, solution structure, and cardioactivity. Peptides 2010, 31, 1292–1300. [Google Scholar] [CrossRef]
- Bihari, M.; Russell, T.P.; Hoagland, D.A. Dissolution and dissolved state of cytochrome c in a neat, hydrophilic ionic liquid. Biomacromolecules 2010, 11, 2944–2948. [Google Scholar]
- Constantinescu, D.; Weingartner, H.; Herrmann, C. Protein denaturation by ionic liquids and the Hofmeister series: A case study of aqueous solutions of ribonuclease A. Angew. Chem. Int. Ed. 2007, 46, 8887–8889. [Google Scholar]
- Noritomi, H.; Suzuki, K.; Kikuta, M.; Kato, S. Catalytic activity of α-chymotrypsin in enzymatic peptide synthesis in ionic liquids. Biochem. Eng. J. 2009, 47, 27–30. [Google Scholar] [CrossRef]
- Wang, Z.; Xiao, H.; Han, Y.; Jiang, P.; Zhou, Z. The effect of four imidazolium ionic liquids on hen egg white lysozyme solubility. J. Chem. Eng. Data 2011, 56, 1700–1703. [Google Scholar]
- Summers, C.A.; Flowers, R.A., II. Protein renaturation by the liquid organic salt ethylammonium nitrate. Protein Sci. 2000, 9, 2001–2008. [Google Scholar]
- Buchfink, R.; Tischer, A.; Patil, G.; Rudolph, R.; Lange, C. Ionic liquids as refolding additives: Variation of the anion. J. Biotechnol. 2010, 150, 64–72. [Google Scholar]
- Sasmal, D.K.; Mondal, T.; Sen Mojumdar, S.; Choudhury, A.; Banerjee, R.; Bhattacharyya, K. An FCS study of unfolding and refolding of CPM-labeled human serum albumin: Role of ionic liquid. J. Phys. Chem. B 2011, 115, 13075–13083. [Google Scholar]
- Vasantha, T.; Attri, P.; Venkatesu, P.; Devi, R.S.R. Thermodynamic contributions of peptide backbone unit from water to biocompatible ionic liquids at T = 298.15 K. J. Chem. Thermodyn. 2012, 45, 122–136. [Google Scholar] [CrossRef]
- Wehofsky, N.; Wespe, C.; Cerovsky, V.; Pech, A.; Hoess, E.; Rudolph, R.; Bordusa, F. Ionic liquids and proteases: A clean alliance for semisynthesis. ChemBioChem 2008, 9, 1493–1499. [Google Scholar]
- Ternois, J.; Ferron, L.; Coquerel, G.; Guillen, F.; Plaquevent, J.-C. Ionic Liquids: New Opportunities for the Chemistry of Amino Acids, Peptides, and Pharmaceutical Compounds. In Ionic Liquid Applications: Pharmaceuticals, Therapeutics, and Biotechnology; American Chemical Society, Oxford University Press: Cary, NC, USA, 2010; Volume 1038, pp. 13–24. [Google Scholar]
- Malhotra, S.V.; Zhang, C.; Wang, H. Enzymatic synthesis of dipeptides in ionic liquids. Lett. Org. Chem. 2010, 7, 168–171. [Google Scholar]
- Ferron, L.; Guillen, F.; Coste, S.; Coquerel, G.; Cardinaël, P.; Schwartz, J.; Paris, J.-M.; Plaquevent, J.-C. Design and scalable synthesis of new chiral selectors. Part 1: Synthesis and characterization of a new constrained cyclopeptide from unnatural bulky amino acids. Tetrahedron 2011, 67, 6036–6044. [Google Scholar]
- Petiot, P.; Charnay, C.; Martinez, J.; Puttergill, L.; Galindo, F.; Lamaty, F.; Colacino, E. Synthesis of a new hydrophilic poly(ethylene glycol)-ionic liquid and its application in peptide synthesis. Chem. Commun. (Camb) 2010, 46, 8842–8844. [Google Scholar]
- He, X.; Chan, T.H. Structurally defined imidazolium-type ionic oligomers as soluble/solid support for peptide synthesis. Org. Lett. 2007, 9, 2681–2684. [Google Scholar]
- Bonnette, F.; Mincheva, Z.; Lavastre, O. Functionalized ionic liquids as new supports for peptide coupling and traceless catalyzed carbon-carbon coupling reactions. Comb. Chem. High Throughput Screen. 2006, 9, 229–232. [Google Scholar]
- Mincheva, Z.; Bonnette, F.; Lavastre, O. Ionic liquid supports stable under conditions of peptide couplings, deprotections and traceless Suzuki reactions. Collect. Czech. Chem. Commun. 2007, 72, 417–434. [Google Scholar]
- Guillen, F.; Brégeon, D.; Plaquevent, J.-C. (S)-Histidine: The ideal precursor for a novel family of chiralaminoacid and peptidic ionic liquids. Tetrahedron Lett. 2006, 47, 1245–1248. [Google Scholar] [CrossRef]
- Chen, L.; Zheng, M.F.; Zhou, Y.; Liu, H.; Jiang, H.L. Ionic-liquid-supported total synthesis of sansalvamide a peptide. Synth.Commun. 2008, 38, 239–248. [Google Scholar]
- Roche, C.; Pucheault, M.; Vaultier, M.; Commerçon, A. Onium salt supported peptide synthesis. Tetrahedron 2010, 66, 8325–8334. [Google Scholar]
- Cho, H.-J.; Lee, S.-M.; Jung, S.; Lee, T.-K.; Yoon, H.-J.; Lee, Y.-S. Ionic liquid incorporated polystyrene resin for solid-phase peptide synthesis. Tetrahedron Lett. 2011, 52, 1459–1461. [Google Scholar]
- Bolton, R. Radiohalogen incorporation into organic systems. J. Label. Compd. Radiopharm. 2002, 45, 485–528. [Google Scholar]
- Moroder, L.; Besse, D.; Musiol, H.J.; RudolphBohner, S.; Siedler, F. Oxidative folding of cystine-rich peptides vs. regioselective cysteine pairing strategies 1. Biopolymers 1996, 40, 207–234. [Google Scholar] [CrossRef]
- Moroder, L.; Musiol, H.J.; Gotz, M.; Renner, C. Synthesis of single- and multiple-stranded cystine-rich peptides. Biopolymers 2005, 80, 85–97. [Google Scholar]
- Moroder, L.; Musiol, H.-J.; Schaschke, N.; Chen, L.; Hargittal, B.; Barany, G. Methods of Organic Chemistry, Synthesis of Peptides and Peptidomimetics; Thieme-Verlag, S., Goodman, M., Felix, A., Moroder, L., Toniolo, C., Eds.; Houben-Weyl, Georg Thieme Verlag: Stuttgart, Germany, 2003; Volume E22a, pp. 384–423. [Google Scholar]
- Besser, D.; Muller, B.; Agricola, I.; Reissmann, S. Synthesis of differentially protected N-acylated reduced pseudodipeptides as building units for backbone cyclic peptides. J. Pept. Sci. 2000, 6, 130–138. [Google Scholar]
- Reissmann, S.; Imhof, D. Development of conformationally restricted analogues of bradykininand somatostatin using constrained amino acids and different types of cyclization. Curr.Med. Chem. 2004, 11, 2823–2844. [Google Scholar]
- Buczek, P.; Buczek, O.; Bulaj, G. Total chemical synthesis and oxidative folding of delta-conotoxin PVIA containing an N-terminal propeptide. Biopolymers 2005, 80, 50–57. [Google Scholar]
- Angell, Y.M.; Alsina, J.; Albericio, F.; Barany, G. Practical protocols for stepwise solid-phase synthesis of cysteine-containing peptides. J. Pept. Res. 2002, 60, 292–299. [Google Scholar]
- Han, Y.; Albericio, F.; Barany, G. Occurrence and minimization of cysteineracemization during stepwise solid-phase peptide synthesis. J. Org. Chem. 1997, 62, 4307–4312. [Google Scholar]
- DeLa Cruz, R.; Whitby, F.G.; Buczek, O.; Bulaj, G. Detergent-assisted oxidative folding of delta-conotoxins. J. Pept. Res. 2002, 61, 202–212. [Google Scholar]
- Fuller, E.; Green, B.R.; Catlin, P.; Buczek, O.; Nielsen, J.S.; Olivera, B.M.; Bulaj, G. Oxidative folding of conotoxins sharing an identical disulfide bridging framework. FEBS J. 2005, 272, 1727–1738. [Google Scholar]
- Lin Huang, J.; Noss, M.E.; Schmidt, K.M.; Murray, L.; Bunagan, M.R. The effect of neat ionic liquid on the folding of short peptides. Chem. Commun. 2011, 47, 8007–8009. [Google Scholar]
- Debeljuh, N.; Barrow, C.J.; Henderson, L.; Byrne, N. Structure inducing ionic liquids-enhancement of alpha helicity in the Abeta(1–40) peptide from Alzheimer’s disease. Chem. Commun. 2011, 47, 6371–6373. [Google Scholar]
- Dawson, P.E.; Muir, T.W.; Clark-Lewis, I.; Kent, S.B. Synthesis of proteins by native chemical ligation. Science 1994, 266, 776–779. [Google Scholar]
- Kent, S.B. Total chemical synthesis of proteins. Chem. Soc. Rev. 2009, 38, 338–351. [Google Scholar]
- Muir, T.W.; Dawson, P.E.; Kent, S.B. Protein synthesis by chemical ligation of unprotected peptides in aqueous solution. Methods Enzymol. 1997, 289, 266–298. [Google Scholar]
- Wieland, T.; Bokelmann, E.; Bauer, L.; Lang, H.U.; Lau, H. Über Peptidsynthesen. 8. Mitteilung Bildung von S-haltigen Peptiden durch intramolekulare Wanderung von Aminoacylresten. Liebigs Ann. Chem. 1953, 583, 129–149. [Google Scholar] [CrossRef]
- Romanelli, A.; Shekhtman, A.; Cowburn, D.; Muir, T.W. Semisynthesis of a segmental isotopically labeled protein splicing precursor: NMR evidence for an unusual peptide bond at the N-extein-intein junction. Proc. Natl. Acad. Sci. USA 2004, 101, 6397–6402. [Google Scholar]
- Böhm, M.; Kühl, T.; Hardes, K.; Coch, R.; Arkona, C.; Schlott, B.; Steinmetzer, T.; Imhof, D. Synthesis and functional characterization of tridegin and its analogues: Inhibitors and substrates of factor XIIIa. ChemMedChem 2012, 7, 326–333. [Google Scholar]
- Hackeng, T.M.; Griffin, J.H.; Dawson, P.E. Protein synthesis by native chemical ligation: Expanded scope by using straightforward methodology. Proc. Natl. Acad. Sci. USA 1999, 96, 10068–10073. [Google Scholar]
- Johnson, E.C.; Kent, S.B. Insights into the mechanism and catalysis of the native chemical ligation reaction. J. Am. Chem. Soc. 2006, 128, 6640–6646. [Google Scholar]
- Dawson, P.E.; Churchill, M.J.; Ghadiri, M.R.; Kent, S.B. Modulation of reactivity in NCL through the use of thiol additives. J. Am. Chem. Soc. 1997, 119, 4325–4329. [Google Scholar]
- Escher, S.E.; Klüver, E.; Adermann, K. Fmoc-based synthesis oft he human CC chemokine CCL14/HCC-1 by SPPS and native chemical ligation. Lett.Pept. Sci. 2002, 8, 349–357. [Google Scholar]
- Han, D.; Row, K. Recent applications of ionic liquids in separation technology. Molecules 2010, 15, 2405–2426. [Google Scholar]
- Polyakova, Y.; Koo, Y.; Row, K. Application of ionic liquids as mobile phase modifier in HPLC. Biotechnol. Bioprocess Eng. 2006, 11, 1–6. [Google Scholar]
- Polyakova, Y.; Koo, Y.; Row, K. Application of ionic liquids of some bioactive molecules in RP-HPLC. Rev. Anal. Chem. 2007, 26, 77–98. [Google Scholar]
- Wang, Y.; Tian, M.; Bi, W.; Row, K. Application of ionic liquids in high performance reversed-phase chromatography. Int. J. Mol. Sci. 2009, 10, 2591–2610. [Google Scholar]
- Chitta, K.; Van Meter, D.; Stalcup, A. Separation of peptides by HPLC using a surface-confined ionic liquid stationary phase. Anal.Bioanal. Chem. 2010, 396, 775–781. [Google Scholar]
- Marszall, M.; Kaliszan, R. Application of ionic liquids in liquid chromatography. Crit. Rev. Anal. Chem. 2007, 37, 127–140. [Google Scholar]
- Abraham, M.; Poole, C.; Poole, S. Solute effects on reversed-phase thin-layer chromatography A linear free energy relationship analysis. J. Chromatogr. A 1996, 749, 201–209. [Google Scholar]
- Abraham, M.; Treiner, C.; Roses, M.; Rafols, C.; Ishihama, Y. Linear free energy relationship analysis of microemulsionelectrokinetic chromatographic determination of lipophilicity. J. Chromatogr. A 1996, 752, 243–249. [Google Scholar]
- Van Meter, D.; Sun, Y.; Parker, K.; Stalcup, A. Retention characteristics of a new butylimidazolium-based stationary phase. Part II: Anion exchange and partitioning. Anal. Bioanal. Chem. 2008, 390, 897–905. [Google Scholar] [CrossRef]
- Qiu, H.; Jiang, S.; Liu, X. N-methylimidazolium anion-exchange stationary phase for high-performance liquid chromatography. J. Chromatogr. A 2006, 1103, 265–270. [Google Scholar] [CrossRef]
- Van Meter, D.; Oliver, N.; Carle, A.; Dehm, S.; Ridgway, T.; Stalcup, A. Characterization of surface-confined ionic liquid stationary phases: Impact of cation and anion identity on retention. Anal.Bioanal. Chem. 2009, 393, 283–294. [Google Scholar]
- Kaliszan, R.; Marszall, M.; Markuszewski, M.; Baczek, T.; Pernak, J. Suppression of deleterious effects of free silanols in liquid chromatography by imidazoliumtetrafluoroborate ionic liquids. J. Chromatogr. A 2004, 1030, 263–271. [Google Scholar] [CrossRef]
- Marszall, M.; Baczek, T.; Kaliszan, R. Reduction of silanophilic interactions in liquid chromatography with the use of ionic liquids. Anal. Chim. Acta 2005, 547, 172–178. [Google Scholar]
- He, L.; Zhang, W.; Zhao, L.; Liu, X.; Jiang, S. Effect of 1-alkyl-3-methylimidazolium-based ionic liquids as the eluent on the separation of ephedrines by liquid chromatography. J. Chromatogr. A 2003, 1007, 39–45. [Google Scholar] [CrossRef]
- Marszall, M.; Baczek, T.; Kaliszan, R. Evaluation of the silanol-suppressing potency of ionic liquids. J. Sep. Sci. 2006, 29, 1138–1145. [Google Scholar]
- Baczek, T.; Sparzak, B. Ionic liquids as novel solvent additives to separate peptides. Z. Naturforsch C 2006, 61, 827–832. [Google Scholar]
- Baczek, T.; Marszall, M.P.; Kaliszan, R.; Walijewski, L.; Makowiecka, W.; Sparzak, B.; Grzonka, Z.; Wisniewska, K.; Juszczyk, P. Behavior of peptides and computer-assisted optimization of peptides separations in a normal-phase thin-layer chromatography system with and without the addition of ionic liquid in the eluent. Biomed.Chromatogr. 2005, 19, 1–8. [Google Scholar]
- Mowthorpe, S.; Clench, M.; Cricelius, A.; Richards, D.; Parr, V.; Tetler, L. Matrix-assisted laser desorption/ionisation time-of-flight thin layer chromatography mass spectrometry—A rapid method for impurity testing. Rapid Commun. Mass Spectrom. 1999, 13, 264–270. [Google Scholar]
- Todd, P.J.; Schaaff, T.G.; Chaurand, P.; Caprioli, R.M. Organic ion imaging of biological tissue with secondary ion mass spectrometry and matrix-assisted laser desorption/ionization. J. Mass Spectrom. 2001, 36, 355–369. [Google Scholar]
- Jackson, S.; Wang, H.; Woods, A. Direct tissue analysis of phospholipids in rat brain using MALDI-TOFMS and MALDI-ion mobility-TOFMS. J. Am. Soc. Mass Spectrom. 2005, 16, 133–138. [Google Scholar]
- Jones, J.J.; Batoy, S.M.; Wilkins, C.L.; Liyanage, R.; Lay, J.O., Jr. Ionic liquid matrix-induced metastable decay of peptides and oligonucleotides and stabilization of phospholipids in MALDI FTMS analyses. J. Am. Soc. Mass Spectrom. 2005, 16, 2000–2008. [Google Scholar]
- Towers, M.W.; McKendrick, J.E.; Cramer, R. Introduction of 4-chloro-alpha-cyanocinnamic acid liquid matrices for high sensitivity UV-MALDI MS. J. Proteome Res. 2010, 9, 1931–1940. [Google Scholar]
- Zabet-Moghaddam, M.; Kruger, R.; Heinzle, E.; Tholey, A. Matrix-assisted laser desorption/ ionization mass spectrometry for the characterization of ionic liquids and the analysis of amino acids, peptides and proteins in ionic liquids. J. Mass Spectrom. 2004, 39, 1494–1505. [Google Scholar]
- Mank, M.; Stahl, B.; Boehm, G. 2,5-Dihydroxybenzoic acid butylamine and other ionic liquid matrixes for enhanced MALDI-MS analysis of biomolecules. Anal.Chem. 2004, 76, 2938–2950. [Google Scholar]
- Tholey, A.; Heinzle, E. Ionic (liquid) matrices for matrix-assisted laser desorption/ionization mass spectrometry-applications and perspectives. Anal.Bioanal. Chem. 2006, 386, 24–37. [Google Scholar]
- Zabet-Moghaddam, M.; Heinzle, E.; Lasaosa, M.; Tholey, A. Pyridinium-based ionic liquid matrices can improve the identification of proteins by peptide mass-fingerprint analysis with matrix-assisted laser desorption/ionization mass spectrometry. Anal. Bioanal. Chem. 2006, 384, 215–224. [Google Scholar]
- Tholey, A.; Zabet-Moghaddam, M.; Heinzle, E. Quantification of peptides for the monitoring of protease-catalyzed reactions by matrix-assisted laser desorption/ionization mass spectrometry using ionic liquid matrixes. Anal. Chem. 2006, 78, 291–297. [Google Scholar]
- Tholey, A. Ionic liquid matrices with phosphoric acid as matrix additive for the facilitated analysis of phosphopeptides by matrix-assisted laser desorption/ionization mass spectrometry. Rapid Commun.Mass Spectrom. 2006, 20, 1761–1768. [Google Scholar]
- Palmblad, M.; Cramer, R. Liquid matrix deposition on conductive hydrophobic surfaces for tuning and quantitation in UV-MALDI mass spectrometry. J. Am. Soc. Mass Spectrom. 2007, 18, 693–697. [Google Scholar]
- Hortal, A.; Hurtado, P.; Martinez-Haya, B. Matrix-assisted laser desorption mass spectrometry of gas-phase peptide-metal complexes. Appl. Phys. A: Mater. Sci. Process. 2008, 93, 935–939. [Google Scholar] [CrossRef]
- Calvano, C.; Carulli, S.; Palmisano, F. Aniline/alpha-cyano-4-hydroxycinnamic acid is a highly versatile ionic liquid for matrix-assisted laser desorption/ionization mass spectrometry. Rapid Commun.Mass Spectrom. 2009, 23, 1659–1668. [Google Scholar]
- Crank, J.; Armstrong, D. Towards a second generation of ionic liquid matrices (ILMs) for MALDI-MS of peptides, proteins, and carbohydrates. J. Am. Soc. Mass Spectrom. 2009, 20, 1790–1800. [Google Scholar] [CrossRef]
- Ullmer, R.; Rizzi, A. Use of a novel ionic liquid matrix for MALDI-MS analysis of glycopeptides and glycans out of total tryptic digests. J. Mass Spectrom. 2009, 44, 1596–1603. [Google Scholar]
- Giménez, E.; Benavente, F.; Barbosa, J.; Sanz-Nebot, V. Ionic liquid matrices for MALDI-TOF-MS analysis of intact glycoproteins. Anal.Bioanal. Chem. 2010, 398, 357–365. [Google Scholar]
- Fitzgerald, J.J.; Kunnath, P.; Walker, A.V. Matrix-enhanced secondary ion mass spectrometry (ME SIMS) using room temperature ionic liquid matrices. Anal. Chem. 2010, 82, 4413–4419. [Google Scholar] [CrossRef]
- Sun, L.; Tao, D.; Han, B.; Ma, J.; Zhu, G.; Liang, Z.; Shan, Y.; Zhang, L.; Zhang, Y. Ionic liquid 1-butyl-3-methyl imidazoliumtetrafluoroborate for shotgun membrane proteomics. Anal.Bioanal. Chem. 2011, 399, 3387–3397. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Tietze, A.A.; Heimer, P.; Stark, A.; Imhof, D. Ionic Liquid Applications in Peptide Chemistry: Synthesis, Purification and Analytical Characterization Processes. Molecules 2012, 17, 4158-4185. https://doi.org/10.3390/molecules17044158
Tietze AA, Heimer P, Stark A, Imhof D. Ionic Liquid Applications in Peptide Chemistry: Synthesis, Purification and Analytical Characterization Processes. Molecules. 2012; 17(4):4158-4185. https://doi.org/10.3390/molecules17044158
Chicago/Turabian StyleTietze, Alesia A., Pascal Heimer, Annegret Stark, and Diana Imhof. 2012. "Ionic Liquid Applications in Peptide Chemistry: Synthesis, Purification and Analytical Characterization Processes" Molecules 17, no. 4: 4158-4185. https://doi.org/10.3390/molecules17044158
APA StyleTietze, A. A., Heimer, P., Stark, A., & Imhof, D. (2012). Ionic Liquid Applications in Peptide Chemistry: Synthesis, Purification and Analytical Characterization Processes. Molecules, 17(4), 4158-4185. https://doi.org/10.3390/molecules17044158





