Regulation of Inflammatory Cytokines in Lipopolysaccharide-Stimulated RAW 264.7 Murine Macrophage by 7-O-Methyl-naringenin
Abstract
:1. Introduction
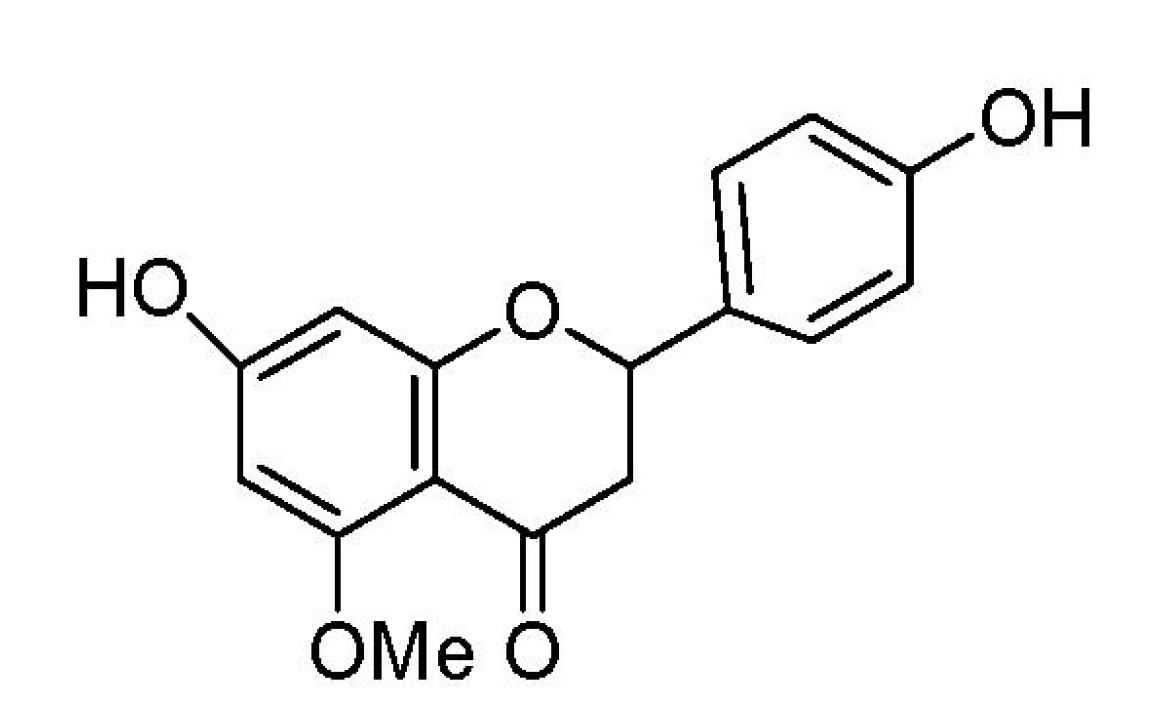
2. Results and Discussion
2.1. Effect of 7-O-Methylnaringenin on Macrophage Toxicity

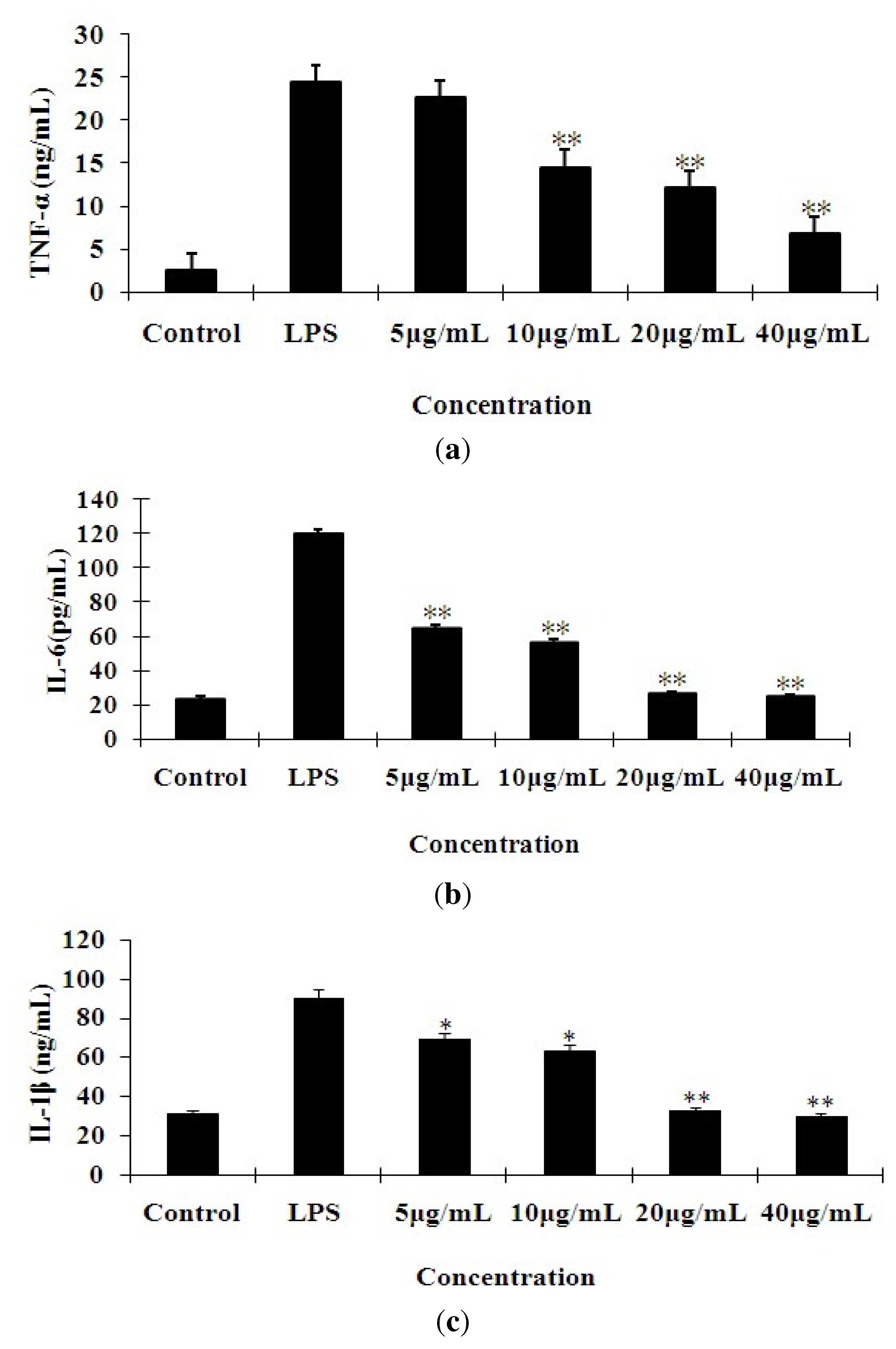
2.2. Effects of 7-O-Methylnaringenin on LPS-Induced Cytokine Production
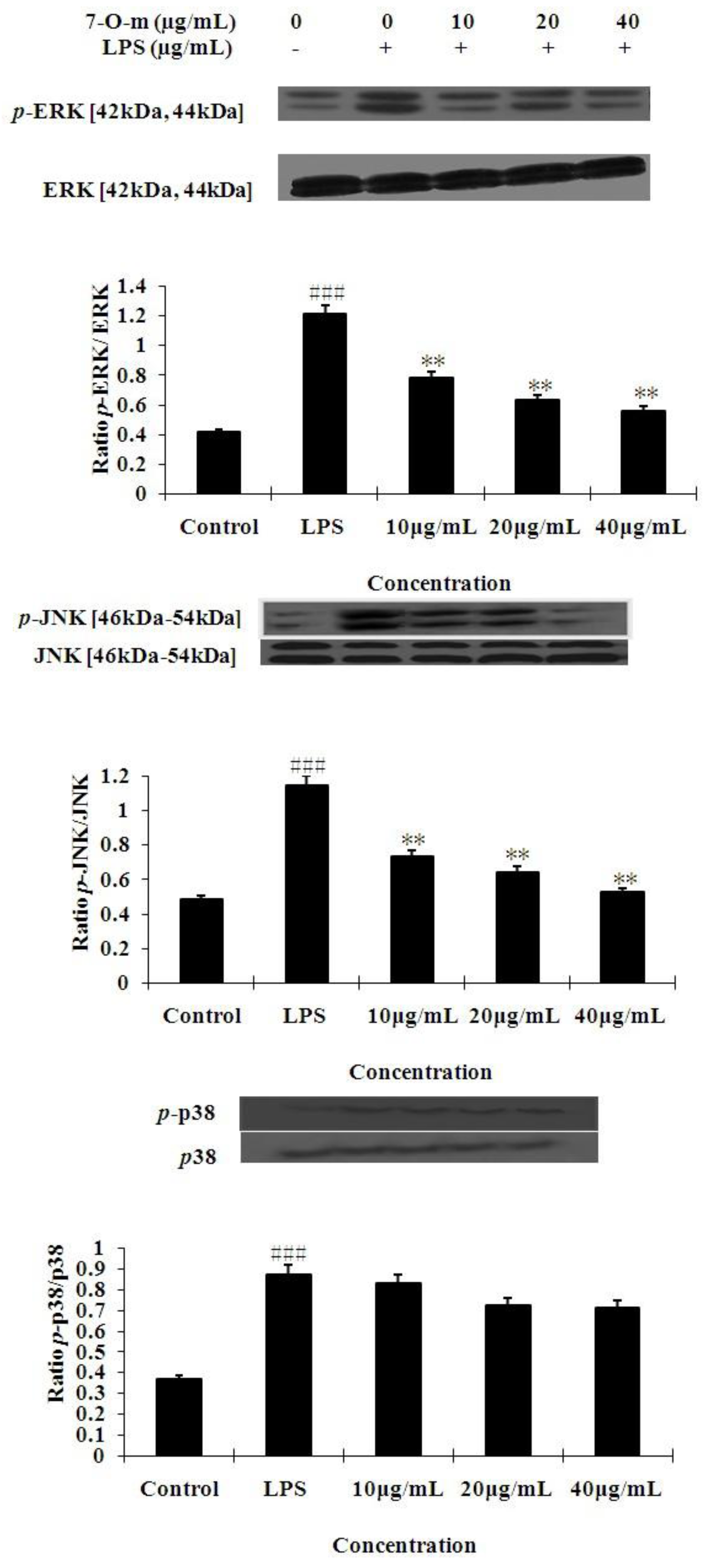
2.3. The Effects of 7-O-Methylnaringenin on LPS-Induced MAPK Pathways Activation
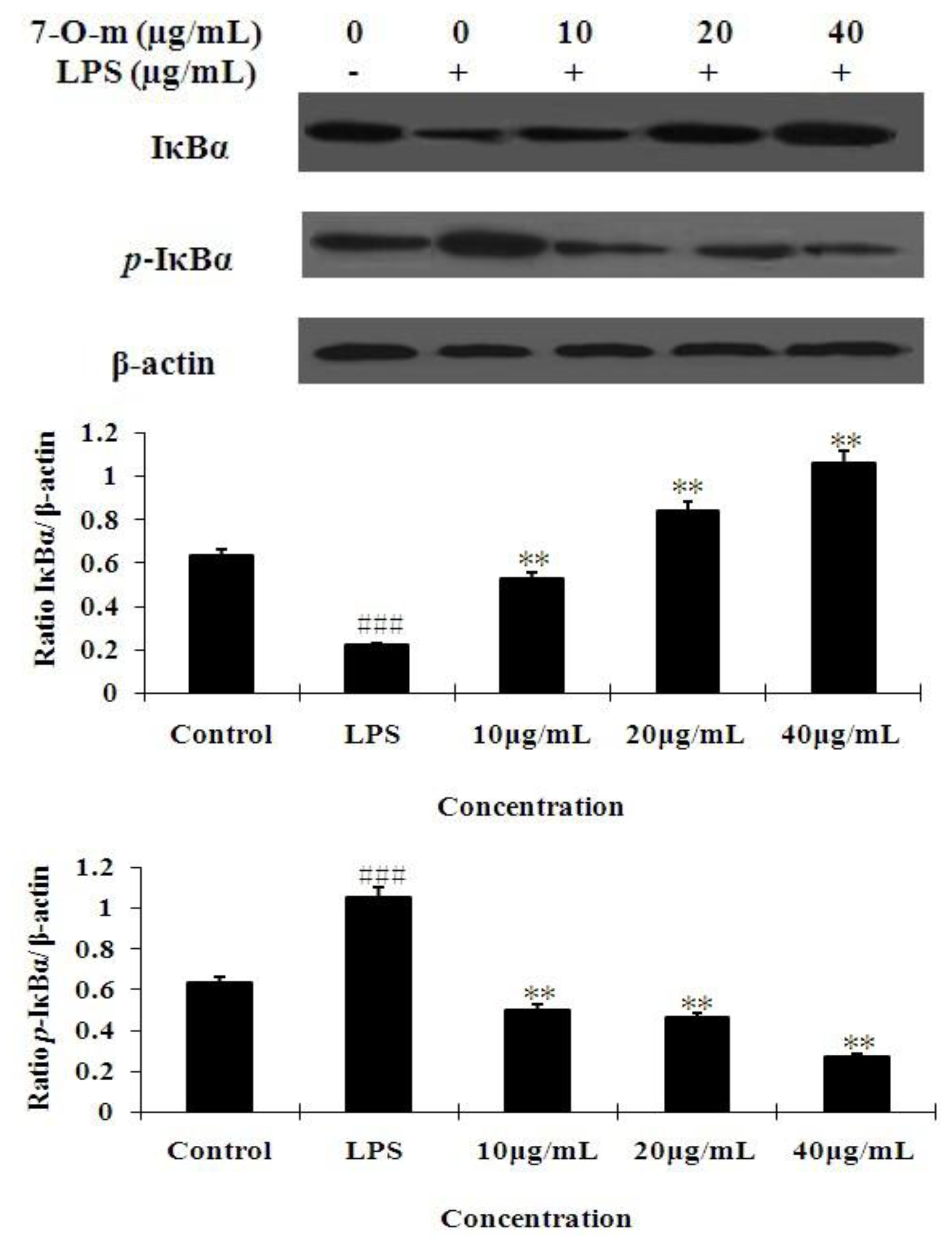
2.4. The Effect of 7-O-Methylnaringenin on LPS-Induced Degradation and Phosphorylation of IκBα
2.5. Discussion
3. Experimental
3.1. Materials
3.2. Preparation of Extract
3.3. Cell Culture and LPS Stimulation
3.4. MTT Assay for Testing Cell Viability
3.5. Measurement of Cytokine Production
3.6. Western Blot Analysis
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
References and Notes
- Feghali, C.A.; Wright, T.M. Cytokines in acute and chronic inflammation. Front. Biosci. 1997, 2, 12–26. [Google Scholar]
- Shimazu, R.; Akashi, S.; Ogata, H.; Nagai, Y.; Fukudome, K.; Miyake, K. MD-2, a molecule that confers lipopolysaccharide responsiveness on Toll-like receptor 4. J. Exp. Med. 1999, 189, 1777–1782. [Google Scholar] [CrossRef]
- Beutler, B.; Krochin, N.; Milsark, I.W.; Luedke, C.; Cerami, A. Control of cachechin (tumor necrosis factor) synthesis: Mechanisms of endotoxin resistance. Science 1986, 232, 977–980. [Google Scholar]
- Morrison, D.C.; Danner, R.L.; Dinarello, C.A.; Munford, R.S.; Natanson, C.; Pollack, M.; Spitzer, J.J.; Ulevitch, R.J.; Vogel, S.N.; McSweegan, E. Bacterial endotoxins and pathogenesis of Gram-negative infections: Current status and future direction. J. Endotoxin Res. 1994, 1, 71–83. [Google Scholar]
- Levine, B.; Kalman, J.; Mayer, L.; Fillit, H.M.; Packer, M. Elevated circulating levels of tumor necrosis factor in severe chronic heart failure. N. Engl. J. Med. 1990, 323, 236–241. [Google Scholar] [CrossRef]
- Bartold, P.M.; Haynes, D.R. Interleukin-6 production by human gingival fibroblasts. J. Periodontal Res. 1991, 26, 339–345. [Google Scholar] [CrossRef]
- van Meir, E.; Sawamura, Y.; Diserens, A.C.; Hamou, M.F.; de Tribolet, N. Human glioblastoma cells release interleukin 6 in vivo and in vitro. Cancer Res. 1990, 50, 6683–6688. [Google Scholar]
- Lu, X.Y.; Wang, Z.C.; Yang, G.Z. The relationships between IL-1, IL-6 and tumor. Guowai Yixue Shengli Bingli Kexue Yu Linchuang Fence 1995, 15, 159–163. [Google Scholar]
- Kielian, T.; Bearden, E.D.; Baldwin, A.C.; Esen, N. IL-1 and TNF-alpha play a pivotal role in the host immune response in a mouse model of Staphylococcus aureus-induced experimental brain abscess. J. Neuropathol. Exp. Neurol. 2004, 63, 381–396. [Google Scholar]
- Morrissey, P.J.; Charrier, K. Treatment of mice with IL-1 before infection increases resistance to a lethal challenge with Salmonella typhimurium. The effect correlates with the resistance allele at the Ity locus. J. Immunol. 1994, 153, 212–219. [Google Scholar]
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomas-Barberan, F.A.; Datta, N.; Singanusong, R. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef]
- Boots, A.W.; Haenen, G.R.; Bast, A. Health effects of quercetin: From antioxidant to nutraceutical. Eur. J. Pharmacol. 2008, 585, 325–337. [Google Scholar] [CrossRef]
- Gomes, A.; Fernandes, E.; Lima, J.L.; Mira, L.; Corvo, M.L. Molecular mechanisms of anti-inflammatory activity mediated by flavonoids. Curr. Med. Chem. 2008, 15, 1586–1605. [Google Scholar] [CrossRef]
- Kawai, M.; Hirano, T.; Higa, S.; Arimitsu, J.; Maruta, M.; Kuwahara, Y. Flavonoids and related compounds as anti-allergic substances. Allergol. Int. 2007, 56, 113–123. [Google Scholar] [CrossRef]
- Cushnie, T.P.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar] [CrossRef]
- Hirpara, K.V.; Aggarwal, P.; Mukherjee, A.J.; Joshi, N.; Burman, A.C. Quercetin and its derivatives: Synthesis, pharmacological uses with special emphasis on anti-tumor properties and prodrug with enhanced bio-availability. Anticancer Agents Med. Chem. 2009, 9, 138–161. [Google Scholar]
- Aalinkeel, R.; Bindukumar, B.; Reynolds, J.L.; Sykes, D.E.; Mahajan, S.D.; Chadha, K.C. The dietary bioflavonoid, quercetin, selectively induces apoptosis of prostate cancer cells by down-regulating the expression of heat shock protein 90. Prostate 2008, 68, 1773–1789. [Google Scholar] [CrossRef]
- Tan, J.; Wang, B.; Zhu, L. Regulation of survivin and Bcl-2 in HepG2 cell apoptosis induced by quercetin. Chem. Biodivers. 2009, 6, 1101–1110. [Google Scholar] [CrossRef]
- Perez-Vizcaino, F.; Duarte, J.; Jimenez, R.; Santos-Buelga, C.; Osuna, A. Antihypertensive effects of the flavonoid quercetin. Pharmacol. Rep. 2009, 61, 67–75. [Google Scholar]
- Saito, T.; Abe, D.; Sekiya, K. Sakuranetin induces adipogenesis of 3T3-L1cells through enhanced expression of PPARgamma2. Biochem. Biophys. Res. Commun. 2008, 372, 835–839. [Google Scholar] [CrossRef]
- Ahmed, M.S.; Galal, A.M.; Ross, S.A.; Ferreira, D.; ElSohly, M.A.; Ibrahim, A.S.; Mossa, J.S.; El-Feraly, F.S. A weakly antimalarial biflavanone from Rhus retinorrhoea. Phytochemistry 2001, 58, 599–602. [Google Scholar] [CrossRef]
- Johnson, G.L.; Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinase. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef]
- Caivano, M. Role of MAP kinase cascades in inducing arginine transporters and nitric oxide synthetase in RAW 264 macrophages. FEBS Lett. 1998, 429, 249–253. [Google Scholar] [CrossRef]
- Bian, Z.M.; Elner, S.G.; Yoshida, A.; Elner, V.M. Human RPE-monocyte co-culture induces chemokine gene expression through activation of MAPK and NIK cascade. Exp. Eye Res. 2003, 76, 573–583. [Google Scholar] [CrossRef]
- Kontoyiannis, D.; Pasparakis, M.; Pizarro, T.T.; Cominelli, F.; Kollias, G. Impaired on/off regulation of TNF biosynthesis in mice lacking TNF AU-rich elements: Implications for joint and gut-associated immunopathologies. Immunity 1999, 10, 387–398. [Google Scholar]
- Kotlyarov, A.; Neininger, A.; Schubert, C.; Eckert, R.; Birchmeier, C.; Volk, H.D.; Gaestel, M. MAPKAP kinase 2 is essential for LPS-induced TNF-alpha biosynthesis. Nat. Cell Biol. 1999, 1, 94–97. [Google Scholar] [CrossRef]
- Dumitru, C.D.; Ceci, J.D.; Tsatsanis, C.; Kontoyiannis, D.; Stamatakis, K.; Lin, J.H.; Patriotis, C.; Jenkins, N.A.; Copeland, N.G.; Kollias, G.; et al. TNF-α induction by LPS is regulated posttranscriptionally via a Tpl2/ERK-dependent pathway. Cell 2000, 103, 1071–1083. [Google Scholar] [CrossRef]
- Baeuerle, P.A.; Baltimore, D. NF-kappa B: Ten years after. Cell 1996, 87, 13–20. [Google Scholar] [CrossRef]
- Phan, H.H.; Cho, K.; Sainz-Lyon, K.S.; Shin, S.; Greenhalgh, D.G. CD14-dependent modulation of NF-κB alternative splicing in lung after burn injury. Gene 2006, 371, 121–129. [Google Scholar] [CrossRef]
- Sample Availability: Samples of 7-O-methylnaringenin are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Soromou, L.W.; Zhang, Z.; Li, R.; Chen, N.; Guo, W.; Huo, M.; Guan, S.; Lu, J.; Deng, X. Regulation of Inflammatory Cytokines in Lipopolysaccharide-Stimulated RAW 264.7 Murine Macrophage by 7-O-Methyl-naringenin. Molecules 2012, 17, 3574-3585. https://doi.org/10.3390/molecules17033574
Soromou LW, Zhang Z, Li R, Chen N, Guo W, Huo M, Guan S, Lu J, Deng X. Regulation of Inflammatory Cytokines in Lipopolysaccharide-Stimulated RAW 264.7 Murine Macrophage by 7-O-Methyl-naringenin. Molecules. 2012; 17(3):3574-3585. https://doi.org/10.3390/molecules17033574
Chicago/Turabian StyleSoromou, Lanan Wassy, Zhichao Zhang, Rongtao Li, Na Chen, Weixiao Guo, Meixia Huo, Shuang Guan, Jing Lu, and Xuming Deng. 2012. "Regulation of Inflammatory Cytokines in Lipopolysaccharide-Stimulated RAW 264.7 Murine Macrophage by 7-O-Methyl-naringenin" Molecules 17, no. 3: 3574-3585. https://doi.org/10.3390/molecules17033574
APA StyleSoromou, L. W., Zhang, Z., Li, R., Chen, N., Guo, W., Huo, M., Guan, S., Lu, J., & Deng, X. (2012). Regulation of Inflammatory Cytokines in Lipopolysaccharide-Stimulated RAW 264.7 Murine Macrophage by 7-O-Methyl-naringenin. Molecules, 17(3), 3574-3585. https://doi.org/10.3390/molecules17033574



