A Facile Synthesis of New Monoazo Disperse Dyes Derived from 4-Hydroxyphenylazopyrazole-5-amines: Evaluation of Microwave Assisted Dyeing Behavior
Abstract
:1. Introduction
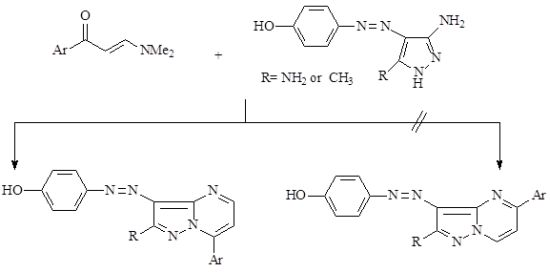
2. Results and Discussion
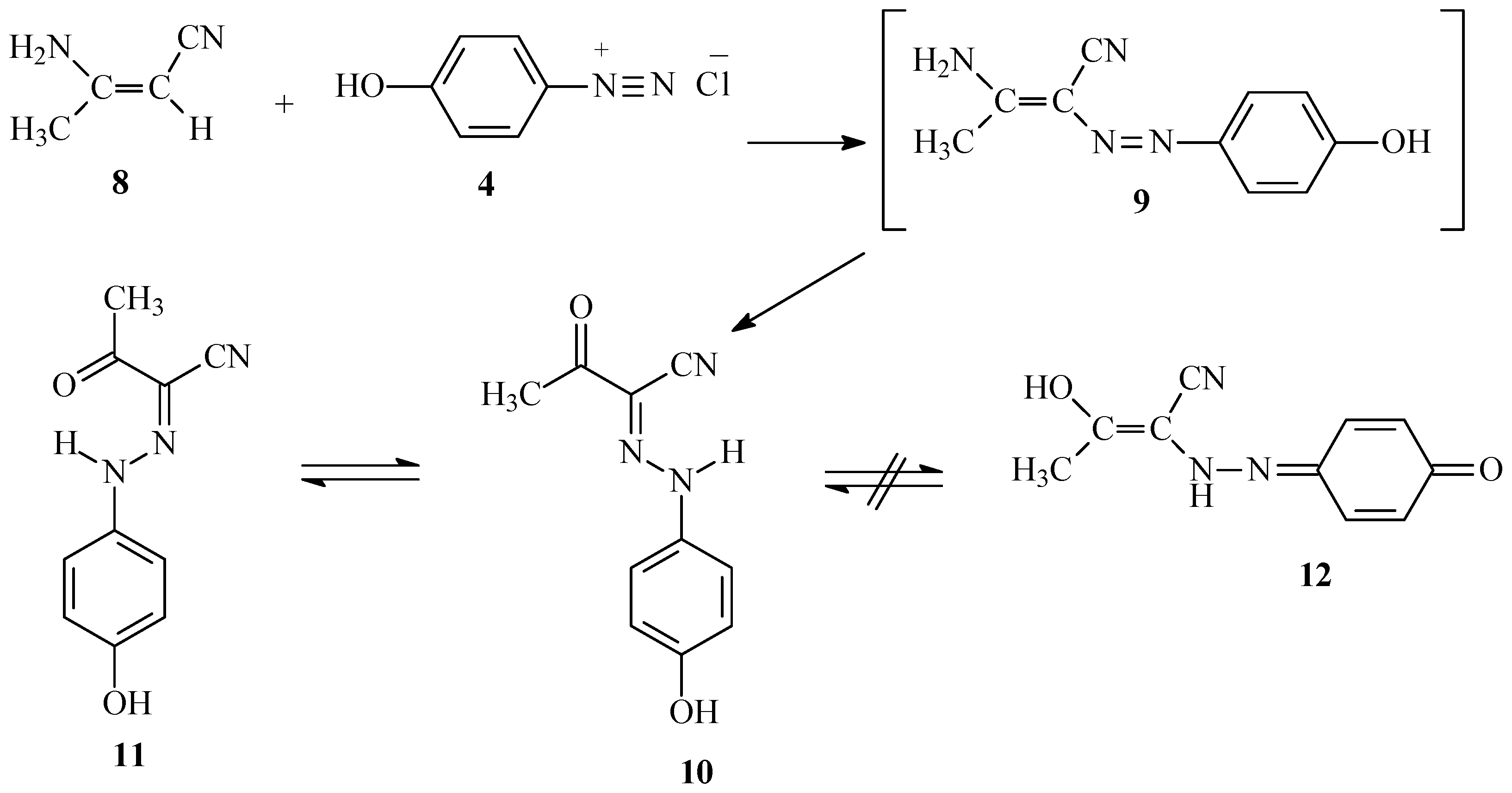
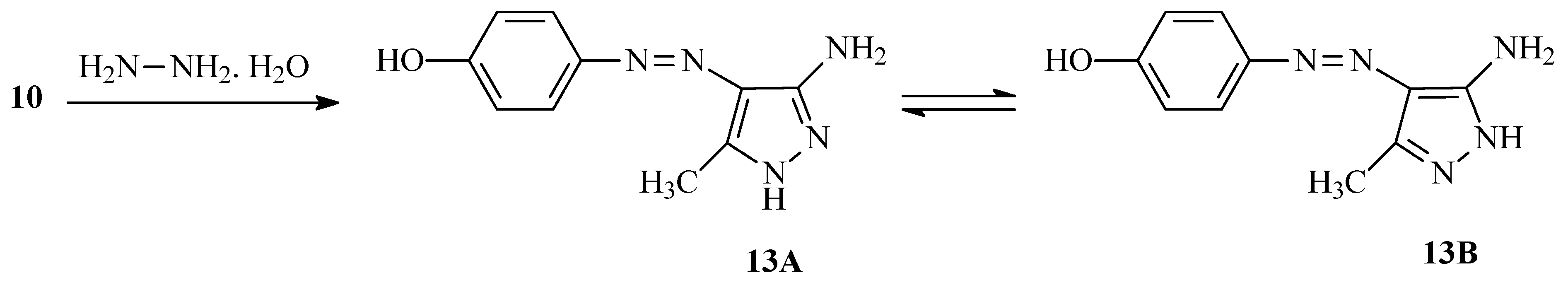
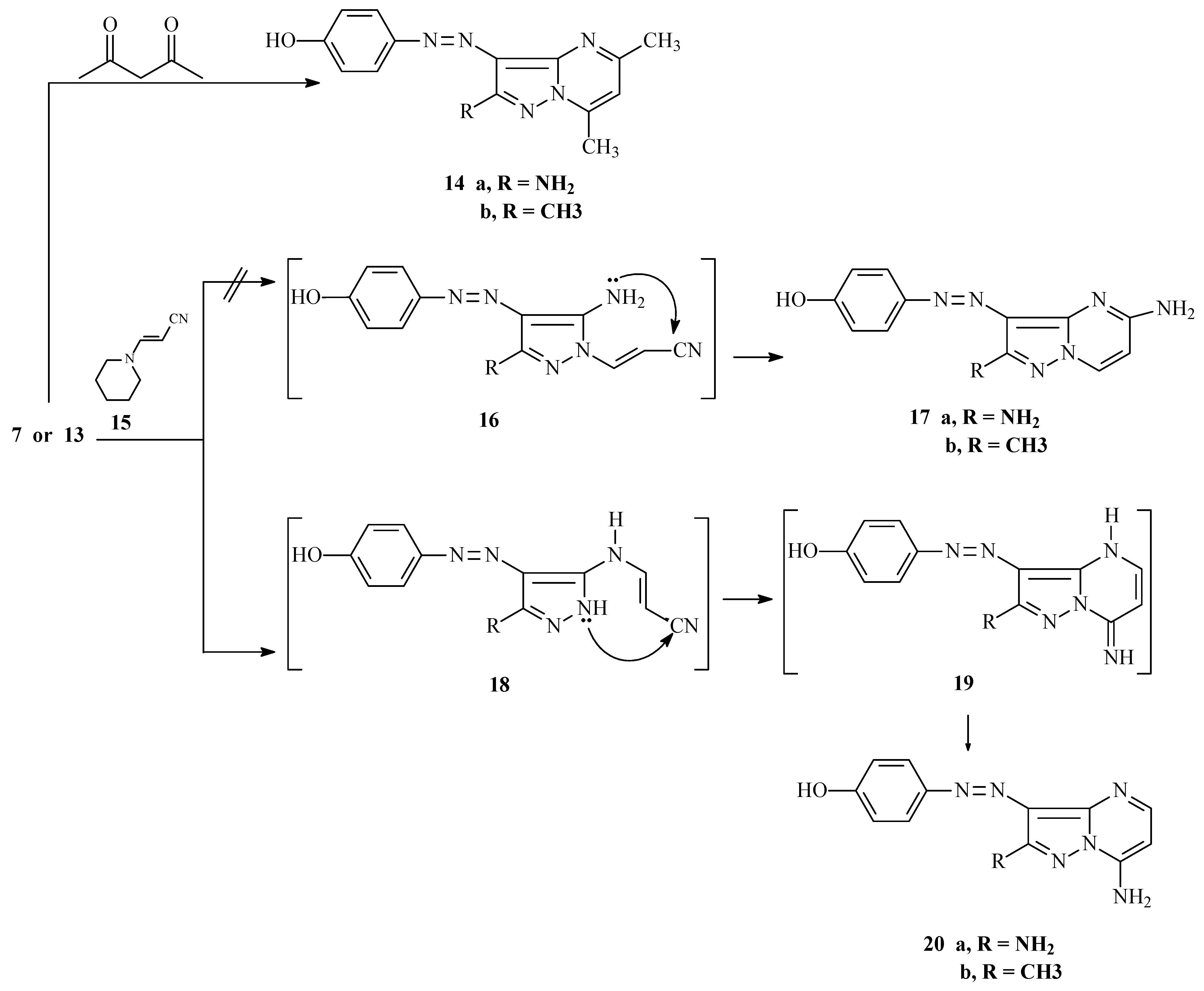
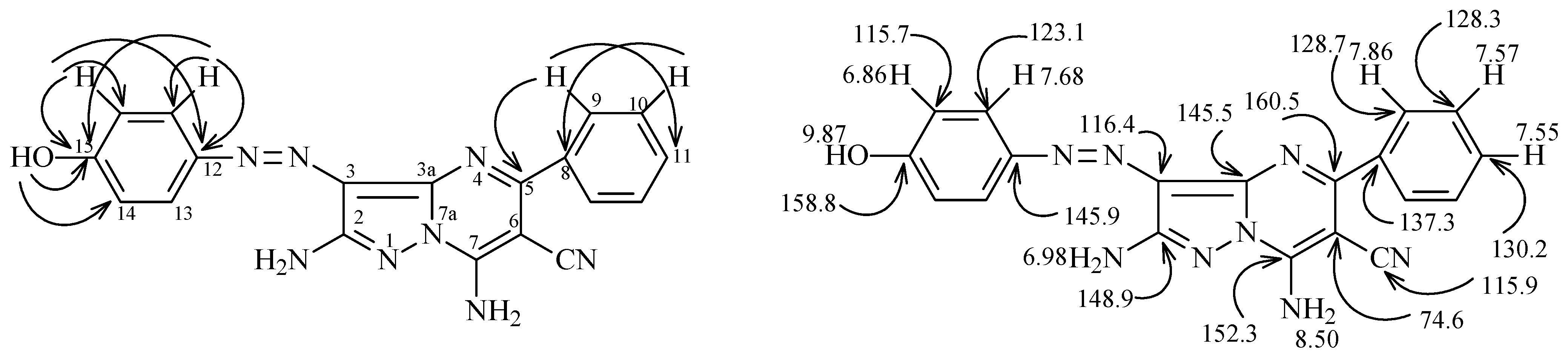
2.1. Synthesis

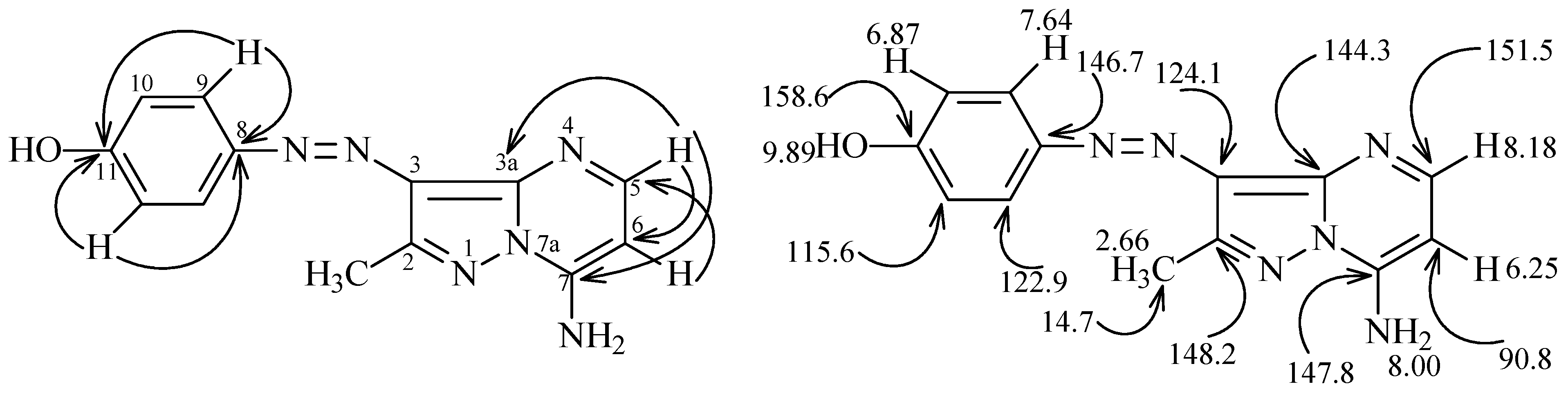

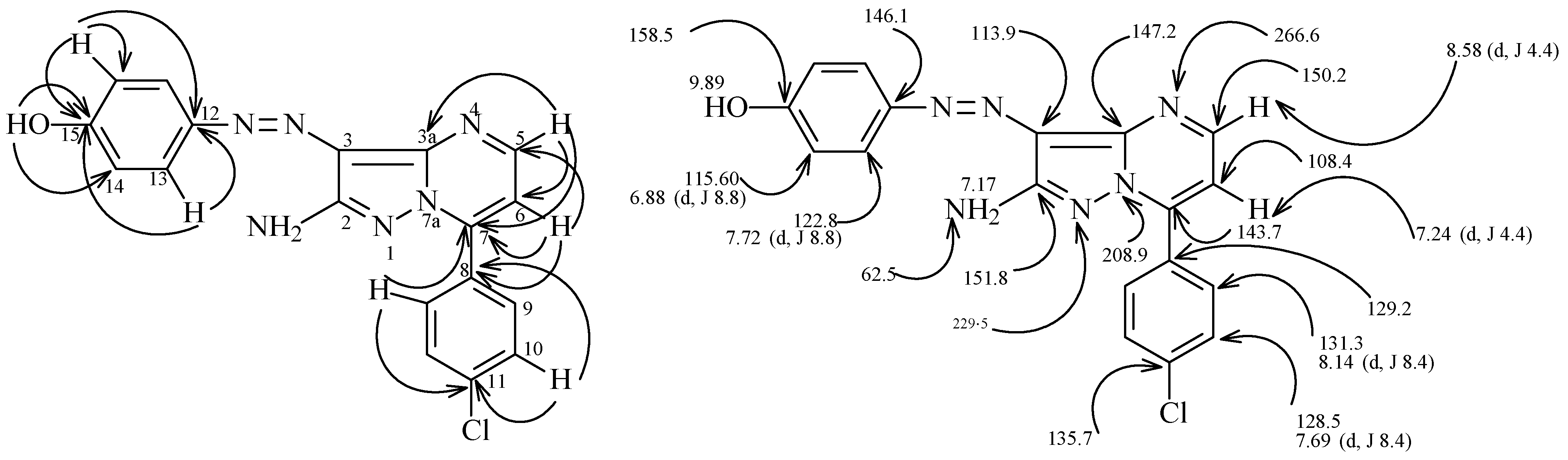
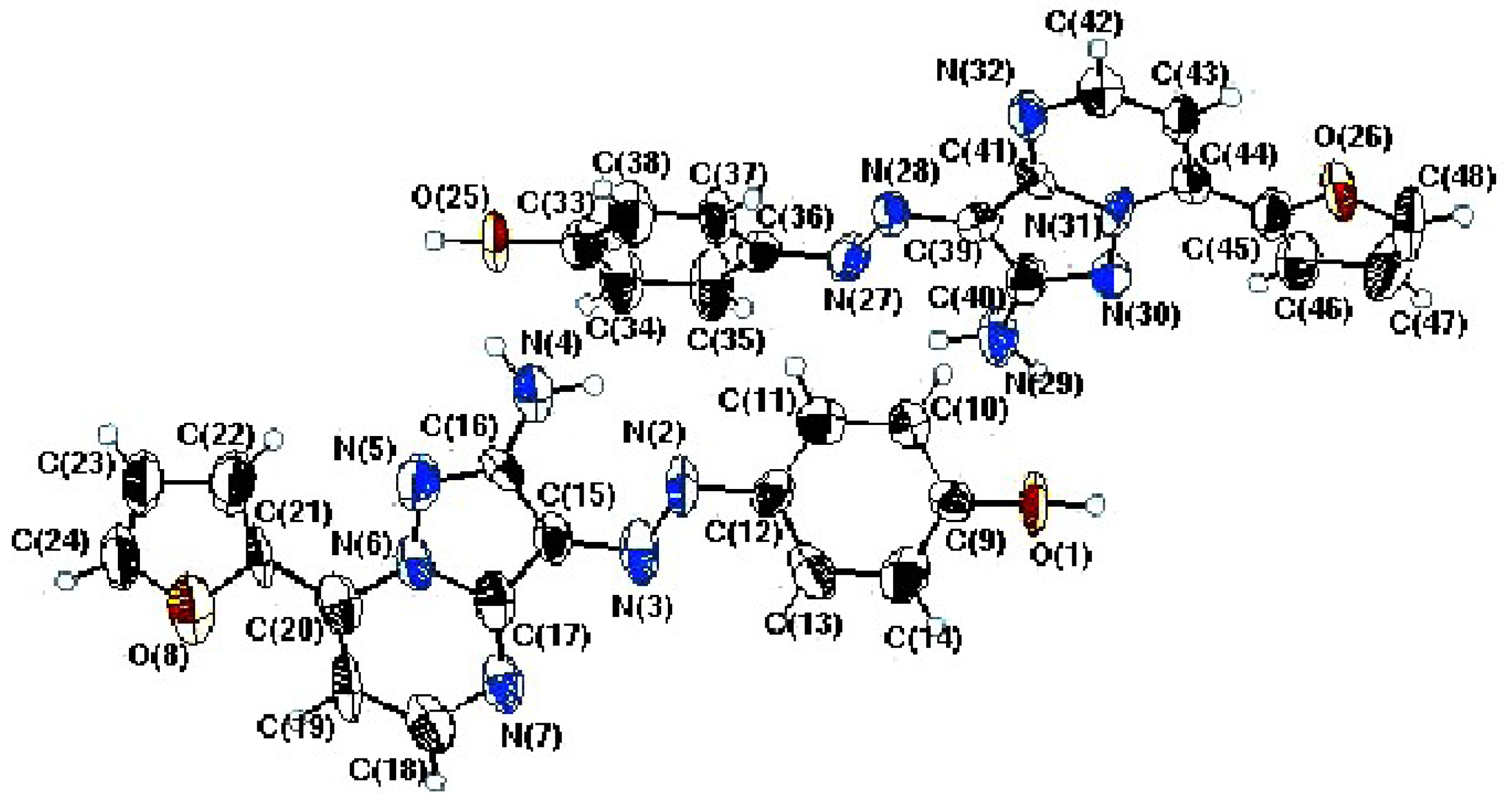
| Bond | Bond length (Å) | Bond | Bond angle (°) |
|---|---|---|---|
| N27—N28 | 1.286 (6) | N31—C44—C45 | 119.7 (6) |
| N31—N30 | 1.373 (6) | C36—N27—N28 | 114.3 (6) |
| N30—C40 | 1.345 (7) | N30—N31—C44 | 126.3 (6) |
| N32—C41 | 1.357 (6) | N31—C41—C39 | 103.7 (7) |
| N32—C42 | 1.324 (7) | C44—N31—C41 | 120.9 (6) |
| N27—C36 | 1.416 (7) | C44—N31—C41 | 120.9 (6) |
| N29—C40 | 1.345 (7) | C40—N29—H29A | 119.9(5) |
| N31—C41 | 1.437 (7) | C40—N29—H29B | 120.1(5) |
| Chemical formula | C16H12N6O2 | Z | 4 |
|---|---|---|---|
| Formula weight | 320.312 | Temperature | 298 K |
| Crystal System | Triclinic | Radiation type | Mo Kα |
| Space group | P-1 | Measured reflections | 5804 |
| a | 8.4725 (4)Å | Independent reflections | 6544 |
| b | 8.5332 (5)Å | Observed reflections | 1068 |
| c | 22.111 (2)Å | Rint | 0.066 |
| α | 97.401 (3)° | R(all) | 0.329 |
| β | 92.591 (3)° | R(gt) | 0.054 |
| γ | 113.776 (7)° | wR(ref) | 0.094 |
| V | 1442.4 (2)Å3 | wR(all) | 0.270 |
| λ | 0.71073 | Parameters | 433 |
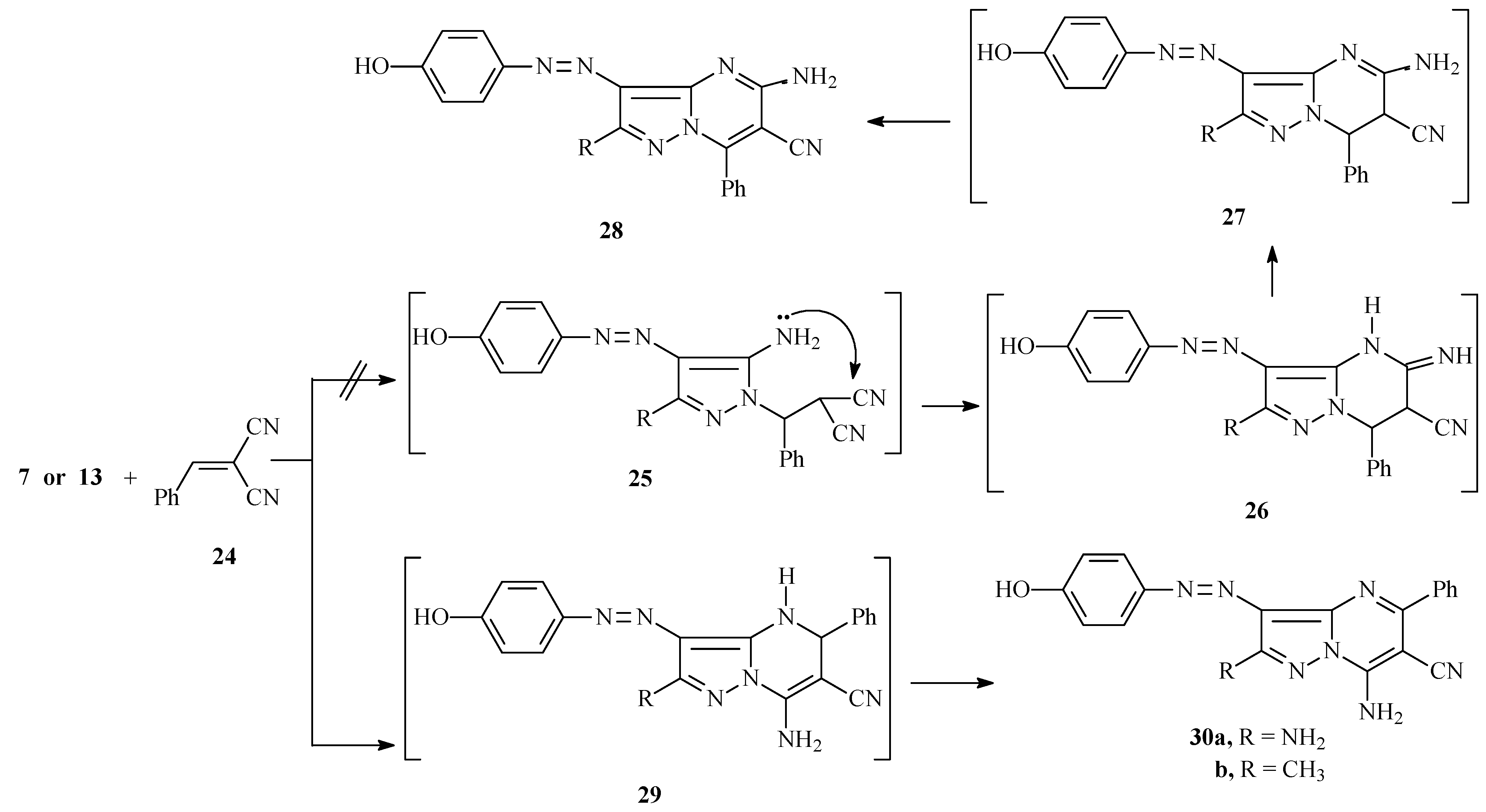
2.2. Dyeing and Fastness Properties
| DyeNo | Molecular weight | Color shade on polyester | Color strength (K/S)% Dye o.m.f. | |||||
|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | |||
| 7 | 234 | Yellowish brown | 0.44 | 0.84 | 1.00 | 1.16 | 1.49 | 1.76 |
| 22a | 330 | Yellow | 7.72 | 9.78 | 10.03 | 13.03 | 13.33 | 16.88 |
| 22b | 344 | Yellow | 14.59 | 15.72 | 15.81 | 17.76 | 21.18 | 24.13 |
| 22c | 364 | Yellow | 10.72 | 14.92 | 16.23 | 17.26 | 21.96 | 24.42 |
| 22d | 320 | Yellowish brown | 15.44 | 21.08 | 23.02 | 25.30 | 26.25 | 28.07 |
| 13 | 217 | Pale brown | 2.18 | 4.35 | 4.88 | 6.43 | 12.86 | 13.10 |
| 22f | 329 | Yellowish orange | 22.12 | 23.55 | 24.04 | 24.41 | 25.20 | 25.67 |
| 22g | 343 | Orange | 20.82 | 20.93 | 21.92 | 22.18 | 23.43 | 23.65 |
| 22h | 363 | Orange | 14.17 | 16.52 | 17.82 | 19.94 | 20.12 | 23.42 |
| Dye | Dye o.m.f. % | Wash fastness a,b | Persipiration fastness | Light fastness | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Alkaline | Acedic | ||||||||||
| Alt | SC | SW | Alt | SC | SW | Alt | SC | SW | |||
| 7 | 1% | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | 5 | 4 |
| 22a | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22b | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 22c | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22d | 4–5 | 4–5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | |
| 13 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 6 | |
| 22f | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 22g | 5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 22h | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 6 | |
| 7 | 2% | 5 | 5 | 5 | 5 | 5 | 5 | 4–5 | 4 | 5 | 3–4 |
| 22a | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22b | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 22c | 5 | 5 | 5 | 5 | 5 | 5 | 4–5 | 4 | 5 | 3 | |
| 22d | 5 | 4 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 13 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5–6 | |
| 22f | 5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 2–3 | |
| 22g | 5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 22h | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 6 | |
| 7 | 3% | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 |
| 22a | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22b | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22c | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 22d | 5 | 4–5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 13 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | |
| 22f | 5 | 5 | 4–5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 2–3 | |
| 22g | 5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | |
| 22h | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 6 | |
| 7 | 4% | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 |
| 22a | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22b | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22c | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22d | 5 | 3–4 | 3–4 | 5 | 4–5 | 5 | 5 | 4–5 | 5 | 3 | |
| 13 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 4–5 | 5 | 5–6 | |
| 22f | 5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 2–3 | |
| 22g | 4–5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22h | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 6 | |
| 7 | 5% | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 |
| 22a | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22b | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22c | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | |
| 22d | 4–5 | 3–4 | 3–4 | 5 | 5 | 5 | 5 | 5 | 5 | 2 | |
| 13 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5–6 | |
| 22f | 5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 2–3 | |
| 22g | 5 | 5 | 4–5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | |
| 22h | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 6 | |
| 7 | 6% | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 |
| 22a | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | |
| 22b | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3–4 | |
| 22c | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | |
| 22d | 4–5 | 4 | 4 | 5 | 4–5 | 5 | 5 | 5 | 5 | 2 | |
| 13 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5–6 | |
| 22f | 5 | 5 | 4–5 | 4–5 | 4–5 | 4–5 | 5 | 4–5 | 5 | 2–3 | |
| 22g | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | |
| 22h | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 6 | |
2.2.1. Color Strength
2.2.2. Wash Fastness
2.2.3. Light Fastness
2.2.4. Perspiration Fastness
3. Experimental
3.1. General
3.2. General Procedure for the Synthesis of Azo Disperse Dyes
3.3. Synthesis of Compounds 14a–b, 20a,b and 22a–j
3.4. General Procedure for the Preparation of 30a–b
3.5. High Temperature Dyeing Method (HT)
3.5.1. Materials
3.5.2. Dyeing
3.6. Color Measurements and Analyses
3.6.1. Color Measurements of the Dyed Fabrics
3.6.2. Color Fastness Tests
3.6.2.1. Fastness to Washing
3.6.2.2. Fastness to Perspiration
3.6.2.3. Fastness to Light
4. Conclusions
Acknowledgments
- Sample Availability: Samples of compounds 7, 10, 13, 14a, 14b, 20a, b, 22a–j and 30a, 30b are available from the authors.
References and Notes
- Ertan, N. Synthesis of some hetarylazopyrazolone dyes and solvent effects on their absorption spectra. Dyes Pigments 1999, 44, 41–48. [Google Scholar] [CrossRef]
- Khalil, A.K.; Hassan, M.A.; Mohamed, M.M.; El-Sayed, A.M. Metal salt-catalyzed diazocoupling of 3-substituted-1H-pyrazol-2-in-5-ones in aqueous medium. Dyes Pigments 2005, 66, 241–245. [Google Scholar] [CrossRef]
- Genin, M.J.; Biles, C.; Keiser, B.J.; Poppe, S.M.; Swaney, S.M.; Tarpley, W.G. Novel 1,5- diphenylpyrazole nonnucleoside HIV-1 reverse transcriptase inhibitors with enhanced activity versus the delavirdine-resistant P236L mutant: Lead identification and sar of 3- and 4-substituted derivatives. J. Med. Chem. 2000, 43, 1034–1040. [Google Scholar] [CrossRef]
- Kandil, S.S.; Abdel-Hay, F.I.; Issa, R.M. Thermal studies of cobalt(II), nickel(II) and copper(II) complexes of 4-(sulfonylazido phenylazo)pyrazolones. J. Therm. Anal. Calor. 2001, 63, 173–180. [Google Scholar] [CrossRef]
- Tsai, P.C.; Wang, I.J. Synthesis and solvatochromic properties of some disazo dyes derived from pyrazolo[1,5-a]pyrimidine derivatives. Dyes Pigments 2005, 64, 259–264. [Google Scholar] [CrossRef]
- Rangnekar, D.W.; Puro, S.S. Synthesis and dyeing characteristics of new 2-methyl-3-arylazo-6-phenyl-7-amino- and 7-acetamidopyrazolo[1,5-a]pyrimidines. Ind. J. Fibre Textile Res. 1990, 15, 37–38. [Google Scholar]
- Rangnekar, D.W.; Puro, S.S. Synthesis and dyeing characteristics of 3,6-bis(arylazo)pyrazolo-[1,5-a]pyrimidines. Ind. J. Fibre Textile Res. 1990, 15, 23–25. [Google Scholar]
- Krystof, V.; Cankar, P.; Frysova, I.; Slouka, J.; Kontopidis, G.; Dzubak, P.; Hajduch, M.; Srovnal, J.; de Azevedo, W.F.J.; Orsag, M.; et al. 4-Arylazo-3,5-diamino-1H- pyrazole CDK inhibitors: SAR study, crystal structure in complex with CDK2, selectivity, and cellular effects. J. Med. Chem. 2006, 49, 6500–6509. [Google Scholar]
- Ho, Y.W. Synthesis of some new azo pyrazolo[1,5-a]pyrimidine-thieno[2,3-b]pyridine derivatives and their application as disperse dyes. Dyes Pigments 2005, 64, 223–230. [Google Scholar] [CrossRef]
- Tsai, P.C.; Wang, I.J. Synthesis and solvatochromic properties of 3,6-bis-hetarylazo dyes derived from pyrazolo[1,5-a]pyrimidine. Dyes Pigments 2008, 76, 575–581. [Google Scholar] [CrossRef]
- Karcı, F.; Demirçalı, A. Synthesis of disazo pyrazolo[1,5-a]pyrimidines. Dyes Pigments 2007, 74, 288–297. [Google Scholar] [CrossRef]
- Tsai, P.C.; Wang, I.J. A facile synthesis of some new pyrazolo[1,5-a]pyrimidine heterocyclic disazo dyes and an evaluation of their solvatochromic behaviour. Dyes Pigments 2007, 74, 578–584. [Google Scholar] [CrossRef]
- Sayed, A.Z.; Aboul-Fetouh, M.S.; Nassar, H.S. Synthesis, biological activity and dyeing performance of some novel azo disperse dyes incorporating pyrazolo[1,5-a]pyrimidines for dyeing of polyester fabrics. J. Mol. Struct. 2012, 1010, 146–151. [Google Scholar] [CrossRef]
- Al-Etaibi, A.M.; Al-Awadi, N.A.; El-Apasery, M.A.; Ibrahim, M.R. Synthesis of some novel pyrazolo[1,5-a]pyrimidine derivatives and their application as disperse dyes. Molecules 2011, 16, 5182–5193. [Google Scholar] [CrossRef]
- Al-Etaibi, A.M.; El-Apasery, M.A.; Mahmoud, H.M.; Al-Awadi, N.A. One-pot synthesis of disperse dyes under microwave irradiation: Dyebath reuse in dyeing of polyester fabrics. Molecules 2012, 17, b4266–b4280. [Google Scholar] [CrossRef]
- El-Dusouqui, O.M.E.; Abdelkhalik, M.M.; Al-Awadi, N.A.; Dib, H.H.; George, B.J.; Elnagdi, M.H. Chemistry of 2-arylhydrazonals: Utility of substituted 2-arylhydrazono-3-oxoalkanals as precursors for 3-oxoalkanonitriles, 3-aminoisoxazole and 1,2,3- and 1,2,4-triazoles. J. Chem. Res. 2006, 5, 295–302. [Google Scholar]
- CCDC 828902 contains the supplementary crystallographic data for this paper. These data can be obtained free of charge via www.ccdc.cam.ac.uk/conts/retrieving.html (or from the CCDC, 12 Union Road, Cambridge CB2 1EZ, UK; fax: +44 1223 336033; Email: deposit@ccdc.cam.ac.uk).
- Elfahham, H.A.; Elgemeie, G.E.H.; Ibraheim, Y.R.; Elnagdi, M.H. Studies on 3,5-diaminopyrazoles: New routes for the synthesis of new pyrazoloazines and pyrazoloazoles. Liebig. Ann. Chem. 1988, 8, 819–822. [Google Scholar]
- Andrews, B.A.K.; Blanchard, E.J.; Reinhardt, R.M. Fabric whiteness retention in durable press finishing with citric acid. Text. Chem. Color. 1993, 25, 52–54. [Google Scholar]
- El-Kholy, Y.M.; Abdel-Hafiz, S.A.; Ahmed, S.H. Synthesis and dyeing properties of novel disperse dyes. Part 2: Pyrazole disperse dye derivatives. J. Soc. Dyers Colour. 1998, 114, 45–48. [Google Scholar]
- Peters, R.H.; Sumner, H.H. The affinities of vat dyes in relation to their constitutions. J. Soc. Dyers Colour. 1955, 71, 130–138. [Google Scholar]
- Giles, C.H.; Rahman, S.M.K. Studies on high fastness to light in coloring matters in hydrophobic substrates. Disperse dyes in cellulose acetates and polyesters. Textile Res. J. 1961, 31, 1012–1019. [Google Scholar] [CrossRef]
- Shakra, S.; Ghattas, A.A.G. The effect of hydrogen bond on the light fastness of azo dyes. Kolorisztikai Ertesito 1979, 21, 287–294. [Google Scholar]
- Ashkar, S.M.; El-Apasery, M.A.; Touma, M.M.; Elnagdi, M.H. Synthesis of some novel biologically active disperse dyes derived from 4-methyl-2,6-dioxo-1-propyl-1,2,5,6-tetrahydropyridine-3-carbonitrile as coupling component and their colour assessment on polyester fabrics. Molecules 2012, 17, 8822–8831. [Google Scholar]
- Chipalkatti, H.R.; Desai, N.F.; Giles, C.H.; Macaulay, N. The influence of the substrate upon the light fading of azo dyes. J. Soc. Dyers Colour. 1954, 70, 487–501. [Google Scholar]
- Chrysler, L.P. Methods of Test for Color Fastness of Textiles and Leather, 7th ed; Bradford: London, UK, 1990; pp. 89–94. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Al-Etaibi, A.M.; El-Apasery, M.A.; Ibrahim, M.R.; Al-Awadi, N.A. A Facile Synthesis of New Monoazo Disperse Dyes Derived from 4-Hydroxyphenylazopyrazole-5-amines: Evaluation of Microwave Assisted Dyeing Behavior. Molecules 2012, 17, 13891-13909. https://doi.org/10.3390/molecules171213891
Al-Etaibi AM, El-Apasery MA, Ibrahim MR, Al-Awadi NA. A Facile Synthesis of New Monoazo Disperse Dyes Derived from 4-Hydroxyphenylazopyrazole-5-amines: Evaluation of Microwave Assisted Dyeing Behavior. Molecules. 2012; 17(12):13891-13909. https://doi.org/10.3390/molecules171213891
Chicago/Turabian StyleAl-Etaibi, Alya M., Morsy A. El-Apasery, Maher R. Ibrahim, and Nouria A. Al-Awadi. 2012. "A Facile Synthesis of New Monoazo Disperse Dyes Derived from 4-Hydroxyphenylazopyrazole-5-amines: Evaluation of Microwave Assisted Dyeing Behavior" Molecules 17, no. 12: 13891-13909. https://doi.org/10.3390/molecules171213891
APA StyleAl-Etaibi, A. M., El-Apasery, M. A., Ibrahim, M. R., & Al-Awadi, N. A. (2012). A Facile Synthesis of New Monoazo Disperse Dyes Derived from 4-Hydroxyphenylazopyrazole-5-amines: Evaluation of Microwave Assisted Dyeing Behavior. Molecules, 17(12), 13891-13909. https://doi.org/10.3390/molecules171213891