Evaluation of Deoxyribonucleic Acid Toxicity Induced by the Radiopharmaceutical 99mTechnetium-Methylenediphosphonic Acid and by Stannous Chloride in Wistar Rats
Abstract
:1. Introduction
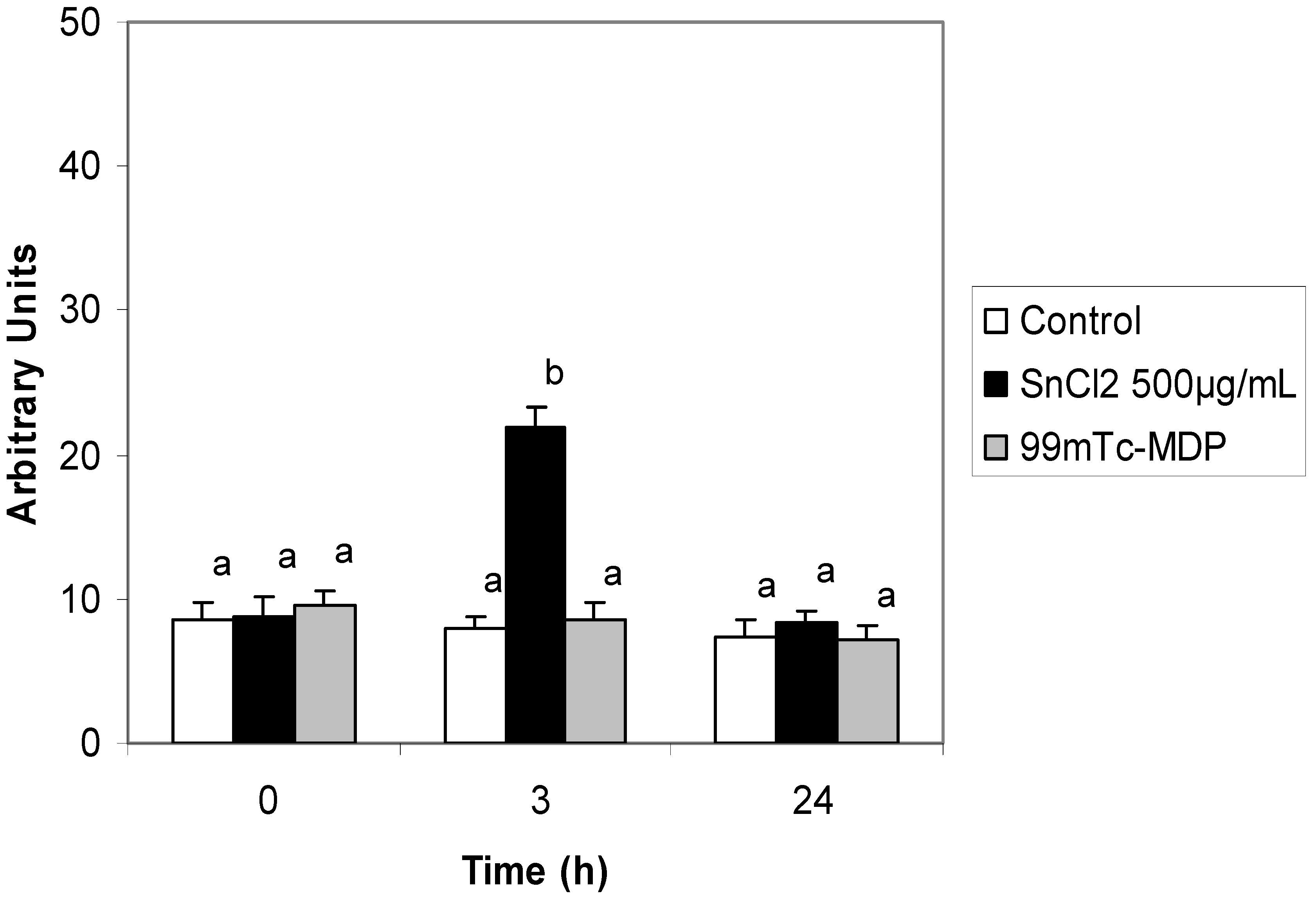
2. Results and Discussion
| Treatments | No. of Animals | Individual data a | N | % | Mean ± SD |
|---|---|---|---|---|---|
| NaCl 0.9% | 5 | 1.2, 2.0, 1.3, 1.2, 2.1 | 15 | 0.15 | 1.50 b ± 0.85 |
| Cyclophosphamide 50 mg/kg b.w. | 5 | 18.15, 22.13, 15.17, 20.12, 16.14 | 162 | 1.62 | 16.20 ± 3.12 |
| SnCl2 500 μg/mL | 5 | 2.1, 1.3, 2.3, 2.2, 3.3 | 22 | 0.22 | 2.20 b,c ± 0.79 |
| 99mTc-MDP 100 μCi | 5 | 1.2, 2.1, 2.3, 2.2, 2.1 | 18 | 0.18 | 1.80 b,c ± 0.63 |
3. Experimental
3.1. Reagents
3.2. Animals
3.3. Comet Assay
3.4. Micronucleus Test
3.5. Statistical Analysis
4. Conclusion
Acknowledgements
Conflict of Interest
References
- Narra, V.R.; Sastry, K.S.R.; Howell, R.W.; Goddu, S.M.; Strand, S.E.; Rao, D.V. Relative biological effectiveness of 99mTc radiopharmaceuticals. Med. Phys. 1994, 12, 21–26. [Google Scholar]
- Saha, G.B. Fundamentals of Nuclear Pharmacy; Springer Verlag: New York, NY, USA, 2010. [Google Scholar]
- Hladik, W.B.; Saha, G.B.; Study, K.T. Essentials of Nuclear Medicine Science; Williams and Wilkins: Baltimore, MD, USA, 1987. [Google Scholar]
- Silva, C.R.; Valsa, J.O.; Caniné, M.S.; Caldeira-de-Araujo, A.; Bernardo-Filho, M. Evaluation of technetium-99m decay on Escherichia coli inactivation: Effects of physical or chemical agents. Yale J. Biol. Med. 1998, 71, 7–14. [Google Scholar]
- Rader, J.I. Anti-nutritive effects of dietary tin. Adv. Exp. Med. Biol. 1991, 289, 509–524. [Google Scholar]
- Budavari, S. The Merck Index: An Encyclopedia of Chemicals, Drugs and Biologicals, 13rd ed; Merck & Co.: Rahway, NJ, USA, 2001. [Google Scholar]
- Boogaard, P.J.; Boisset, M.; Blunden, S.; Davies, S.; Ong, T.J.; Taverne, J.P. Comparative assessment of gastrointestinal irritant potency in man of tin (II) chloride and tin migrated from packaging. Food Chem. Toxicol. 2003, 41, 1663–1670. [Google Scholar] [CrossRef]
- White, D.J. “Return” to stannous fluoride dentifricies. J. Clin. Dent. 1995, 6, 29–36. [Google Scholar]
- Tavss, E.A.; Fisher, S.W.; Campbell, S.; Bonta, Y.; Darcy-Siegel, J.; Blackwell, B.L.; Volpe, A.R.; Miller, S.E. The scientific rationale and development of an optimized dentifrice for the treatment of dentin hypersensitivity. Am. J. Dent. 2004, 17, 61–70. [Google Scholar]
- McLean, J.R.N.; Blakey, D.H.; Douglas, G.R.; Kaplan, J.R. The effect of stannous and stannic (tin) chloride on DNA in Chinese hamster ovary cells. Mutat. Res. 1983, 119, 195–201. [Google Scholar] [CrossRef]
- El-Demerdash, F.M.; Yousef, M.I.; Zoheir, M.A. Stannous chloride induces alteratios in enzymes activities, lipid peroxidation and histopathology in male rabbit: antioxidant role of vitamin C. Food Chem. Toxicol. 2005, 43, 1743–1752. [Google Scholar] [CrossRef]
- Hallas, L.E.; Cooney, J.J. Tin and tin-resistant microorganisms in Chesapeake Bay. Appl. Environ. Microbiol. 1981, 41, 466–471. [Google Scholar]
- Raper, E.S. Complexes of heterocyclic thione donors. Coord. Chem. Rev. 1985, 61, 115–184. [Google Scholar] [CrossRef]
- Larsson, A.; Kinnby, B.; Kunsberg, R.; Peszkowski, M.J.; Warfuinge, G. Irritant and sensitizing potential of copper, Mercury and tin salts in experimental contact stomatitis of rat oral mucosa. Contact Dermat. 1990, 23, 146–153. [Google Scholar] [CrossRef]
- Ashby, J.; Tennant, R.W. Definitive relationships among chemical and mutagenicity for 301 chemicals tested by the U.S. NTP. Mutat. Res. 1991, 257, 229–306. [Google Scholar] [CrossRef]
- Hattori, T.; Maehashi, H. Augmentation of calcium influx by stannous chloride at mouse motor nerve terminals. Res. Commun. Mol. Pathol. Pharmacol. 1994, 84, 253–256. [Google Scholar]
- Hattori, T.; Maehashi, H.; Miyazawa, T.; Naito, M. Potentiation by stannous chloride of calcium entry into osteoblastic MC3T3-E1 cells through voltage-dependent L-type calcium channels. Cell Calcium 2001, 30, 67–72. [Google Scholar] [CrossRef]
- Yousef, M.I. Protective role of ascorbic acid to enhance reprodutive performance of male rabbits treated with stannous chloride. Toxicology 2005, 207, 81–89. [Google Scholar] [CrossRef]
- Nishioka, H. Mutagenic activities of metal compounds in bacteria. Mutat. Res. 1975, 31, 185–189. [Google Scholar] [CrossRef]
- Kada, T.; Hirano, K.; Shirasu, Y. Chemical Mutagens: Principles and Methods for Their Detection; Holander, A., Serres, F.J., Eds.; Plenum Press: New York, NY, USA, 1980; pp. 149–173. [Google Scholar]
- Singh, I. Induction of reverse mutation and mitotic gene conversion by some metal compounds in Saccharomyces cerevisae. Mutat. Res. 1983, 117, 149–152. [Google Scholar] [CrossRef]
- Olivier, P.; Marzin, D. Study of genotoxic potential of 48 inorganic derivates with the SOS chromotest. Mutat. Res. 1987, 189, 263–269. [Google Scholar] [CrossRef]
- Cabral, R.E.C.; Leitão, A.; Lage, C.; Caldeira-de-Araujo, A.; Bernardo-Filho, M.; Dantas, F.J.S.; Cabral-Neto, J. Mutational potentiality of stannous chloride: An important reducing agent in the 99mTc-radiopharmaceuticals. Mutat. Res. 1998, 408, 129–135. [Google Scholar] [CrossRef]
- Pungartnik, C.; Viau, C.; Picada, J.; Caldeira-de-Araujo, A.; Henriques, J.A.P.; Brendel, M. Genotoxicity of stannous chloride in yeast and bacteria. Mutat. Res. 2005, 583, 146–157. [Google Scholar] [CrossRef]
- Dantas, F.J.S.; Moraes, M.O.; De Mattos, J.C.P.; Bezerra, R.J.A.C.; Carvalho, E.F.; Bernardo-Filho, M.; Caldeirade-Araujo, A. Stannous chloride mediates single strand breaks in plasmid DNA through reactive oxygen species formation. Toxicol. Lett. 1999, 110, 129–136. [Google Scholar] [CrossRef]
- Dantas, F.J.S.; Moraes, M.O.; Carvalho, E.F.; Valsa, J.O.; Bernardo-Filho, M.; Caldeira-de-Araujo, A. Lethality induced by stannous chloride on Escherichia coli AB1157: Participation of reactive oxygen species. Food Chem. Toxicol. 1996, 34, 959–962. [Google Scholar] [CrossRef]
- De Mattos, J.C.P.; Dantas, F.J.S.; Bezerra, R.J.A.C.; Bernardo-Filho, M.; Cabral-Neto, J.B.; Lage, C.; Leitão, A.C.; Caldeira-de-Araujo, A. Damage induced by stannous chloride in plasmid DNA. Toxicol. Lett. 2000, 116, 159–163. [Google Scholar] [CrossRef]
- De Mattos, J.C.P.; Lage, C.; Dantas, F.J.S.; Moraes, M.O.; Nunes, A.P.M.; Bezerra, R.J.A.C.; Faria, M.V.C.; Leitão, A.C.; Caldeira-de-araujo, A. Interaction of stannous chloride leads to alteration in DNA, triphosphate nucleotides and isolated bases. Mol. Cell. Biochem. 2005, 280, 173–179. [Google Scholar] [CrossRef]
- Assis, M.L.B.; Neto, J.B.C.; Souza, J.E.Q.; Caldeira-de-Araujo, A.; Bernardo-Filho, M. Stannous chloride and the glucoheptonic acid effect: study of a kit used in nuclear medicine. Cancer Lett. 1998, 130, 127–131. [Google Scholar] [CrossRef]
- Sethi, R.S.; Mittal, B.; Bhattacharya, A.; Singh, B. Tc99m MDP bone SPECT in a case of osteomyelitis of the skull. Indian J. Nucl. Med. 2010, 25, 25–26. [Google Scholar] [CrossRef]
- Guedes, A.P.; Cardoso, V.N.; De Mattos, J.C.P.; Dantas, F.J.S.; Matos, V.C.; Silva, J.C.F.; Bezerra, R.J.A.C.; Caldeira-de-Araujo, A. Cytotoxic and genotoxic effects induced by stannous chloride associated to nuclear medicine kits. Nucl. Med. Biol. 2006, 33, 915–921. [Google Scholar] [CrossRef]
- Balamurugan, K.; Schaffner, W. Copper homeostasis in eukaryotes: Teetering on a tightrope. Biochim. Biophys. Acta 2006, 1763, 737–746. [Google Scholar]
- Gunshin, H.; Mackenzie, B.; Berger, U.V.; Gunshin, Y.; Romero, M.F.; Boron, W.F.; Nussberger, S.; Gollan, J.L.; Hediger, M.A. Cloning and characterization of a mammalian proton-coupled metal-ion transporter. Nature 1997, 388, 482–488. [Google Scholar] [CrossRef]
- Knopfel, M.; Zhao, L.; Garrick, M.D. Transport of divalent transition-metal ions is lost in small-intestinal tissue of b/b Belgrade rats. Biochemistry 2005, 44, 3454–3465. [Google Scholar] [CrossRef]
- Canonne-Hergaux, F.; Zhang, A.S.; Ponka, P.; Gros, P. Characterization of the iron transporter DMT1 (NRAMP2/DCT1) in red blood cells of normal and anemic mk/mk mice. Blood 2001, 13, 3823–3830. [Google Scholar]
- Kakhlon, O.; Cabantchik, Z.I. The labile iron pool: Characterization, Measurement, and Participation in cellular processes. Free Radic. Biol. Med. 2002, 33, 1037–1046. [Google Scholar] [CrossRef]
- Speit, G.; Hartmann, A. The comet assay (single cell gel test) - a sensitive genotoxicity test for the detection of DNA damage and repair. In Methods in Molecular Biology,113 DNA-Repair Protocols:Eukaryotic Systems; Henderson, D.S., Ed.; Human Press Inc.: Totowa, NY, USA, 1999; pp. 203–212. [Google Scholar]
- Dantas, F.J.S.; De Mattos, J.C.P.; Moraes, M.O.; Viana, M.E.; Lage, C.; Cabral-Neto, J.B.; Caldeira-deAraujo, A. Genotoxic effects of stannous chloride (SnCl2) in K562 cell line. Food Chem. Toxicol. 2002, 40, 1493–1498. [Google Scholar] [CrossRef]
- Nunes, A.P.M.; Ferreira-Machado, S.C.; Nunes, R.M.; Dantas, F.J.S.; De Mattos, J.C.P.; Caldeira-de-Araujo, A. Analysis of genotoxic potentiality of stevioside by comet assay. Food Chem. Toxicol. 2007, 45, 662–666. [Google Scholar] [CrossRef]
- Miyakoshi, Y.; Suzuki, Y.; Ooida, M.; Takahashi, A.; Tsukui, M. Micronucleus astrocytes test using cultured new born rat. Ind. Health 1999, 37, 95–102. [Google Scholar] [CrossRef]
- Ribeiro, L.R.; Salvadori, D.M.F.; Marques, E.K. Mutagênese Ambiental; Ed Ulbra: Canoas, RS, Brazil, 2003. [Google Scholar]
- Aguila, M.B.; Pinheiro, A.R.; Mandarim-de-Lacerda, C.A. Spontaneously hypertensive rats left ventricular cardiomyocyte loss attenuation through different edible oils long-term intake. Int. J. Cardiol. 2005, 100, 461–466. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Mattos, J.C.P.; Matos, V.C.d.; Rodrigues, M.P.; Oliveira, M.B.N.d.; Dantas, F.J.S.; Santos-Filho, S.D.; Bernardo-Filho, M.; Caldeira-de-Araujo, A. Evaluation of Deoxyribonucleic Acid Toxicity Induced by the Radiopharmaceutical 99mTechnetium-Methylenediphosphonic Acid and by Stannous Chloride in Wistar Rats. Molecules 2012, 17, 12974-12983. https://doi.org/10.3390/molecules171112974
De Mattos JCP, Matos VCd, Rodrigues MP, Oliveira MBNd, Dantas FJS, Santos-Filho SD, Bernardo-Filho M, Caldeira-de-Araujo A. Evaluation of Deoxyribonucleic Acid Toxicity Induced by the Radiopharmaceutical 99mTechnetium-Methylenediphosphonic Acid and by Stannous Chloride in Wistar Rats. Molecules. 2012; 17(11):12974-12983. https://doi.org/10.3390/molecules171112974
Chicago/Turabian StyleDe Mattos, José Carlos Pelielo, Vanessa Coutinho de Matos, Michelle Pinheiro Rodrigues, Marcia Betânia Nunes de Oliveira, Flavio José S. Dantas, Sebastião David Santos-Filho, Mario Bernardo-Filho, and Adriano Caldeira-de-Araujo. 2012. "Evaluation of Deoxyribonucleic Acid Toxicity Induced by the Radiopharmaceutical 99mTechnetium-Methylenediphosphonic Acid and by Stannous Chloride in Wistar Rats" Molecules 17, no. 11: 12974-12983. https://doi.org/10.3390/molecules171112974
APA StyleDe Mattos, J. C. P., Matos, V. C. d., Rodrigues, M. P., Oliveira, M. B. N. d., Dantas, F. J. S., Santos-Filho, S. D., Bernardo-Filho, M., & Caldeira-de-Araujo, A. (2012). Evaluation of Deoxyribonucleic Acid Toxicity Induced by the Radiopharmaceutical 99mTechnetium-Methylenediphosphonic Acid and by Stannous Chloride in Wistar Rats. Molecules, 17(11), 12974-12983. https://doi.org/10.3390/molecules171112974






