Synthesis and Cytotoxicity Evaluation of 13-n-Alkyl Berberine and Palmatine Analogues as Anticancer Agents
Abstract
:1. Introduction
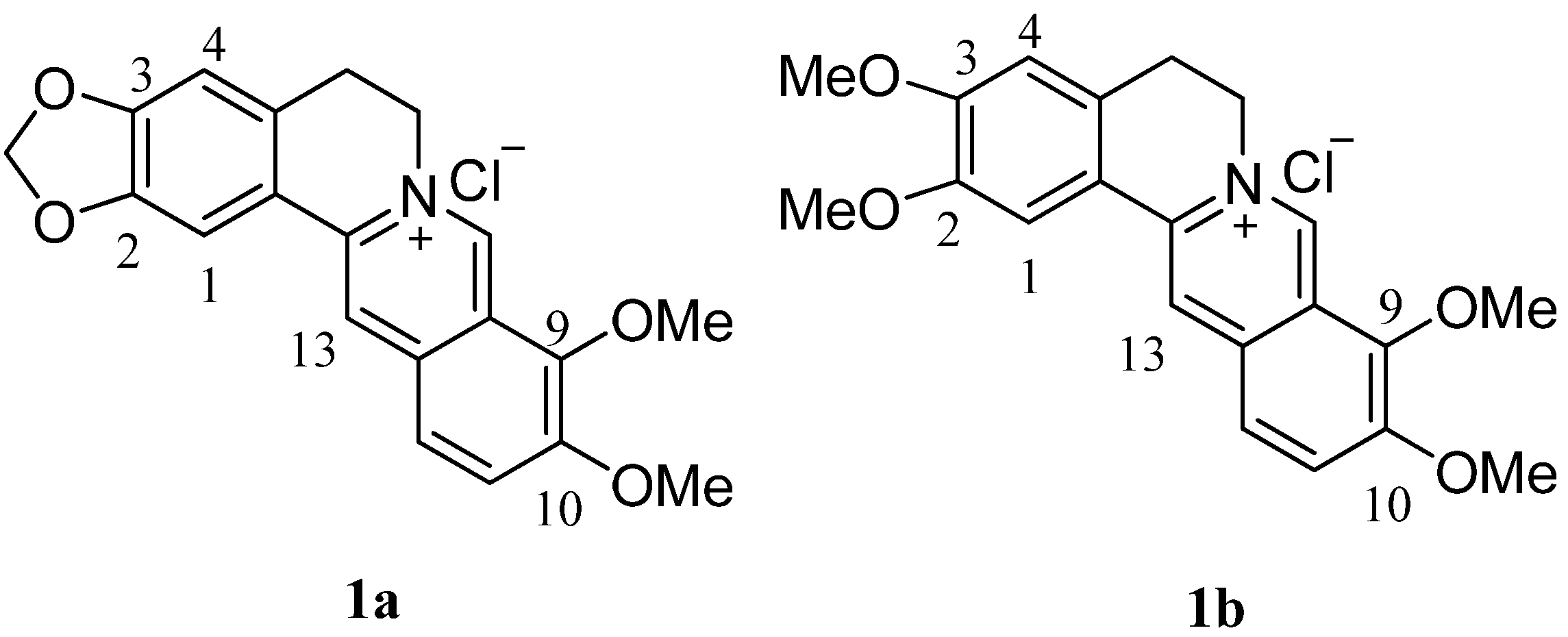
2. Results and Discussion
2.1. Chemistry
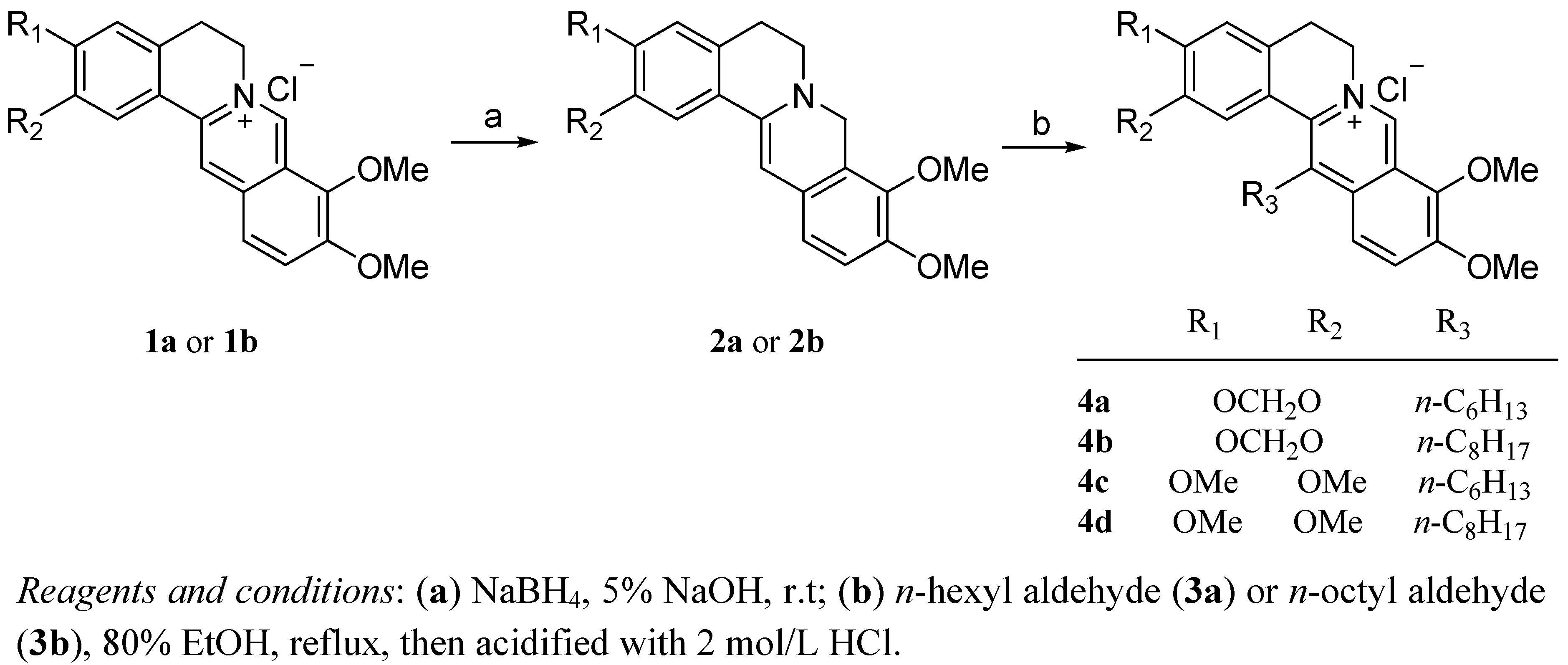
2.2. Biology
2.2.1. In Vitro Cytotoxic Effects
| Comp. | IC50 (μM) a | ||||||
|---|---|---|---|---|---|---|---|
| 7701QGY | SMMC7721 | HepG2 | CEM | CEM/VCR | K III | Lewis | |
| 4a | 3.28 ± 0.27 | 0.37 ± 0.07 | 4.74 ± 0.48 | 3.64 ± 0.35 | 9.65 ± 2.37 | 25.47 ± 2.95 | 2.26 ± 0.49 |
| 4b | 1.79 ± 0.26 | 0.04 ± 0.02 | 3.16 ± 0.39 | 0.37 ± 0.03 | 5.19 ± 0.64 | 13.58 ± 2.84 | 0.86 ± 0.10 |
| 4c | 10.09 ± 1.98 | 0.68 ± 0.05 | 5.54 ± 0.24 | 1.97 ± 0.09 | 17.54 ± 1.98 | 30.58 ± 1.69 | 2.86 ± 0.54 |
| 4d | 1.08 ± 0.25 | 0.02 ± 0.01 | 2.28 ± 0.37 | 0.16 ± 0.11 | 4.80 ± 0.81 | 10.41 ± 2.58 | 0.34 ± 0.09 |
| berberine | 22.18 ± 1.12 | 2.09 ± 0.25 | 117.63 ± 3.13 | 45.04 ± 1.42 | 120.37 ± 3.84 | 84.29 ± 3.42 | 20.29 ± 4.42 |
| palmatine | ND | 23.19 ± 1.21 | ND | 5.68 ± 0.27 | 230.76 ± 5.21 | 74.13 ± 4.50 | 30.18 ± 2.76 |
2.2.2. In Vivo Anticancer Activity
| Comp. | Dose mg/kg | Injection | Number of mice | Weight of mice (g) | Weight of tumor X±SD (g) | Tumor inhibitory rate (%) | ||
|---|---|---|---|---|---|---|---|---|
| Start | End | Start | End | |||||
| Control | - | iv | 10 | 10 | 19.48 ± 1.45 | 22.19 ± 2.20 | 2.20 ± 0.93 | - |
| CP | 30 | iv | 10 | 10 | 20.23 ± 1.25 | 20.81 ± 2.31 | 0.43 ± 0.28 ** | 80.61 |
| berberine | 30 | ip | 10 | 9 | 20.20 ± 1.26 | 17.66 ± 3.24 | 1.26 ± 0.54 ** | 42.99 |
| 4a | 1 | ip | 10 | 9 | 19.98 ± 1.33 | 17.86 ± 2.25 | 1.32 ± 0.63 ** | 40.00 |
| 2.5 | ip | 10 | 6 | 19.62 ± 1.52 | 18.04 ± 2.54 * | 1.01 ± 0.48 ** | 54.09 | |
| 4b | 1 | ip | 10 | 8 | 20.20 ± 2.22 | 19.17 ± 1.12 | 1.02 ± 0.25 ** | 53.52 |
| 2.5 | ip | 10 | 5 | 19.89 ± 1.54 | 17.11 ± 1.06 | 0.88 ± 0.28 ** | 59.86 | |
| palmatine | 30 | ip | 10 | 10 | 20.56 ± 2.03 | 20.95 ± 2.18 | 1.45 ± 0.31 ** | 34.09 |
| 4c | 5 | ip | 10 | 10 | 20.33 ± 1.28 | 20.45 ± 1.45 | 1.43 ± 0.44 ** | 34.88 |
| 10 | ip | 10 | 9 | 19.16 ± 1.21 | 18.88 ± 2.60 * | 1.16 ± 0.44 ** | 47.40 | |
| 4d | 5 | ip | 10 | 9 | 20.72 ± 1.45 | 19.15 ± 2.75 | 1.28 ± 0.38 ** | 42.05 |
| 10 | ip | 10 | 5 | 20.04 ± 1.57 | 18.63 ± 3.20 | 1.08 ± 0.42 ** | 50.96 | |
3. Experimental
3.1. General
3.2. Chemistry: General Procedure for the Synthesis of Compounds 4a–d
3.2. Pharmacology
3.2.1. MTT Assay
3.2.2. Anticancer Activity in Vivo
3.2.3. Statistical Analysis
4. Conclusions
Acknowledgments
References
- Rabbani, G.H.; Butler, T.; Knight, J.; Sanyal, S.C.; Alam, K. Randomized controlled trial of berberine sulfate therapy for diarrhea due to enterotoxigenic E. coli and Vibrio cholerae. J. Infect. Dis. 1987, 155, 979–984. [Google Scholar] [CrossRef]
- Zhou, H.Y.; Mineshita, S. The Effect of Berberine Chloride on Experimental Colitis in Rats In Vivo and In Vitro. J. Pharmacol. Exp. Ther. 2000, 294, 822–829. [Google Scholar]
- Iwasa, K.; Kamigauchi, M.; Ueki, M.; Taniguchi, M. Antibacterial activity and tructure-activity relationships of berberine analogs. Eur. J. Med. Chem. 1996, 31, 469–478. [Google Scholar] [CrossRef]
- Samosorn, S.; Tanwirat, B.; Muhamad, N.; Casadei, G.; Tomkiewicz, D.; Lewis, K.; Suksamrarn, A.; Prammananan, T.; Gornall, K.C.; Beck, J.L.; et al. Antibacterial activity of berberine-NorA pump inhibitor hybrids with a methylene ether linking group. Bioorg. Med. Chem. 2009, 17, 3866–3872. [Google Scholar] [CrossRef]
- Park, K.D.; Lee, J.H.; Kim, S.H.; Kang, T.H.; Moon, J.S.; Kim, S.U. Synthesis of 13-(substituted benzyl) berberine and berberrubine derivatives as antifungal agents. Bioorg. Med. Chem. Lett. 2006, 16, 3913–3916. [Google Scholar] [CrossRef]
- Iwasa, K.; Kim, H.S.; Wataya, Y.; Lee, D.U. Antimalarial activity and structure-activity relationships of protoberberine alkaloids. Eur. J. Med. Chem. 1998, 13, 65–69. [Google Scholar]
- Vennerstrom, J.L.; Lovelace, J.K.; Waits, V.B.; Hanson, W.L.; Klayman, D.L. Berberine derivatives as antileishmanial drugs. Antimicrob. Agents Chemother. 1990, 34, 918–921. [Google Scholar] [CrossRef]
- Letasiová, S.; Jantová, S.; Cipák, L.; Múcková, M. Berberine-antiproliferative activity in vitro and induction of apoptosis/necrosis of the U937 and B16 cells. Cancer Lett. 2006, 239, 254–262. [Google Scholar] [CrossRef]
- Sun, Y.Y.; Xun, K.L.; Wang, Y.T.; Chen, X.P. A systematic review of the anticancer properties of berberine, A natural product from Chinese herbs. Anticancer Drugs 2009, 20, 757–769. [Google Scholar] [CrossRef]
- Tang, J.; Feng, Y.B.; Tsao, S.; Wang, N.; Curtain, R.; Wang, Y.W. Berberine and Coptidis Rhizoma as novel antineoplastic agents: A review of traditional use and biomedical investigations. J. Ethnopharmacol. 2009, 126, 5–7. [Google Scholar] [CrossRef]
- Li, X.K.; Motwani, M.; Tong, W.; Bornmann, W.; Schwartz, G.K. Huanglian, A Chinese Herbal Extract, Inhibits Cell Growth by Suppressing the Expression of Cyclin B1 and Inhibiting CDC2 Kinase Activity in Human Cancer Cell. Mol. Pharmacol. 2000, 58, 1287–1293. [Google Scholar]
- Kim, S.; Choi, J.H.; Kim, J.B.; Nam, S.J.; Yang, J.H.; Kim, J.H.; Lee, J.E. Berberine Suppresses TNF-α-induced MMP-9 and Cell Invasion through Inhibition of AP-1 Activity in MDA-MB-231 Human Breast Cancer Cells. Molecules 2008, 13, 2975–2985. [Google Scholar] [CrossRef]
- Jiang, H.L.; Wang, X.; Huang, L.; Luo, Z.H.; Su, T.; Ding, K.; Li, X.S. Benzenediol-berberine hybrids: Multifunctional agents for Alzheimer’s disease. Bioorg. Med. Chem. 2011, 19, 7228–7235. [Google Scholar] [CrossRef]
- Ji, H.F.; Shen, L. Berberine: A Potential Multipotent Natural Product to Combat Alzheimer’s Disease. Molecules 2011, 16, 6732–6740. [Google Scholar] [CrossRef]
- Bodiwala, H.S.; Sabde, S.; Mitra, D.; Bhutani, K.K. Synthesis of 9-substituted derivatives of berberine as anti-HIV agents. Eur. J. Med. Chem. 2011, 46, 1045–1049. [Google Scholar] [CrossRef]
- Hayashi, K.; Minoda, K.; Nagaoka, Y.; Hayashi, T.; Uesatob, S. Antiviral activity of berberine and related compounds against human cytomegalovirus. Bioorg. Med. Chem. Lett. 2007, 17, 1562–1564. [Google Scholar] [CrossRef]
- Yang, P.; Song, D.Q.; Li, Y.H.; Kong, W.J.; Wang, Y.X.; Gao, L.M.; Liu, S.Y.; Cao, R.Q.; Jiang, J.D. Synthesis and structure-activity relationships of berberine analogues as a novel class of low-density-lipoprotein receptor up-regulators. Bioorg. Med. Chem. Lett. 2008, 18, 4675–4677. [Google Scholar]
- Tang, L.Q.; Wei, W.; Chen, L.M.; Liu, S. Effects of berberine on diabetes induced by alloxan and a high-fat/high-cholesterol diet in rats. J. Ethnopharmacol. 2006, 108, 109–115. [Google Scholar] [CrossRef]
- Chen, W.H.; Pang, J.Y.; Qin, Y.; Peng, Q.; Cai, Z.; Jiang, Z.H. Synthesis of linked berberine dimers and their remarkably enhanced DNA-binding affinities. Bioorg. Med. Chem. Lett. 2005, 15, 2689–2692. [Google Scholar] [CrossRef]
- Franceschin, M.; Rossetti, L.; D’Ambrosio, A.; Schirripa, S.; Bianco, A.; Ortaggi, G.; Savino, M.; Schultes, C.; Neidle, S. Natural and synthetic G-quadruplex interactive berberine derivatives. Bioorg. Med. Chem. Lett. 2006, 16, 1707–1711. [Google Scholar]
- Zhang, W.J.; Ou, T.M.; Lu, Y.J.; Huang, Y.Y.; Wu, W.B.; Huang, Z.S.; Zhou, J.L.; Wong, K.Y.; Gu, L.Q. 9-Substituted berberine derivatives as G-quadruplex stabilizing ligands in telomeric DNA. Bioorg. Med. Chem. 2007, 15, 5493–5501. [Google Scholar]
- Bbadra, K.; Kumar, G.S. Therapeutic Potential of Nucleic Acid-Binding Isoquinoline Alkaloids: Binding Aspects and Implications for Drug Design. Med. Res. Rev. 2011, 31, 821–862. [Google Scholar] [CrossRef]
- Islam, M.M.; Basu, A.; Hossain, M.; Sureshkumar, G.; Hotha, S.; Kumar, G.S. Enhanced DNA Binding of 9-ω-Amino Alkyl Ether Analogs from the Plant Alkaloid Berberine. DNA Cell Biol. 2011, 30, 123–133. [Google Scholar] [CrossRef]
- Islam, M.M.; Basu, A.; Kumar, G.S. Binding of 9-O-(ω-amino) alkyl ether analogues of the plant alkaloid berberine to poly(A): Insights into self-structure induction. Med. Chem. Commun. 2011, 2, 631–637. [Google Scholar] [CrossRef]
- Basu, A.; Jaisankar, P.; Kumar, G.S. Synthesis of novel 9-O-N-aryl/aryl-alkyl amino carbonyl methyl substituted berberine analogs and evaluation of DNA binding aspects. Bioorg. Med. Chem. 2012, 20, 2498–2505. [Google Scholar] [CrossRef]
- Bhowmik, D.; Hossain, M.; Buzzetti, F.; D’Auria, R.; Lombardi, P.; Kumar, G.S. Biophysical Studies on the Effect of the 13 Position Substitution of the Anticancer Alkaloid Berberine on Its DNA Binding. J. Phys. Chem. 2012, 116, 2314–2324. [Google Scholar]
- Chang, Y.L.; Usami, S.; Hsieh, M.T.; Jiang, M.J. Effects of palmatine on isometric force and intracellular calcium levels of arterial smooth muscle. Life Sci. 1999, 64, 597–606. [Google Scholar] [CrossRef]
- Vennerstrom, J.L.; Klayman, D.L. Protoberberine Alkaloids as Antimalarials. J. Med. Chem. 1988, 31, 1084–1087. [Google Scholar] [CrossRef]
- Kuo, C.L.; Chou, C.C.; Yung, B.Y. Berberine complexes with DNA in the berberine-induced apoptosis in human leukemic HL-60 cells. Cancer Lett. 1995, 93, 193–200. [Google Scholar] [CrossRef]
- Iwasa, K.; Kamigauchi, M.; Sugiura, M.; Nanba, H. Antimicrobial Activity of Some 13-Alkyl Substituted Protoberberinium Salts. Planta Med. 1997, 63, 196–198. [Google Scholar] [CrossRef]
- Wei, H.L.; Wang, S.Q.; Xu, F.; Xu, L.F.; Zheng, J.R.; Chen, Y. Evaluation of a 13-hexyl-berberine hydrochloride topical gel formulation. Drug Dev. Ind. Pharm. 2012, 38, 1–6. [Google Scholar] [CrossRef]
- Skehan, P.; Storeng, R.; Scudiero, D.; Monks, A.; McMahon, J.; Vistica, D.; Warren, J.T.; Bokesch, H.; Kenney, S.; Boyd, M.R. New Colorimetric Cytotoxicity Assay for Anticancer-Drug Screening. J. Natl. Cancer. Inst. 1990, 82, 1107–1112. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Zhang, L.; Li, J.; Ma, F.; Yao, S.; Li, N.; Wang, J.; Wang, Y.; Wang, X.; Yao, Q. Synthesis and Cytotoxicity Evaluation of 13-n-Alkyl Berberine and Palmatine Analogues as Anticancer Agents. Molecules 2012, 17, 11294-11302. https://doi.org/10.3390/molecules171011294
Zhang L, Li J, Ma F, Yao S, Li N, Wang J, Wang Y, Wang X, Yao Q. Synthesis and Cytotoxicity Evaluation of 13-n-Alkyl Berberine and Palmatine Analogues as Anticancer Agents. Molecules. 2012; 17(10):11294-11302. https://doi.org/10.3390/molecules171011294
Chicago/Turabian StyleZhang, Lei, Jingjing Li, Fei Ma, Shining Yao, Naisan Li, Jing Wang, Yongbin Wang, Xiuzhen Wang, and Qizheng Yao. 2012. "Synthesis and Cytotoxicity Evaluation of 13-n-Alkyl Berberine and Palmatine Analogues as Anticancer Agents" Molecules 17, no. 10: 11294-11302. https://doi.org/10.3390/molecules171011294
APA StyleZhang, L., Li, J., Ma, F., Yao, S., Li, N., Wang, J., Wang, Y., Wang, X., & Yao, Q. (2012). Synthesis and Cytotoxicity Evaluation of 13-n-Alkyl Berberine and Palmatine Analogues as Anticancer Agents. Molecules, 17(10), 11294-11302. https://doi.org/10.3390/molecules171011294




