Formulation and Evaluation of Celastrol-Loaded Liposomes
Abstract
:1. Introduction
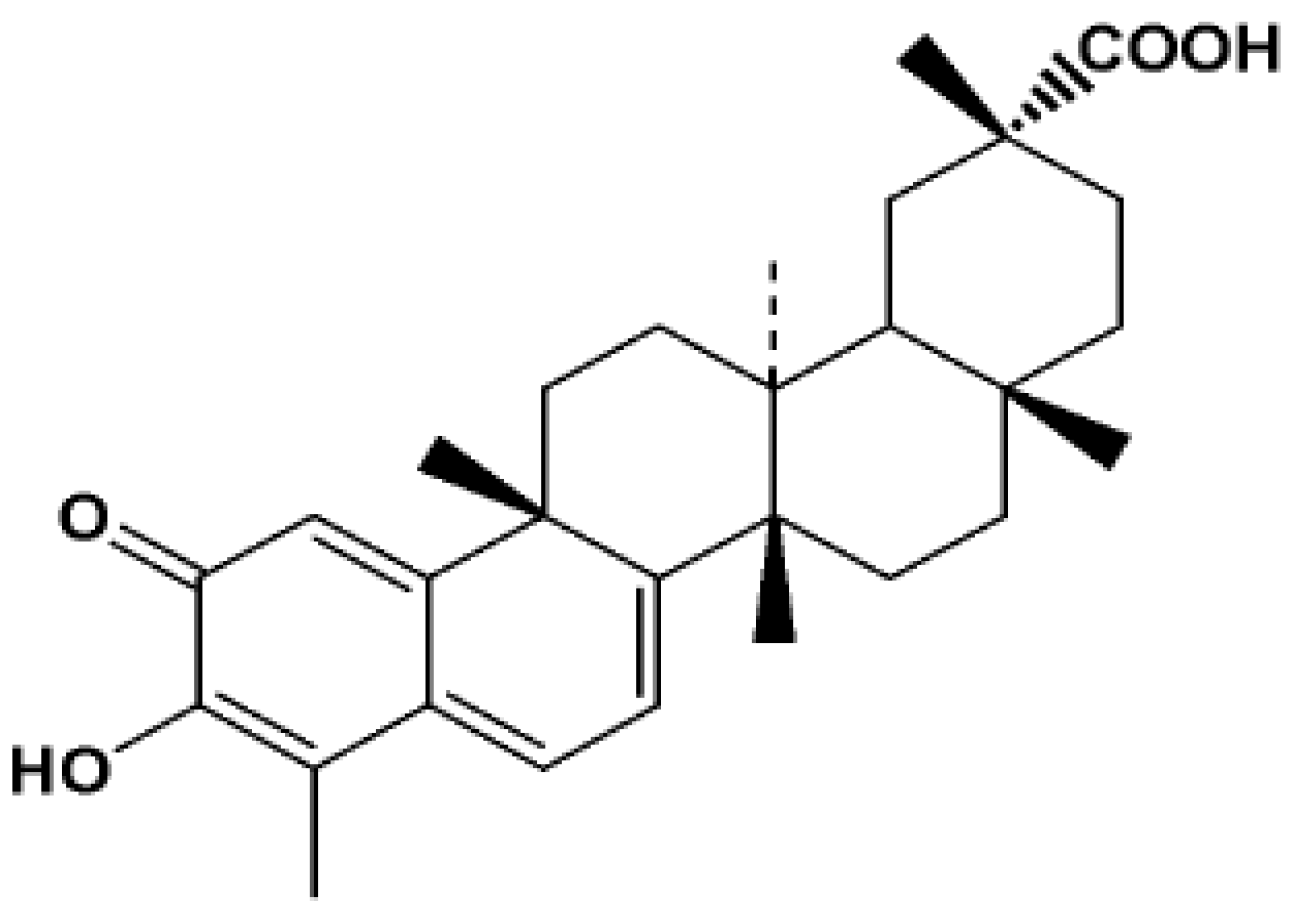
2. Results and Discussion
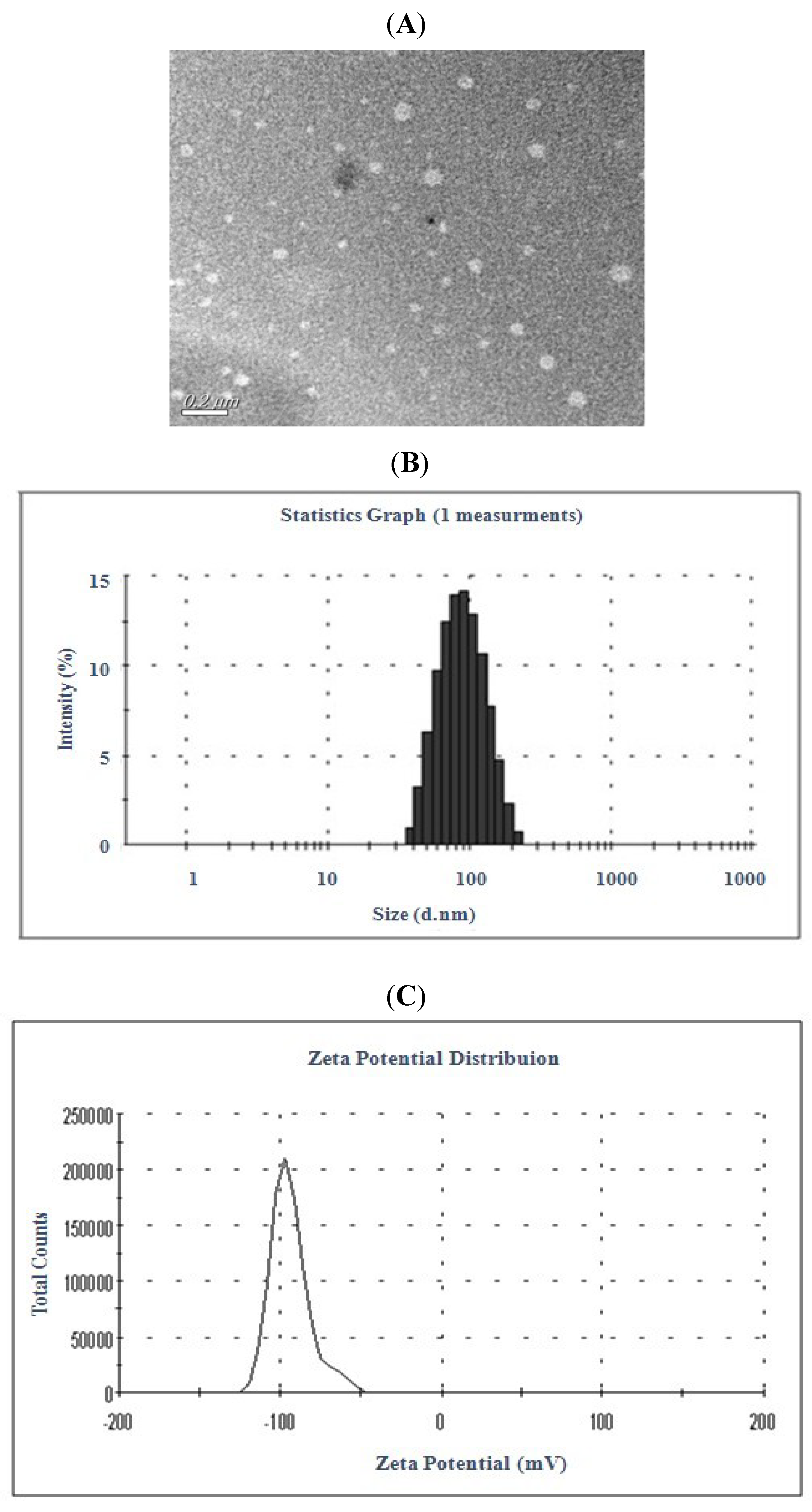
2.1. Preparation and Characteristic of Liposomal Celastrol
 ± s, n = 3).
± s, n = 3).
| Surfactants | Tween-80 | P188 | PEG400 | SD |
|---|---|---|---|---|
| EE% | 94.67 ± 3.58 | 92.36 ± 3.84 | 76.35 ± 2.03 | 81.23 ± 2.47 |
| Injection velocity (mL/min) | 0.1 | 0.5 | 1.0 | 2.0 |
|---|---|---|---|---|
| EE% | 90.81 ± 2.80 | 94.67 ± 3.58 | 94.26 ± 3.78 | 91.31 ± 2.96 |
| Average Size (nm) | 71.31 ± 0.43 | 63.62 ± 0.44 | 61.53 ± 0.46 | 97.41 ± 0.71 |
| Batch | Celastrol | Cholesterol | Tween-80 | Aqueous Phase Volume | EE |
|---|---|---|---|---|---|
| Weight | Weight | Concentration | |||
| (mg) | (mg) | (mg/mL) a | (mL) | (% w/w) b | |
| 1 | 10 | 40 | 0 | 20 | 79.42 ± 2.45 |
| 2 | 10 | 80 | 0.5 | 30 | 98.98 ± 2.16 |
| 3 | 10 | 120 | 1.0 | 40 | 89.77 ± 2.43 |
| 4 | 10 | 160 | 2.0 | 50 | 80.62 ± 2.37 |
| 5 | 20 | 40 | 0.5 | 40 | 88.54 ± 2.32 |
| 6 | 20 | 80 | 0 | 50 | 85.35 ± 2.52 |
| 7 | 20 | 120 | 2.0 | 20 | 79.89 ± 2.61 |
| 8 | 20 | 160 | 1.5 | 30 | 85.71 ± 2.59 |
| 9 | 30 | 40 | 1.5 | 50 | 85.38 ± 2.78 |
| 10 | 30 | 80 | 2.0 | 40 | 93.55 ± 2.59 |
| 11 | 30 | 120 | 0 | 30 | 84.18 ± 2.45 |
| 12 | 30 | 160 | 1.0 | 20 | 87.13 ± 2.53 |
| 13 | 40 | 40 | 2.0 | 20 | 75.60 ± 2.19 |
| 14 | 40 | 80 | 1.5 | 30 | 95.57 ± 2.24 |
| 15 | 40 | 120 | 1.0 | 50 | 89.25 ± 2.53 |
| 16 | 40 | 160 | 0 | 40 | 80.82 ± 2.62 |
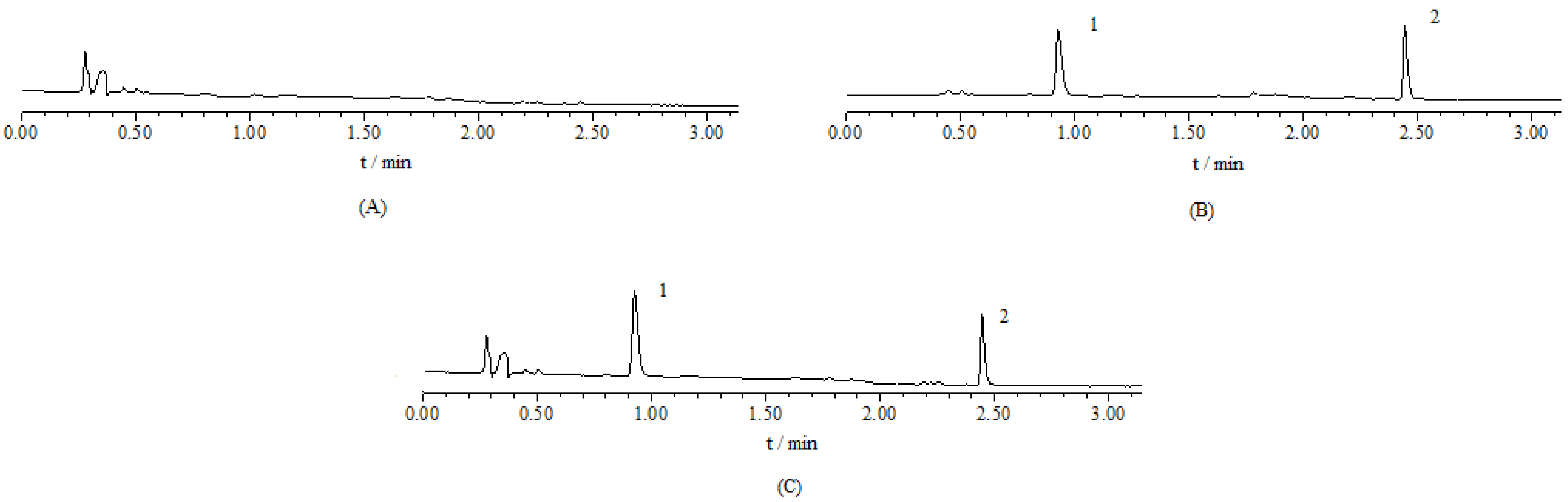
2.2. Intestinal Absorption Activity Comparison
| Concentration (μM) | k (×10−3 s−1) | |||
|---|---|---|---|---|
| pH 5.5 | pH 6.5 | pH 7.4 | pH 8.0 | |
| 4 | 2.11 | 1.71 | 1.54 | 1.39 |
| 8.3 | 2.57 | 2.49 | 2.38 | 2.03 |
| 20 | 3.31 | 2.17 | 1.97 | 1.56 |
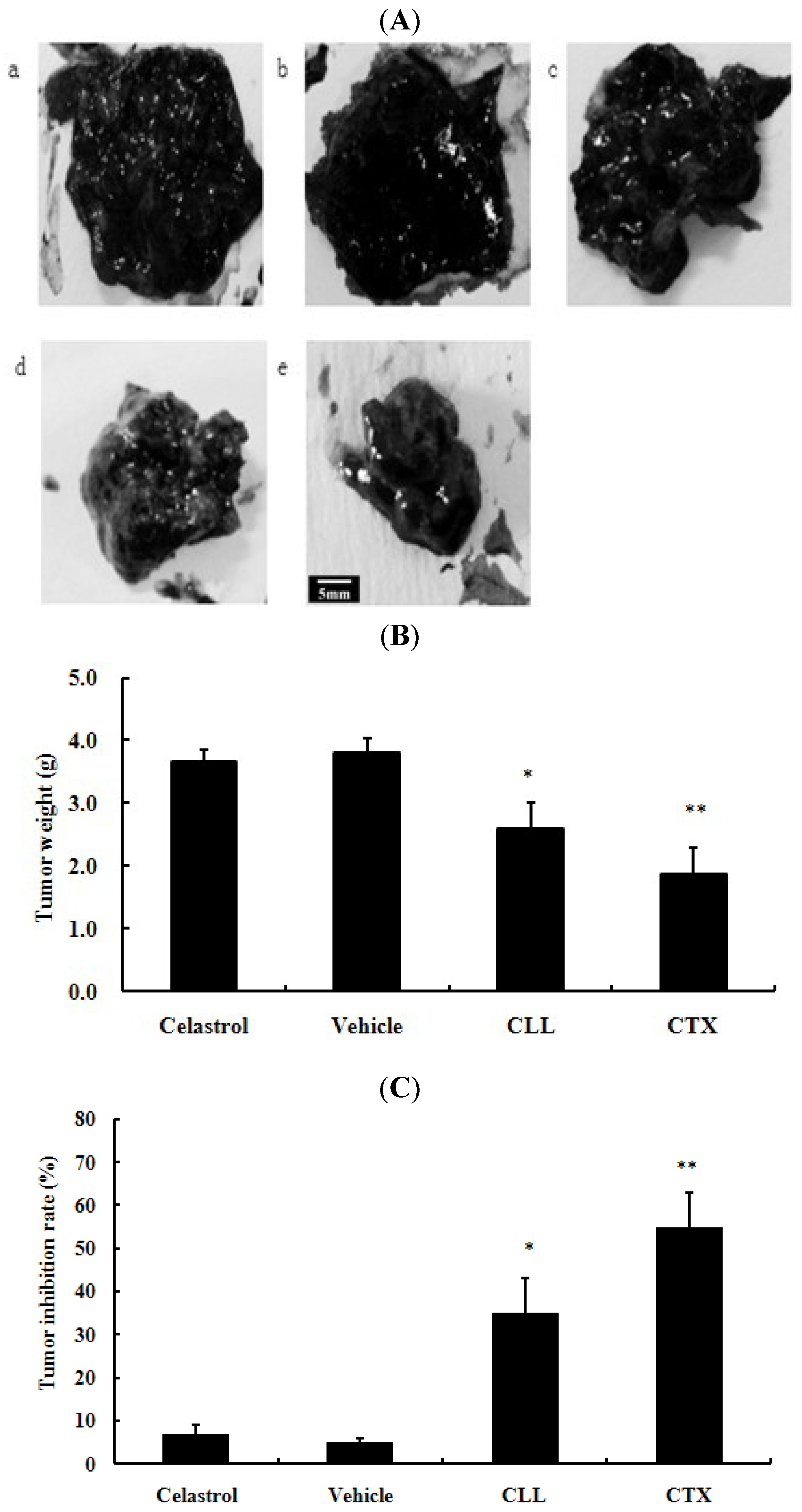
2.3. Celastrol-Loaded Liposomes Inhibited Tumor Growth

3. Experimental
3.1. Chemicals
3.2. Animals
3.3. Liposome Preparation by Ethanol Injection Method
3.4. Morphological Study by Transmission Electron Microscopy
3.5. Particle Size and Zeta-Potential Analysis of Liposomes
3.6. Determination of Encapsulation Efficiency
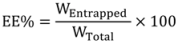
3.7. Animal Surgery
3.8. Four Site Single-Pass Rat Intestinal Perfusion Experiment
3.9. UPLC Analysis of Intestinal Perfusate
3.10. Perfusate Preparations and Stability Test of Celastrol in the Perfusate
3.11. Data Analysis
3.12. Effect of Celastrol-Loaded Liposomes on Tumor Growth in C57BL/6 Mice
3.13. Statistical Analysis
4. Conclusions
Acknowledgments
- Sample Availability: Samples of the compound celastrol are available from the authors.
References
- Liu, Z.; Ma, L.; Zhou, G. The main anticancer bullets of the Chinese medicinal herb, thunder god vine. Molecules 2011, 16, 5283–5297. [Google Scholar] [CrossRef]
- Abbas, S.; Bhoumik, A.; Dahl, R.; Vasile, S.; Krajewski, S.; Cosford, N.D.P.; Ronai, Z.A. Preclinical studies of celastrol and acetyl isogambogic acid in melanoma. Clin. Cancer Res. 2007, 13, 6769–6778. [Google Scholar]
- Peng, B.; Xu, L.; Cao, F.; Wei, T.; Yang, C.; Uzan, G.; Zhang, D. HSP90 inhibitor, celastrol, arrests human monocytic leukemia cell U937 at G0/G1 in thiol-containing agents reversible way. Mol. Cancer 2010, 9, 79. [Google Scholar]
- Ge, P.; Ji, X.; Ding, Y.; Wang, X.; Fu, S.; Meng, F.; Jin, X.; Ling, F.; Luo, Y. Celastrol causes apoptosis and cell cycle arrest in rat glioma cells. Neurol. Res. 2010, 32, 94–100. [Google Scholar] [CrossRef]
- Pang, X.; Yi, Z.; Zhang, J.; Lu, B.; Sung, B.; Qu, W.; Aggarwal, B.B.; Liu, M. Celastrol suppresses angiogenesis-mediated tumo B.r growth through inhibition of AKT/mammalian target of rapamycin pathway. Cancer Res. 2010, 70, 1951–1959. [Google Scholar] [CrossRef]
- Yang, H.; Chen, D.; Cui, Q.; Yuan, X.; Dou, Q. Celastrol, a triterpene extracted from the Chinese “Thunder of God Vine”, is a potent proteasome inhibitor and suppresses human prostate cancer growth in nude mice. Cancer Res. 2006, 66, 4758–4765. [Google Scholar] [CrossRef]
- Dai, Y.; DeSano, J.T.; Meng, Y.; Ji, Q.; Ljungman, M.; Lawrence, T.S.; Xu, L. Celastrol potentiates radiotherapy by impairment of DNA damage processing in human prostate cancer. Int. J. Radiat. Oncol. Biol. Phys. 2009, 74, 1217–1225. [Google Scholar] [CrossRef]
- Lee, J.H.; Koo, T.H.; Yoon, H.; Jung, H.; Jin, H.; Lee, K.; Hong, Y.; Lee, J.J. Inhibition of NF-kappa B activation through targeting I kappa B kinase by celastrol, a quinone methide triterpenoid. Biochem. Pharmacol. 2006, 72, 1311–1321. [Google Scholar] [CrossRef]
- Yang, F.; Jin, C.; Jiang, Y.; Li, J.; Di, Y.; Ni, Q.; Fu, D. Liposome based delivery systems in pancreatic cancer treatment: From bench to bedside. Cancer Treat. Rev. 2011. [Google Scholar] [CrossRef]
- Muthu, M.S.; Singh, S. Targeted nanomedicines: Effective treatment modalities for cancer, AIDS and brain disorders. Nanomedicine (Lond) 2009, 4, 105–118. [Google Scholar] [CrossRef]
- Gunasekera, U.A.; Pankhurst, Q.A.; Douek, M. Imaging applications of nanotechnology in cancer. Target Oncol. 2009, 4, 169–181. [Google Scholar] [CrossRef]
- Barenholz, Y. Liposome application: Problems and prospects. Curr. Opin. Colloid Interface Sci. 2001, 6, 66–77. [Google Scholar] [CrossRef]
- Domazou, A.S.; Luisi, P.L. Size distribution of spontaneously formed liposomes by the alcohol injection method. J. Liposome Res. 2002, 12, 205–220. [Google Scholar] [CrossRef]
- Isele, U.; van Hoogevest, P.; Hilfiker, R.; Capraro, H.G.; Schieweck, K.; Leuenberger, H. Large-scale production of liposomes containing monomeric zinc phthalocyanine by controlled dilution of organic solvents. J. Pharm. Sci. 1994, 11, 1608–1616. [Google Scholar]
- Sonar, S.; D’Souza, S.E.; Mishra, K.P. A simple one-step protocol for preparing small-sized doxorubicin-loaded liposomes. J. Environ. Pathol. Toxicol. Oncol. 2008, 27, 181–189. [Google Scholar] [CrossRef]
- Justo, O.R.; Moraes, A.M. Kanamycin incorporation in lipid vesicles prepared by ethanol injection designed for tuberculosis treatment. J. Pharm. Pharmacol. 2005, 57, 23–30. [Google Scholar]
- Schubert, M.A.; Müller-Goymann, C.C. Solvent injection as a new approach for manufacturing lipid nanoparticles-evaluation of the method and process parameters. Eur. J. Pharm. Biopharm. 2003, 55, 125–131. [Google Scholar] [CrossRef]
- Müller, R.H.; Jacobs, C.; Kayser, O. Nanosuspensions as particulate drug formulations in therapy: Rationale for development and what we can expect for the future. Adv. Drug. Deliv. Rev. 2001, 47, 3–19. [Google Scholar] [CrossRef]
- Batzri, S.; Korn, E.D. Single bilayer liposomes prepared without sonication. Biochim. Biophys. Acta 1973, 298, 1015–1019. [Google Scholar] [CrossRef]
- Zhuang, C.Y.; Li, N.; Wang, M.; Zhang, X.N.; Pan, W.S.; Peng, J.J.; Pan, Y.S.; Tang, X. Preparation and characterization of vinpocetine loaded nanostructured lipid carriers (NLC) for improved oral bioavailability. Int. J. Pharm. 2010, 394, 179–185. [Google Scholar] [CrossRef]
- Fry, D.W.; White, J.C.; Goldman, I.D. Rapid separation of low molecular weight solutes from liposomes without dilution. Anal. Biochem. 1978, 90, 809–815. [Google Scholar] [CrossRef]
- New, R.R.C. Liposomes: A Practical Approach; Oxford University Press: New York, NY, USA, 1990; pp. 33–104. [Google Scholar]
- El-Maghraby, G.M.; Williams, A.C.; Barry, B.W. Skin delivery of 5-fluorouracil from ultradeformable and standard liposomes in-vitro. J. Pharm. Pharmacol. 2001, 53, 1069–1077. [Google Scholar]
- Abuasal, B.; Sylvester, P.W.; Kaddoumi, A. Intestinal absorption of -tocotrienol is mediated by Niemann-Pick C1-like 1: In situ rat intestinal perfusion studies. Drug Metab. Dispos. 2010, 38, 939–945. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, J.; Jia, X.; Tan, X.; Hu, M. Role of intestinal hydrolase in the absorption of prenylated flavonoids present in yinyanghuo. Molecules 2011, 16, 1336–1348. [Google Scholar] [CrossRef]
- Feng, L.; Jia, X.; Zhu, M.; Chen, Y.; Shi, F. Antioxidant activities of total phenols of prunella vulgaris L. in vitro and in tumor-bearing mice. Molecules 2010, 15, 9145–9156. [Google Scholar] [CrossRef]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Song, J.; Shi, F.; Zhang, Z.; Zhu, F.; Xue, J.; Tan, X.; Zhang, L.; Jia, X. Formulation and Evaluation of Celastrol-Loaded Liposomes. Molecules 2011, 16, 7880-7892. https://doi.org/10.3390/molecules16097880
Song J, Shi F, Zhang Z, Zhu F, Xue J, Tan X, Zhang L, Jia X. Formulation and Evaluation of Celastrol-Loaded Liposomes. Molecules. 2011; 16(9):7880-7892. https://doi.org/10.3390/molecules16097880
Chicago/Turabian StyleSong, Jie, Feng Shi, Zhenhai Zhang, Fenxia Zhu, Jing Xue, Xiaobin Tan, Luyong Zhang, and Xiaobin Jia. 2011. "Formulation and Evaluation of Celastrol-Loaded Liposomes" Molecules 16, no. 9: 7880-7892. https://doi.org/10.3390/molecules16097880
APA StyleSong, J., Shi, F., Zhang, Z., Zhu, F., Xue, J., Tan, X., Zhang, L., & Jia, X. (2011). Formulation and Evaluation of Celastrol-Loaded Liposomes. Molecules, 16(9), 7880-7892. https://doi.org/10.3390/molecules16097880




