Effect of ASP2151, a Herpesvirus Helicase-Primase Inhibitor, in a Guinea Pig Model of Genital Herpes
Abstract
:1. Introduction
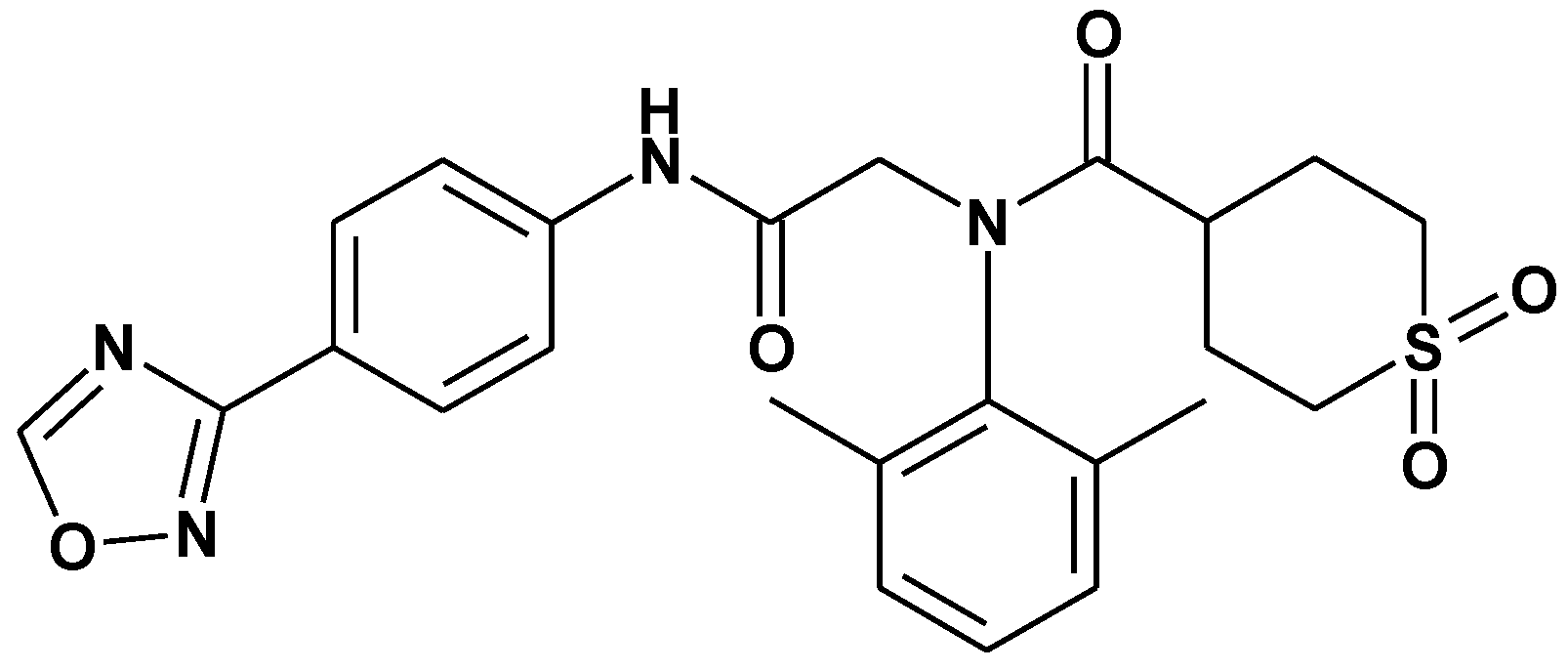
2. Results and Discussion
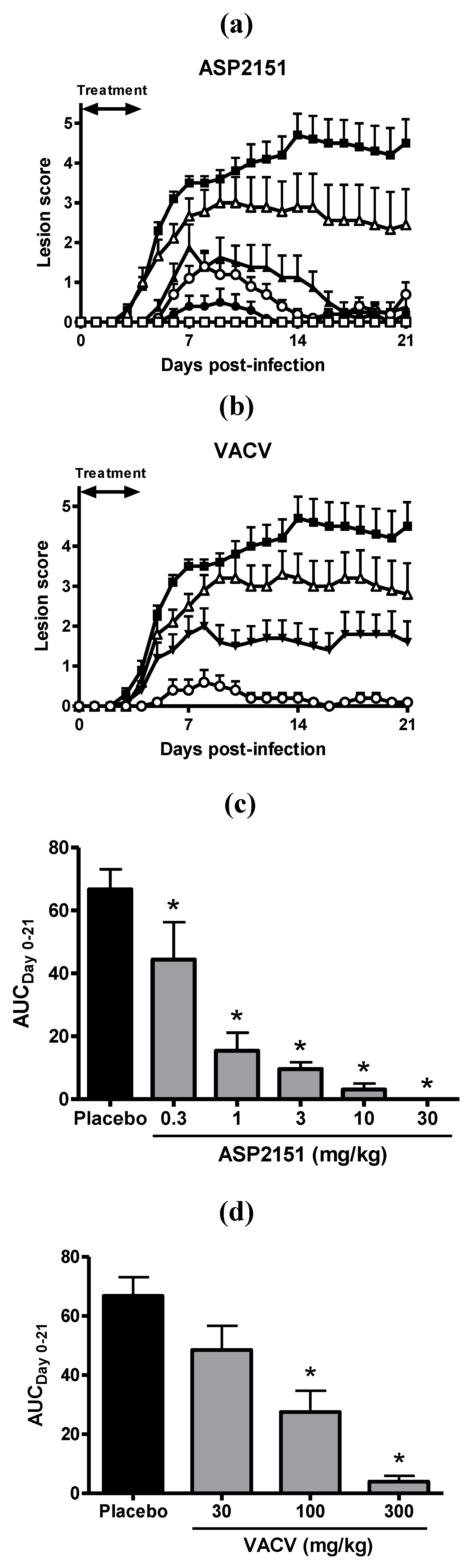
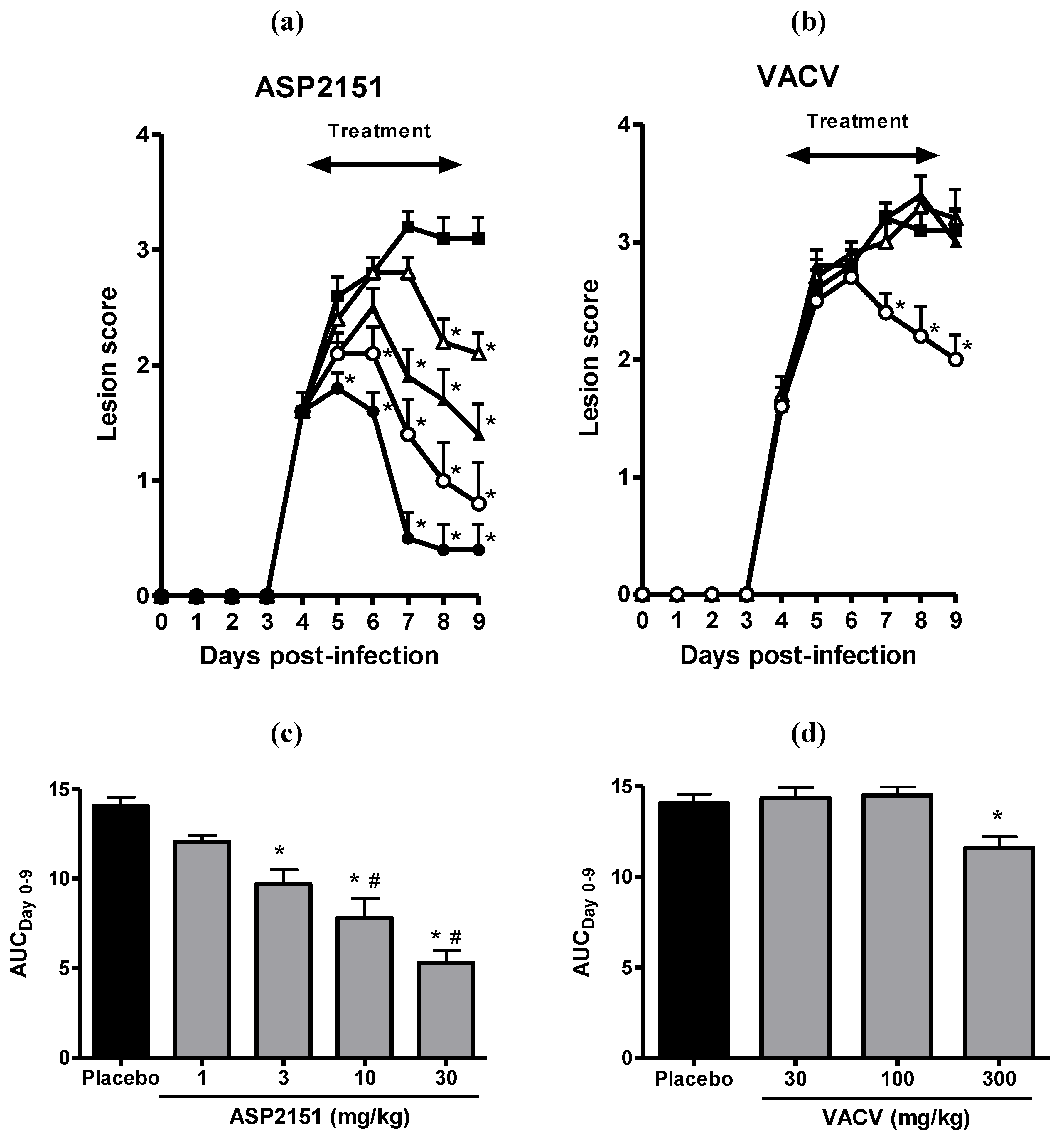
2.3. Therapeutic Efficacies of ASP2151 and VACV on Genital Herpes Symptoms in HSV-2-Infected Guinea Pigs
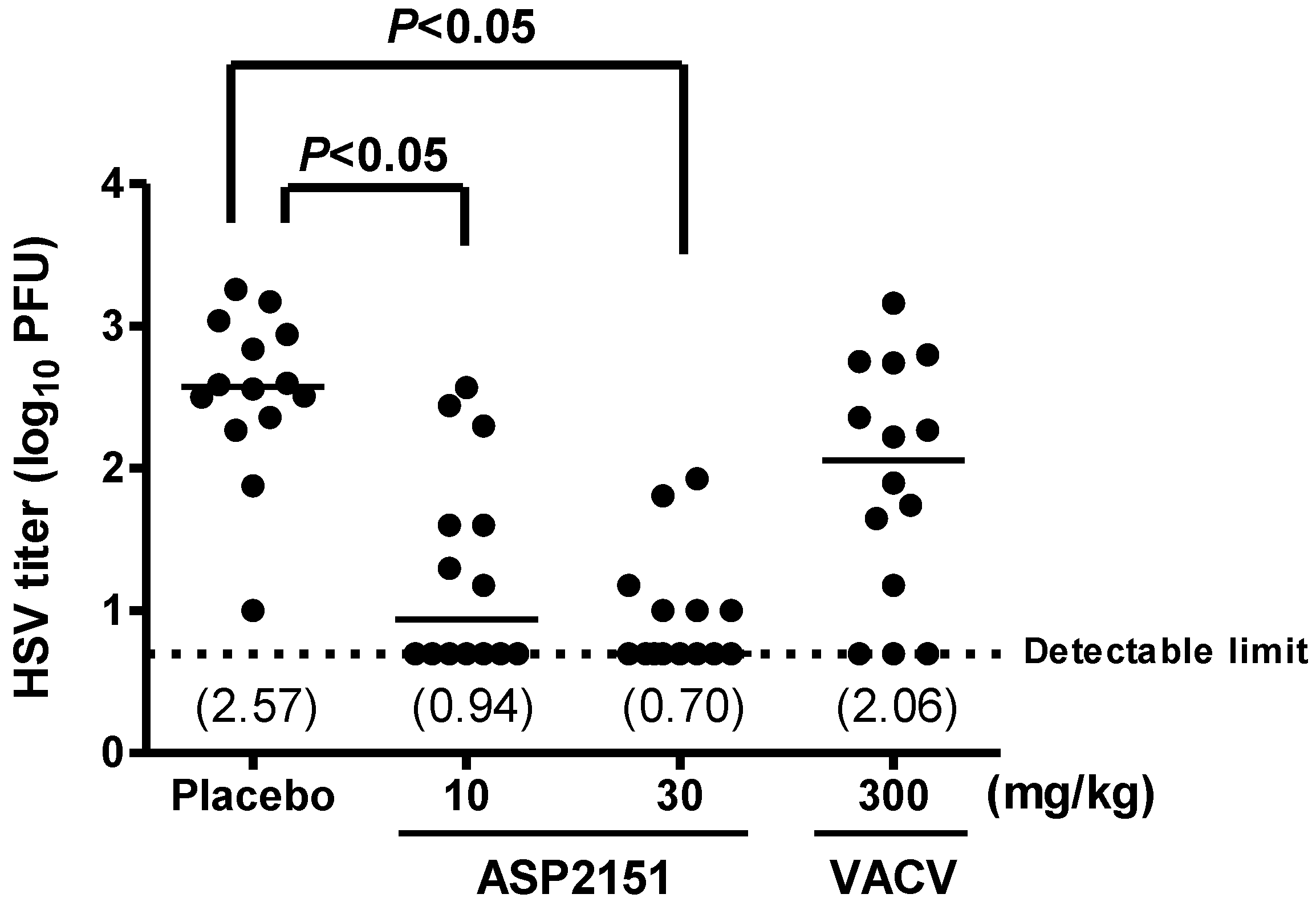
2.4. Therapeutic Efficacies of ASP2151 and VACV on Viral Shedding
2.5. Discussion
3. Experimental Section
3.1. Drugs
3.2. Viruses and Cell Lines
3.3. Plaque Reduction and Cytotoxicity Assays
3.5. Virus Detection
3.6. Statistical Analyses
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Pellet, P.E.; Roizman, B. The family Herpesviridae: A brief introduction. In Fields Virology, 5th; Knipe, D., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; Volume 2, pp. 2479–2499. [Google Scholar]
- Benedetti, J.; Corey, L.; Ashley, R. Recurrence rates in genital herpes after symptomatic first-episode infection. Ann. Intern. Med. 1994, 121, 847–854. [Google Scholar]
- Kinghorn, G.R. Epidemiology of genital herpes. J. Int. Med. Res. 1994, 22 (Suppl. 1), 14–23. [Google Scholar]
- Whitley, R.J.; Roizman, B. Herpes simplex virus infections. Lancet 2001, 357, 1513–1518. [Google Scholar] [CrossRef]
- Ashley, R.L.; Wald, A. Genital herpes: Review of the epidemic and potential use of type-specific serology. Clin. Microbiol. Rev. 1999, 12, 1–8. [Google Scholar]
- Whitley, R.J.; Kimberlin, D.W.; Roizman, B. Herpes simplex viruses. Clin. Infect. Dis. 1998, 26, 541–553. [Google Scholar] [CrossRef]
- Lafferty, W.E. The changing epidemiology of HSV-1 and HSV-2 and implications for serological testing. Herpes 2002, 9, 51–55. [Google Scholar]
- Wald, A.; Zeh, J.; Selke, S.; Ashley, R.L.; Corey, L. Virologic characteristics of subclinical and symptomatic genital herpes infections. N. Engl. J. Med. 1995, 333, 770–775. [Google Scholar] [CrossRef]
- Lafferty, W.E.; Coombs, R.W.; Benedetti, J.; Critchlow, C.; Corey, L. Recurrences after oral and genital herpes simplex virus infection. N. Engl. J. Med. 1987, 316, 1444–1449. [Google Scholar] [CrossRef]
- Engelberg, R.; Carrell, D.; Krantz, E.; Corey, L.; Wald, A. Natural history of genital herpes simplex virus type 1 infection. Sex Transm. Dis. 2003, 30, 174–177. [Google Scholar] [CrossRef]
- De Clercq, E. Antivirals for the treatment of herpesvirus infections. J. Antimicrob. Chemother. 1993, 32 (Suppl. A), 121–132. [Google Scholar]
- De Clercq, E.; Field, H.J. Antiviral prodrugs - the development of successful prodrug strategies for antiviral chemotherapy. Br. J. Pharmacol. 2006, 147, 1–11. [Google Scholar] [CrossRef]
- Crumpacker, C.S.; Schaffer, P.A. New anti-HSV therapeutics target the helicase-primase complex. Nat. Med. 2002, 8, 327–328. [Google Scholar] [CrossRef]
- Reichman, R.C.; Badger, G.J.; Mertz, G.J.; Corey, L.; Richman, D.D.; Connor, J.D.; Redfield, D.; Savoia, M.C.; Oxman, M.N.; Bryson, Y.; Tyrrell, D.L.; Portnoy, J.; Creigh-Kirk, T.; Keeney, R.E.; Ashikaga, T.; Dolin, R. Treatment of recurrent genital herpes simplex infections with oral acyclovir: a controlled trial. JAMA 1984, 251, 2103–2107. [Google Scholar]
- Spruance, S.L.; Stewart, J.C.; Rowe, N.H.; McKeough, M.B.; Wenerstrom, G.; Freeman, D.J. Treatment of recurrent herpes simplex labialis with oral acyclovir. J. Infect. Dis. 1990, 161, 185–190. [Google Scholar] [CrossRef]
- Chono, K.; Katsumata, K.; Kontani, T.; Kobayashi, M.; Sudo, K.; Yokota, T.; Konno, K.; Shimizu, Y.; Suzuki, H. ASP2151, a novel helicase-primase inhibitor, possesses antiviral activity against varicella-zoster virus and herpes simplex virus types 1 and 2. J. Antimicrob. Chemother. 2010, 65, 1733–1741. [Google Scholar] [CrossRef]
- Crute, J.J.; Grygon, C.A.; Hargrave, K.D.; Simoneau, B.; Faucher, A.M.; Bolger, G.; Kibler, P.; Liuzzi, M.; Cordingley, M.G. Herpes simplex virus helicase-primase inhibitors are active in animal models of human disease. Nat. Med. 2002, 8, 386–391. [Google Scholar] [CrossRef]
- Kleymann, G.; Fischer, R.; Betz, U.A.; Hendrix, M.; Bender, W.; Schneider, U.; Handke, G.; Eckenberg, P.; Hewlett, G.; Pevzner, V.; Baumeister, J.; Weber, O.; Henninger, K.; Keldenich, J.; Jensen, A.; Kolb, J.; Bach, U.; Popp, A.; Maben, J.; Frappa, I.; Haebich, D.; Lockhoff, O.; Rubsamen-Waigmann, H. New helicase-primase inhibitors as drug candidates for the treatment of herpes simplex disease. Nat. Med. 2002, 8, 392–398. [Google Scholar]
- Baumeister, J.; Fischer, R.; Eckenberg, P.; Henninger, K.; Ruebsamen-Waigmann, H.; Kleymann, G. Superior efficacy of helicase-primase inhibitor BAY 57-1293 for herpes infection and latency in the guinea pig model of human genital herpes disease. Antiviral Chem. Chemother. 2007, 18, 35–48. [Google Scholar]
- Betz, U.A.K.; Fischer, R.; Kleymann, G.; Hendrix, M.; Rubsamen-Waigmann, H. Potent in vivo antiviral activity of the herpes simplex virus primase-helicase inhibitor BAY 57-1293. Antimicrob. Agents Chemother. 2002, 46, 1766–1772. [Google Scholar] [CrossRef]
- Lucas, B.A.; Wiesendanger, W.; Schmidt-Ruppin, K.H. Genital herpres in guinea pigs. Archives Virol. 1974, 44, 153–155. [Google Scholar]
- Whitley, R.J. Herpes simplex viruses. In Fields Virology, 5th; Knipe, D., Howley, P.M., Eds.; Lippincott Williams & Wilkins: Philadelphia, PA, USA, 2007; Volume 2, pp. 2501–2601. [Google Scholar]
- Landry, M.L.; Myerson, D.; Bull, C. Recurrent genital infection in the guinea pig: differences between herpes simplex types 1 and 2. Intervirology 1992, 34, 169–179. [Google Scholar]
- Stanberry, L.R. Animal-models and HSV latency. Semin. Virol. 1994, 5, 213–219. [Google Scholar] [CrossRef]
- Stanberry, L.R.; Kern, E.R.; Richards, J.T.; Abbott, T.M.; Overall, J.C., Jr. Genital herpes in guinea pigs: pathogenesis of the primary infection and description of recurrent disease. J. Infect. Dis. 1982, 146, 397–404. [Google Scholar] [CrossRef]
- Stanberry, L.R.; Kit, S.; Myers, M.G. Thymidine kinase-deficient herpes simplex virus type 2 genital infection in guinea pigs. J. Virol. 1985, 55, 322–328. [Google Scholar]
- Stanberry, L.R.; Kern, E.R.; Richards, J.T.; Overall, J.C., Jr. , Recurrent genital herpes simplex virus infection in guinea pigs. Intervirology 1985, 24, 226–231. [Google Scholar] [CrossRef]
- Bourne, N.; Ireland, J.; Stanberry, L.R.; Bernstein, D.I. Effect of undecylenic acid as a topical microbicide against genital herpes infection in mice and guinea pigs. Antiviral Res. 1999, 40, 139–144. [Google Scholar] [CrossRef]
- Bernstein, D.I.; Harrison, C.J.; Tomai, M.A.; Miller, R.L. Daily or weekly therapy with resiquimod (R-848) reduces genital recurrences in herpes simplex virus-infected guinea pigs during and after treatment. J. Infect Dis. 2001, 183, 844–849. [Google Scholar] [CrossRef]
- Bourne, N.; Bravo, F.J.; Ashton, W.T.; Meurer, L.C.; Tolman, R.L.; Karkas, J.D.; Stanberry, L.R. Assessment of a selective inhibitor of herpes simplex virus thymidine kinase (L-653,180) as therapy for experimental recurrent genital herpes. Antimicrob. Agents Chemother. 1992, 36, 2020–2024. [Google Scholar] [CrossRef]
- Bravo, F.J.; Stanberry, L.R.; Kier, A.B.; Vogt, P.E.; Kern, E.R. Evaluation of HPMPC therapy for primary and recurrent genital herpes in mice and guinea pigs. Antiviral Res. 1993, 21, 59–72. [Google Scholar] [CrossRef]
- Jennings, R.; Smith, T.L.; Myhren, F.; Phillips, J.; Sandvold, M.L. Evaluation of a novel, anti-herpes simplex virus compound, acyclovir elaidate (P-4010), in the female guinea pig model of genital herpes. Antimicrob. Agents Chemother. 1999, 43, 53–61. [Google Scholar]
- Rose, W.A., II.; Tuthill, C.; Pyles, R.B. An immunomodulating dipeptide, SCV-07, is a potential therapeutic for recurrent genital herpes simplex virus type 2 (HSV-2). Int. J. Antimicrob. Agents 2008, 32, 262–266. [Google Scholar] [CrossRef]
- Miller, R.L.; Imbertson, L.M.; Reiter, M.J.; Gerster, J.F. Treatment of primary herpes simplex virus infection in guinea pigs by imiquimod. Antiviral Res. 1999, 44, 31–42. [Google Scholar] [CrossRef]
- Landry, M.L.; Lucia, H.L.; Hsiung, G.D.; Pronovost, A.D.; Dann, P.R.; August, M.J.; Mayo, D.R. Effect of acyclovir on genital infection with herpes simplex virus types 1 and 2 in the guinea pig. Am. J. Med. 1982, 73, 143–150. [Google Scholar] [CrossRef]
- Pronovost, A.D.; Lucia, H.L.; Dann, P.R.; Hsiung, G.D. Effect of acyclovir on genital herpes in guinea pigs. J. Infect. Dis. 1982, 145, 904–908. [Google Scholar] [CrossRef]
- Myerson, D.; Hsiung, G.D. Prophylactic and therapeutic treatment with acyclovir of genital herpes in the guinea pig. Proc. Soc. Exp. Biol. Med. 1983, 174, 147–152. [Google Scholar]
- Corey, L.; Wald, A.; Patel, R.; Sacks, S.L.; Tyring, S.K.; Warren, T.; Douglas, J.M.; Paavonen, J.; Morrow, R.A.; Beutner, K.R.; Stratchounsky, L.S.; Mertz, G.; Keene, O.N.; Watson, H.A.; Tait, D.; Vargas-Cortes, M.; Stud, V.H.T. Once-daily valacyclovir to reduce the risk of transmission of genital herpes. N. Engl. J. Med. 2004, 350, 11–20. [Google Scholar]
- Wald, A.; Corey, L.; Cone, R.; Hobson, A.; Davis, G.; Zeh, J. Frequent genital herpes simplex virus 2 shedding in immunocompetent women. Effect of acyclovir treatment. J. Clin. Invest. 1997, 99, 1092–1097. [Google Scholar] [CrossRef]
- Gupta, R.; Wald, A.; Krantz, E.; Selke, S.; Warren, T.; Vargas-Cortes, M.; Miller, G.; Corey, L. Valacyclovir and acyclovir for suppression of shedding of herpes simplex virus in the genital tract. J. Infect. Dis. 2004, 190, 1374–1381. [Google Scholar] [CrossRef]
- Kern, E.R. Acyclovir treatment of experimental genital herpes simplex virus infections. Am. J. Med. 1982, 73, 100–108. [Google Scholar] [CrossRef]
- De Clercq, E.; Descamps, J.; Ogata, M.; Shigeta, S. In vitro susceptibility of varicella-zoster virus to E-5-(2-bromovinyl)-2'-deoxyuridine and related compounds. Antimicrob. Agents. Chemother. 1982, 21, 33–38. [Google Scholar] [CrossRef]
- Sample Availability: Not available.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Katsumata, K.; Chono, K.; Sudo, K.; Shimizu, Y.; Kontani, T.; Suzuki, H. Effect of ASP2151, a Herpesvirus Helicase-Primase Inhibitor, in a Guinea Pig Model of Genital Herpes. Molecules 2011, 16, 7210-7223. https://doi.org/10.3390/molecules16097210
Katsumata K, Chono K, Sudo K, Shimizu Y, Kontani T, Suzuki H. Effect of ASP2151, a Herpesvirus Helicase-Primase Inhibitor, in a Guinea Pig Model of Genital Herpes. Molecules. 2011; 16(9):7210-7223. https://doi.org/10.3390/molecules16097210
Chicago/Turabian StyleKatsumata, Kiyomitsu, Koji Chono, Kenji Sudo, Yasuaki Shimizu, Toru Kontani, and Hiroshi Suzuki. 2011. "Effect of ASP2151, a Herpesvirus Helicase-Primase Inhibitor, in a Guinea Pig Model of Genital Herpes" Molecules 16, no. 9: 7210-7223. https://doi.org/10.3390/molecules16097210
APA StyleKatsumata, K., Chono, K., Sudo, K., Shimizu, Y., Kontani, T., & Suzuki, H. (2011). Effect of ASP2151, a Herpesvirus Helicase-Primase Inhibitor, in a Guinea Pig Model of Genital Herpes. Molecules, 16(9), 7210-7223. https://doi.org/10.3390/molecules16097210