Antimicrobial and Antioxidant Activities of New Metal Complexes Derived from 3-Aminocoumarin
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
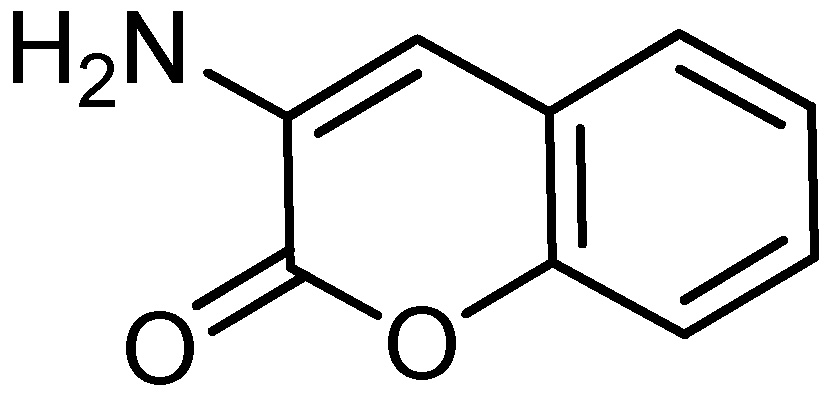
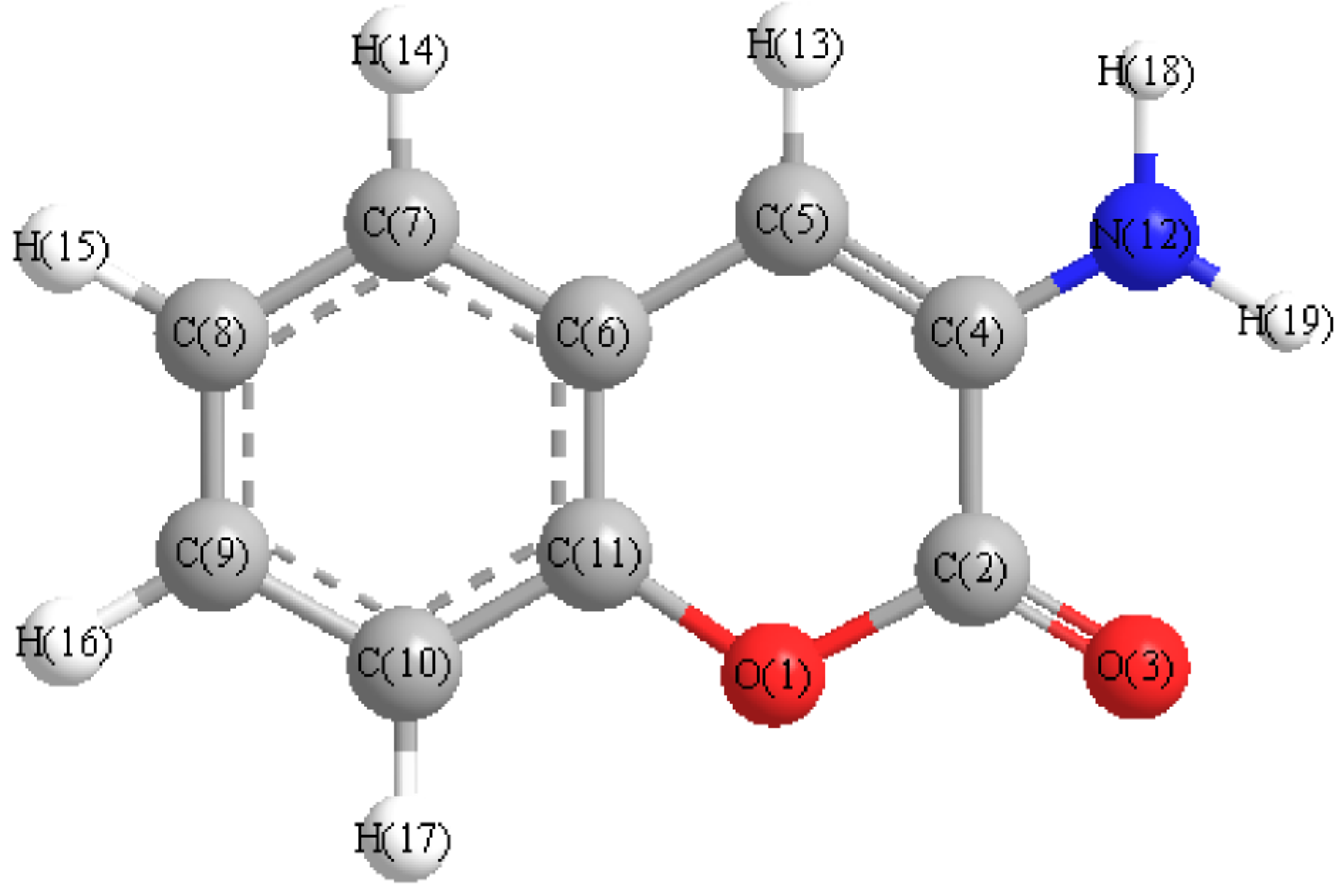
| Atom | Charge | Atom | Charge | Atom | Charge |
|---|---|---|---|---|---|
| O(1) | −0.0576918 | C(7) | −0.115268 | H(13) | 0.0181674 |
| C(2) | 0.546493 | C(8) | −0.0752691 | H(14) | 0.0223331 |
| O(3) | −0.580016 | C(9) | −0.101239 | H(15) | 0.0253347 |
| C(4) | 0.144636 | C(10) | −0.110194 | H(16) | 0.0249427 |
| C(5) | −0.359074 | C(11) | 0.197216 | H(17) | 0.0281505 |
| C(6) | 0.00167107 | N(12) | 0.205349 | H(18) | 0.092316 |
| H(19) | 0.0921441 | − | − | − |
| No. | Compounds | M:L | M.P. °C | Yield % | Elemental analysis (calculated) | Elemental analysis (found) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| C% | H% | N% | M% | C% | H% | N% | M% | |||||
| C1 | [Cr(L)2Cl2]Cl | 1:2 | 189 | 40 | 44.98 | 2.94 | 5.83 | 10.82 | 45.02 | 2.42 | 5.30 | 10.65 |
| C2 | [Ni(L)2Cl2] | 1:2 | 221d | 55 | 47.84 | 3.12 | 6.20 | 12.99 | 47.59 | 3.07 | 6.09 | 12.53 |
| C3 | [Cu(L)2Cl2] | 1:2 | 209d | 67 | 47.33 | 3.09 | 6.13 | 13.91 | 46.98 | 2.80 | 5.92 | 13.99 |
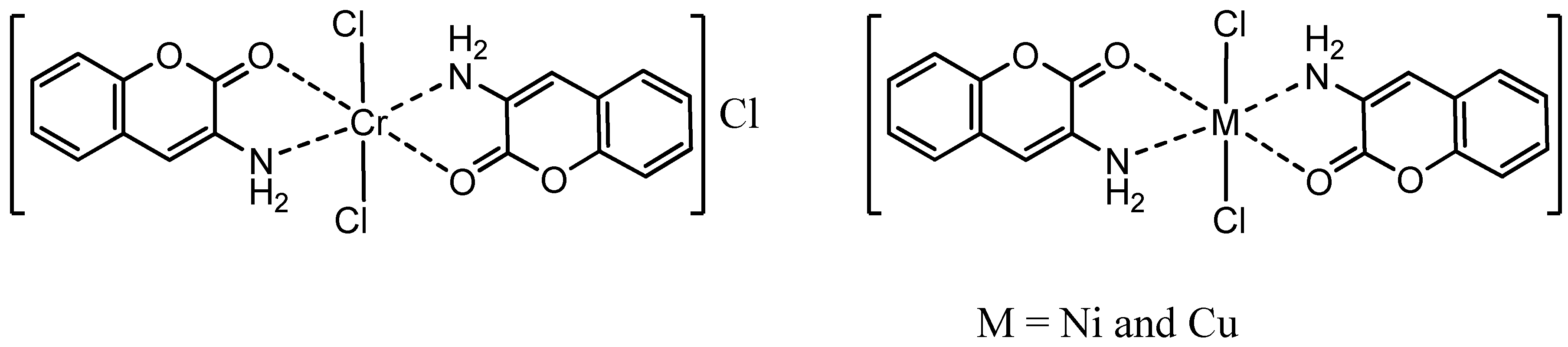
2.1.1. Elemental Analysis
2.1.2. Infrared Spectra
| NO. | N–H | C=O | M–Cl | νM–N | νM–O |
|---|---|---|---|---|---|
| C1 | 3315, 3201 | 1691 | 343 | 467 | 503 |
| C2 | 3310, 3181 | 1701 | 330 | 455 | 501 |
| C3 | 3326, 3217 | 1685 | 330 | 459 | 486 |
2.1.3. 1H-NMR Spectra
2.1.4. Molar Conductance
| No. | λmax cm−1 | Magnetic moment µ (B.M.) | ʌ ohm 1 cm2 mol−1 | Structure |
|---|---|---|---|---|
| C1 | 15217, 20112, 32125 | 4.1 | 90 | Octahedral |
| C2 | 15890, 31915 | 2.6 | 20 | Octahedral |
| C3 | 12117, 15812, 22076 | 1.6 | 22 | Octahedral |
2.1.5. Magnetic Moment and UV-Vis Spectra
2.1.6. Suggested Stereostructures of the Complexes
2.2. Pharmacology
2.2.1. Antibacterial Activity
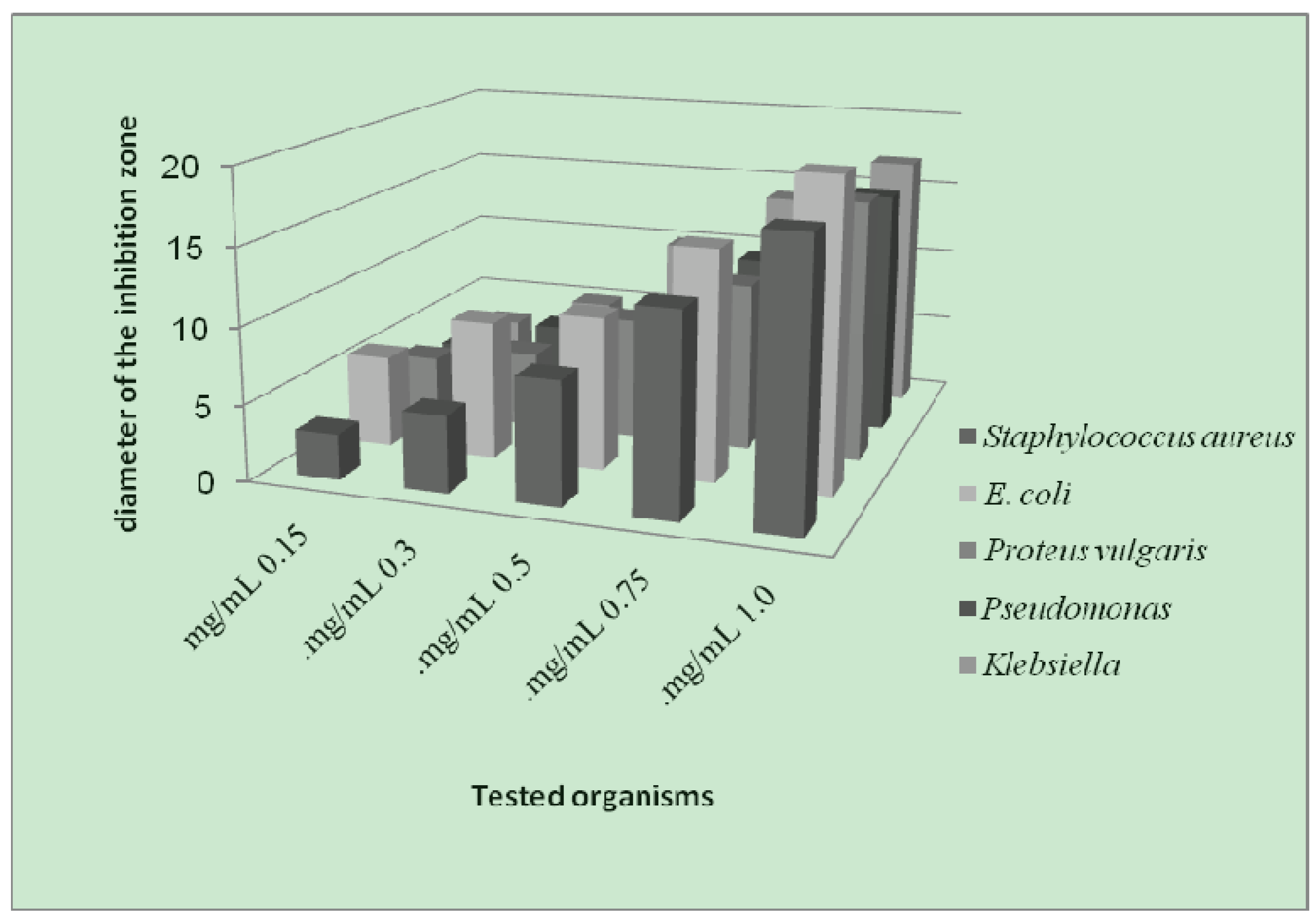

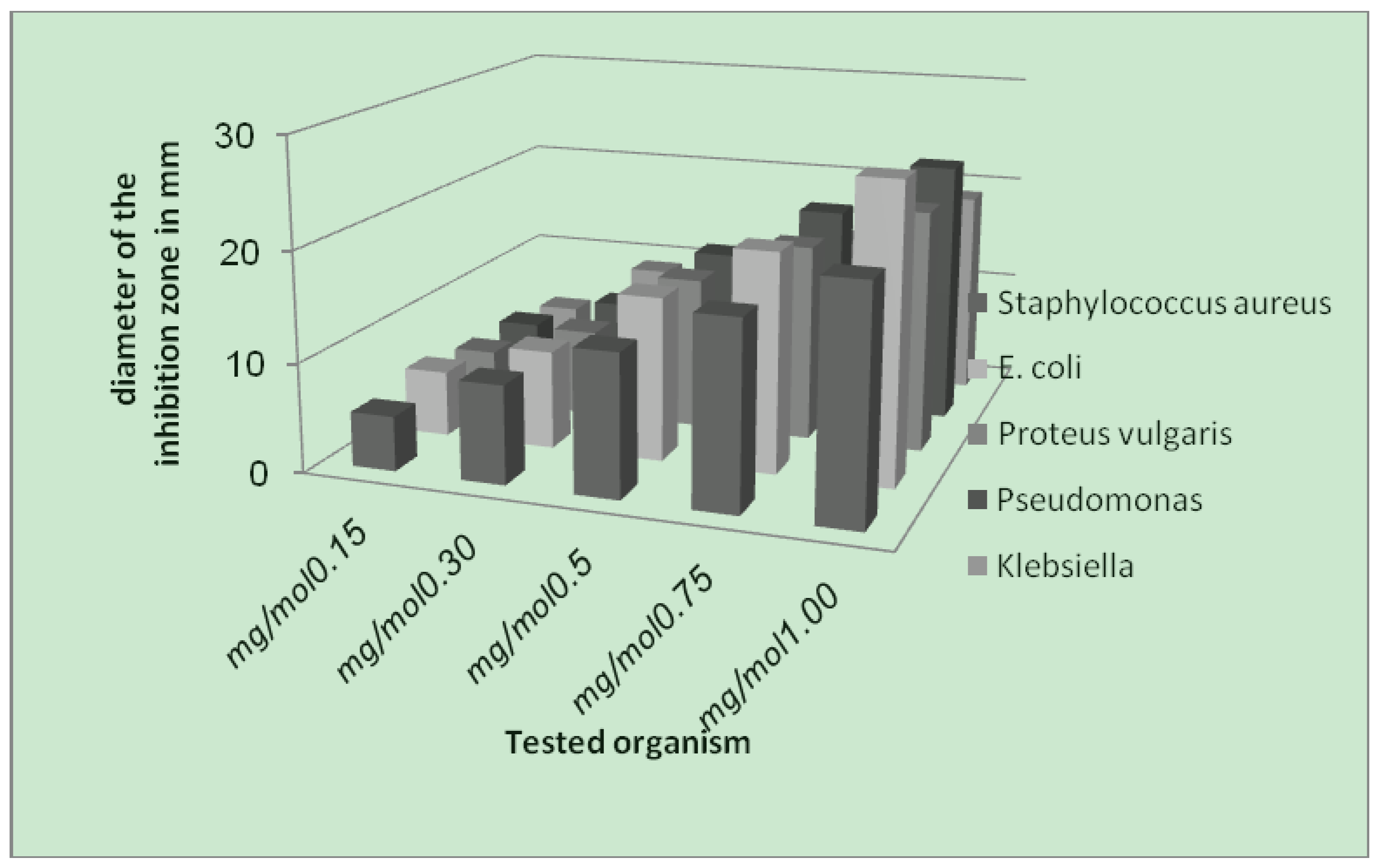
- (1). Generally, the result of prepared complexes exhibited antibacterial activity toward E. coli bacteria was more than the inhibition on other types of bacteria.
- (2). The copper complex has more activity toward all the kinds of tested bacteria, compared to other complexes. The stability of the copper complex and the coupling with the ligand may be the reasons for this activity against bacteria.
2.2.2. Antifungal Activities
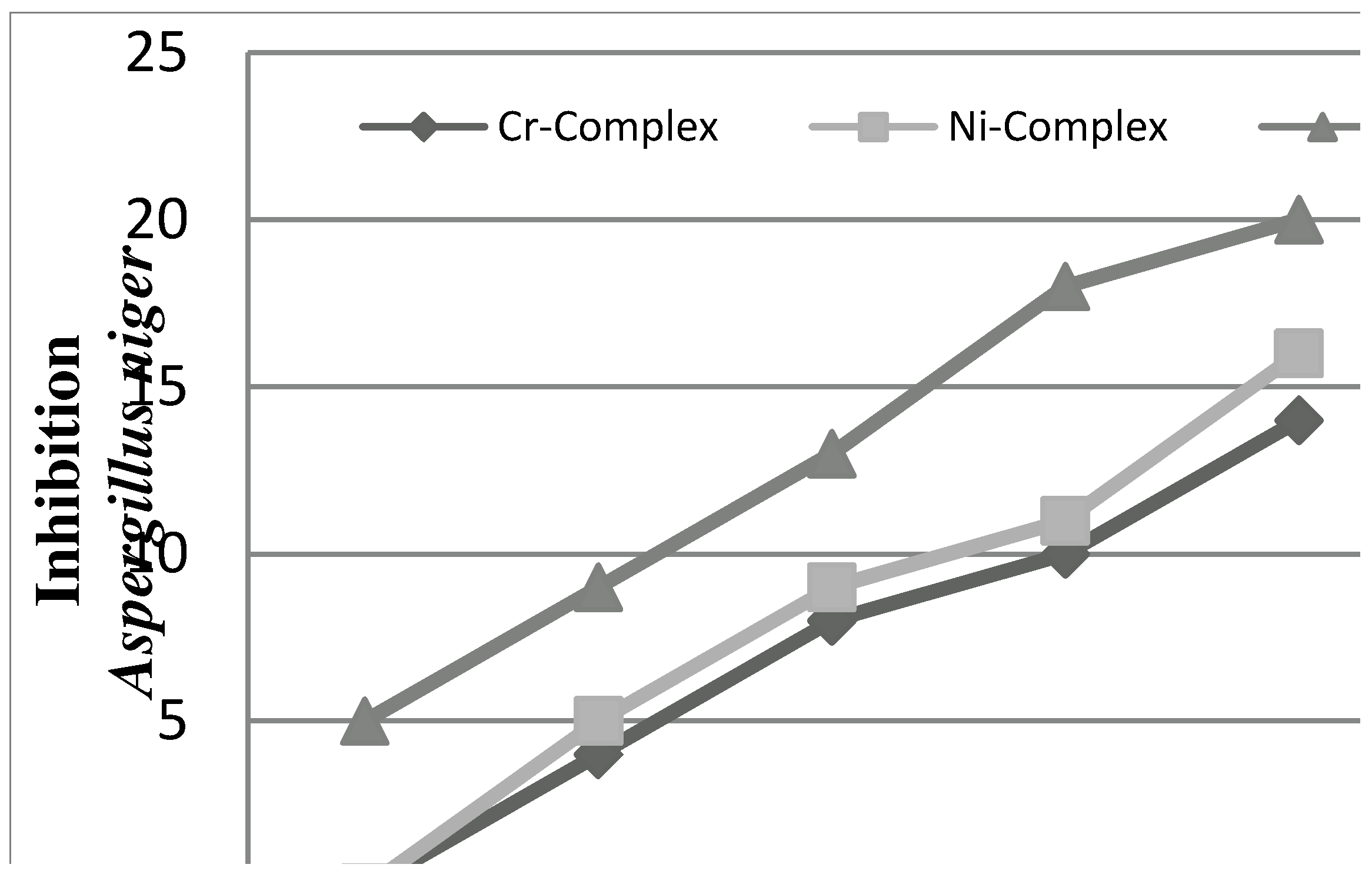
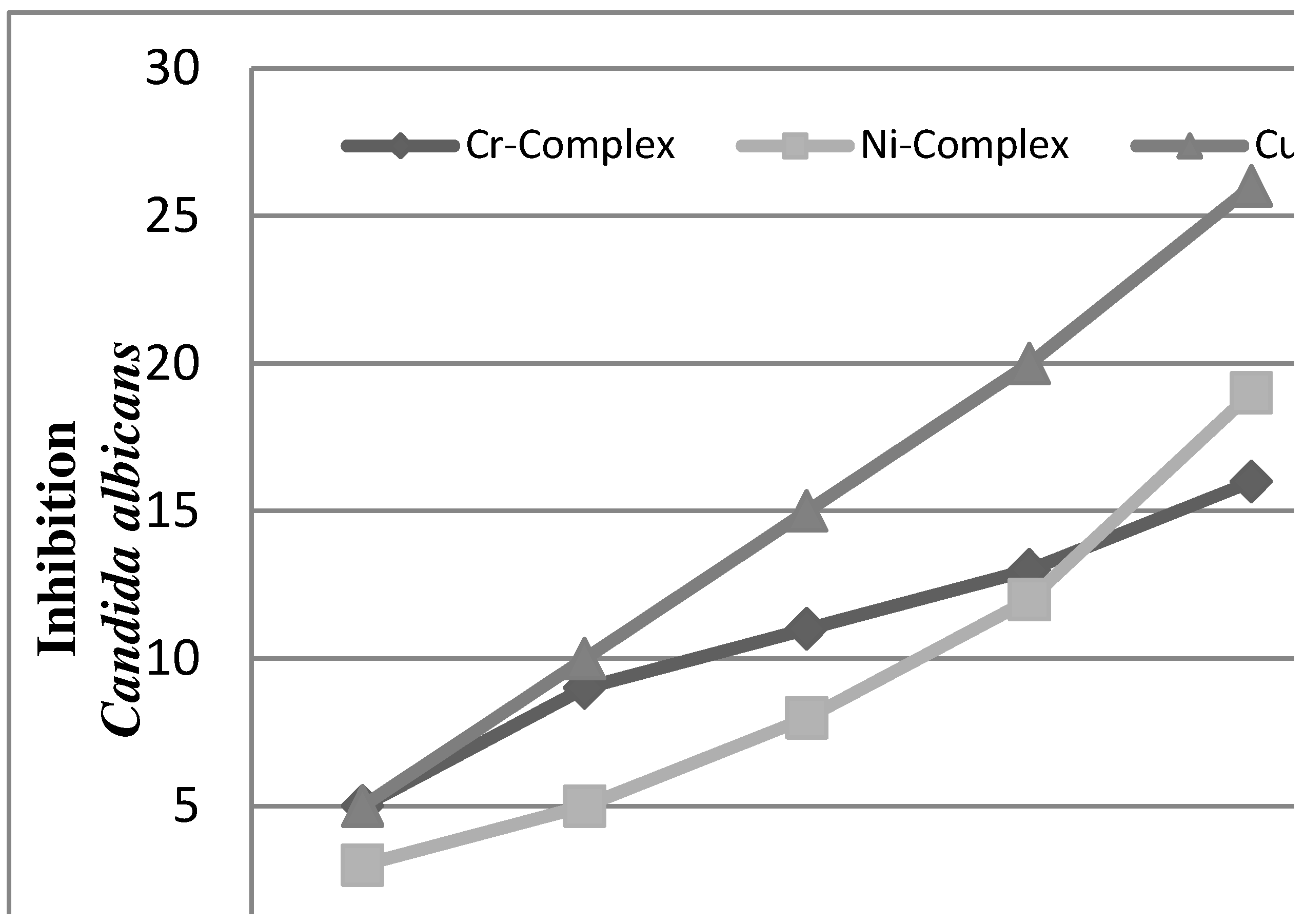
2.2.3. Radical Scavenging Activity
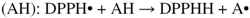
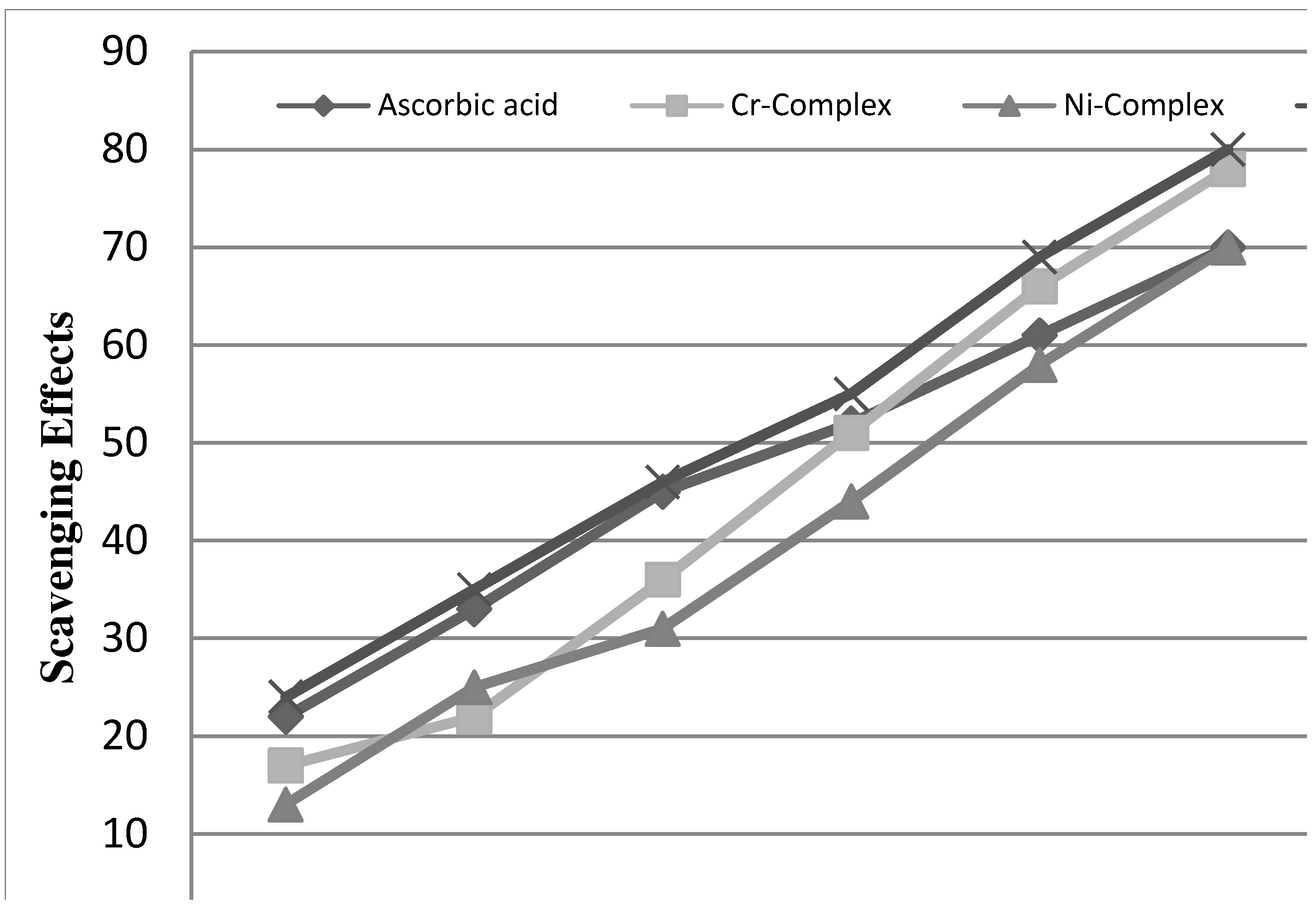
3. Experimental
3.1. General
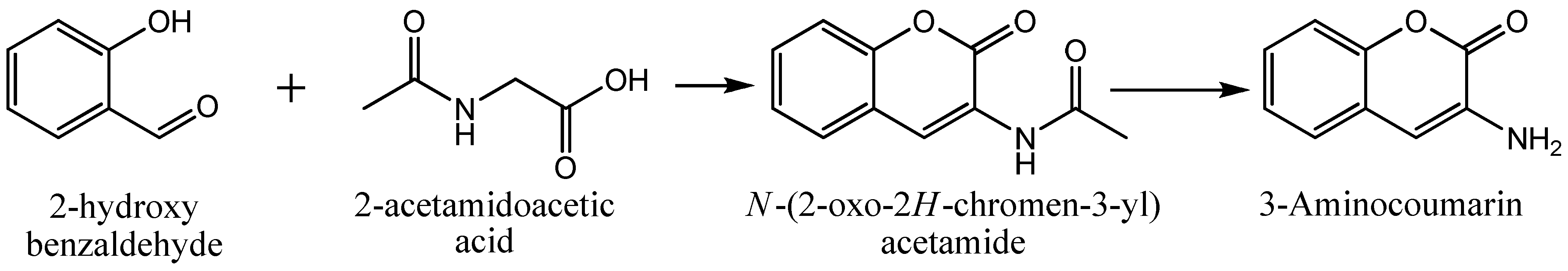
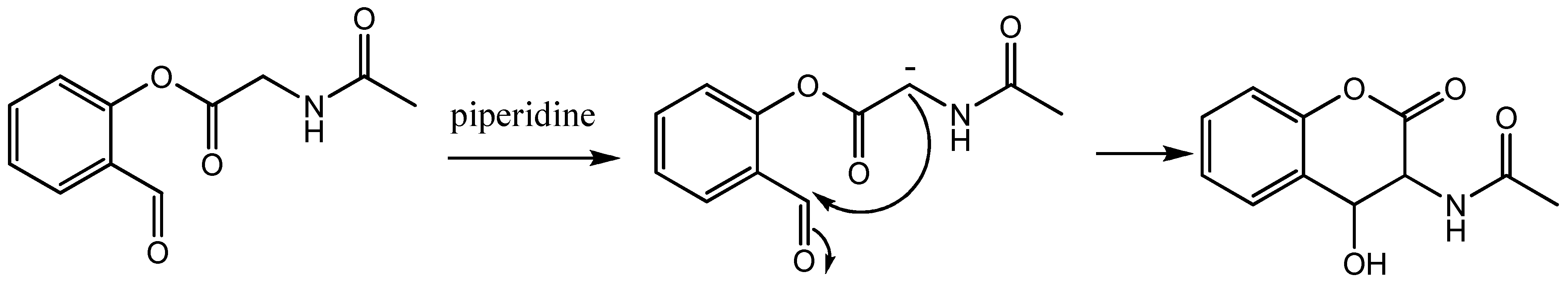
3.2. Chemistry
3.2.1. Synthesis of the Ligand
3.2.2. Synthesis of the Complexes
3.2.3. Study of Complex Formation in Solution
3.3. Pharmacology
3.3.1. Evaluation of Antibacterial Activities
3.3.2. Evaluation of Antifungal Assay
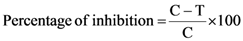
3.3.3. Evaluation of Antioxidant Activity

4. Conclusions
References
- Raman, N.; Sakthivel, A.; Rajasekaran, K. Synthesis and spectral characterization of antifungal sensitive Schiff base transition metal complexes. Mycobiology 2007, 35, 150–153. [Google Scholar] [CrossRef]
- Rice, S.A.; Givskov, M.; Steinberg, P.; Kjelleberg, S. Bacterial signals and antagonists: The interaction between bacteria and higher organisms. J. Mol. Microbiol. Biotechnol. 1999, 1, 23–31. [Google Scholar]
- Ironmonger, A.; Whittaker, B.; Andrew, J.; Baron, B.; Chris, J.; Alison, E.; Ashcroft, G.; Nelson, A. Scanning conformational space with a library of stereo- and regiochemically diverse aminoglycoside derivatives: The discovery of new ligands for RNA hairpin sequences. Org. Biomol. Chem. 2007, 5, 1081–1086. [Google Scholar] [CrossRef]
- Kumar, G.; Kumar, D.; Devi, S.; Verma, R.; Johari, R. Synthesis, spectral characterization of biologically active compounds derived from oxalyldihydrazide and 5-tert-butyl-2-hydroxy-3-(3-phenylpent-3-yl)benzaldehyde and their Cu(II), Ni(II) and Co(II) Complexes. Int. J. Eng. Sci. Technol. 2011, 3, 1630–16354. [Google Scholar]
- Parmer, S.; Kumar, R. Substituted quinazolone hydrazides as possible antituberculous agents. J. Med. Chem. 1968, 11, 635–636. [Google Scholar]
- Reddy, Y.D.; Somayajulu, V.V. Synthesis, spectra and physiological activity of 7H-pyrano[3,2-c]benzoxazole-7-ones. J. Ind. Chem. Soc. 1981, 58, 599–601. [Google Scholar]
- Soine, T.O. Naturally occuring coumarins and related physiological activities. J. Pharm. Sci. 1964, 53, 231–264. [Google Scholar] [CrossRef]
- Jund, L.; Corse, J.; King, A.D.; Bayne, H.; Mihara, K. Antimicrobial properties of 6,7-dihydroxy-, 7,8-dihydroxy-, 6-hydroxy- and 8- hydroxycoumarins. Phytochemicals 1971, 10, 2971–2974. [Google Scholar] [CrossRef]
- Abd Allah, O.A. Synthesis and biological studies of some benzopyrano[2,3-c]pyrazole derivatives. II Farmacogn. 2000, 55, 641–649. [Google Scholar] [CrossRef]
- Abdel-Al, E.H.; Al-Ashamawi, M.I.; Abd El-Fattah, B. Synthesis and antimicrobial testing of certain oxadiazoline and triazole derivatives. Die Pharm. 1983, 38, 833–838. [Google Scholar]
- Bhamaria, R.P.; Bellare, R.A.; Deliwala, C.V. In intro effect of 1-acyl-4-alkyl-(or aryl)-thiosemicarbazides 1-(5-chlorosalicylidine)-4-alkyl-(or aryl)-thiosemicarbazones and some hydrazones of 5-chlorosalicylaldehyde against pathogenic bacteria including mycobacterium tuberculosis (H37Rv). Indian J. Exp. Biol. 1968, 6, 62–63. [Google Scholar]
- Dutta, M.M.; Goswani, B.N.; Kataky, J.C.S. Studies on biologically active heterocycles. Part I. Synthesis and antifungal activity of some new aroyl hydrazones and 2,5-disubstituted-1,3,4-oxadiazoles. J. Heterocycl. Chem. 1986, 23, 793–795. [Google Scholar] [CrossRef]
- Gupta, A.K.S.; Garg, M.; Chandra, U. Synthesis of some new Mannich bases derived from substituted benzimidazole, benzoxazol-2-one, benzoxazol-2-thione, oxadiazol-2-thiones and their biological activities. J. Indian Chem. Soc. 1979, 56, 1230–1232. [Google Scholar]
- Wenner, W. Antitubercular agents. Derivatives of pyridinecarboxylic acid hydrazides. J. Org. Chem. 1953, 18, 1333–1337. [Google Scholar] [CrossRef]
- Mansour, A.K.; Eid, M.M.; Khalil, N. Synthesis and reactions of some new heterocyclic carbohydrazides and related compounds as potential anticancer agents. Molecules 2003, 8, 744–755. [Google Scholar] [CrossRef]
- Hoult, J.R.S.; Paya, M. Pharmacological and biochemical actions of simple coumarins: Natural products with therapeutic potential. Gen. Pharmacol. 1996, 27, 713–722. [Google Scholar] [CrossRef]
- Matsumoto, P.; Hanawalt, C. Histone H3 and heat shock protein GRP78 are selectively cross-linked to DNA by photoactivated gilvocarcin V in human fibroblasts. Cancer Res. 2000, 60, 3921–3926. [Google Scholar]
- Kelly, V.P.; Ellis, E.M.; Manson, M.M. Chemoprevention of aflatoxin B1 hepatocarcinogenesis by coumarin, a natural benzopyrone that is a potent inducer of aflatoxin B1-aldehyde reductase, the glutathione S-transferase A5 and P1 subunits, and NAD(P)H: Quinone oxidoreductase in rat liver. Cancer Res. 2000, 60, 957–969. [Google Scholar]
- Kirkiacharian, S.; Thuy, D.T.; Sicsic, S.; Bakhchinian, R.; Kurkjian, R.; Tonnaire, T. Structure-activity relationships of some 3-substituted-4-hydroxycoumarins as HIV-1 protease inhibitors. II Farmacogn. 2002, 57, 703–708. [Google Scholar] [CrossRef]
- Suzuki, D.; Yu, M.; Xie, L.; Morris, S.L.; Lee, K.H. Recent progress in the development of coumarin derivatives as potent anti-HIV agents. Med. Res. Rev. 2003, 23, 322–345. [Google Scholar] [CrossRef]
- Marshall, E.M.; Ryles, M.; Butler, K.; Weiss, L. Treatment of advanced renal cell carcinoma (RCC) with coumarin and cimetidine: longterm follow-up of patients treated on a phase I trial. J. Cancer Res. Clin. Oncol. 1994, 120, 535–538. [Google Scholar]
- Fylaktakidou, K.C.; Hadjipavlou-Litina, D.J.; Litinas, K.E.; Nicolaides, D.N. Natural and synthetic coumarin derivatives with anti-inflammatory/antioxidant activities. Curr. Pharm. Des. 2004, 10, 3813–3833. [Google Scholar] [CrossRef]
- Tianzhi, Y.; Yuling, Z.; Duowang, F. Synthesis, crystal structure and photoluminescence of 3-(1-benzotriazole)-4-methyl-coumarin. J. Mol. Struct. 2006, 791, 18–22. [Google Scholar] [CrossRef]
- Yu, T.Z.; Zhao, Y.L.; Ding, X.S.; Fan, D.W.; Qian, L.; Dong, W.K. Synthesis, crystal structure and photoluminescent behaviors of 3-(1H-benzotriazol-1-yl)-4-methylbenzo[7,8]coumarin. J. Photochem. Photobiol. A 2007, 188, 245–251. [Google Scholar] [CrossRef]
- Ray, D.; Bharadwaj, P.K. A coumarin-derived fluorescence probe selective for magnesium. Inorg. Chem. 2008, 7, 2252–2254. [Google Scholar]
- Trenor, S.R.; Shultz, A.R.; Love, B.J.; Long, T.E. Coumarins in polymers: From light harvesting to photo-cross-linkable tissue scaffolds. Chem. Rev. 2004, 104, 3059–3077. [Google Scholar] [CrossRef]
- Tianzhi, Y.; Peng, Z.; Yuling, Z.; Hui, Z.; Jing, M.; Duowang, F. Synthesis and photoluminescent properties of two novel tripodal compounds containing coumarin moieties. Spectrochim. Acta Part A 2009, 73, 168–173. [Google Scholar] [CrossRef]
- Ruikui, C.; Xichuan, Y.; Haining, T.; Licheng, S. Tetrahydroquinoline dyes with different spacers for organic dye-sensitized solar cells. J. Photochem. Photobiol. A 2007, 189, 295–300. [Google Scholar] [CrossRef]
- Yu, T.Z.; Zhang, P.; Zhao, Y.L.; Zhang, H.; Meng, J.; Fan, D.W.; Dong, W.K. Photoluminescence and electroluminescence of a tripodal compound containing 7-diethylamino-coumarin moiety. J. Phys. D: Appl. Phys. 2008, 41, 235406–235413. [Google Scholar]
- Tang, C.W.; VanSlyke, S.A. Organic electroluminescent diodes. Appl. Phys. Lett. 1987, 51, 913–915. [Google Scholar] [CrossRef]
- Musa, A.Y.; Khadom, A.A.; Kadhum, A.A.H.; Mohamad, A.B.; Takriff, M.S. Kinetic behavior of mild steel corrosion inhibition by 4-amino-5-phenyl-4H-1,2,4-trizole-3-thiol. J. Taiwan Inst. Chem. Eng. 2010, 41, 126–128. [Google Scholar] [CrossRef]
- Tianzhi, Y.; Peng, Z.; Yuling, Z.; Hui, Z.; Jing, M.; Duowang, F. Synthesis, characterization and high-efficiency blue electroluminescence based on coumarin derivatives of 7-diethylaminocoumarin-3-carboxamide. Org. Electron. 2009, 10, 653–660. [Google Scholar] [CrossRef]
- Asish, R.; Arunima, M.; Raghunath, S. Synthesis of biologically potent new 3-(heteroaryl)aminocoumarin derivatives via Buchwald–Hartwig C–N coupling. Tetrahedron Lett. 2010, 51, 1099–1102. [Google Scholar] [CrossRef]
- Amit, A.; Jamie, K.; Chad, C.; Natasha, D.; Graham, J. Hydrolysis-free synthesis of 3-aminocoumarins. Tetrahedron Lett. 2007, 48, 5077–5080. [Google Scholar] [CrossRef]
- Yadav, L.D.S.; Singh, S.; Rai, V.K. A one-pot [Bmim]OH-mediated synthesis of 3-benzamidocoumarins. Tetrahedron Lett. 2009, 50, 2208–2212. [Google Scholar] [CrossRef]
- Guru, P. Microbial Studies on Some Coordination Compound of Metals with Tetracycline. J. Appl. Chem. Res. 2010, 12, 7–16. [Google Scholar]
- Mishra, A.P.; Mishra, R.K.; Shrivastava, S.P. Structural and antimicrobial studies of coordination compounds of VO(II), Co(II), Ni(II) and Cu(II) with some Schiff bases involving 2-amino-4-chlorophenol. J. Serbian Chem. Soc. 2009, 74, 523–535. [Google Scholar] [CrossRef]
- Al-Amiery, A.A.; Al-Majedy, K.; Abdulhadi, S.A. Design, synthesis and bioassay of novel metal complexes of 3-amino-2-methylquinazolin-4(3H)-one. Afr. J. Pure Appl. Chem. 2009, 3, 218–227. [Google Scholar]
- Sengupta, S.K.; Pandey, O.P.; Srivastava, B.K.; Sharma, V.K. Synthesis, structural and biochemical aspects of titanocene and zirconocene chelates of acetylferrocenyl thiosemicarbazones. Transit. Met. Chem. 1998, 23, 349–353. [Google Scholar] [CrossRef]
- Rohaya, A.; Abdul Manaf, A.; Daud, A.; Israf, T.; Nor, H.; Khozirah, S.; Nordin, H. Antioxidant, radical-scavenging, anti-inflammatory, cytotoxic and antibacterial activities of methanolic extracts of some Hedyotis species. Life Sci. 2005, 76, 1953–1964. [Google Scholar] [CrossRef]
- Soares, J.R.; Dinis, T.C.P.; Cunha, A.P.; Almeida, L.M. Antioxidant activities of some extracts of thymus zygis. Free Radic. Res. 1997, 26, 469–478. [Google Scholar] [CrossRef]
- Duh, P.D.; Tu, Y.Y.; Yen, G.C. Antioxidant activity of water extract of Harng Jyur (Chyrsanthemum morifolium Ramat). Lebensm. Wiss. Technol. 1999, 32, 269–277. [Google Scholar] [CrossRef]
- Matthaus, B. Antioxidant activity of extracts obtained from residues of different oilseeds. J. Agric. Food Chem. 2002, 50, 3444–3452. [Google Scholar]
- Bukhari, S.B.; Memon, S.; Mahroof-Tahir, M.; Bhanger, M.I. Synthesis, characterization and antioxidant activity copper-quercetin complex. Spectrochim. Acta A Mol. Biomol. Spectrosc. 2009, 71, 1901–1906. [Google Scholar] [CrossRef]
- Gabrielska, J.; Soczynska-Kordala, M.; Hladyszowski, J.; Zylka, R.; Miskiewicz, J.; Przestalski, S. Antioxidative effect of quercetin and its equimolar mixture with phenyltin compounds on liposome membranes. J. Agric. Food Chem. 2006, 54, 7735–7746. [Google Scholar] [CrossRef]
- Bukhari, S.B.; Memon, S.M.; Tahir, M.M.; Bhanger, I. Synthesis, characterization and investigation of antioxidant activity of cobalt-quercetin complex. J. Mol. Struct. 2008, 892, 39–46. [Google Scholar] [CrossRef]
- Tripathy, K.P.; Mukerjee, K.A. A Facile synthesis of 3-acylaminocoumarins. Indian J. Chem. 1987, 26, 61–62. [Google Scholar]
- Al-Amiery, A.A.; Al-Majedy, K.; Abdulreazak, H.; Abood, H. Synthesis, characterization, theoretical crystal structure and antibacterial activities of some transition metal complexes of the thiosemicarbazone (Z)-2-(pyrrolidin-2-ylidene)hydrazinecarbothioamide. Bioinorg. Chem. Appl. 2011, 2011. [Google Scholar] [CrossRef]
- Al-Majedy, K.; Al-Amiery, A.A.; Almoussaoy, H.H.; Khweter, R. Novel analytical method for the determination of theophylline in pharmaceutical preparations. J. Appl. Sci. Res. 2011, 7, 470–475. [Google Scholar]
- Al-Amiery, A.A.; Mohammed, A.; Ibrahim, H.; Abbas, A. Study the biological activities of tribulus terrestris extracts. World Acad. Sci. Eng. Technol. 2009, 57, 433–435. [Google Scholar]
- Daw, Z.Y.; EL-Baroty, G.S.; Mahmoud, A.E. Inhibition of Aspergillus parasiticus growth and aflatoxin production by some essential oils. Chem. Mikrobiol. Technol. Lebensm. 1994, 16, 129–135. [Google Scholar]
- Myiut, S.; Daud, W.R.W.; Mohamed, A.B.; Kadhum, A.A.H. Gas chromatographic determination of eugenol in ethanol extract of cloves. J. Chromatogr. B: Biomed. Appl. 1996, 676, 193–195. [Google Scholar]
- Chen, Y.; Wong, M.; Rosen, R.; Ho, C. 2,2-Diphenyl-1-picrylhydrazyl radical scavenging active components from Polygonum multiflorum Thunb. J. Agric. Food Chem. 1999, 47, 2226–2228. [Google Scholar] [CrossRef]
- Sample Availability: Samples of 3-aminocoumarin and its complexes are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kadhum, A.A.H.; Mohamad, A.B.; Al-Amiery, A.A.; Takriff, M.S. Antimicrobial and Antioxidant Activities of New Metal Complexes Derived from 3-Aminocoumarin. Molecules 2011, 16, 6969-6984. https://doi.org/10.3390/molecules16086969
Kadhum AAH, Mohamad AB, Al-Amiery AA, Takriff MS. Antimicrobial and Antioxidant Activities of New Metal Complexes Derived from 3-Aminocoumarin. Molecules. 2011; 16(8):6969-6984. https://doi.org/10.3390/molecules16086969
Chicago/Turabian StyleKadhum, Abdul Amir H., Abu Bakar Mohamad, Ahmed A. Al-Amiery, and Mohd S. Takriff. 2011. "Antimicrobial and Antioxidant Activities of New Metal Complexes Derived from 3-Aminocoumarin" Molecules 16, no. 8: 6969-6984. https://doi.org/10.3390/molecules16086969
APA StyleKadhum, A. A. H., Mohamad, A. B., Al-Amiery, A. A., & Takriff, M. S. (2011). Antimicrobial and Antioxidant Activities of New Metal Complexes Derived from 3-Aminocoumarin. Molecules, 16(8), 6969-6984. https://doi.org/10.3390/molecules16086969




