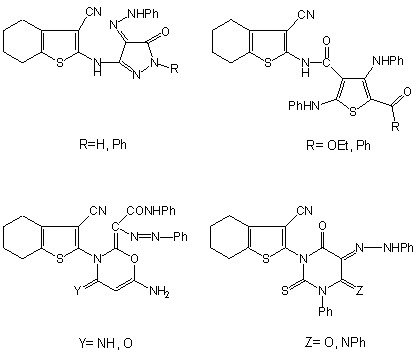
Design and Synthesis of Novel Antimicrobial Acyclic and Heterocyclic Dyes and Their Precursors for Dyeing and/or Textile Finishing Based on 2-N-Acylamino-4,5,6,7-tetrahydro-benzo[b]thiophene Systems
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemistry
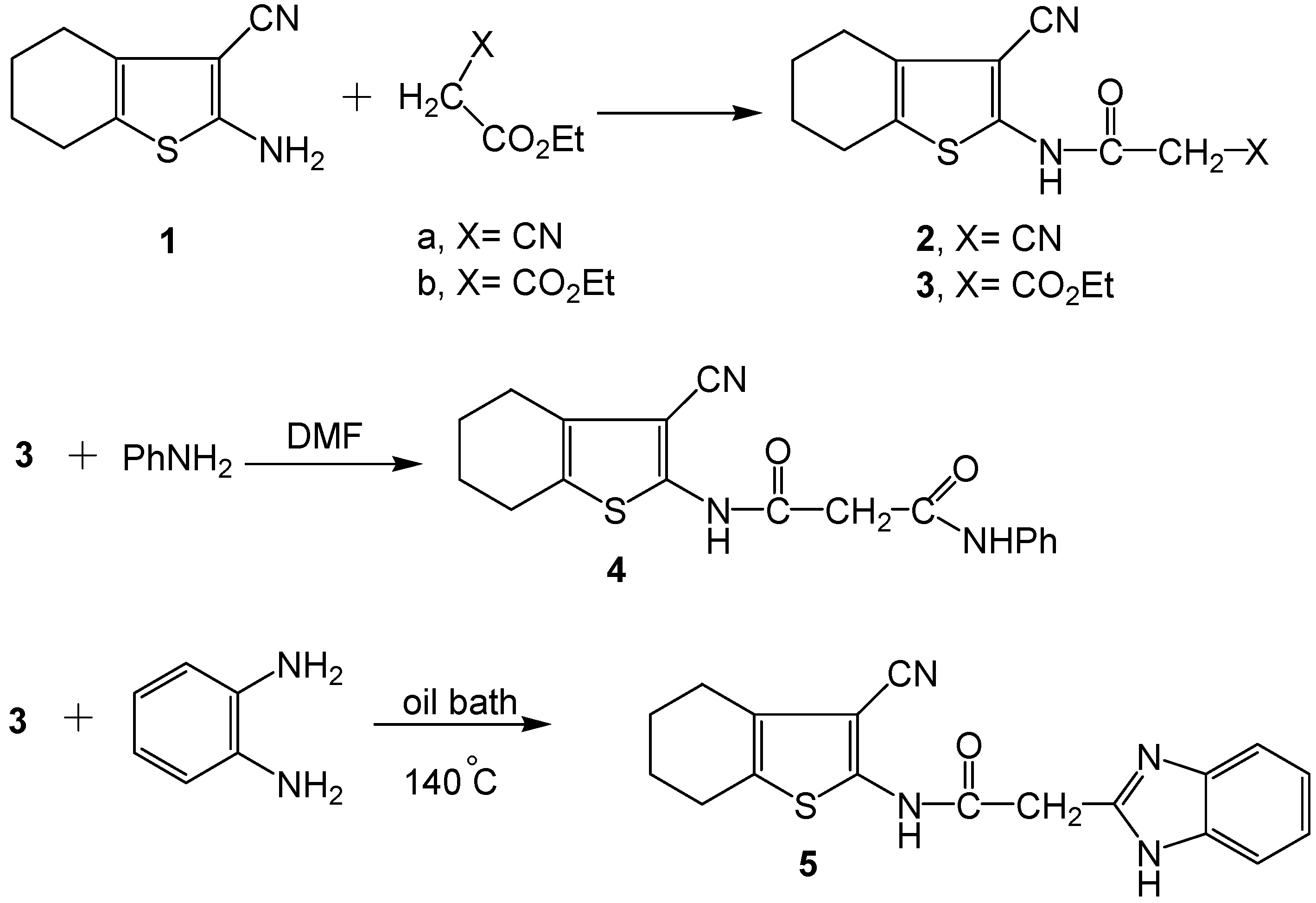

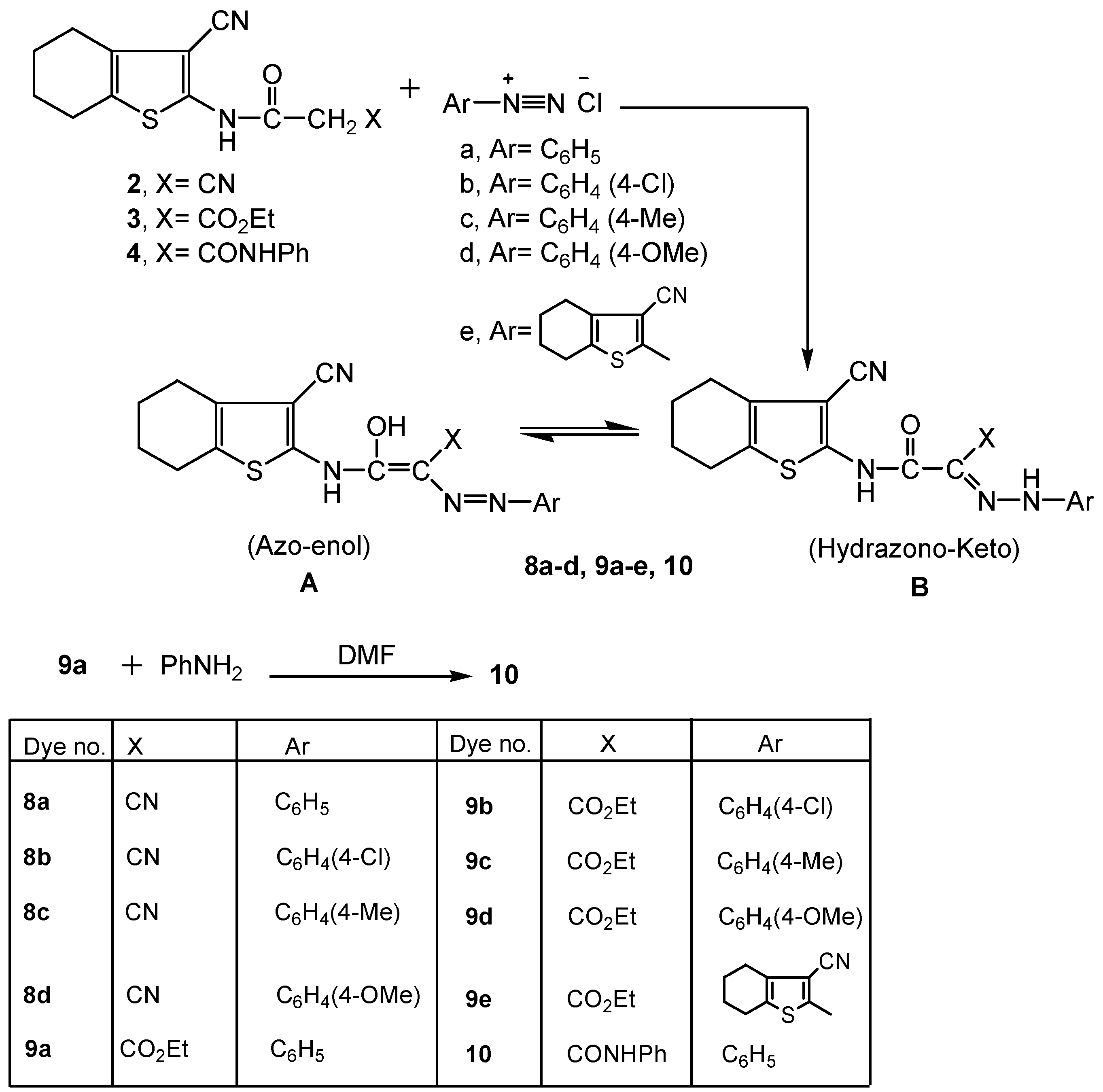
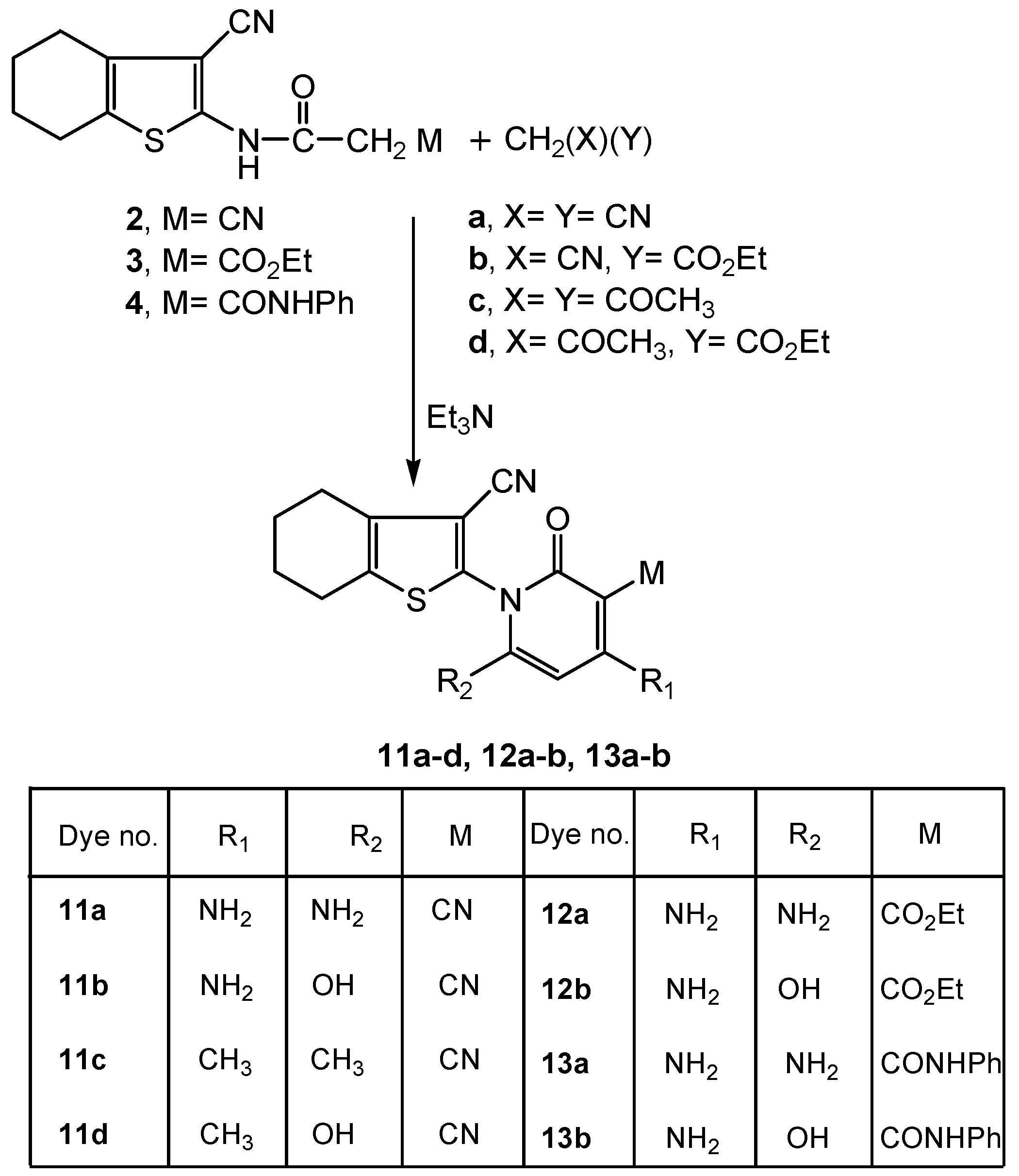
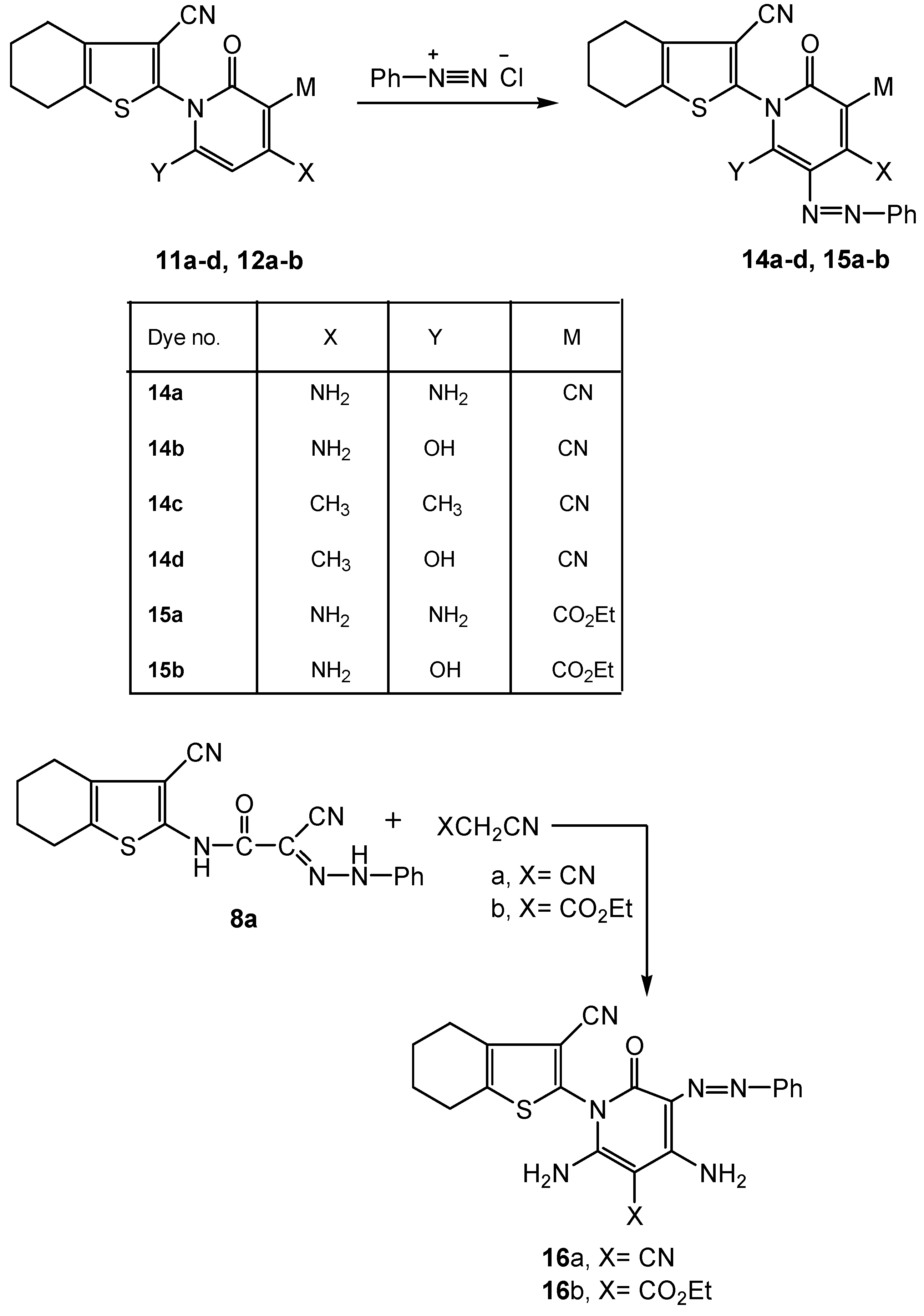
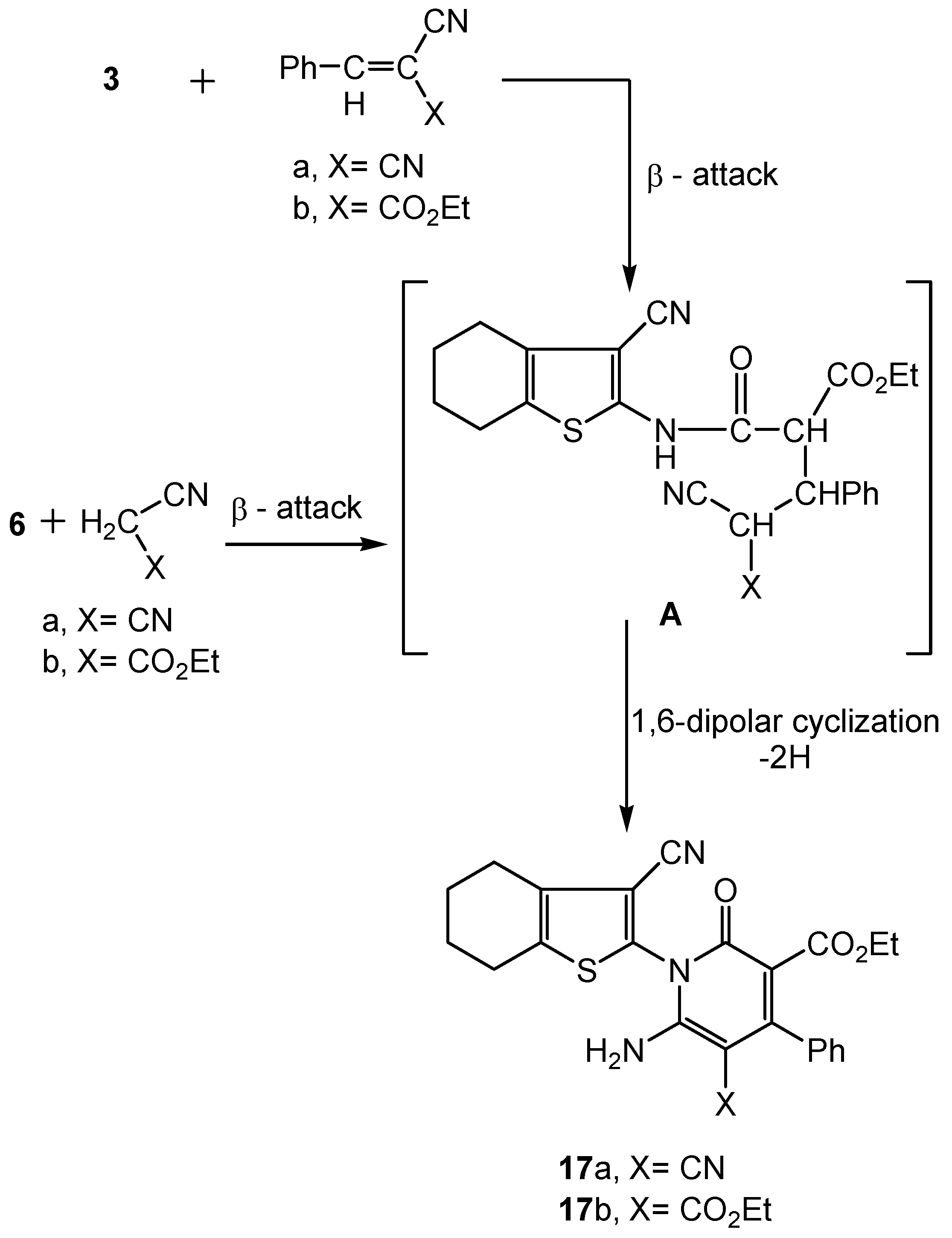
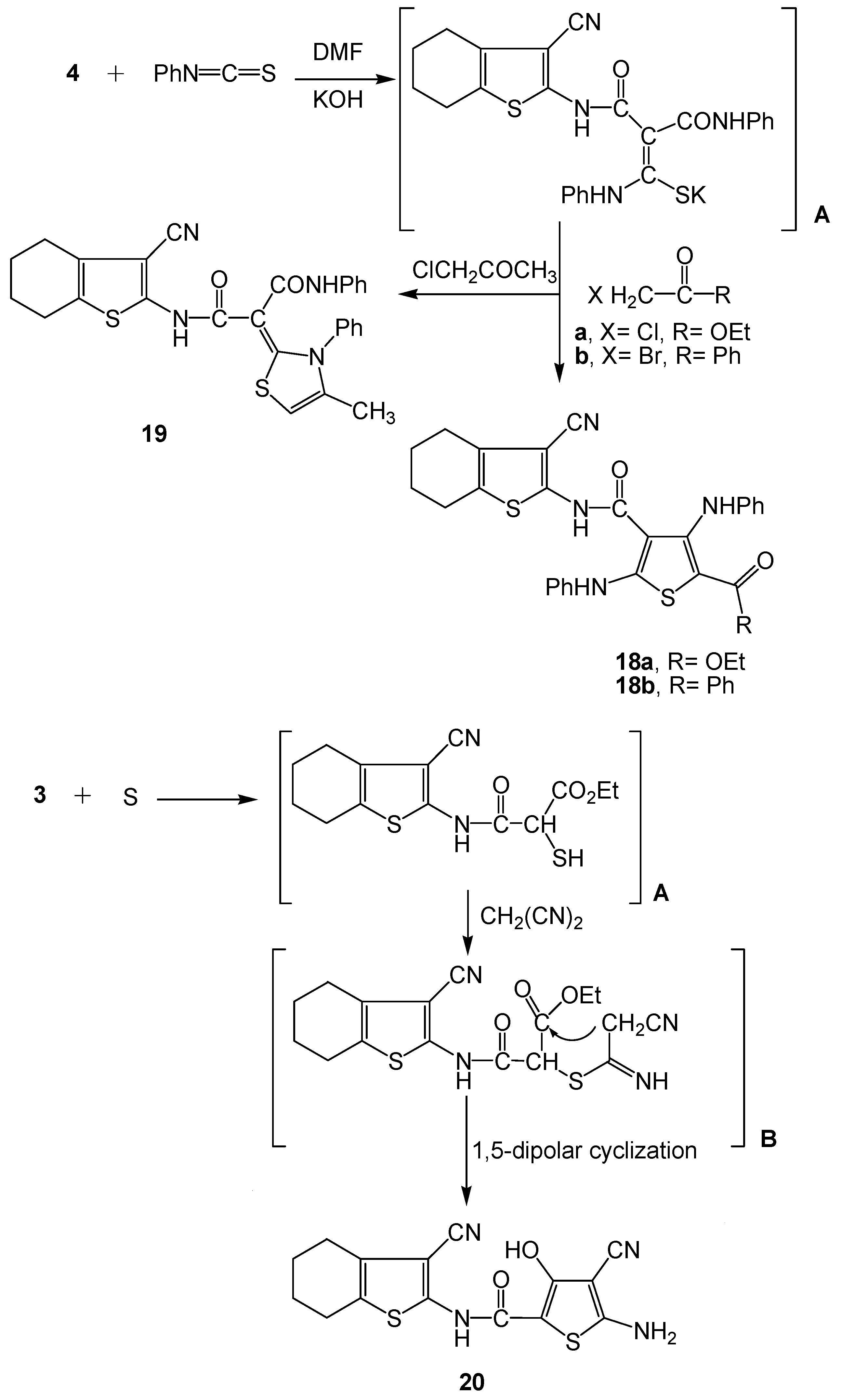
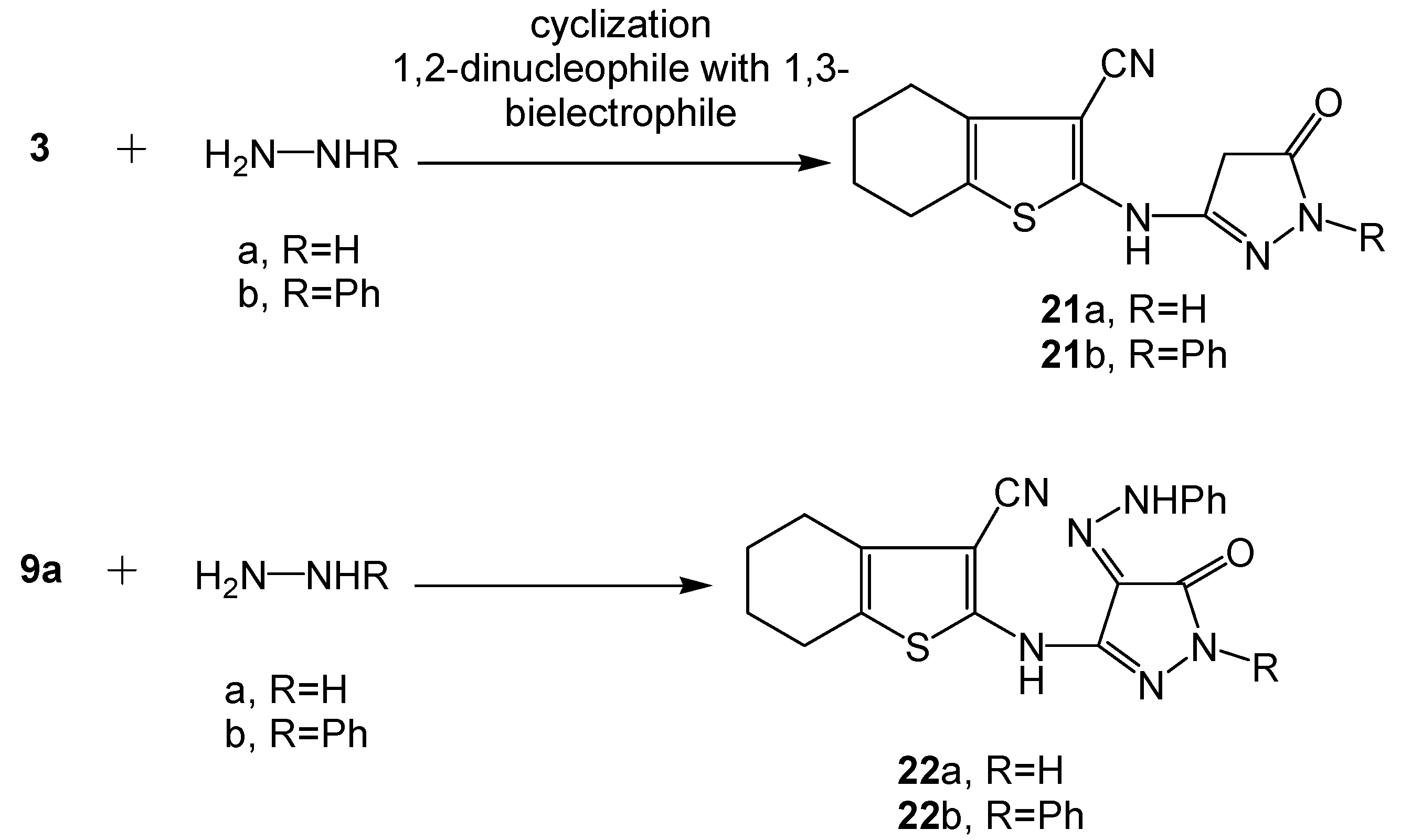
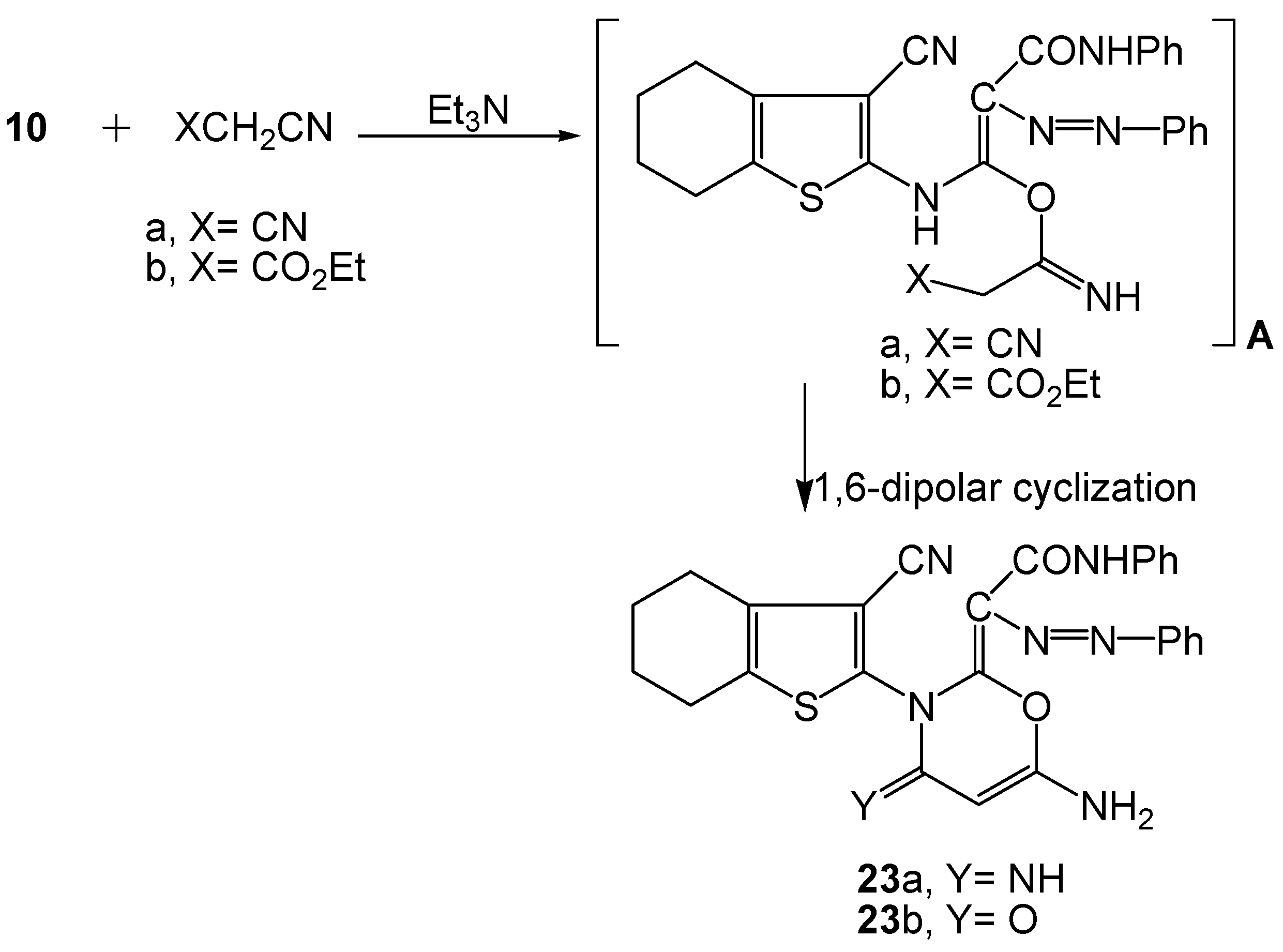
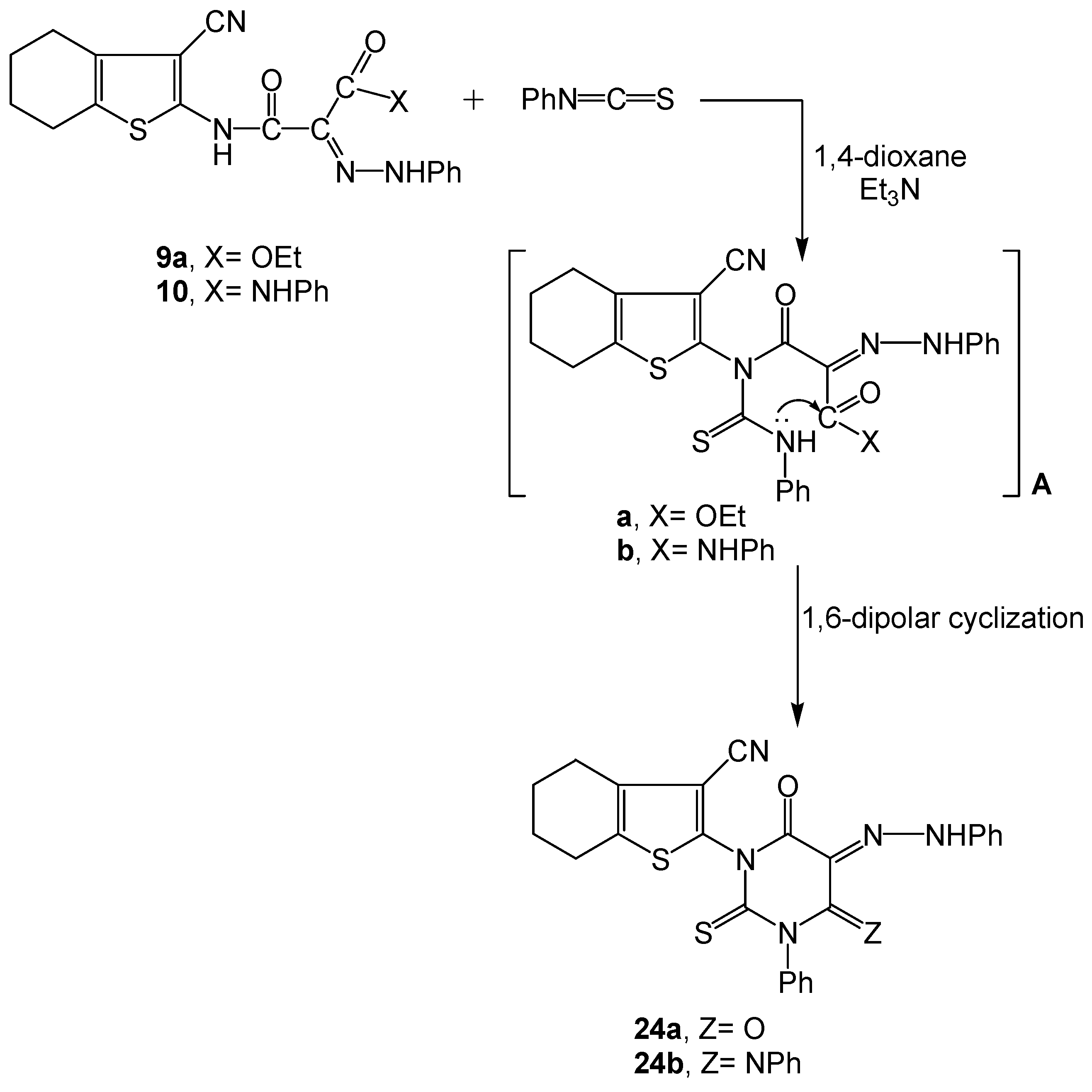
2.2. Spectral Characterization, Colour Assessment and Dyeing Properties
| Dye | K/S** | Fastness to Rubbing | Washing fastness | Fastness to Perspiration | Light | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sample | at λmax = | at 90 ºC | Acidic | Alkaline | λmax | |||||||
| Dyed * | 400 nm | Dry | Wet | Alteration | Staining | Alteration | Staining | Alteration | Staining | nm | ||
| 8a | N | 24.01 | 3 | 4 | 3-4 | 3-4 | 5 | 4-5 | 5 | 4 | 3 | 741, |
| A | 4.78 | 4 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | 392, | |
| P | 6.61 | 4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | 223 | |
| 8b | N | 21.74 | 3 | 3-4 | 3 | 3-4 | 4-5 | 4-5 | 4-5 | 4-5 | 3-4 | 740, |
| A | 7.72 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 408, | |
| P | 11.87 | 3 | 3-4 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3 | 224 | |
| 8c | N | 24.13 | 3-4 | 4 | 3 | 3-4 | 5 | 4-5 | 5 | 4 | 3 | 741, |
| A | 9.29 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | 404, | |
| P | 13.81 | 3-4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | 225 | |
| 8d | N | 20.56 | 3-4 | 3 | 3 | 4 | 5 | 5 | 5 | 5 | 2-3 | 741, |
| A | 9.12 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | 419, | |
| P | 12.79 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | 224 | |
| 9a | N | 10.33 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | 740 |
| A | 4.44 | 3-4 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 4-5 | 359 | |
| P | 7.02 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | 237 | |
| 9b | N | 13.34 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 740 |
| A | 4.59 | 3-4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | 364 | |
| P | 7.80 | 3-4 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | 247 | |
| 9c | N | 8.73 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | 647, |
| A | 3.91 | 4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 364, | |
| P | 9.08 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 242 | |
| 9d | N | 14.40 | 3 | 3-4 | 4-5 | 3 | 5 | 5 | 5 | 5 | 3 | 827 |
| A | 4.82 | 3-4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 323, | |
| P | 8.75 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 229 | |
| 9e | N | 9.96 | 3-4 | 3 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3 | 740, |
| A | 3.03 | 3 | 4 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3 | 301, | |
| P | 2.94 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 205 | |
| 10 | N | 19.84 | 3 | 3-4 | 4-555 | 4-5 | 5 | 5 | 5 | 5 | 3 | 740, |
| A | 9.40 | 3-4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 3 | 390, | ||
| P | 15.91 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 3-4 | 238 | ||
| 14a | N | 22.93 | 5 | 4-5 | 3-4 | 3-4 | 5 | 4-5 | 5 | 4-5 | 3 | 741, |
| A | 8.87 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 405, | |
| P | 15.52 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 224 | |
| 14b | N | 24.01 | 4-5 | 5 | 3 | 4 | 5 | 4-5 | 5 | 4-5 | 3 | 740, |
| A | 9.66 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 405, | |
| P | 16.08 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 232 | |
| 14c | N | 11.46 | 3 | 3-4 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 2-3 | 740 |
| A | 5.77 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | 401 | |
| P | 7.00 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 2-3 | ||
| 14d | N | 24.78 | 4-5 | 4 | 4 | 3 | 5 | 4-5 | 5 | 4 | 3 | 740, |
| A | 8.98 | 3-4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 4-5 | 3-4 | 401, | |
| P | 12.53 | 3-4 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 222 | |
| 15a | N | 17.19 | 3-4 | 4 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3 | 740, |
| A | 6.08 | 3-4 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 219 | |
| P | 11.07 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | ||
| 15b | N | 14.94 | 3 | 3-4 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3 | 740, |
| A | 6.51 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 224 | |
| P | 9.94 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | ||
| 16a | N | 25.19 | 3 | 3-4 | 4 | 4 | 5 | 4-5 | 5 | 4-5 | 3 | 741, |
| A | 9.04 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 395, | |
| P | 14.31 | 3-4 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 221 | |
| 16b | N | 23.28 | 3-4 | 4 | 3-4 | 3-4 | 4-5 | 4-5 | 4-5 | 4-5 | 3 | 741, |
| A | 7.83 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 4-5 | 3-4 | 400, | |
| P | 11.40 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 223 | |
| 22a | N | 16.62 | 3 | 3-4 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3-4 | 741, |
| A | 7.06 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 4-5 | 370, | |
| P | 11.07 | 3 | 3 | 5 | 5 | 5 | 5 | 5 | 5 | 4-5 | 224 | |
| 22b | N | 18.32 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 741, |
| A | 8.32 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 4-5 | 364, | |
| P | 13.02 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 4-5 | 224 | |
| 23a | N | 15.52 | 4 | 5 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3 | 741, |
| A | 6.87 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 5 | 3 | 235 | |
| P | 9.71 | 4 | 4-5 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | ||
| 23b | N | 19.93 | 3 | 3-4 | 4-5 | 4-5 | 5 | 5 | 5 | 5 | 3 | 740, |
| A | 8.94 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 235 | |
| P | 14.45 | 3 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | ||
| 24a | N | 5.56 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 741, |
| A | 4.22 | 3-4 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 4-5 | 230 | |
| P | 4.43 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 4-5 | ||
| 24b | N | 18.24 | 3-4 | 3-4 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | 740, |
| A | 8.22 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 3-4 | 223 | |
| P | 12.98 | 3 | 4 | 5 | 5 | 5 | 5 | 5 | 5 | 4 | ||
2.2.1. Spectral Characterization
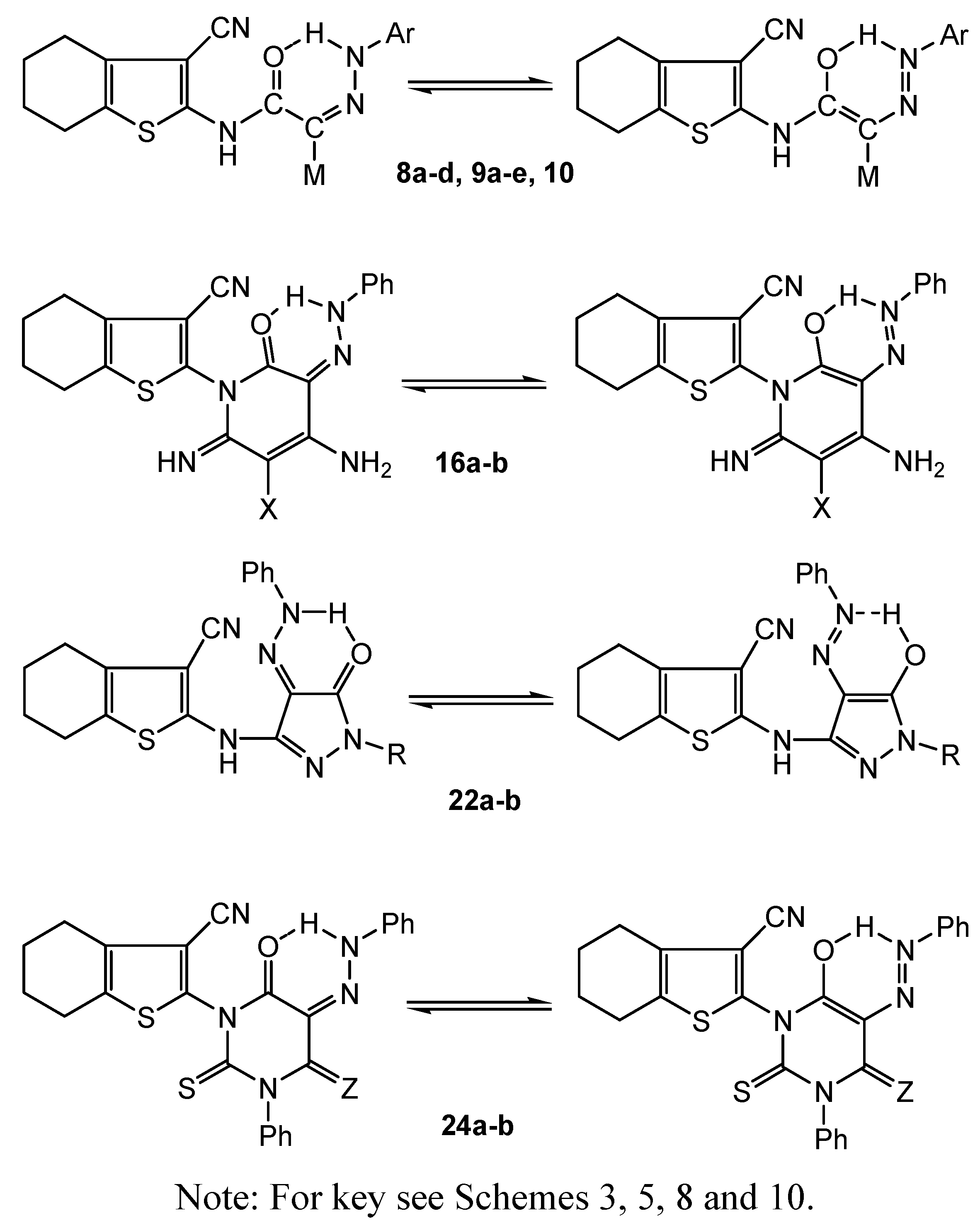
2.2.2. Colour Assessment and Dyeing Properties
2.3. Biology
2.3.1. Antimicrobial Evaluation of the Newly Synthesized Compounds
| Compound | E. coli | B. cereus | B. subtilis | C. albicans |
|---|---|---|---|---|
| 3 | NA | 4.85 | 12.00 | 10.00 |
| 4 | 18.51 | 22.16 | 20.18 | 10.00 |
| 5 | 6.60 | 17.81 | 6.80 | 8.00 |
| 6 | NA | 14.62 | 4.18 | 0.60 |
| 7 | 6.58 | 8.48 | 4.46 | 12.38 |
| 8a | NA | 4.22 | 10.89 | 12.42 |
| 8b | NA | 4.39 | 4.33 | 12.77 |
| 8c | NA | 0.08 | 2.22 | 6.44 |
| 8d | NA | 3.03 | 0.68 | 20.50 |
| 9a | NA | 16.32 | 6.22 | 0.40 |
| 9b | NA | 12.30 | 4.22 | 12.55 |
| 9c | NA | 6.03 | 12.42 | 4.55 |
| 9d | NA | 12.34 | 6.13 | 0.40 |
| 9e | NA | 2.25 | 14.00 | 16.00 |
| 10 | NA | 7.39 | 4.33 | 12.77 |
| 12a | 16.77 | 6.28 | 22.48 | 4.66 |
| 12b | 4.55 | 10.33 | 2.26 | 10.58 |
| 13a | 12.57 | 8.00 | 4.62 | 33.00 |
| 13b | NA | 4.76 | 8.66 | 8.22 |
| 14a | 16.60 | 12.42 | 4.01 | 0.20 |
| 14b | 0.36 | 1.16 | 9.67 | 0.04 |
| 14c | 10.50 | 4.77 | 8.80 | 30.55 |
| 14d | 4.72 | 19.22 | 6.80 | 8.06 |
| 15a | 10.21 | 2.36 | 0.22 | 0.20 |
| 15b | NA | 12.30 | 4.22 | 12.55 |
| 16a | NA | 1.25 | 8.23 | 2.24 |
| 16b | NA | 0.08 | 2.22 | 6.44 |
| 17a | NA | 2.19 | 6.44 | 4.48 |
| 17b | 10.55 | 6.88 | 5.38 | 8.79 |
| 18a | 6.44 | 8.46 | 6.42 | 0.61 |
| 18b | 8.99 | 12.68 | 6.77 | 2.87 |
| 19 | 6.48 | 8.18 | 22.30 | 18.42 |
| 20 | 10.33 | 4.14 | 10.33 | 6.08 |
| 21a | NA | 12.34 | 6.13 | 0.40 |
| 21b | 8.22 | 6.03 | 12.42 | 4.55 |
| 22a | 8.48 | 6.08 | 18.30 | 16.48 |
| 22b | NA | 6.18 | 4.03 | 0.81 |
| 23a | NA | 6.88 | 5.38 | 6.99 |
| 23b | 6.98 | 6.68 | 4.36 | 8.26 |
| 24a | 19.04 | 8.36 | 10.60 | 18.26 |
| 24b | 12.41 | 8.52 | 2.01 | 0.80 |
| Ampicillin | 6.25 | 3.13 | 12.50 | – |
| Cycloheximide | – | – | – | 12.50 |
2.3.2. Antimicrobial Evaluation of the Dyed Fabrics
| Dye | Sample Dyed * | E. coli | B. cereus | B. subtilis | C. albicans | P. aeruginosa | S. aureus |
|---|---|---|---|---|---|---|---|
| 8a | N | 20 | 18 | 17 | 27 | 17 | 15 |
| 8b | A | 12 | 16 | 18 | 22 | 12 | 16 |
| 8c | P | 15 | 12 | 16 | 23 | 19 | 18 |
| 8d | A | 12 | 10 | 13 | 22 | 14 | 14 |
| 9a | A | 15 | 11 | 15 | 22 | 18 | 15 |
| 9b | P | 10 | 12 | 11 | 20 | 12 | 16 |
| 9c | P | 18 | 11 | 10 | 28 | 14 | 18 |
| 9d | N | 20 | 14 | 18 | 22 | 18 | 12 |
| 9e | N | 18 | 20 | 18 | 28 | 20 | 15 |
| 10 | P | 16 | 15 | 16 | 25 | 16 | 20 |
| 14a | A | 16 | 11 | 10 | 18 | 14 | 15 |
| 14b | P | 14 | 13 | 15 | 19 | 14 | 17 |
| 14c | N | 14 | 12 | 14 | 19 | 14 | 17 |
| 14d | N | 10 | 10 | 17 | 28 | 14 | 14 |
| 15a | P | 14 | 12 | 14 | 19 | 13 | 16 |
| 15b | A | 18 | 14 | 18 | 16 | 20 | 22 |
| 16a | N | 19 | 19 | 19 | 25 | 20 | 20 |
| 16b | A | 20 | 20 | 18 | 20 | 19 | 19 |
| 22a | N | 15 | 18 | 16 | 24 | 14 | 18 |
| 22b | A | 15 | 14 | 15 | 24 | 16 | 19 |
| 23a | P | 15 | 13 | 15 | 24 | 15 | 15 |
| 23b | A | 18 | 18 | 18 | 20 | 19 | 15 |
| 24a | P | 19 | 19 | 19 | 30 | 20 | 20 |
| 24b | N | 14 | 14 | 12 | 20 | 16 | 16 |
3. Experimental
3.1. General
3.1.1. Equipment
3.1.2. Materials and Chemicals
3.2. Chemistry
3.2.1. Reaction of 4,5,6,7-Tetrahydrobenzo[b] Thiophene Derivatives 3 and 4 with Benzaldehyde: Synthesis of 6 and 7
3.2.2. Reaction of 4,5,6,7-Tetrahydrobenzo[b]thiophene Derivative 2 with Aryl Diazonium Chloride Salts: Synthesis of 8a-d
3.2.3. Synthesis of N-(3-cyano-4,5,6,7-tetrahydrobenzo[b]thiophen-2-yl)-malonamic Acid Ethyl Ester Azo Derivatives 9a-d
3.2.4. Synthesis of Functionalized 4,5,6,7-Tetrahydrobenzo[b]thiophen-2-yl-3-cyano-, 3-Carboxylic Acid Ethyl Esters or 3-Carboxylic Acid Phenyl Amides 11a-d, 12a-b or 13a-b
3.2.4.1. Synthesis of 12a-b, 13a-b
3.2.5. Synthesis of Functionalized 4,5,6,7-Tetrahydrobenzo[b]thiophen-2-yl-5-phenylazo Pyridine Derivatives 14a-d and 15a-b
3.2.6. Synthesis of Functionalized 4,5,6,7-Tetrahydrobenzo[b]thiophen-2-yl-2-phenyl Azo Pyridine Derivatives 16a-b
3.2.7. Synthesis of Functionalized 4,5,6,7-Tetrahydrobenzo[b]thiophen-2-yl-2-oxopyridine Derivatives 17a-b
3.2.8. Synthesis of Functionalized 4,5,6,7-Tetrahydrobenzo[b]thiophen-2-yl-thiophene Derivatives 18a-b and Thiazole Derivative 19
3.2.9. Synthesis of 5-Amino-4-cyano-3-hydroxy-thiophene-2-carboxylic acid (3-cyano-4,5,6,7-tetra-hydrobenzo[b]thiophen-2-yl)-amide (20)
3.2.10. Synthesis of Functionalized 5-Oxopyrazole Derivatives 21a-b and the Respective Hydrazono Dyes 22a-b
3.2.11. Synthesis of Functionalized 4,5,6,7-Tetrahydrobenzo[b]thiophen-2-yl-oxazin-ylidine Azo Dyes 23a-b
3.2.12. Synthesis of Functionalized Pyrimidine Phenyl Hydrazone Dyes 24a-b
3.3. Spectral Characterization, Colour Assessment and Dyeing Properties
3.3.1. Dyeing Procedure
3.3.2. Colour Strength
3.3.3. Fastness Properties
3.3.3.1. Colour Fastness to Washing
3.3.3.2. Colour Fastness to Rubbing
3.3.3.3. Colour Fastness to Perspiration
3.3.3.4. Colour Fastness to Light
3.4. Biology
3.4.1. Antimicrobial Activity of the Synthesized Dyed and Dye Intermediate
3.4.2. Antimicrobial Activity of the Dyed Fabrics
3.4.2.1. Media Used
3.4.2.2. Antimicrobial Activity Test
4. Conclusions
Acknowledgments
- Sample Availability: Samples of the compounds 3-24b are available from the authors.
References and Notes
- Basaif, S.A.; Hassan, M.A.; Gobouri, A.A. AlCl3-catalyzed diazocoupling of 1-(aryl/het-aryl)-3-phenyl-1H-pyrazol-2-in-5-ones in aqueous medium. Synthesis of hetaryl-azopyrazol-ones and their application as disperse dyes. Dyes Pigm. 2007, 72, 387–391. [Google Scholar] [CrossRef]
- Ho, Y.W. Synthesis of some new azo pyrazolo[1,5-a]pyrimidine-thieno[2,3-b]pyridine derivatives and their application as disperse dyes. Dyes Pigm. 2005, 64, 223–230. [Google Scholar] [CrossRef]
- Rădulescu, C.; Hossu, A.M.; Ioniţă, I. Disperse dyes derivatives from compact condensed system 2-aminothiazolo[5,4-c]pyridine: Synthesis and characterization. Dyes Pigm. 2006, 71, 123–129. [Google Scholar] [CrossRef]
- Rădulescu, C.; Hossu, A.M.; Ioniţă, I.; Moater, E.I. Synthesis and characterization of new cationic dyes for synthetic fibres. Dyes Pigm. 2008, 76, 366–371. [Google Scholar] [CrossRef]
- Shams, H.Z.; Helal, M.H.; Mohamed, F.A.; Abd-Elhafiz, S.A. A novel synthesis of 1-oxathieno[3/, 2/-3,4]pyrazolo[1,2-a] pyrazole azo dye systems for dyeing of synthetic and modified cellulose fibers. Pigm. Resin Technol. 2001, 30, 158–167. [Google Scholar]
- Shams, H.Z.; Helal, M.H.; Mohamed, F.A. A novel synthesis of polyfunctionally substituted pyrazolo [1,2-a] pyrazole, and thieno [3′,2′-3,4] pyrazolo [1,2-a] pyrazole systems. Phosphorous Sulfur Silicon 2001, 174, 255–267. [Google Scholar] [CrossRef]
- Shams, H.Z.; Youssef, Y.A.; Mohamed, F.A.; El-Zawahry, M.M.; Helal, M.H.; El-kharadly, E.A. Synthesis and application of novel disazo pyrazolo[1,2-a]pyrazole reactive dyes. Pigm. Resin Technol. 2009, 38, 372–379. [Google Scholar] [CrossRef]
- Shams, H.Z.; Helal, M.H.; Samir, I.M.; Mohareb, R.M. Synthesis of new disperse azodyes based on oxopyridine core. Pigm. Resin Technol. 2008, 37, 299–307. [Google Scholar] [CrossRef]
- Isak, S.J.; Eyring, E.M.; Spikes, J.D.; Meekins, P.A. Direct blue dye solutions: Photo properties. J. Photochem. Photobiol. A 2000, 134, 77–85. [Google Scholar] [CrossRef]
- Dinçalp, H.; Toker, F.; Durucasu, İ.; Avcıbaşı, N.; Ιcli, S. New thiophene-based azo ligands containing azo methine group in the main chain for the determination of copper(II) ions. Dyes Pigm. 2007, 75, 11–24. [Google Scholar] [CrossRef]
- Li, X.; Wu, Y.; Gu, D.; Gan, F. Spectral, thermal and optical properties of metal(II)-azo complexes for optical recording media. Dyes Pigm. 2010, 86, 182–189. [Google Scholar] [CrossRef]
- Shindy, H.A.; El-Maghraby, M.A.; Eissa, F.M. Synthesis, photosensitization and antimicrobial activity of certain oxadiazine cyanine dyes. Dyes Pigm. 2006, 70, 110–116. [Google Scholar] [CrossRef]
- Ho, M.S.; Barrett, C.; Paterson, J.; Esteghamatian, M.; Natansohn, A.; Rochon, P. Synthesis and optical properties of poly{(4-nitrophenyl)-[3-[N-[2-(methacryloyloxy)ethyl]-carbazolyl]]-diazene}. Macromolecules 1996, 29, 4613–4618. [Google Scholar] [CrossRef]
- Boogers, J.A.F.; Klaase, P.T.A.; de Vlieger, J.J.; Alkema, D.P.W.; Tinnemans, A.H.A. Cross-linked polymer materials for nonlinear optics. 1. UV-cured acrylic monomers bearing azobenzene dyes. Macromolecules 1994, 27, 197–204. [Google Scholar] [CrossRef]
- Yin, S.; Xu, H.; Shi, W.; Geo, Y.; Song, Y.; Lam, J.W.Y.; Tang, B.Z. Synthesis and optical properties of polyacetylenes containing nonlinear optical chromophores. Polymer 2005, 46, 7670–7677. [Google Scholar]
- Katz, H.E.; Singer, K.D.; Sohn, J.E.; Dirk, C.W.; King, L.A.; Gordon, H.M. Greatly enhanced second-order nonlinear optical susceptibilities in donor-acceptor organic molecules. J. Am. Chem. Soc. 1987, 109, 6561–6563. [Google Scholar]
- Freeman, H.S.; Sokolwska, J.; Jolanta, S. Developments in dyestuff chemistry. Rev. Prog. Color 1999, 29, 8–22. [Google Scholar]
- Chao, Y.C.; Yeh, C.W. The influence of long-chain alkyl groups on the colour, dyeing and fastness properties of 2-(and 1-)phenylazo-1-(and 2-) naphthol acid dyes. Dyes Pigm. 1993, 23, 285–296. [Google Scholar] [CrossRef]
- Chao, Y.C.; Chang, M.J.; Chang, C.H. Water-repellent acid dyes: The influence of the perfluorobutamido group on the colour, dyeing and fastness properties of 2-(p-alkyl) phenylazo-1-naphthol acid dyes. Dyes Pigm. 1998, 39, 183–191. [Google Scholar] [CrossRef]
- Rajagopal, R.; Seshadri, S. Light-stabilised azo dyes containing a built-in ultraviolet absorber residue. Dyes Pigm. 1988, 9, 233–241. [Google Scholar] [CrossRef]
- Freeman, H.S.; Posey, J.C., Jr. An approach to the design of lightfast disperse dyes-analogs of disperse yellow 42. Dyes Pigm. 1992, 20, 171–195. [Google Scholar] [CrossRef]
- Freeman, H.S.; Mason, M.E.; Lye, J. Disperse dyes containing a built-in oxalanilide stabilizer. Dyes Pigm. 1999, 42, 53–63. [Google Scholar] [CrossRef]
- Singh, R.; Jain, A.; Panwar, S.; Gupta, D.; Khare, S.K. Antimicrobial activity of some natural dyes. Dyes Pigm. 2005, 66, 99–102. [Google Scholar] [CrossRef]
- Dev, V.R.G.; Venugopal, J.; Sudha, S.; Deepika, G.; Ramakrishna, S. Dyeing and antimicrobial characteristics of chitosan treated wool fabrics. Carbohydr. Polym. 2009, 75, 646–650. [Google Scholar] [CrossRef]
- Dong, Y.; Wang, J.; Liu, P. Dyeing and finishing of cotton fabric in a single bath with reactive dyes and citric acid. Coloration Technol. 2001, 117, 262–265. [Google Scholar] [CrossRef]
- Raheel, M.; Guo, C. Single-Step dyeing and formaldehyde-free durable press finishing of cotton fabric. Text. Res. J. 1998, 68, 571–577. [Google Scholar] [CrossRef]
- Choi, H.M.; Srinivasan, M.; Morris, N.M. Single-step dyeing and finishing treatment of cotton with 1,2,3,4-butanetetracarboxylic acid. J. Appl. Polym. Sci. 1994, 54, 2107–2118. [Google Scholar]
- Sharma, P.; Gahlot, M.A. Comparative study of simultaneous dyeing-finishing and two-step dyeing finishing on silk fabric. Man-Made Text. India 2002, 45, 257–259. [Google Scholar]
- Shyu, J.P.; Chen, C.C. Dyeing N-methylol group containing acid dyes using the pad/dry cure process. Am. Dyestuff. Rep. 1992, 81, 60–62. [Google Scholar]
- Nasr, H.I.; Abdel-Fattah, S.H.; El Kharadly, E.A. Simultaneous dye and finish fixation on a radiation-induced N-methylolacrylamide-treated wool fabrics. Radiat. Phys. Chem. 1980, 16, 491–492. [Google Scholar]
- Ma, M.; Sun, G. Antimicrobial cationic dyes. Part 3: Simultaneous dyeing and antimicrobial finishing of acrylic fabrics. Dyes Pigm. 2005, 66, 33–41. [Google Scholar] [CrossRef]
- Sun, Y.; Sun, G. Durable and regenerable antimicrobial textile materials prepared by a continuous grafting process. J. Appl. Polym. Sci. 2002, 84, 1592–1599. [Google Scholar] [CrossRef]
- Kim, Y.H.; Sun, G. Functional finishing of acrylic and cationic dyeable fabrics: Intermolecular interactions. Text. Res. J. 2002, 72, 1052–1056. [Google Scholar] [CrossRef]
- Alnajjar, A.-A.; Abdelkhalik, M.M.; Al-Enezi, A.; Elnagdi, M.H. Enaminones as building blocks in heterocyclic synthesis: Reinvestigating the product structures of enaminones with malononitrile. A novel route to 6-substituted-3-oxo-2,3-dihydropyridazine-4-carboxylic acids. Molecules 2009, 14, 68–77. [Google Scholar]
- Al-Mousawi, S.M.; El-Apasery, M.A.; Elnagdi, M.H. Enaminones in heterocyclic synthesis: A novel route to tetrahydropyrimidines, dihydropyridines, triacylbenzenes and naphthofurans under microwave irradiation. Molecules 2010, 15, 58–67. [Google Scholar]
- Šimůnek, P.; Macháček, V. The structure and tautomerism of azo coupled β-Enaminones. Dyes Pigm. 2010, 86, 197–205. [Google Scholar] [CrossRef]
- Elassar, A.-Z.A.; El-Khair, A.A. Recent developments in the chemistry of enaminones. Tetrahedron 2003, 59, 8463–8480. [Google Scholar] [CrossRef]
- Elassar, A.-Z.A.; Dib, H.H.; Al-Awadi, N.A.; Elnagdi, M.H. Chemistry of carbofunctionally substituted hydrazones. ARKIVOC 2007, ii, 272–315. [Google Scholar]
- Shams, H.Z.; Mohareb, R.M.; Elkholy, Y.M.; Azam, R.A. Synthetic potentialities of thiophene systems in heterocyclic synthesis: A novel synthesis of thieno [2,3-b] pyridine derivatives. Phosphorus. Sulfur Silicon 1999, 155, 215–233. [Google Scholar] [CrossRef]
- Wardakhan, W.W.; Shams., H.Z.; Moustafa, H.E. Synthesis of polyfunctionally substituted thiophene, thieno[2,3-b]pyrimidine and thieno[2,3-d] pyrimidine derivatives. Phosphorus Sulfur Silicon 2005, 180, 1815–1827. [Google Scholar] [CrossRef]
- Shams, H.Z.; Mohareb, R.M.; Helal, M.H.; Mahmoud, A.E. Synthesis, structure elucidation, and biological evaluation of some fused and/or pendant thiophene, pyrazole, imidazole, thiazole, triazole, triazine, and coumarin systems based on cyanoacetic 2-[(benzoyl-amino)thioxomethyl]-hydrazide. Phosporus Sulfur Silicon 2007, 182, 237–263. [Google Scholar] [CrossRef]
- Shams, H.Z.; Mohareb, R.M.; Helal, M.H.; Mahmoud, A.E. Novel synthesis and antitumor evaluation of polyfunctionally substituted heterocyclic compounds derived from 2-cyano-N-(3-cyano-4,5,6,7-tetrahydrobenzo[b]thiophen-2-yl)-acetamide. Molecules 2011, 16, 52–73. [Google Scholar]
- Gewald, K.; Schindler, R. Cyclisierungen mit cyanthioacetamid in gegenwart von schwefel. J. Prakt. Chem. 1990, 332, 223–228. [Google Scholar] [CrossRef]
- Mohareb, R.M.; Aziz, S.I.; Sayed, N.I.A.; Shams, H.Z. Reactions of benzoyl isothiocyanate with active methylene reagents: A novel synthesis of thiophene, thiazoline and thieno[2,3-d]pyrimidine derivatives. J. Chin. Chem. Soc. 1992, 39, 181–187. [Google Scholar]
- Hawkey, P.M.; Lewis, D.A. Medical Bacteriology − A Practical Approach; Oxford University Press: London, UK, 1994; pp. 181–194. [Google Scholar]
- Rameshkumar, N.; Ashokkumar, M.; Subramanian, E.H.; Ilavarasan, R.; Sridhar, S.K. Synthesis of 6-fluoro-1,4-dihydro-4-oxo-quinoline-3-carboxylic acid derivatives as potential antimicrobial agents. Eur. J. Med. Chem. 2003, 38, 1001–1004. [Google Scholar] [CrossRef]
- Liu, S.; Ma, J.; Zhao, D. Synthesis and characterization of cationic monoazo dyes incorporating quaternary ammonium salts. Dyes Pigm. 2007, 75, 255–262. [Google Scholar] [CrossRef]
- Trotman, E.R. Dyeing and Chemical Technology of Textile Fibers, 6th ed; John Wiley & Sons Inc: London, UK/ Melbourne, Australia/ Auckland, NZ, 1984; pp. 306–309. [Google Scholar]
- Society of Dyer and Colourists, Standard Methods for the Determination of the Colour Fastness of Textiles and Leather, 4th ed; The England Society: Bradford, England, UK, 1978.
- Gewald, K. Synthesis of 4-amino-thiono[2,3-b]pyridine. Monatshefte Chem. 1979, 110, 1189–1196. [Google Scholar] [CrossRef]
- Mohareb, R.M.; El-Omran, F.A.; Ho, J.Z. The reaction of 2-amino-3-cyano-4,5,6,7-tetrahydrobenzo[b]thiophene with diethyl malonate: Synthesis of coumarin, pyridine, and thiazole derivatives. Heteroatom. Chem. 2001, 12, 168–175. [Google Scholar] [CrossRef]
- Ericsson, H.M.; Sherris, J.C. Antibiotic sensitivity testing: Report of an international collaborative study. Acta Path. Microbiol. Scand. 1971, Suppl. 217. Sec. B.. [Google Scholar]
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Shams, H.Z.; Mohareb, R.M.; Helal, M.H.; Mahmoud, A.E.-S. Design and Synthesis of Novel Antimicrobial Acyclic and Heterocyclic Dyes and Their Precursors for Dyeing and/or Textile Finishing Based on 2-N-Acylamino-4,5,6,7-tetrahydro-benzo[b]thiophene Systems. Molecules 2011, 16, 6271-6305. https://doi.org/10.3390/molecules16086271
Shams HZ, Mohareb RM, Helal MH, Mahmoud AE-S. Design and Synthesis of Novel Antimicrobial Acyclic and Heterocyclic Dyes and Their Precursors for Dyeing and/or Textile Finishing Based on 2-N-Acylamino-4,5,6,7-tetrahydro-benzo[b]thiophene Systems. Molecules. 2011; 16(8):6271-6305. https://doi.org/10.3390/molecules16086271
Chicago/Turabian StyleShams, Hoda Zaki, Rafat Milad Mohareb, Maher Helmy Helal, and Amira El-Sayed Mahmoud. 2011. "Design and Synthesis of Novel Antimicrobial Acyclic and Heterocyclic Dyes and Their Precursors for Dyeing and/or Textile Finishing Based on 2-N-Acylamino-4,5,6,7-tetrahydro-benzo[b]thiophene Systems" Molecules 16, no. 8: 6271-6305. https://doi.org/10.3390/molecules16086271
APA StyleShams, H. Z., Mohareb, R. M., Helal, M. H., & Mahmoud, A. E.-S. (2011). Design and Synthesis of Novel Antimicrobial Acyclic and Heterocyclic Dyes and Their Precursors for Dyeing and/or Textile Finishing Based on 2-N-Acylamino-4,5,6,7-tetrahydro-benzo[b]thiophene Systems. Molecules, 16(8), 6271-6305. https://doi.org/10.3390/molecules16086271