General
Melting points are reported uncorrected and were determined on a Gallenkamp apparatus. The Infrared spectra were recorded on a Jasco FT/IR-6300 FT-IR using KBr disks.
1H-NMR and
13C-NMR spectra were measured on a Bruker DPX 400 MHz and Bruker AVANCE ΙΙ 600 MHz spectrometers, with DMSO-d
6 or CDCl
3 as solvent using TMS as an internal standard. The methods used for the purpose of NMR assignment were COSY, HSQC and HMBC. The chemical shifts are expressed as δ unit in parts per million (ppm) and TMS = 0.00 ppm. The following abbreviation are used: s = singlet, d = doublet, t = triplet; q = quartet; m = multiple; br. = broad. Mass spectra were measured on GC/MS DFS, THERMO instrument. Microanalyses were performed on a CHNS-Vario Micro Cube analyzer, Single crystal X-ray crystallography was perfomed using a Rigaku Rapid ΙΙ located at the Chemistry Department of Kuwait University. Compound
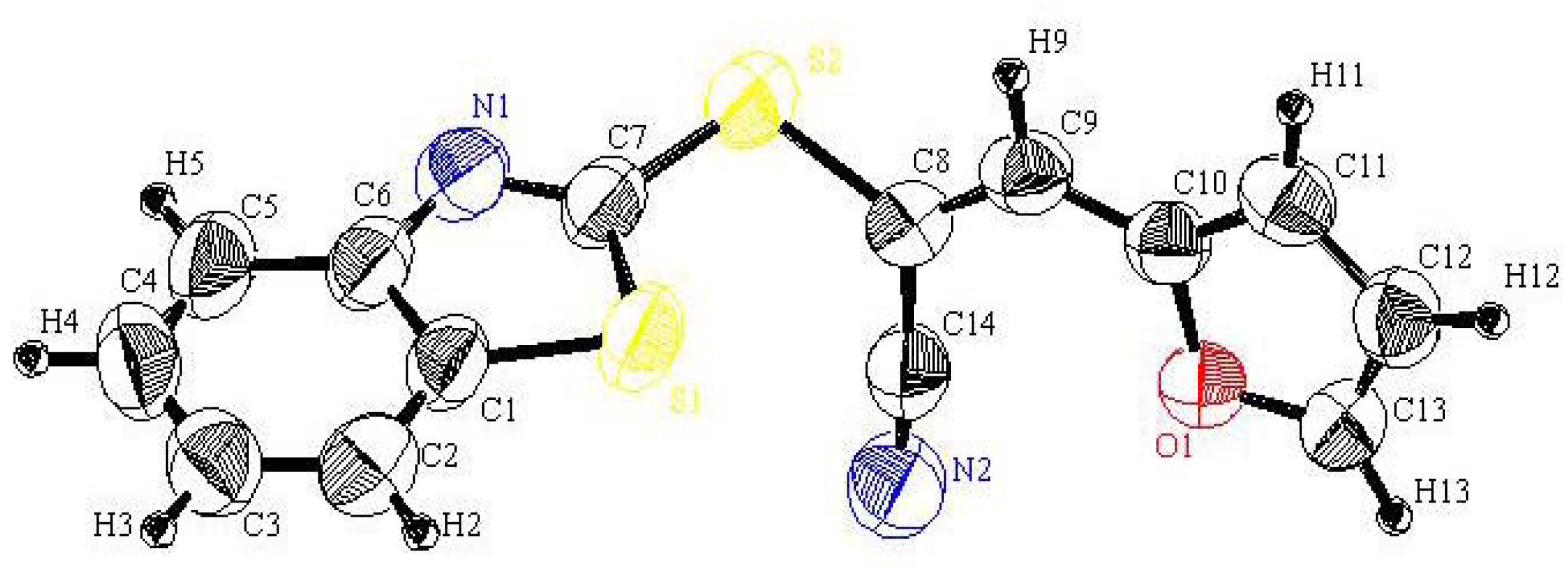
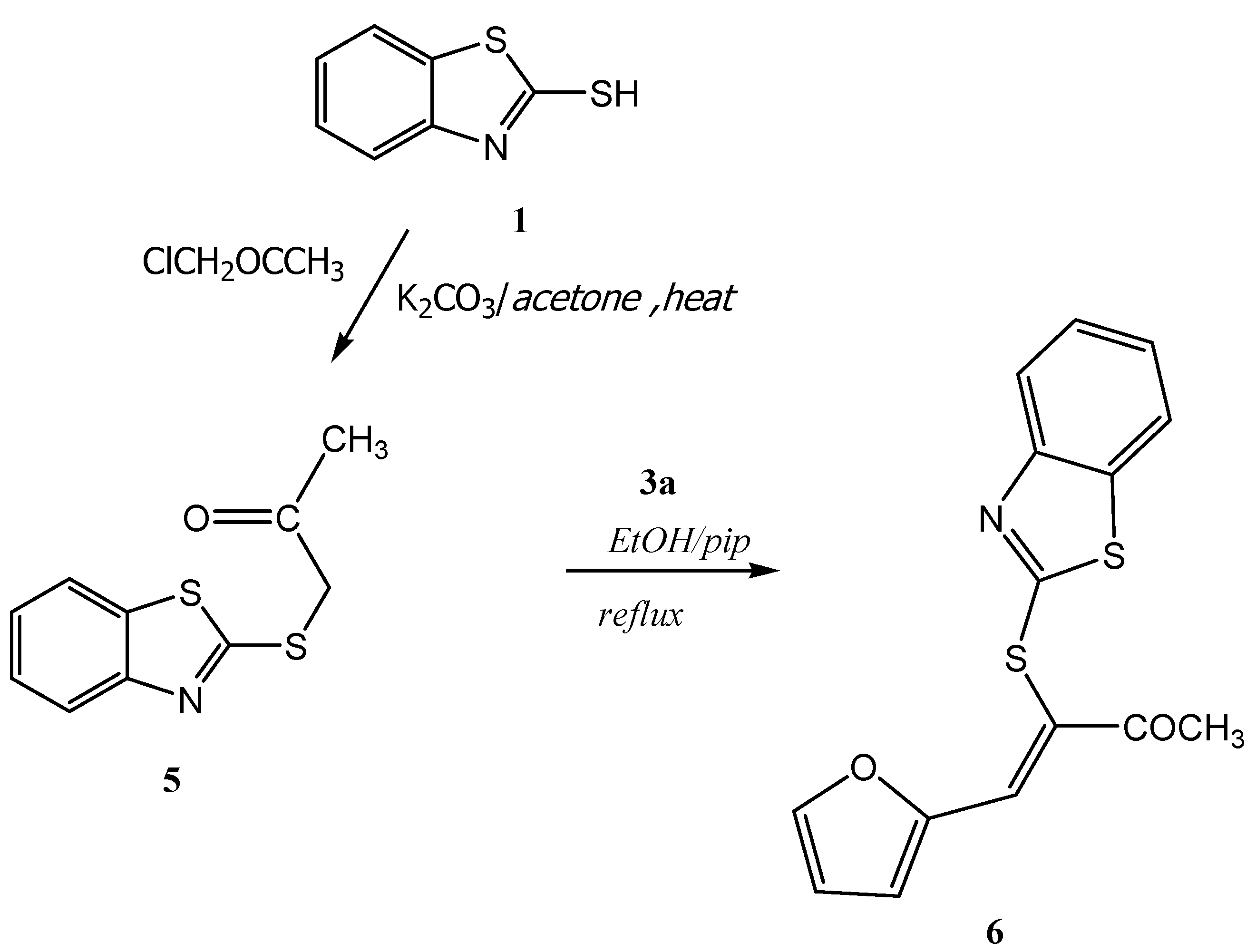
5 was prepared according to our recent reference [
15] and its X-ray data was reported in reference [
19].
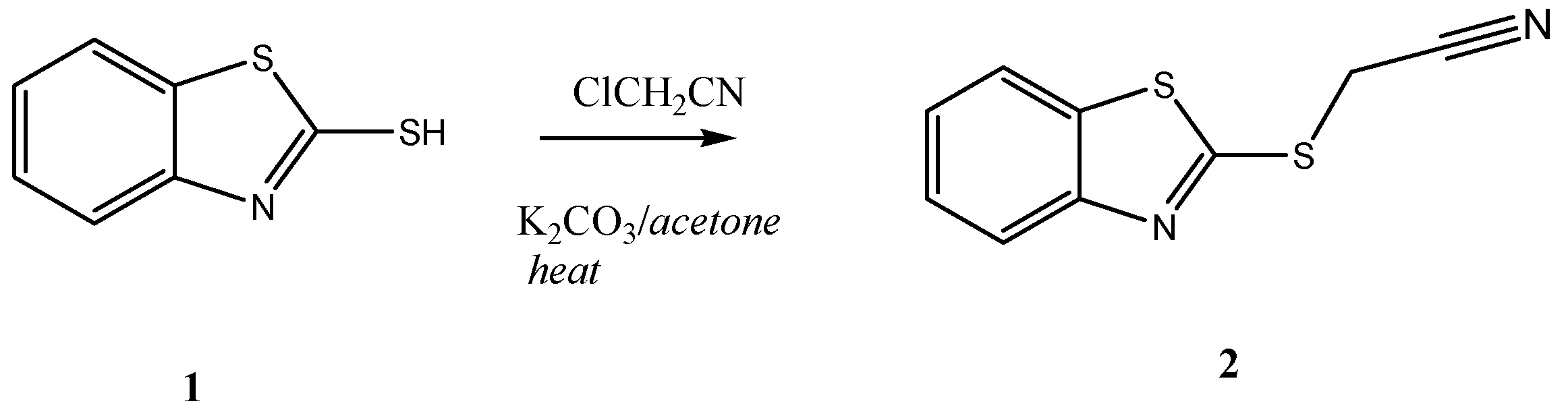
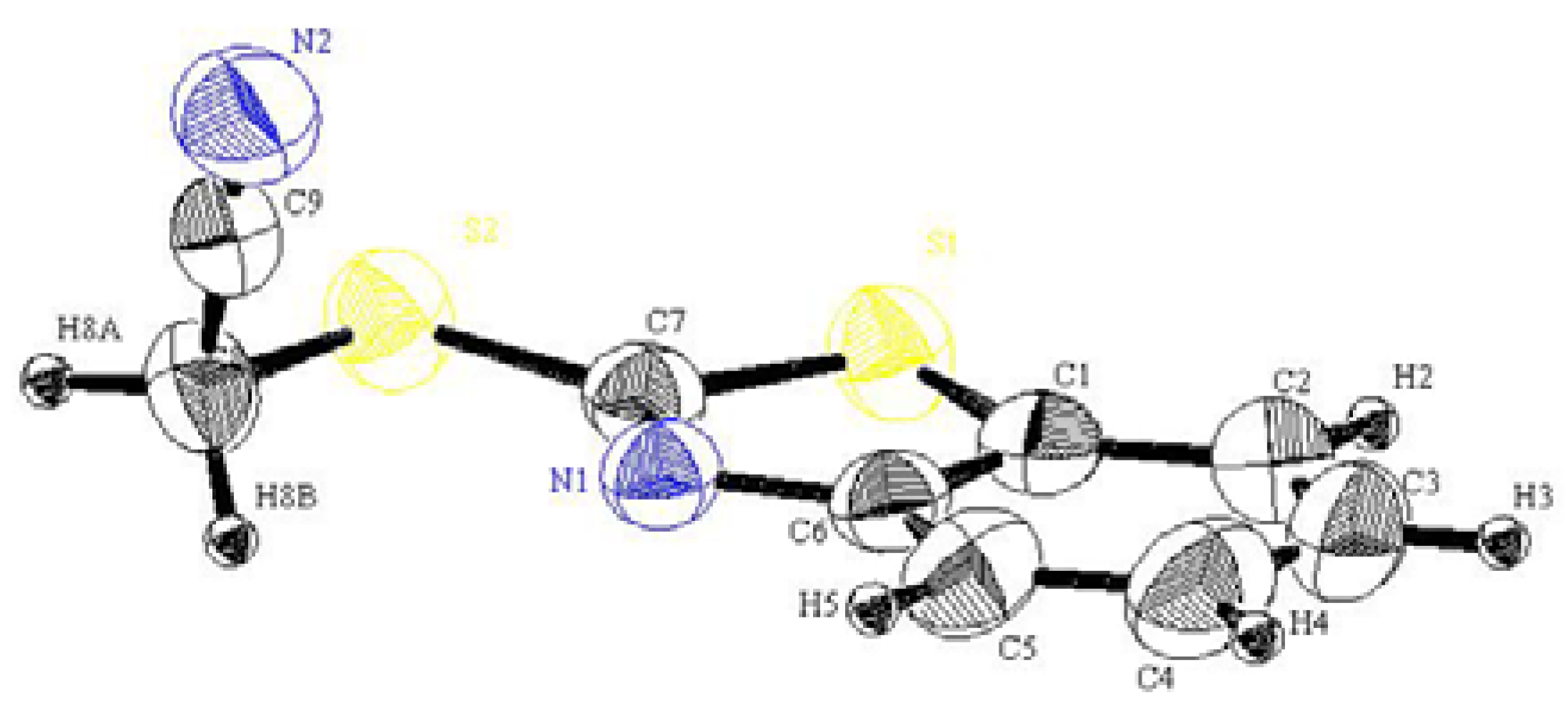
2-(Benzothiazol-2-ylthio) acetonitrile (2): A mixture of 1 (1.67 g, 10.0 mmol), chloroacetonitrile (0.63 g, 10.0 mmol), and anhydrous potassium carbonate (1.38 g, 10.0 mmol) in acetone (100 mL), were heated in water bath for 2 h. The solvent was then evaporated under reduced pressure. The solid product, so formed, was collected by filtration and crystallized from ethanol as brown crystals. Yield: 1.85 g (90%), mp. 70–72 °C; FT-IR: νmax/cm−1: 2243 (CN); 1H-NMR (DMSO-d6): δH 4.58 (s, 2H, CH2), 7.43 (t, 1H, J = 8.0 Hz, H-5'), 7.52 (t, 1H, J = 8.0 Hz , H-6'), 7.95 (d, 1H, J = 8.0 Hz, H-7'), 8.08 (d, 1H, J = 8.0 Hz, H-4'); 13C-NMR (DMSO-d6): δC 163.8 (C-2'), 152.7 (C-3a'), 135.6 (C-7a'), 127.1 (C-6'), 125.5 (C-5'), 122.6 (C-4'), 122.0 (C-7'), 117.9 (CN), 18.6 (CH2) ppm; MS m/z (%) 206 [M+, 100%]. Anal. Calcd. for C9H6N2S2 (206.28): C, 52.40; H, 2.93; N, 13.57%. Found: C, 52.29; H, 3.19; N, 13.45%.
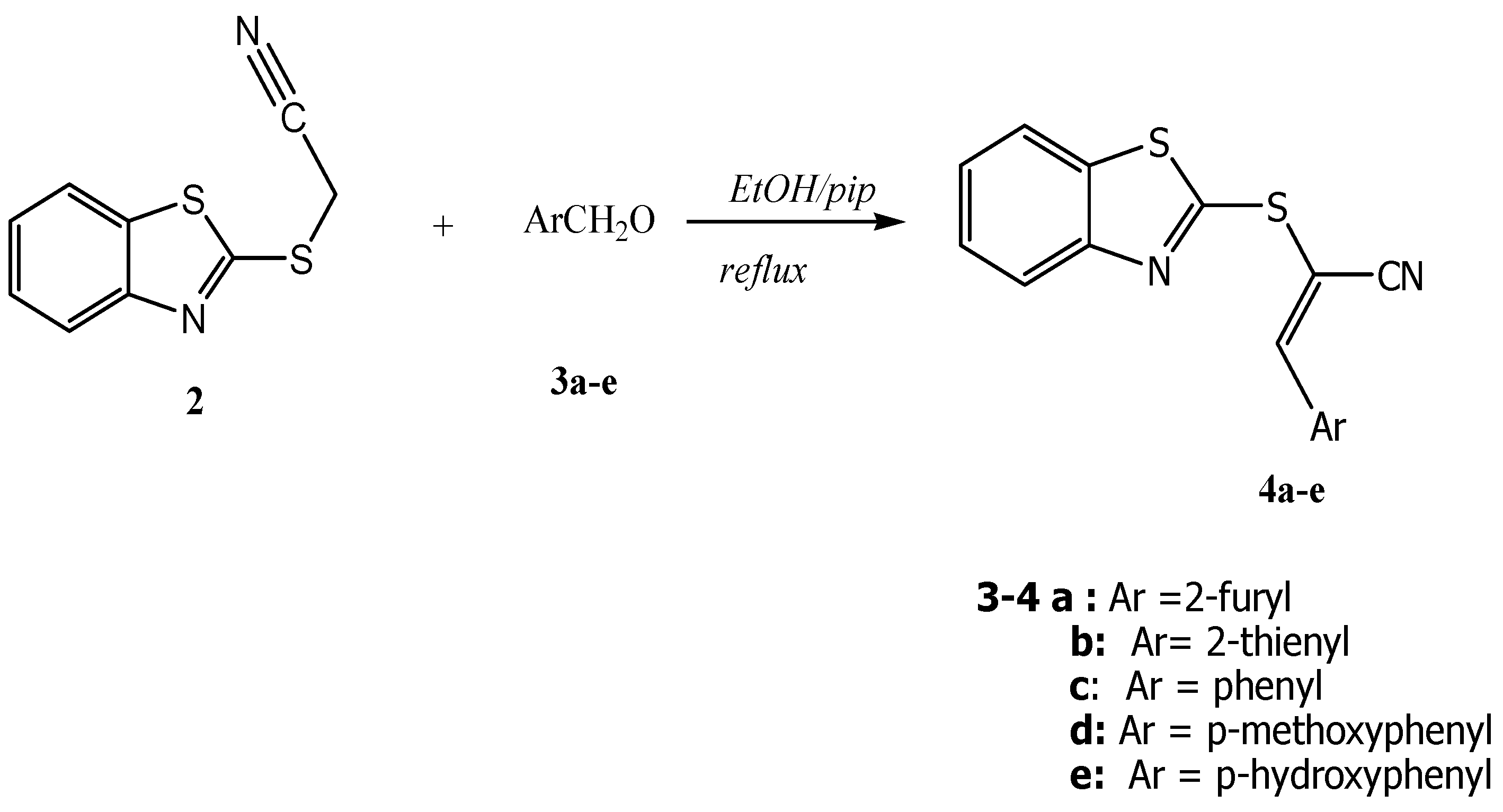
General Procedure for Synthesis of 3-Aryl-2-(benzothiazol-2'-ylthio) acrylonitriles 4a–e. A mixture of 2 (2.06 g, 10.0 mmol) and aromatic aldehydes 3a–e (10.0 mmol) in ethanol (20 mL) containing a few drops of piperidine was refluxed for 4 h. The reaction was allowed to cool to room temperature for 24 h. The solid product so formed was collected by filtration and crystallized from the appropriate solvent.
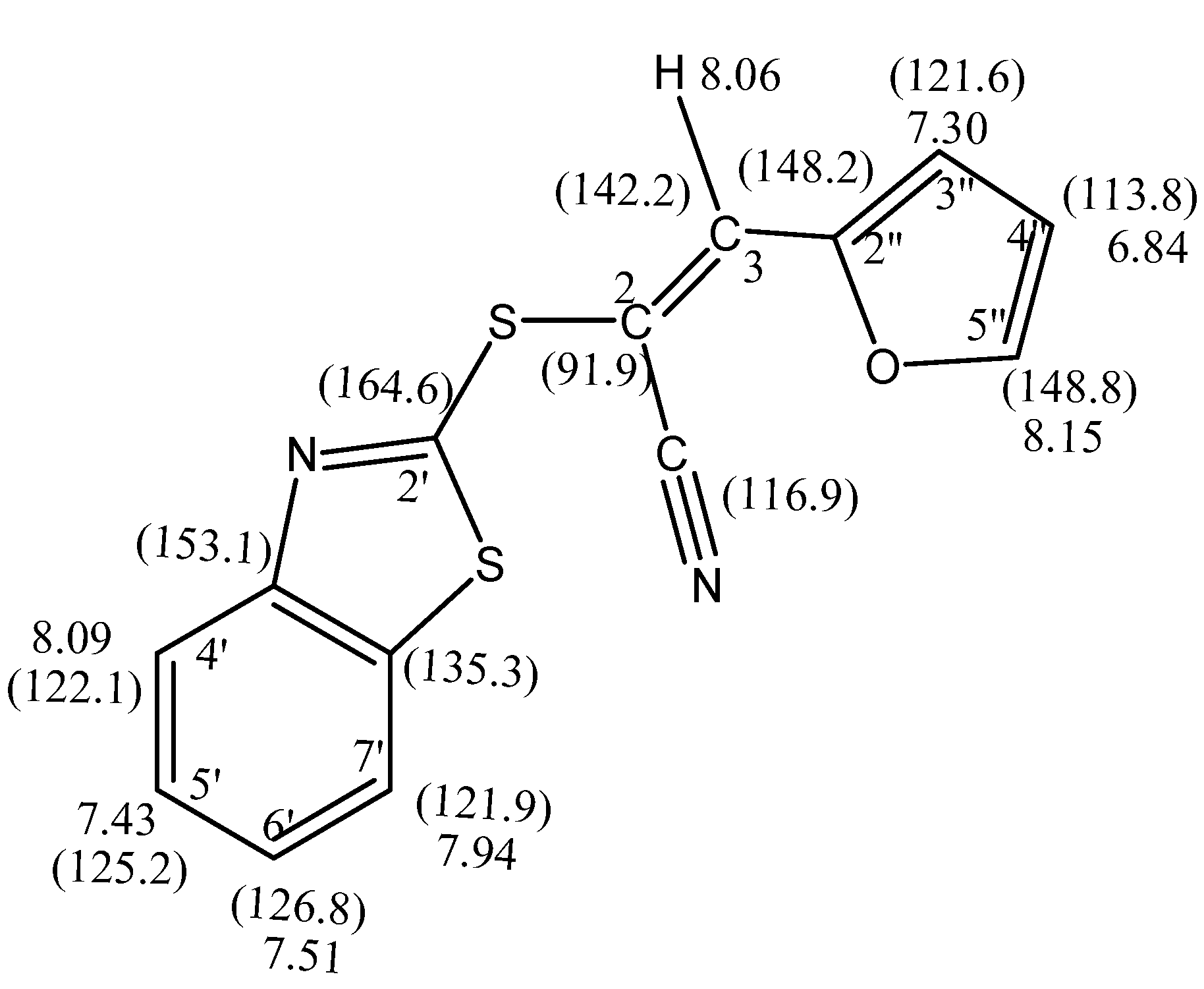
(E)-2-(Benzothiazol-2'-ylthio)-3-(furan-2''-yl) acrylonitrile (4a): This compound was crystallized from ethanol as brown crystals. Yield: 2.24g (79%) yield, mp. 134–136 °C; FT-ir: νmax/cm−1: 2209 (CN); 1H-NMR (DMSO-d6): 6.84 (dd, 1H, J = 3.6 & 1.2 Hz, H-4''), 7.30 (d, 1H, J = 3.6 Hz, H-3''), 7.43 (t, J = 8.2 Hz, H-5'), 7.51 (t, 1H, J = 8.0 Hz, H-6'), 7.94 (d, 1H, J = 8.2 Hz, H-7'), 8.06 (s, 1H, H-3), 8.07 (d, 1H, J = 8.0 Hz, H-4'), 8.15 (d, 1H, J = 1.2 Hz, H-5''); 13C-NMR (DMSO-d6): δC 164.6 (C-2'), 153.1 (3a'), 148.8 (C-5"), 148.2 (C-2''), 142.2 (C-3), 135.3 (C-7a'), 126.8 (C-6'), 125.2 (C-5'), 122.1 (C-4'), 121.9 (C-7'), 121.6 (C-3''), 116.9 (CN), 113.8 (C-4''), 91.9 (C-2) ppm. MS m/z (%) 284 [M+, 18%]. Anal. Calcd. for C14H8N2OS2 (284.36): C, 59.13; H, 2.84; N, 9.85; S, 22.55%. Found: C, 58.95; H, 2.81; N, 10.07; S, 22.56%.
(E)-2-(Benzothiazol-2'-ylthio)-3-(thiophen-2''-yl) acrylonitrile (4b). This compound was crystallized from a 3:2 mixture of ethanol/diethyl ether as yellow crystals. Yield: 2.4 g (88%), mp. 98–100 °C; FT-IR: νmax/cm−1: 2210 (CN); 1H-NMR (DMSO-d6): δH 7.35 (dd, 1H, J = 4.8 & 3.5 Hz, H-4''), 7.43 (t, 1H, J = 7.6 Hz, H-5'), 7.52 (t, 1H, J = 7.6 Hz, H-6'), 7.89 (d, 1H, J = 3.5 Hz, H-3''), 7.95 (d, 1H, J = 8.0 Hz, H-7'), 8.09 (d, 1H, J = 8.0 Hz, H-4'), 8.14 (d, 1H, J = 4.8 Hz, H-5''), 8.52 ppm (s, 1H, H-3); 13C-NMR (DMSO-d6): δC 164.8 (C-2'), 153.2 (C-3a'), 150.4 (C-3), 137.5 (C-3''), 135.9 (C-2''), 135.3 (C-7a'), 135.1 (C-5''), 128.6 (C-4''), 126.8 (C-6'), 125.2 (C-5'), 122.1 (C-4'), 121.9 (C-7'), 117.1 (CN), 92.3 (C-3) ppm; MS m/z (%) 300 [M+, 14%]. Anal. Calcd. for C14H8N2S3 (300.42): C, 55.97; H, 2.68; N, 9.32; S, 32.01%. Found: C, 56.04; H, 2.52; N, 9.54; S, 32.08%.
(E)-2-(Benzothiazol-2'-ylthio)-3-phenyl acrylonitrile (4c). This compound was crystallized from a 2:1 mixture of ethanol/diethyl ether as yellow crystals. Yield: 2.08 g (71%) , mp. 103–105 °C; FT-IR: νmax/cm−1: 2206 (CN); 1H-NMR (DMSO-d6): δH 7.38 (t, 1H, J = 7.8 Hz, H-5'), 7.49 (t, 1H, J = 8.4 Hz, H-6' ), 7.58–7.63 (m, 3H, H-3'', H-4'' & H-5''), 7.96 (d, 1H, J = 8.4 Hz, H-7), 7.97 (d, 2H, J = 8.4 Hz, H-2''& H-6''), 8.10 (d, 1H, J = 8.0 Hz, H-4'), 8.29 ppm (s, 1H vinylic-H); 13C-NMR (DMSO-d6): δC 163.7 (C-2'), 157.0 (C-3), 153.1 (C-3a'),135.4 (C-7a'), 132.6 (C-4''), 130.8 (C-1''), 129.5 (C-2'' & C-6''), 128.9 (C-3'' & C-5''),126.8 (C-6'),125.5 (C-5'), 122.2 (C-4'), 122.0 (C-7'), 116.8 (CN), 97.5 (C-2) ppm. MS m/z (%) 294 [M+, 83%]. Anal. Calcd. for C16H10N2S2, (294.39) requires: C, 65.28 ; H, 3.42; N, 9.52; S, 21.78%. Found: C, 64.98; H, 3.25; N, 9.73; S, 22.03%.
(E)-2-(Benzothiazol-2'-ylthio)-3-(4''-methoxyphenyl) acrylonitrile (4d). This compound was crystallized from ethanol as brown crystals. Yield: 2.62 g (81%), mp. 95–97 °C. FT-IR: νmax/cm−1: 2202 (CN); 1H-NMR (DMSO-d6): δH 3.87 (s, 3H, OCH3), 7.15 (d, 2H, J = 9.0 Hz, H-3'' & H-5''), 7.42 (t, 1H, J = 7.8 Hz, H-5'), 7.51 (t, 1H, J = 7.8 Hz, H-6'), 7.94 (d, 1H, J = 7.8 Hz, H-7'), 8.00 (d, 2H, J = 8.4 Hz, H-2’’ & H-6’’), 8.06 (d, 1H, J = 7.8 Hz, H-4'), 8.17 ppm (s, 1H, H-3); 13C-NMR (DMSO-d6): δC 165.0 (C-2'), 162.9 (C-4''), 157.3 (C-3), 153.3 (C-3a'), 135.3 (C-7a'), 132.1 (C-2'', C-6"), 126.7 (C-6'), 125.1 (C-5'), 124.9 (C-1''), 122.1 (C-4'), 121.9 (C-7'), 117.5 (CN), 114.8 (C-3'' & C-5"), 92.8 (C-2), 55.7 (OCH3) ppm; MS m/z (%) 324 [M+, 20% ]. Anal. Calcd. for C17H12N2OS2 (324.42): C, 62.94; H, 3.73; N, 8.63; S, 19.76%. Found: C, 63.06; H, 3.63; N, 8.87; S, 20.06%.
(E)-2-(Benzothiazol-2'-ylthio)-3-(4''-hydroxyphenyl) acrylonitrile (4e). This compound was crystallized from a 2:1 mixture of ethanol/diethyl ether as yellow crystals. Yield: 2.32 (75%), mp. 113–114 °C ; FT-IR: νmax/cm−1: 3417 (OH), 2200 (CN); 1H-NMR (DMSO-d6): δH 6.97 (d, 2H, J = 8.4 Hz, H-3'' & H-5''), 7.39 (t, 1H, J = 8.4 Hz, H-5'), 7.50 (t, 1H, J = 8.4 Hz, H-6'), 7.91 (d, 2H, J = 8.4 Hz, H-2'' & H-6''), 7.93 (d, 1H, J = 8.4, Hz, H-7'), 8.06 (d, 1H, J = 8.4 Hz, H-4'), 8.12 (s, 1H, H-3), 9.05 (bs., 1H, OH, D2O exchangeable); 13C-NMR (DMSO-d6): δC 165.6 (C-2'), 162.2 (C-4''), 158.0 (C-3), 153.3 (C-3a'), 135.2 (C-7a'), 132.5 (C-2'' & C-6''), 126.7 (C-6'), 125.0 (C-5'), 123.0 (C-1''), 122.1 (C-4'), 121.8 (C-7'), 117.8 (CN), 116.3 (C-3'' & C-5''), 91.2 (C-2) ppm; MS m/z (%) 310 [M+, 44%]. Anal. Calcd. for C16H10N2OS2 (310.39): C, 61.91; H, 3.25 ; N, 9.03; S, 20.66%. Found: C, 61.63; H, 3.40; N, 9.27; S, 20.92%.
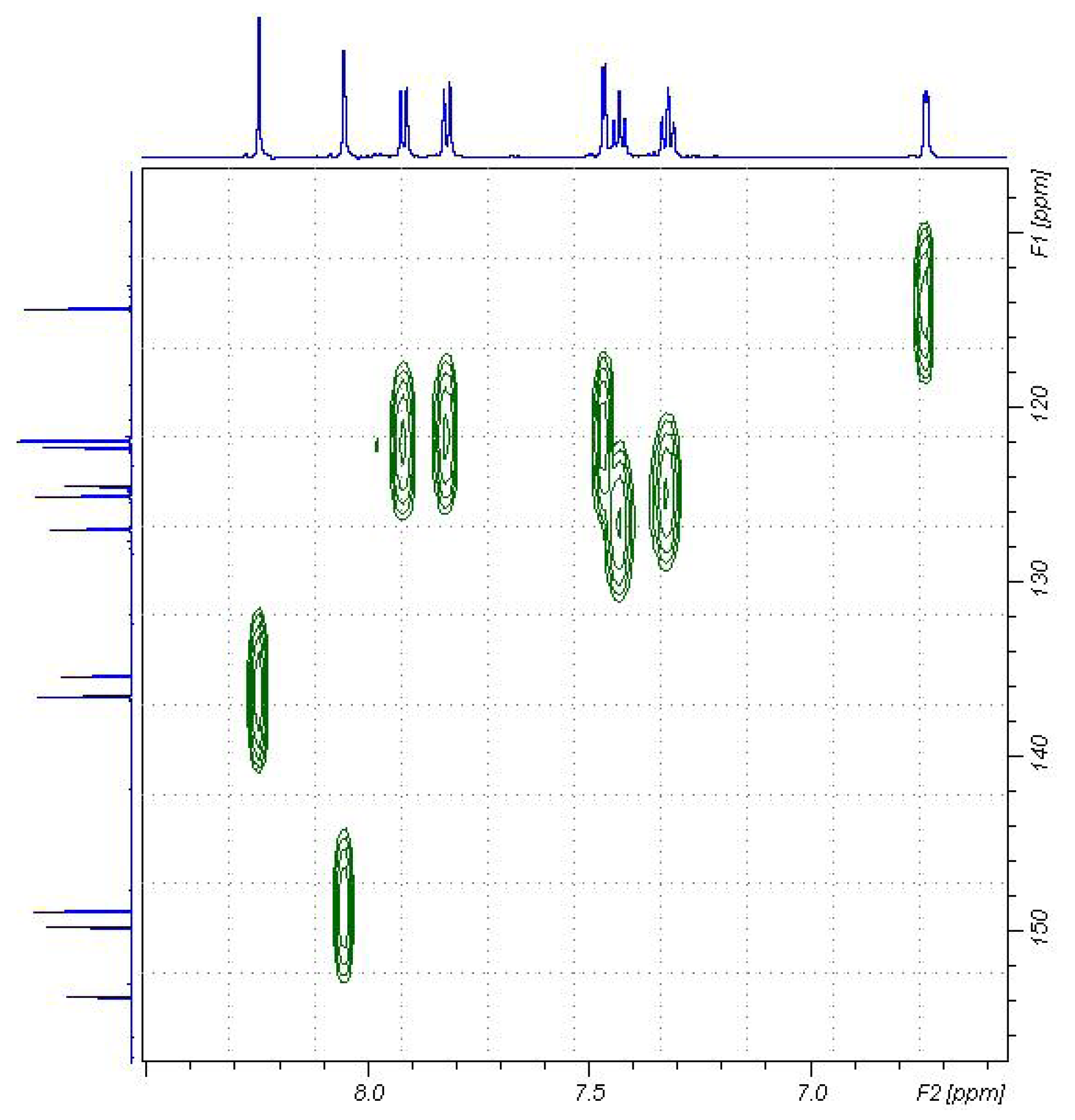
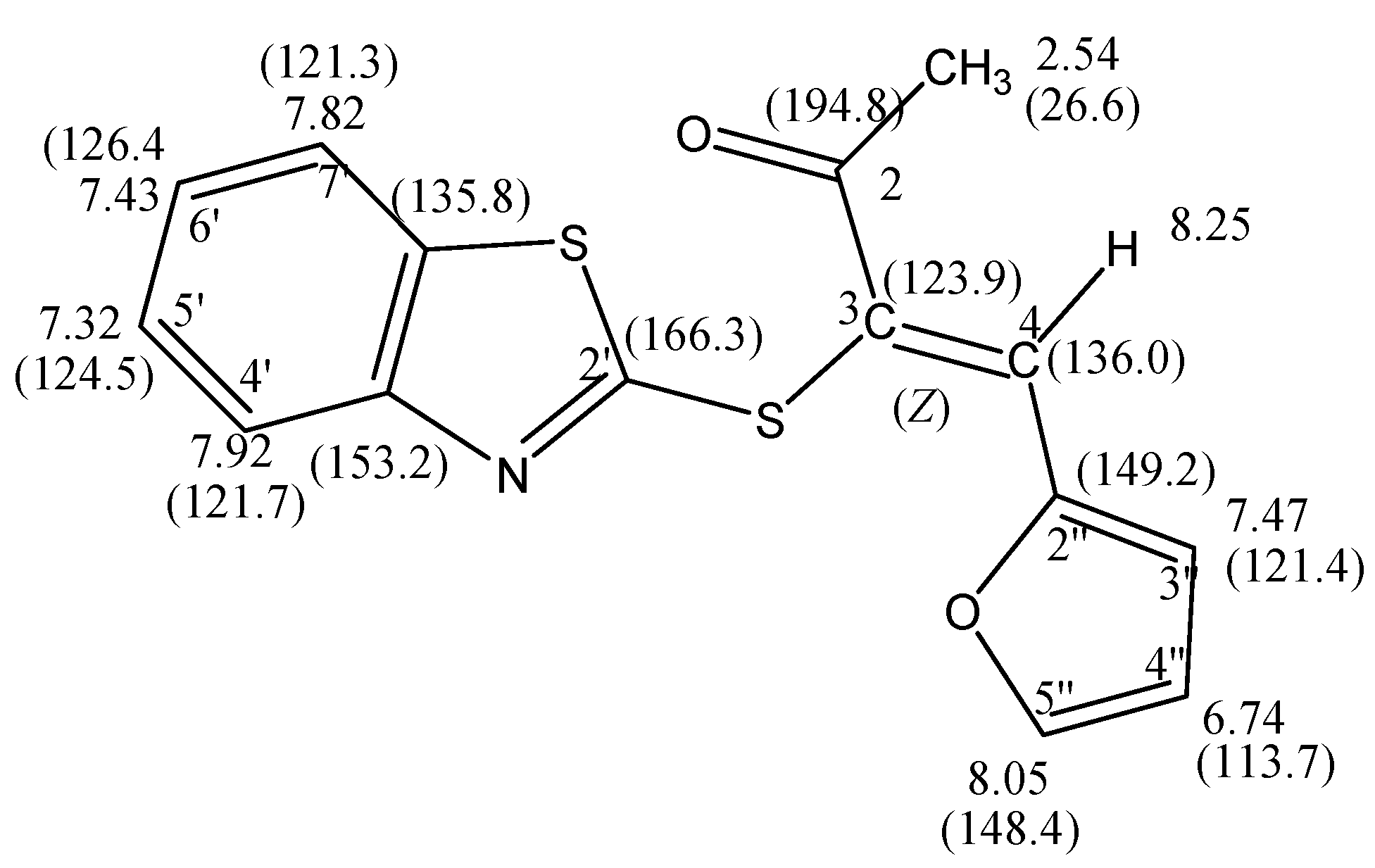
(Z)-3-(Benzothiazol-2'-ylthio)-4-(furan-2''-yl)-3-buten-2-one (6): A mixture of 5 (2.33 g, 10.0 mmol) and furan-2-carbaldehyde 3a (10.0 mmol) in ethanol (20 mL) containing a few drops of piperidine was refluxed to 4 h. The reaction was allowed to cool to room temperature for 24 h. The solid product so formed was collected by filtration and crystallized from the ethanol as yellow crystals. Yield: 2.16 g (72%) yield, mp. 85–87 °C ; FT-IR: νmax/cm−1: 1662 (CO); 1H-NMR (DMSO-d6): 2.54 (s, 3H, CH3), 6.74 (dd, 1H, J = 3.6 & 1.2 Hz, H-4''), 7.32 (t, 1H, J = 8.0 Hz, H-5'), 7.43 (t, 1H, J = 8.0 Hz, H-6'), 7.47 (d, 1H, J = 3.6 Hz, H-3''), 7.82 (d, 1H, J = 8.0 Hz, H-7'), 7.92 (d, 1H, J = 8.0 Hz, H-4'), 8.05 (d, 1H, J = 1.6 Hz, H-5''), 8.25 (s, 1H, H-4); 13C-NMR (DMSO-d6): δC 194.8 (C-2), 166.3 (C-2'), 153.2 (C-3a'), 149.2 (C-2''), 148.4 (C-5''), 136.0 (C-4), 135.8 (C-7a'),126.4 (C-6'), 124.5 (C-5'), 123.9 (C-3), 121.7 (C-4'), 121.4 (C-3''), 121.3 (C-7') ,113.7 (C-4''), 26.6 (CH3) ppm; MS m/z (%) 301 [M+, 30%]. Anal. Calcd. for C15H11NO2S2 (301.38): C, 59.78; H, 3.67; N, 4.64%. Found: C, 59.41; H, 3.68; N, 4.80%.
3-(2'-Oxopropylthio)-3-phenylquinazolin-4(3H)-one (9): A mixture 8 (2.54 g, 10.0 mmol), chloro-acetone (0.79 g, 10.0 mmol), and anhydrous potassium carbonate (1.38 g, 10.0 mmol) in acetone (100 mL) were heated in water bath for 2 h. The solvent was then evaporated under reduced pressure. The solid product, so formed, was collected by filtration and crystallized from ethanol as yellow crystals. Yield: 2.4 g (79%), mp. 140–142 °C. FT-IR: νmax/cm−1: 1725 (CO ketone), 1683 (CO amide); 1H-NMR (DMSO-d6): δH 2.33 (s, 3H, CH3) ,4.06 (s, 2H, CH2), 7.29 (d, 1 Hz, J = 7.6 Hz, H-5), 7.35 (t, 1H, J = 7.6 Hz, H-7), 7.63–7.41 (m, 5H, phenyl–H), 7.81 (t, 1H, J = 8.0 Hz, H-6), 8.08 (d, 1H, J = 7.6Hz, H-8); 13C-NMR (DMSO-d6): δC 202.5 (C-2'), 161.1 (C-4), 157.3 (C-2), 147.5 (C-8a), 136.4 (C-6), 135.4 (C-1''), 130.4, 129.8, 129.5 (phenyl carbons), 127.1 (C-8), 126.5 (C-7), 126.4 (C-5), 123.9 (C-4a), 42.6 (C-1'), 28.7 (CH3) ppm; MS m/z (%) 310 [M+, 12%]. Anal. Calcd. for C17H14N2O2S(310.37): C, 65.79, H, 4.55, N, 9.03%. Found: C, 65.65, H, 4.30, N, 9.06%.
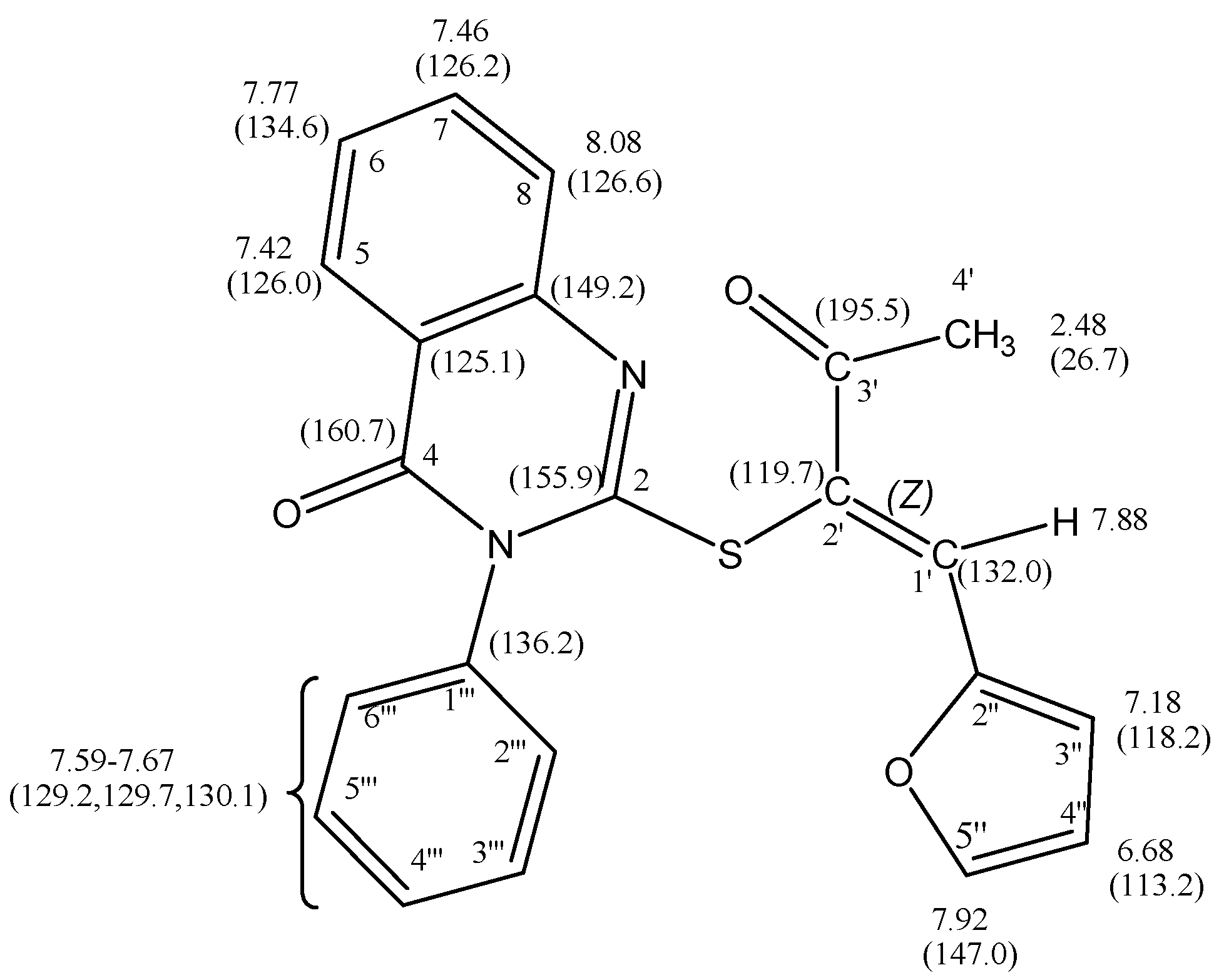
(Z)-2-(1'-(Furan-2''-yl)-3'-oxobut-1''-en-2-ylthio)-3-phenylquinazolin-4(3H)-one (10): A mixture of 9 (3.10 g, 10 mmol) and 3a (0.83 g, 10 mmol) in ethanol (20 mL) containing a few drops of piperidine was refluxed for 4 h. The reaction was allowed to cool to room temperature for 24 h. The solid product so formed was collected by filtration and crystallized from ethanol as brown crystals. Yield: 2.83 g (73%) yield, mp. 190–192 °C; FT-IR: νmax/cm−1: 1675 (CO ketone), 1610 (CO amide); 1H-NMR (DMSO-d6): 2.48 (s, 3H, CH3), 6.68 (dd, 1H, J = 3.6 Hz & 1.8 Hz, H-4''), 7.18 (d, 1H, J = 3.6 Hz, H-3''), 7.42 (d, 1H, J = 8.0 Hz, H-5), 7.46 (t, 1H, J = 8.0 Hz, H-7), 7.67–7.59 (m, 5H, phenyl–H),7.77 (t, 1H, J = 8.0 Hz, H-6), 7.88 (s, 1H, H-1'), 7.92 (d, 1H, J = 1.8 Hz, H-5''), 8.08 (d, 1H, J = 8.0 Hz, H-8) ppm; 13C-NMR (DMSO-d6): δC 195.5 (C-3'), 160.7 (C-4), 155.9 (C-2), 149.2 (C-8a), 147.0 (C-5''), 146.8 (C-2''), 136.2 (C-1'''), 134.9 (C-6), 132.0 (C-1', vinylic–H), 130.1, 129.7, 129.2 (phenyl carbons), 126.6 (C-8), 126.2 (C-7), 126.0 (C-5), 125.1 (C-4a), 119.7 (C-2'), 118.2 (C-3''), 113.2 (C-4''), 26.7 (CH3) ppm; MS m/z (%) 388 [M+, 82]. Anal. Calcd. for C22H16N2O3S( 388.44): C, 68.02; H, 4.15; N, 7.21; S, 8.25%. Found: C, 67.78; H, 4.26; N, 7.16, S, 8.32%.