1. Introduction
Flos Daturae is the dry flower of
Datura metel L. (Solanaceae), which widely distributed in China [
1]. Flos Daturae, known as “baimantuoluo” or “yangjinhua” in China, has been used for centuries in Traditional Chinese Medicine for the treatment of asthma, convulsions, pain, and rheumatism [
2]. Flos Daturae has been found to be rich in tropane alkaloids [
2]. Besides, a number of withanolides have also been isolated from Flos Daturae [
3,
4,
5,
6,
7,
8]. Recently Flos Daturae has been used clinically for the treatment of psoriasis in China [
9]. The effective part for psoriasis, namely the non-alkaloid water-soluble fraction of Flos Daturae, has been demonstrated to have anti-inflammatory, anti- skin titillation and anti-anaphylaxis actions by detailed pharmacological experiments [
9]. However, its active constituents and pharmacological effects related to the treatment of psoriasis were not fully elucidated. As a part of a continuing project to study the active constituents of Flos Daturae for psoriasis [
6,
7,
8], we investigated the 50% ethanol eluate fraction from a macroporous resin of the flowers of
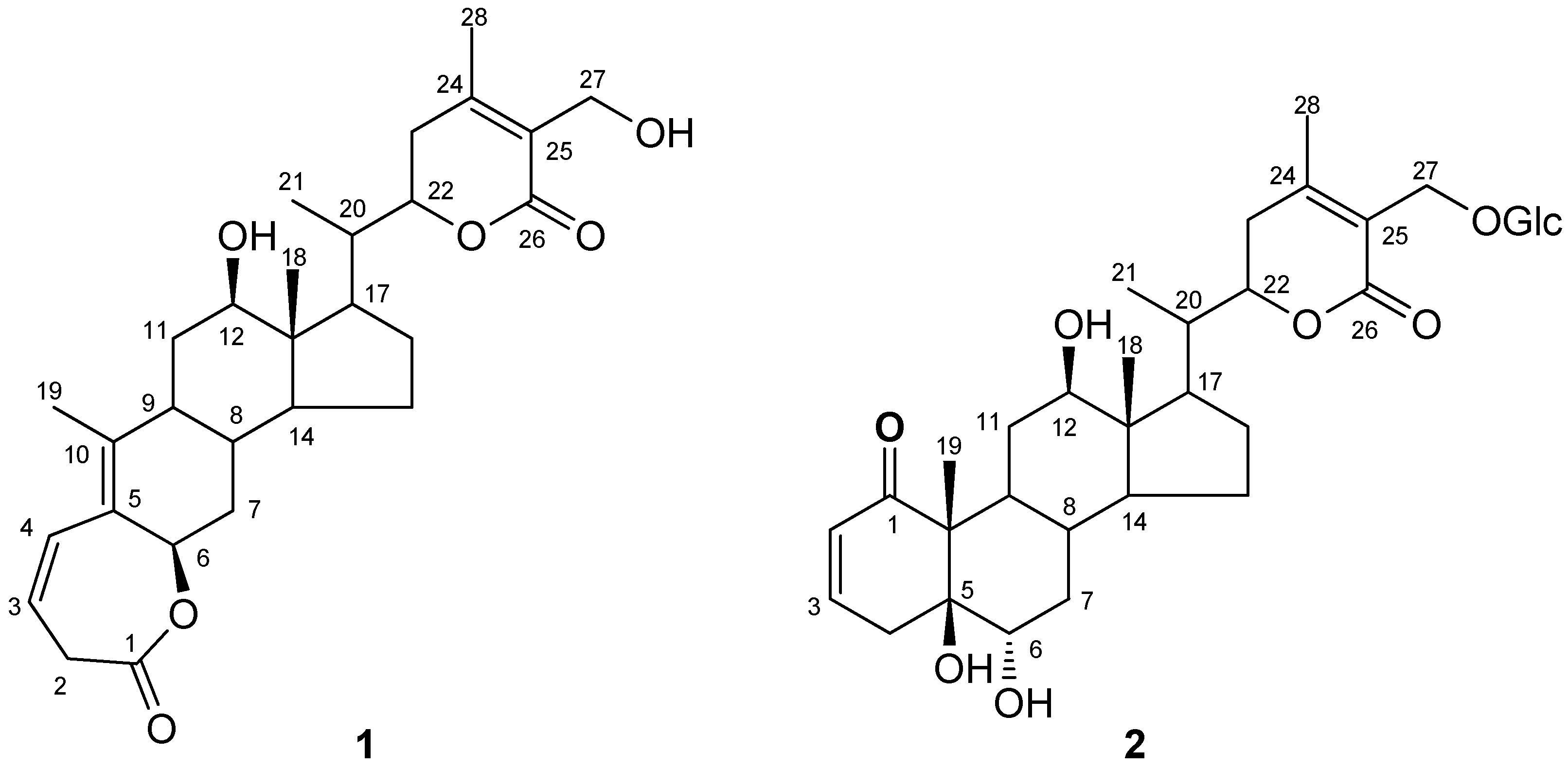
D. metel. Our extraction and separation method can greatly enrich fractions in withanolide compounds so trace withanolides can be isolated. In this paper, we present the isolation and structural characterization of the two new withanolide lactones (
Figure 1) on the basis of the interpretation of spectral data, including 1D, 2D NMR and HRESIMS data. A 1,10-seco withanolide is reported from only the third time in herb plants.
Figure 1.
Structures of 1 and 2.
Figure 1.
Structures of 1 and 2.
2. Results and Discussion
Compound
1 was obtained as a white amorphous powder. Its molecular formula was established as C
28H
38O
6 by the positive HRESIMS, indicating six degrees of unsaturation. The
1H-NMR spectrum of
1 (
Table 1), showed several characteristic signals of the common withanolide steroid. Three signals at
δ 0.78 (3H,
s), 1.18 (3H,
d,
J = 6.8 Hz), 1.84(3H,
s) and 2.10 (3H,
s) were attributed to Me-18, Me-21, Me-19 and Me-28, respectively. A Me-27 signal was missing, and was replaced by two doublets at
δ 4.37 (1H,
d,
J = 11.7 Hz) and 4.30 (1H,
d,
J = 11.7 Hz), suggesting that C-27 was substituted by hydroxyl group. The double doublet at
δ 3.47 (1H,
dd,
J = 10.9, 4.5 Hz) was characteristic for a 12
β-hydroxywithanolide [
4]. H-22 resonated as a double triplet at
δ 4.62 (1H,
dt,
J = 12.9, 3.4 Hz), revealing a
R configuration at C-22 [
5,
6].
The
13C-NMR spectrum of
1 showed resonances for all 28 carbons. The characteristic downfield signal at
δ 175.5 was due to two lactone carbonyls, respectively, along with the characteristic doublets at
δ 126.3, 157.9 and 168.6 were attributed to C-24, C-25 and C-26 of the
α,β-unsaturation-
γ-lactone ring respectively, in the ring E. The signals at
δ 118.1, 130.2, 126.5 and 142.5 for the vinylic carbons at C-3, C-4, C-5, C-10 respectively, in the ring A. The typical signals at
δ 74.8, 78.6, 80.8, and 56.3 were assigned to the oxygenated carbons at C-6, C-12, C-22 and C-27, respectively. The signals appearing at
δ 8.1, 15.6, 15.3 and 20.2 were assigned to the Me-18, Me-19, Me-21 and Me-28, respectively. In addition, a signal at
δH 5.30 (1H,
br. s) together with the signal in the
13C NMR spectrum of a keto-carbonyl of C-1 upfield of
δC 175.5 due to an ester group of CO-O-CH indicated that
1 possesses a 1,10-
seco-steroid skeleton [
10,
11]. Further support this assumption was obtained from the fact that the methylene protons at C-2 [
δ 4.03 (1H,
br. d,
J =17.4 Hz) and 3.08 (1H,
dd,
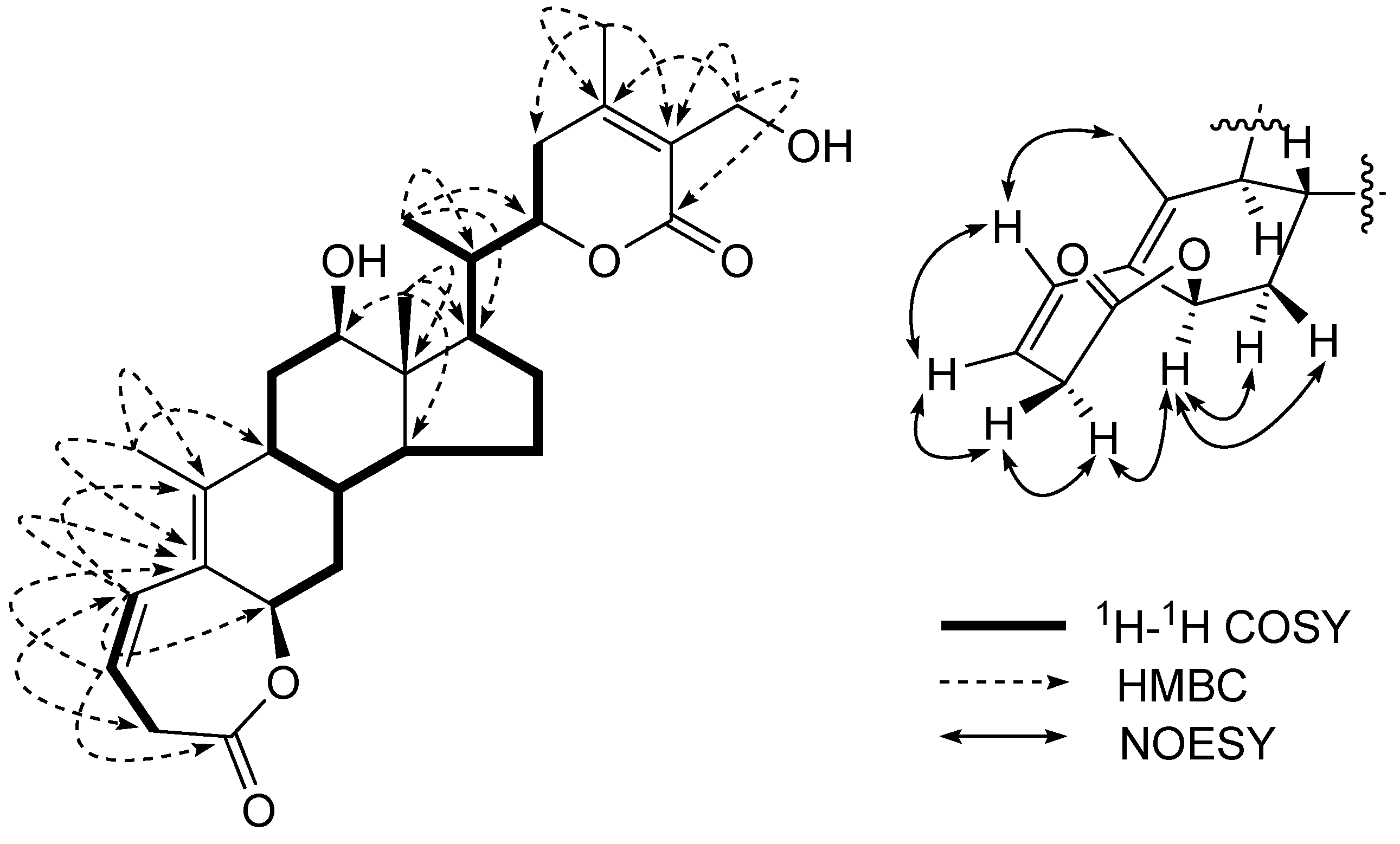
J =17.4, 8.7 Hz)] were unusually shifted to a low field, suggesting the methylene to be situated between an ester carbonyl group and carbon-carbon double bond. Based on this finding and HMBC correlations, between C-1 (
δC 175.5) and H-2
α, H-2
β, and H-3, between H-4 (6.62 (1H,
dd,
J = 11.4, 3.1 Hz) and C-2, C-5, C-6, and C-10, and between C-5 and Me-19, as shown in
Figure 2. Thus, a seven-membered
β,
γ-unsaturated lactonic moiety was determined in ring A.
The
β-configuration of the lactone bond at C-6 was established by a NOESY experiment (
Figure 2). The NOESY spectrum showed the correlation between H-6 (1H,
br. s) and H-2
α, H-7
α and H-7
β, indicating that H-6 has the same configuration as H-2
α. Since a small coupling between H-2
α and H-3 in the
1H NMR spectrum was observed due to an approximate 90°, a
β-configuration of the lactone bond at C-6 was inferred, which is in agreement with those of 1,10-seco steroids [
10,
11]. Therefore, the structure of
1 was deduced as (12
β,6
β,22
R)-1,10-seco-6,12,27-trihydroxy-26-oxo-witha-3,5,24-trienolide-1-oic acid-
ε-lactone, which was named baimantuoluoline G.
Table 1.
1H and 13C-NMR data of 1 and 2 in CD3OD at 400 MHz and 100 MHz, J in Hz.
Table 1.
1H and 13C-NMR data of 1 and 2 in CD3OD at 400 MHz and 100 MHz, J in Hz.
| No. | 1 | | 2 | |
|---|
| δH | δC | δH | δC |
|---|
| 1 | | 175.5 | | 207.0 |
| 2 | 4.03 (1H, br. d, J = 17.4, α-H) | 35.9 | 5.77 (1H, dd, J = 10.0, 2.4) | 128.9 |
| | 3.08 (1H, dd, J = 17.4, 8.7, β-H) | | | |
| 3 | 5.53 (1H, dd, J = 11.4, 8.7) | 118.1 | 6.66 (1H, ddd, J = 10.0, 5.2, 2.0) | 143.5 |
| 4 | 6.62 (1H, dd, J = 11.4, 3.1) | 130.2 | 3.24 (1H, dt, J = 20.0, 2.4) | 36.5 |
| | | | 2.05 (1H, dd, J = 20.0,5.2) | |
| 5 | | 126.5 | | 78.2 |
| 6 | 5.30 (1H, br. s) | 74.8 | 3.52 (1H, t, J = 2.0) | 75.2 |
| 7 | 2.03 (1H, m) | 33.7 | | 33.8 |
| | 1.43 (1H, m) | | | |
| 8 | 1.50 (1H, m) | 33.5 | | 30.5 |
| 9 | 1.77 (1H, m) | 47.2 | 1.87 (1H, m) | 41.0 |
| 10 | | 142.5 | | 52.8 |
| 11 | 2.14 (1H, dt, J = 16.1, 4.2) | 36.9 | 2.42 (1H, dt, J = 12.4, 4.0) | 33.8 |
| | 1.36 (1H, m) | | 1.36 (1H, m) | |
| 12 | 3.47 (1H, dd, J = 10.9, 4.5) | 78.6 | 3.47 (1H, dd, J = 11.2, 4.4) | 78.7 |
| 13 | | 49.1 | | 49.0 |
| 14 | 1.25 (1H, m) | 53.8 | 1.25 (1H, m) | 55.3 |
| 15 | 1.72 (1H, m) | 23.8 | 1.76 (1H, m) | 24.6 |
| | 1.30 (1H, m) | | 1.31 (1H, m) | |
| 16 | 1.73 (1H, m) | 27.7 | 1.76 (1H, m) | 27.6 |
| | 1.60 (1H, m) | | 1.54 (1H, m) | |
| 17 | 1.61 (1H, m) | 54.5 | 1.56 (1H, m) | 55.0 |
| 18 | 0.78 (3H, s) | 8.1 | 0.76(3H, s) | 8.0 |
| 19 | 1.84 (3H, s) | 15.6 | 1.30(3H, s) | 16.1 |
| 20 | 2.01 (1H, m) | 38.9 | 2.05 (1H, m) | 38.9 |
| 21 | 1.18 (3H, d, J = 6.8) | 15.3 | 1.18 (3H, d, J = 6.8) | 15.5 |
| 22 | 4.62 (1H, dt, J = 12.9, 3.4) | 80.8 | 4.60 (1H, dt, J = 13.6, 4.0) | 80.9 |
| 23 | 2.55 (1H, dd, J = 18.0, 13.2) | 32.5 | 2.58 (1H, dd, J = 18.0, 13.2) | 32.3 |
| | 2.24 (1H, dd, J = 18.0, 3.2) | | 2.21 (1H, dd, J = 18.0, 3.2) | |
| 24 | | 157.9 | | 160.5 |
| 25 | | 126.3 | | 123.6 |
| 26 | | 168.6 | | 168.7 |
| 27 | 4.37 (1H, d, J = 11.7) | 56.3 | 4.46 (1H, d, J = 10.8) | 63.5 |
| | 4.30 (1H, d, J = 11.7) | | 4.61 (1H, d, J = 10.8) | |
| 28 | 2.10 (3H, s) | 20.2 | 2.13 (3H, s) | 20.8 |
| 1′ | | | 4.31 (1H, d, J = 8.0) | 103.9 |
| 2′ | | | 3.16 (1H, t, J = 8.0) | 75.0 |
| 3′ | | | 3.26 (1H, m) | 78.0 |
| 4′ | | | 3.29 (1H, m) | 71.5 |
| 5′ | | | 3.24 (1H, m) | 78.0 |
| 6′ | | | 3.85 (1H, dd, J = 12.0, 2.0) | 62.7 |
| | | | 3.67 (1H, dd, J = 12.0, 5.2) | |
Figure 2.
Key 1H-1H COSY and HMBC correlations of 1.
Figure 2.
Key 1H-1H COSY and HMBC correlations of 1.
Compound
2 was obtained as a white amorphous powder and showed positive results for the
Molish reagent, which was indicative of a withanolide glycoside. Its molecular formula was established as C
34H
50O
12 by the positive HRESIMS, indicating 10 degrees of unsaturation. The
1H-NMR spectrum of
2 showed distinct resemblance to those of baimantuoluoside G ((5
α,6
β,22R)-5,6,27-trihydroxy-1-oxowitha-2,24-dienolide-27-O
-β-glucopyranoside) [
6]. The only notable difference was the change of H-12 signal appeared as a double doublet at
δ 3.47 (1H,
dd,
J = 11.2, 4.4 Hz), indicating that C-12 was substituted by a hydroxyl group. The
13C-NMR (DEPT) spectrum showed an additional downfield C-atom signal at
δ C 78.7 in
2, which was affirmatively assigned to the C-12. On the basis of above data, the structure of
2 was identified to be (5
β,6
α,12
β,22
R)-5,6,12,27-tetrahydroxy-1,26-dioxo-witha-2,24-dienolide-27-O-
β-glucopyranoside, and was named baimantuoluoside H.
3. Experimental
3.1. General
Preparative HPLC (Waters, Delta 600-2487) was performed on a Hypersil-ODS II (10 μm, 20 × 300 mm, Yilite, Dalian, People’s Republic of China) with Waters Empower software. IR spectra were measured in KBr discs on a Shimadzu FTIR-8400S spectrometer. HRESIMS were carried out on Waters Xevo QTOF mass spectrometer with Masslynx V4.11 software. NMR data were recorded in CD3OD on Bruker DPX 400 spectrometer at 400 MHz for (1H) and 100 MHz (13C) with Xwin-NMR software version 2.6; chemical shifts δ in ppm rel. to SiMe4 as internal standard, coupling constant J in Hz. The pulse conditions were as follows: for the 1H-NMR spectra, spectrometer frequency (SF) 400.13 MHz, acquisition time (AQ) 2.0447731s, number of transients (NS) 64, receiver gain (RG) 128, temperature (TE) 303.0 K, dwell time (DW) 62.400 μs, per scan delay (DE) 7.00 μs, dummy scans (DS) 0; for the 13C-NMR spectrum, SF 100.62 MHz, AQ 0.65 s, NS 1430, RG 14596.5, TE 303.0 K, DW 19.900 μs, DE 28.00 μs, DS 0; for the COSY spectrum, SF 400.13 MHz, NS 16, DS 16, pulse (P1) 10.8 μs, TE 303.0 K, RG 574.7, DW 170.400 μs, DE 7.00 μs; for the NOE experiments, SF 400.13 MHz, NS 64, DE 7.00 μs; for the HMBC spectrum, SF 400.13 MHz, AQ 0.1745396 s, RG 16384, NS 64, DW 170.400 μs, DS 16, DE 7.00 μs, TE 303.0 K; and for the HSQC spectrum, SF 400.13 MHz, AQ 0.1745396 s, NS 32, DS 16, DE 7.00 μs, DW 170.400 μs, RG 16384, TE 303.0 K.
3.2. Plant Material
The dry flowers of D. metel were collected in Nanjing city of Jiangsu Province of China in September 2002, and identified by Prof. Zhenyue Wang. A voucher specimen (No. 2002035) is deposited at the Herbarium of Heilongjiang University of Chinese Medicine, China.
3.3. Extraction and Isolation
The dried flowers (30 kg) of D. metel L. were extracted with 70% EtOH under reflux (2 × 100 L) for 2.5 h (each), and the combined solution was filtered and evaporated under vacuum to a syrup (45 °C), followed by suspension in H2O (500 L). The suspension was acidified with 0.1% HCl, and then filtered and exchange for Styrene-DVB (001×7). The exchanged solution was passed through AB-8 crosslinked polystyrene, and sequentially eluted with H2O, 50% EtOH, and 95% EtOH, respectively. 50% EtOH elution was concentrated under vacuum to yield a syrup (52.0 g) and this crude residue was subjected to silica gel (300 mesh, 50 × 8 cm, flow rate 10 mL/min) and eluted successively with 6 L of CHCl3/MeOH during each gradient (10:1→1:1) to give 10 fractions (Fr. 1–10). Fr. 7 (5 g) continues silica gel chromatography (300 mesh, 10 × 2 cm, flow rate 1 mL/min) eluted with 600 mL of CHCl3/MeOH during each gradient (5:1→1:1) to afford a number of sub-fractions A1-A13. Compounds 1 (15 mg, tR = 37.2 min) and 2 (21 mg, tR = 12.3 min) were obtained from the sub-fraction A4 (0.9 g) with MeOH/H2O (2:3) by prep. HPLC chromatography on a Hypersil-ODS II column (10 μm, 20 × 300 mm, flow rate 8 mL/min).
Baimantuoluoline G (
1). White amorphous powder, [α]
25D = +17.0 (c = 0.1, MeOH). IR (KBr): ν
max = 3426, 2925, 1708, 1387, 1286, 1135, 1089, 995 cm
−1. HRESIMS (positive):
m/z = 493.2578 (calc. for C
28H
38NaO
6, 493.2566, [M+Na]
+) and 509.2327 (calc. for C
28H
38KO
6, 509.2305, [M+K]
+).
1H- and
13C-NMR: see
Table 1.
Baimantuoluoside H (2): White amorphous powder, [α]
25D = +24 (c = 0.1, MeOH). IR (KBr): ν
max = 3411, 3326, 2942, 2923, 2865, 2590, 1670, 1070, 1030 cm
−1. HRESIMS (positive):
m/z = 673.3224 (calc. for C
34H
50NaO
12, 673.3200, [M+Na]
+) and 689.2948 (calc. for C
34H
50KO
12, 689.2939, [M+K]
+).
1H- and
13C-NMR: see
Table 1.