Catalytic Enantioselective Aryl Transfer to Aldehydes Using Chiral 2,2’-Bispyrrolidine-Based Salan Ligands
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental
3.1. General
3.2. Typical Procedure for the Asymmetric Aryl Transfer Reaction
4. Conclusions
Acknowledgements
References and Notes
- Shafi’ee, A.; Hite, G. Absolute configurations of the pheniramines, methyl phenidates, and pipradrols. J. Med. Chem. 1969, 12, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Ebnǒther, A.; Weber, H.P. Synthesis and absolute configuration of clemastine and its isomers. Helv. Chim. Acta 1976, 59, 2462–2468. [Google Scholar] [CrossRef] [PubMed]
- Meguro, K.; Aizawa, M.; Sohda, T.; Kawamatsu, A.; Nagaoka, A. New 1,4-dihydropyridine derivatives with potent and long-lasting hypotensive effect. Chem. Pharm. Bull. 1985, 33, 3787–3797. [Google Scholar] [CrossRef] [PubMed]
- Toda, F.; Tanaka, K.; Koshiro, K. A new preparative method for optically active diarylcarbinols. Tetrahedron Asymmetry 1991, 2, 873–874. [Google Scholar] [CrossRef]
- Casy, A.F.; Drake, A.F.; Ganellin, C.R.; Mercer, A.D.; Upton, C. Stereochemical studies of chiral H-1 antagonists of histamine: The resolution, chiral analysis, and biological evaluation of four antipodal pairs. Chirality 1992, 4, 356–366. [Google Scholar] [CrossRef] [PubMed]
- Stanev, S.; Rakovska, R.; Berova, N.; Snatzke, G. Synthesis, absolute configuration and circular dichroism of some diarylmethane derivatives. Tetrahedron Asymmetry 1995, 6, 183–198. [Google Scholar] [CrossRef]
- Botta, M.; Summa, V.; Corelli, F.; Di Pietro, G.; Lombardi, P. Synthesis of aryl 2-benzofuranyl and 2-indolyl carbinols of high enantiomeric purity via palladium-catalyzed heteroannulation of chiral arylpropargylic alcohols. Tetrahedron Asymmetry 1996, 7, 1263–1266. [Google Scholar] [CrossRef]
- Torrens, A.; Castrillo, J.A.; Claparols, A.; Redondo, J. Enantioselective synthesis of (R)- and (S)-cizolirtine. Application of oxazaborolidine-catalyzed asymmetric borane reduction to azolyl phenyl ketones. Synlett 1999, 765–767. [Google Scholar] [CrossRef]
- Corey, E.J.; Helal, C.J. Reduction of carbonyl compounds with chiral oxazaborolidine catalysts: A new paradigm for enantioselective catalysis and a powerful new synthetic method. Angew. Chem. Int. Ed. 1998, 37, 1986–2012. [Google Scholar] [CrossRef]
- Corey, E.J.; Bakshi, R.K.; Shibata, S. Highly enantioselective borane reduction of ketones catalyzed by chiral oxazaborolidines. Mechanism and synthetic implications. J. Am. Chem. Soc. 1987, 109, 5551–5553. [Google Scholar] [CrossRef]
- Corey, E.J. New enantioselective routes to biologically interesting compounds. Pure Appl. Chem. 1990, 62, 1209–1216. [Google Scholar] [CrossRef]
- Ohkuma, T.; Koizumi, M.; Ikehira, H.; Yokozawa, T.; Noyori, R. Selective hydrogenation of benzophenones to benzhydrols. Asymmetric synthesis of unsymmetrical diarylmethanols. Org. Lett. 2000, 2, 659–662. [Google Scholar] [CrossRef] [PubMed]
- Noyori, R.; Ohkuma, T. Rapid, productive and stereoselective hydrogenation of ketones. Pure Appl. Chem. 1999, 71, 1493–1501. [Google Scholar] [CrossRef]
- Pu, L.; Yu, H.-B. Catalytic asymmetric organozinc additions to carbonyl compounds. Chem. Rev. 2001, 101, 757–824. [Google Scholar] [CrossRef] [PubMed]
- Bolm, C.; Hildebrand, J.P.; Muńiz, K.; Hermanns, N. Catalyzed asymmetric arylation reactions. Angew. Chem. Int. Ed. 2001, 40, 3284–3308. [Google Scholar] [CrossRef]
- Schmidt, F.; Stemmler, R.T.; Rudolph, J.; Bolm, C. Catalytic asymmetric approaches towards enantiomerically enriched diarylmethanols and diarylmethylamines. Chem. Soc. Rev. 2006, 35, 454–470. [Google Scholar] [PubMed]
- Langer, F.; Schwink, L.; Devasagayaraj, A.; Chavant, P.-Y.; Knochel, P. Preparation of functionalized dialkylzincs via a boron-zinc exchange. Reactivity and catalytic asymmetric addition to aldehydes. J. Org. Chem. 1996, 61, 8229–8243. [Google Scholar] [CrossRef] [PubMed]
- Oppolzer, W.; Radinov, R.N.; Sayed, E.E. Catalytic asymmetric synthesis of macrocyclic (E)-allylic alcohols from ω-alkynals via intramolecular 1-alkenylzinc/aldehyde additions. J. Org. Chem. 2001, 66, 4766–4770. [Google Scholar] [CrossRef] [PubMed]
- Dahmen, S.; Brase, S. [2, 2] Paracyclophane-based N,O-ligands in alkenylzinc additions to Aldehydes. Org. Lett. 2001, 3, 4119–4122. [Google Scholar] [CrossRef] [PubMed]
- Bolm, C.; Rudolph, J. Catalyzed asymmetric aryl transfer reactions to aldehydes with boronic acids as aryl source. J. Am. Chem. Soc. 2002, 124, 14850–14851. [Google Scholar] [CrossRef] [PubMed]
- Rudolph, J.; Schmidt, F.; Bolm, C. Highly enantioselective synthesis of secondary alcohols using triphenylborane. Adv. Synth. Catal. 2004, 346, 867–872. [Google Scholar] [CrossRef]
- Braga, A.L.; Luedtke, D.S.; Vargas, F.; Paixao, M.W. Catalytic enantioselective arylation of aldehydes: Boronic acids as a suitable source of transferable aryl groups. Chem. Commun. 2005, 2512–2514. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.-Y.; Liu, X.-Y.; Zhao, G. Catalyzed asymmetric aryl transfer reactions to aldehydes with boroxines as aryl source. Tetrahedron Asymmetry 2005, 16, 2299–2305. [Google Scholar] [CrossRef]
- Liu, X.-Y.; Wu, X.-Y.; Chai, Z.; Wu, Y.-Y.; Zhao, G.; Zhu, S.-Z. Highly effective and recyclable dendritic ligands for the enantioselective aryl transfer reactions to aldehydes. J. Org. Chem. 2005, 70, 7432–7435. [Google Scholar] [CrossRef] [PubMed]
- Bolm, C.; Schmidt, F.; Zani, L. Synthesis of new chiral hydroxy oxazolines and their use in the catalytic asymmetric phenyl transfer to aldehydes. Tetrahedron Asymmetry 2005, 16, 1367–1376. [Google Scholar] [CrossRef]
- Dahmen, S.; Lormann, M. Triarylborane ammonia complexes as ideal precursors for arylzinc reagents in asymmetric catalysis. Org. Lett. 2005, 7, 4597–4600. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.-F.; Fang, L.; Lin, A.-J.; Pan, Y.; Zhu, C.-J. Highly efficient and facile aryl transfer to aldehydes using ArB(OH)2-GaMe3. Synlett 2009, 3, 495–499. [Google Scholar]
- Ji, J.-X.; Wu, J.; Au-Yeung, T.-T.-L.; Yip, C.W.; Haynes, R.K.; Chan, A.S.C. Highly enantioselective phenyl transfer to aryl aldehydes catalyzed by easily accessible chiral tertiary aminonaphthol. J. Org. Chem. 2005, 70, 1093–1095. [Google Scholar] [CrossRef] [PubMed]
- Ito, K.; Tomita, Y.; Katsuki, T. Enantioselective phenyl transfer to aldehydes using 1,1'-bi-2-naphthol-3,3'-dicarboxamide as chiral auxiliary. Tetrahedron Lett. 2005, 46, 6083–6086. [Google Scholar] [CrossRef]
- Braga, A.L.; Milani, P.; Vargas, F.; Paixāo, M.W.; Sehnem, J.A. Modular chiral thiazolidine catalysts in asymmetric aryl transfer reactions. Tetrahedron Asymmetry 2006, 17, 2793–2797. [Google Scholar] [CrossRef]
- Wu, P.-Y.; Wu, H.-L.; Uang, B.-J. Asymmetric synthesis of functionalized diarylmethanols catalyzed by a new γ-amino thiol. J. Org. Chem. 2006, 71, 833–835. [Google Scholar] [CrossRef] [PubMed]
- Lu, G.; Kwong, F.-Y.; Ruan, J.W.; Li, Y.-M.; Chan, A.S.C. Highly enantioselective addition of in situ prepared arylzinc to aldehydes catalyzed by a series of atropisomeric binaphthyl-derived amino alcohols. Chem. Eur. J. 2006, 12, 4115–4120. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.J.; Sarkar, S.M.; Lee, D.H.; Qiu, H.L. Highly enantioselective aryl transfer to aldehydes: A remarkable effect of sulfur substitution in amino thioacetate ligands. Org. Lett. 2008, 10, 1235–1237. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.-B.; Wu, L.-L.; Xu, J.-Q.; Zong, L.-L.; Hu, H.-W.; Cheng, Y.-X. Enantioselective arylation of aldehydes catalyzed by a soluble optically active polybinaphthols ligand. Tetrahedron Lett. 2008, 49, 6823–6826. [Google Scholar] [CrossRef]
- DeBerardinis, A.M.; Turlington, M.; Pu, L. Activation of functional arylzincs prepared from aryl iodides and highly enantioselective addition to aldehydes. Org. Lett. 2008, 10, 2709–2712. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.-C.; Wang, X.-D.; Ding, X.; Liu, Z.-K. Catalytic asymmetric aryl transfer: Highly enantioselective preparation of (R)- and (S)-diarylmethanols catalyzed by the same chiral ferrocenyl aziridino alcohol. Tetrahedron 2008, 64, 2559–2564. [Google Scholar] [CrossRef]
- Ruan, J.-W.; Lu, G.; Xu, L.-J.; Li, Y.-M.; Chan, A.S.C. Catalytic asymmetric alkynylation and arylation of aldehydes by an H8-binaphthyl-based amino alcohol ligand. Adv. Synth. Catal. 2008, 350, 76–84. [Google Scholar] [CrossRef]
- Wang, M.-C.; Zhang, Q.-J.; Zhao, W.-X.; Wang, X.-D.; Ding, X.; Jing, T.-T.; Song, M.-P. N-(Ferrocenylmethyl)azetidin-2-yl(diphenyl)methanol for catalytic asymmetric addition of organozinc reagents to aldehydes. J. Org. Chem. 2008, 73, 168–176. [Google Scholar] [CrossRef] [PubMed]
- Braga, A.L.; Paixa, M.W.; Westermann, B.; Schneider, P.H.; Wessjohann, L.A. Acceleration of arylzinc formation and its enantioselective addition to aldehydes by microwave irradiation and aziridine-2-methanol catalysts. J. Org. Chem. 2008, 73, 2879–2882. [Google Scholar] [CrossRef] [PubMed]
- Salvi, L.; Kim, J.G.; Walsh, P.J. Practical catalytic asymmetric synthesis of diaryl-, aryl heteroaryl-, and diheteroarylmethanols. J. Am. Chem. Soc. 2009, 131, 12483–12493. [Google Scholar] [CrossRef] [PubMed]
- Wouters, A.D.; Trossini, G.H.G.; Stefani, H.A.; Lüdtke, D.S. Enantioselective arylations catalyzed by carbohydrate-based chiral amino alcohols. Eur. J. Org. Chem. 2010, 2351–2356. [Google Scholar] [CrossRef]
- Alexakis, A.; Mangeney, P. Advanced Asymmetric Synthesis; Stephenson, G.R., Ed.; Chapman & Hall: London, UK, 1996; Chapter 5; p. 93. [Google Scholar]
- Bennani, Y.L.; Hannessian, S. Trans-1,2-diaminocyclohexane derivatives as chiral reagents, scaffolds, and ligands for catalysis: Applications in asymmetric synthesis and molecular recognition. Chem. Rev. 1997, 97, 3161–3195. [Google Scholar] [CrossRef] [PubMed]
- Lucet, D.; Le Gall, T.; Mioskowski, C. The chemistry of vicinal diamines. Angew. Chem. Int. Ed. 1998, 37, 2580–2627. [Google Scholar] [CrossRef]
- Hirama, M.; Oishi, T.; Ito, S. Asymmetric dihydroxylation of alkenes with osmium tetroxide: Chiral N, N'-dialkyl-2,2'-bipyrrolidine complex. Chem. Commun. 1989, 10, 665–666. [Google Scholar] [CrossRef]
- Oishi, T.; Hirama, M. Highly enantioselective dihydroxylation of trans-disubstituted and monosubstituted olefins. J. Org. Chem. 1989, 54, 5834–5835. [Google Scholar] [CrossRef]
- Oishi, T.; Hirama, M.; Sita, L.R.; Masamune, S. Synthesis of chiral 2, 2'-bipyrrolidine derivatives. Synthesis 1991, 789–792. [Google Scholar] [CrossRef]
- Kotsuki, H.; Kuzume, H.; Gohda, T.; Fukuhara, M.; Ochi, M.; Oishi, T.; Hirama, M.; Shirot, M. New convenient, enantiospecific synthesis of (S, S)- and (R, R)-2,2'-bipyrrolidine derivatives. Tetrahedron Asymmetry 1995, 6, 2227–2236. [Google Scholar] [CrossRef]
- Alexakis, A.; Tomassini, A.; Chouillet, C.; Roland, S.; Mangeney, P.; Bernardinelli, G. A new efficient synthesis of (R, R)-2,2'-bipyrrolidine: An interesting chiral 1,2-diamine with C2 symmetry. Angew. Chem. Int. Ed. 2000, 39, 4093–4095. [Google Scholar] [CrossRef]
- Denmark, S.E.; Fu, J.-P.; Lawler, M.J. (R, R)-2,2'- bipyrrolidine and (S, S)-2,2'-bipyrrolidine: Useful ligands for asymmetric synthesis. Org. Synth. 2006, 83, 121–130. [Google Scholar]
- Denmark, S.E.; Fu, J.-P. Catalytic, Enantioselective addition of substituted allylic trichlorosilanes using a rationally-designed 2,2'-bispyrrolidine-based bisphosphoramide. J. Am. Chem. Soc. 2001, 123, 9488–9489. [Google Scholar] [CrossRef] [PubMed]
- Alexakis, A.; Andrey, O. Diamine-catalyzed asymmetric michael additions of aldehydes and ketones to nitrostyrene. Org. Lett. 2002, 4, 3611–3614. [Google Scholar] [CrossRef] [PubMed]
- Andrey, O.; Alexakis, A.; Bernardinelli, G. Asymmetric michael addition of α-Hydroxyketones to nitroolefins catalyzed by chiral diamine. Org. Lett. 2003, 5, 2559–2561. [Google Scholar] [CrossRef] [PubMed]
- Andrey, O.; Alexakis, A.; Tomassini, A.; Bernardinelli, G. The use of N-alkyl-2,2’-bipyrrolidine derivatives as organocatalysts for the asymmetric michael addition of ketones and aldehydes to nitroolefins. Adv. Synth. Catal. 2004, 346, 1147–1168. [Google Scholar] [CrossRef]
- Mossé, S.; Alexakis, A. First organocatalyzed asymmetric michael addition of aldehydes to vinyl Sulfones. Org. Lett. 2005, 7, 4361–4364. [Google Scholar] [CrossRef] [PubMed]
- Alexakis, A.; Tomassini, A.; Andrey, O.; Bernardinelli, G. Diastereoselective alkylation of (arene)tricarbonylchromium and ferrocene complexes using a chiral, C2-symmetrical 1,2-diamine as auxiliary. Eur. J. Org. Chem. 2005, 1332–1339. [Google Scholar] [CrossRef]
- Denmark, S.E.; Fu, J.-P.; Lawler, M.J. Chiral phosphoramide-catalyzed enantioselective addition of allylic trichlorosilanes to aldehydes. Preparative studies with bidentate phosphorus-based amides. J. Org. Chem. 2006, 71, 1523–1536. [Google Scholar] [CrossRef] [PubMed]
- Chen, M.S.; White, M.C. A predictably selective aliphatic C-H oxidation reaction for complex molecule synthesis. Science 2007, 318, 783–787. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, K.; Oldenburg, P.D.; Que, L., Jr. Iron-catalyzed asymmetric olefin cis-dihydroxylation with 97% enantiomeric excess. Angew. Chem. Int. Ed. 2008, 47, 1887–1889. [Google Scholar] [CrossRef] [PubMed]
- Sergeeva, E.; Kopilov, J.; Goldberg, I.; Kol, M. Salan ligands assembled around chiral bipyrrolidine: Predetermination of chirality around octahedral Ti and Zr centres. Chem. Commun. 2009, 21, 3053–3055. [Google Scholar] [CrossRef] [PubMed]
- Sergeeva, E.; Kopilov, J.; Goldberg, I.; Kol, M. 2,2'-Bipyrrolidine versus 1,2-diaminocyclohexane as chiral cores for helically wrapping diamine-diolate ligands. Inorg. Chem. 2009, 48, 8075–8077. [Google Scholar] [CrossRef] [PubMed]
- Jia, Y.-X.; Zhu, S.-F.; Yang, Y.; Zhou, Q.-L. Asymmetric friedel-crafts alkylations of indoles with nitroalkenes catalyzed by Zn(II)-bisoxazoline complexes. J. Org. Chem. 2006, 71, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Arai, T.; Yokoyama, N. Tandem Catalytic Asymmetric friedel-crafts/henry reaction: Control of three contiguous acyclic stereocenters. Angew. Chem. Int. Ed. 2008, 47, 4989–4992. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |
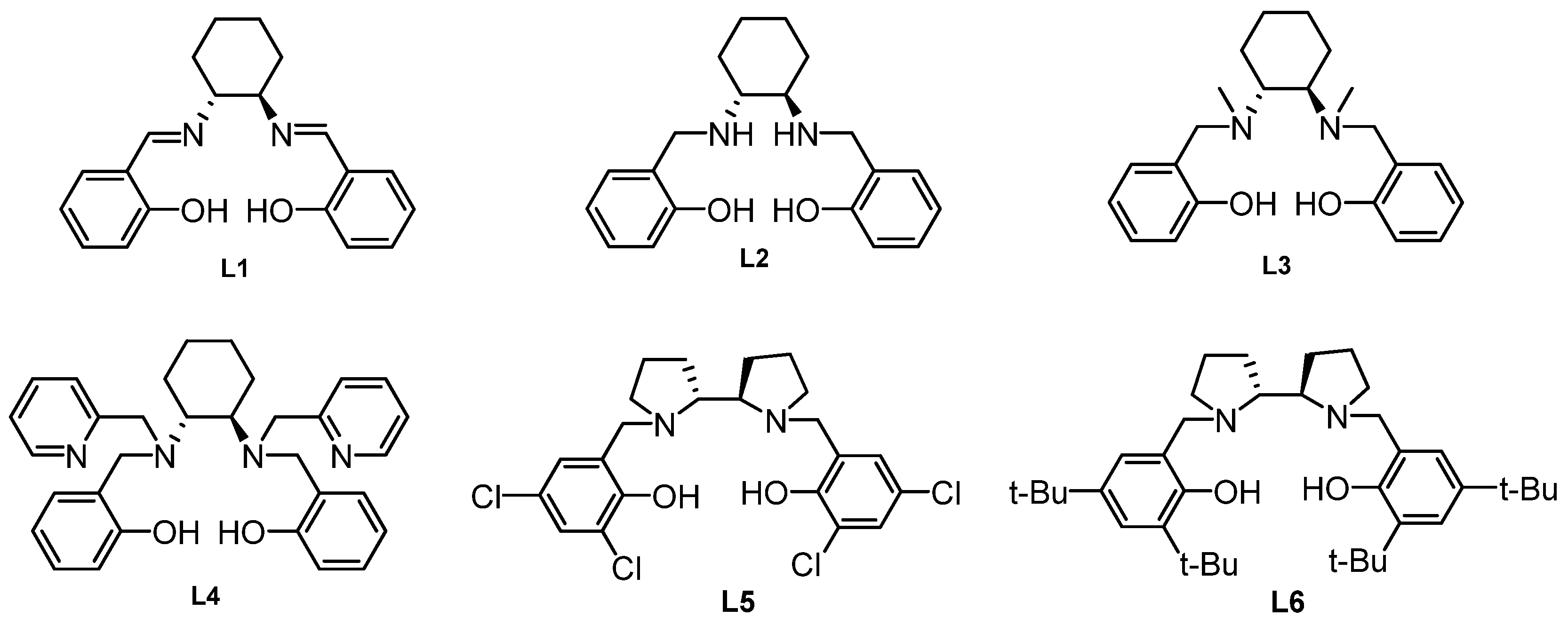
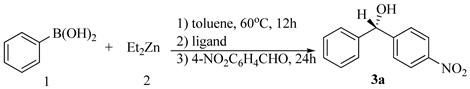
| Entry | Ligand | Mol% | T(°C) | Yield(%)b | Ee(%)c |
|---|---|---|---|---|---|
| 1 | L1 | 10 | 0 | 66 | 6 |
| 2 | L2 | 10 | 0 | 73 | 11 |
| 3 | L3 | 10 | 0 | 69 | 16 |
| 4 | L4 | 10 | 0 | 80 | 3 |
| 5 | L5 | 10 | 0 | 74 | 31 |
| 6 | L6 | 10 | 0 | 84 | 43 |
| 7 | L6 | 10 | -25 | 70 | 63 |
| 8 | L6 | 15 | -25 | 80 | 71 |
| 9 | L6 | 20 | -25 | 88 | 83(S) d |
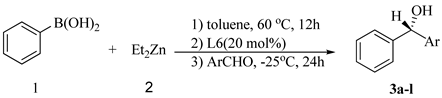
| Entry | Ar | Product | Yield (%)b | Ee (%)c,d |
|---|---|---|---|---|
| 1 | 4-NO2C6H4 | 3a | 88 | 83(S) |
| 2 | 4-ClC6H4 | 3b | 80 | 41(S) |
| 3 | 3-NO2C6H4 | 3c | 91 | 75(S) |
| 4 | 2-NO2C6H4 | 3d | 85 | 41(R) |
| 5 | 2-CF3C6H4 | 3e | 72 | 80(R) |
| 6 | 2-ClC6H4 | 3f | 84 | 73(S) |
| 7 | 3-BrC6H4 | 3g | 85 | 26(S) |
| 8 | 2-MeC6H4 | 3h | 76 | 60(S) |
| 9 | 3-MeC6H4 | 3i | 78 | 52(S) |
| 10 | 4-MeOC6H4 | 3j | 80 | 11(S) |
| 11 | 2-C10H7 | 3k | 80 | 70(S) |
| 12 | PhCH=CH | 3l | 76 | 47(R) |
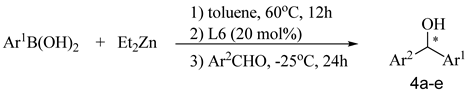
| Entry | Ar1 | Ar2 | Product | Yield (%)b | Ee (%)c |
|---|---|---|---|---|---|
| 1 | 4-Cl C6H4 | 4-NO2C6H4 | 4a | 75 | 71 |
| 2 | 4-Cl C6H4 | 3-BrC6H4 | 4b | 82 | 55 |
| 3 | 4-MeOC6H4 | 4-NO2C6H4 | 4c | 78 | 54(S)d |
| 4 | 4-MeOC6H4 | 3-NO2C6H4 | 4d | 70 | 24 |
| 5 | 3,5-diMeC6H3 | C6H5 | 4e | 70 | 48 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Jia, X.; Lin, A.; Mao, Z.; Zhu, C.; Cheng, Y. Catalytic Enantioselective Aryl Transfer to Aldehydes Using Chiral 2,2’-Bispyrrolidine-Based Salan Ligands. Molecules 2011, 16, 2971-2981. https://doi.org/10.3390/molecules16042971
Jia X, Lin A, Mao Z, Zhu C, Cheng Y. Catalytic Enantioselective Aryl Transfer to Aldehydes Using Chiral 2,2’-Bispyrrolidine-Based Salan Ligands. Molecules. 2011; 16(4):2971-2981. https://doi.org/10.3390/molecules16042971
Chicago/Turabian StyleJia, Xuefeng, Aijun Lin, Zhijie Mao, Chengjian Zhu, and Yixiang Cheng. 2011. "Catalytic Enantioselective Aryl Transfer to Aldehydes Using Chiral 2,2’-Bispyrrolidine-Based Salan Ligands" Molecules 16, no. 4: 2971-2981. https://doi.org/10.3390/molecules16042971




