Antioxidant Therapy as a Potential Approach to Severe Influenza-Associated Complications
Abstract
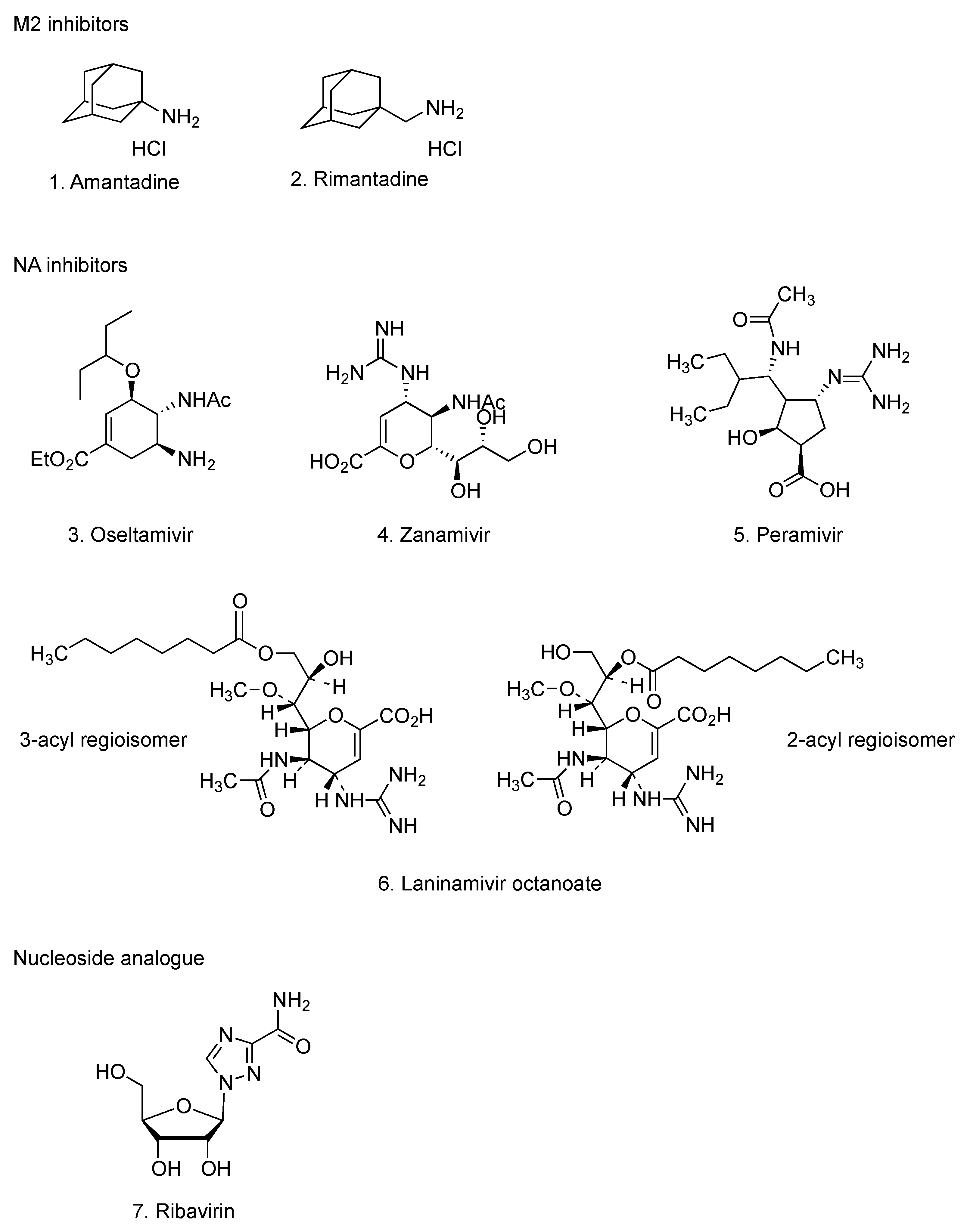
1. Introduction
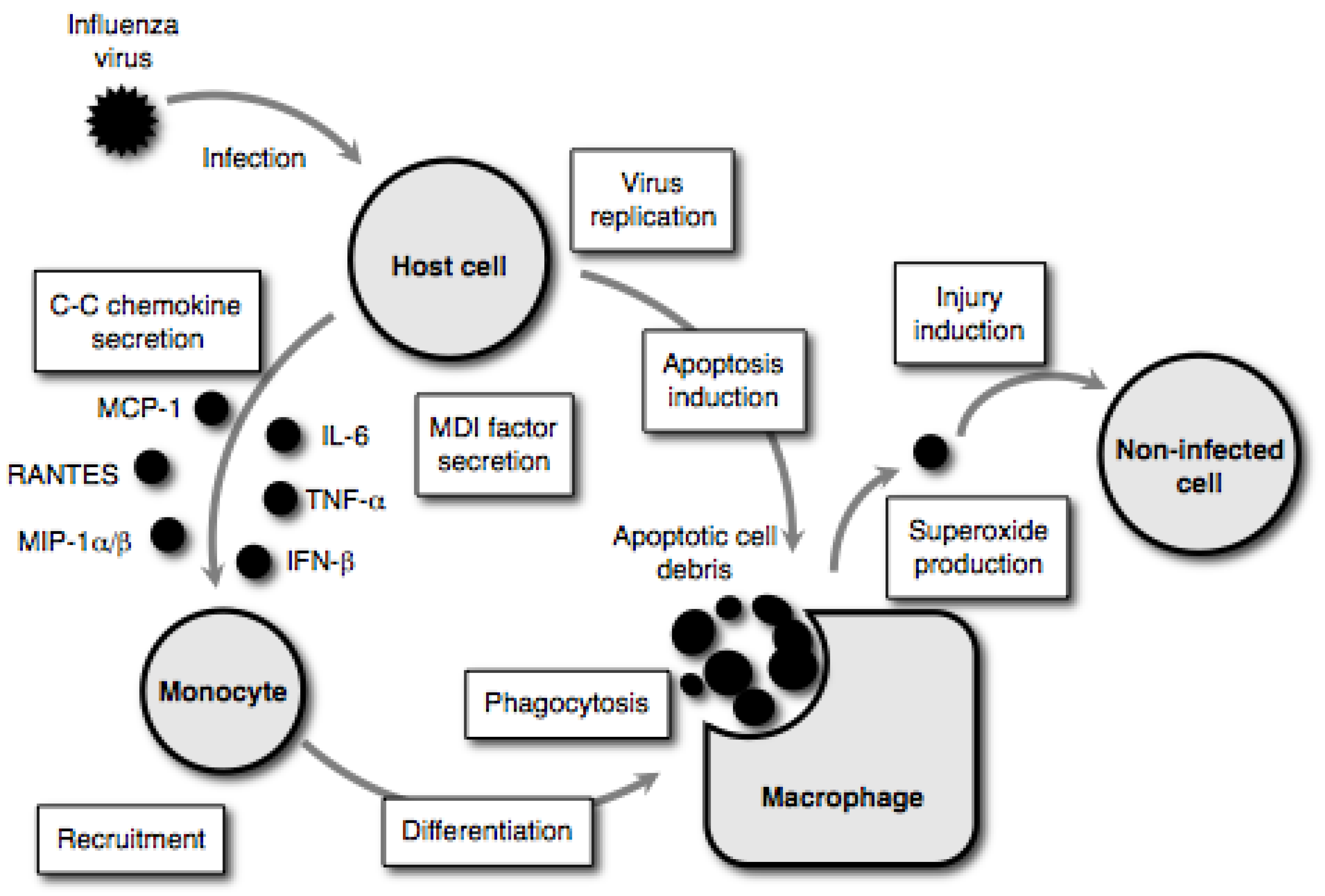
2. Effect of Selected Antioxidants on the Pathogenesis of Influenza Virus Infections
2.1. Superoxide dismutases
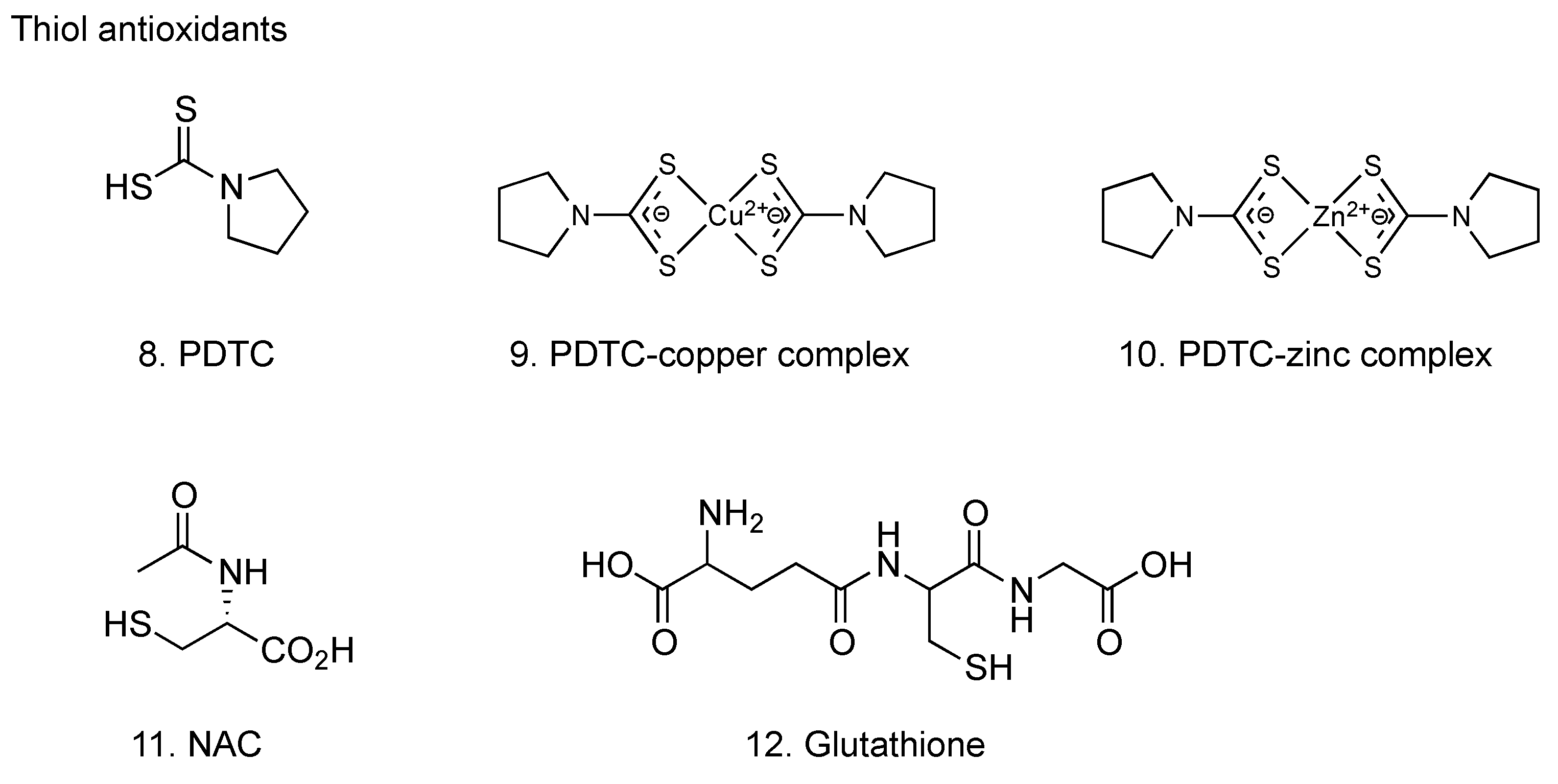
2.2. Thiol antioxidants
2.2.1. Pyrrolidine dithiocarbamate
2.2.2. N-Acetyl-l-cysteine
2.2.3. Glutathione
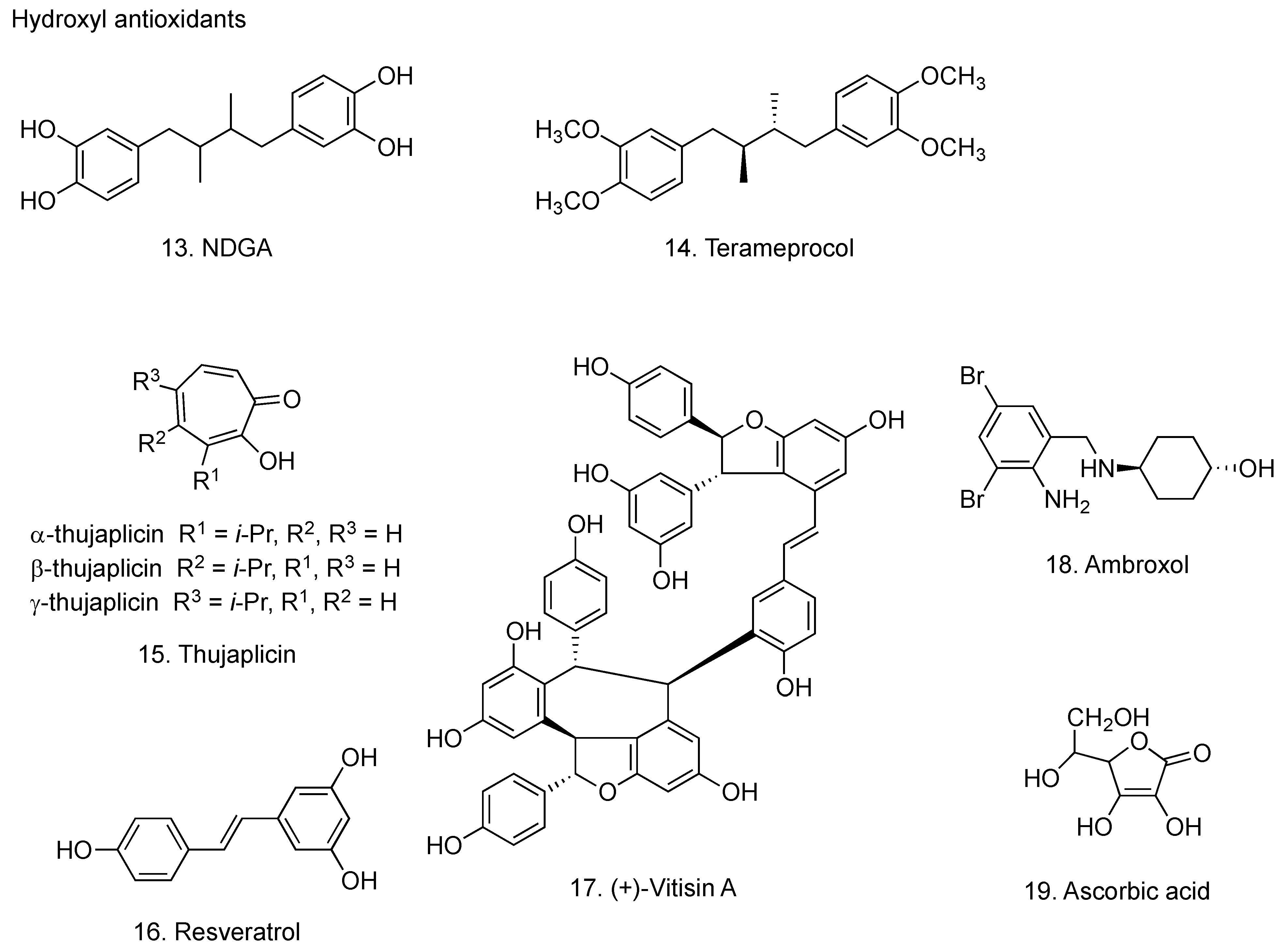
2.3. Hydroxyl antioxidants
2.3.1. Nordihydroguaiaretic acid
2.3.2. Thujaplicin
2.3.3. Resveratrol
2.3.4. Ambroxol
2.3.5. Ascorbic acid
2.4. Flavonoids
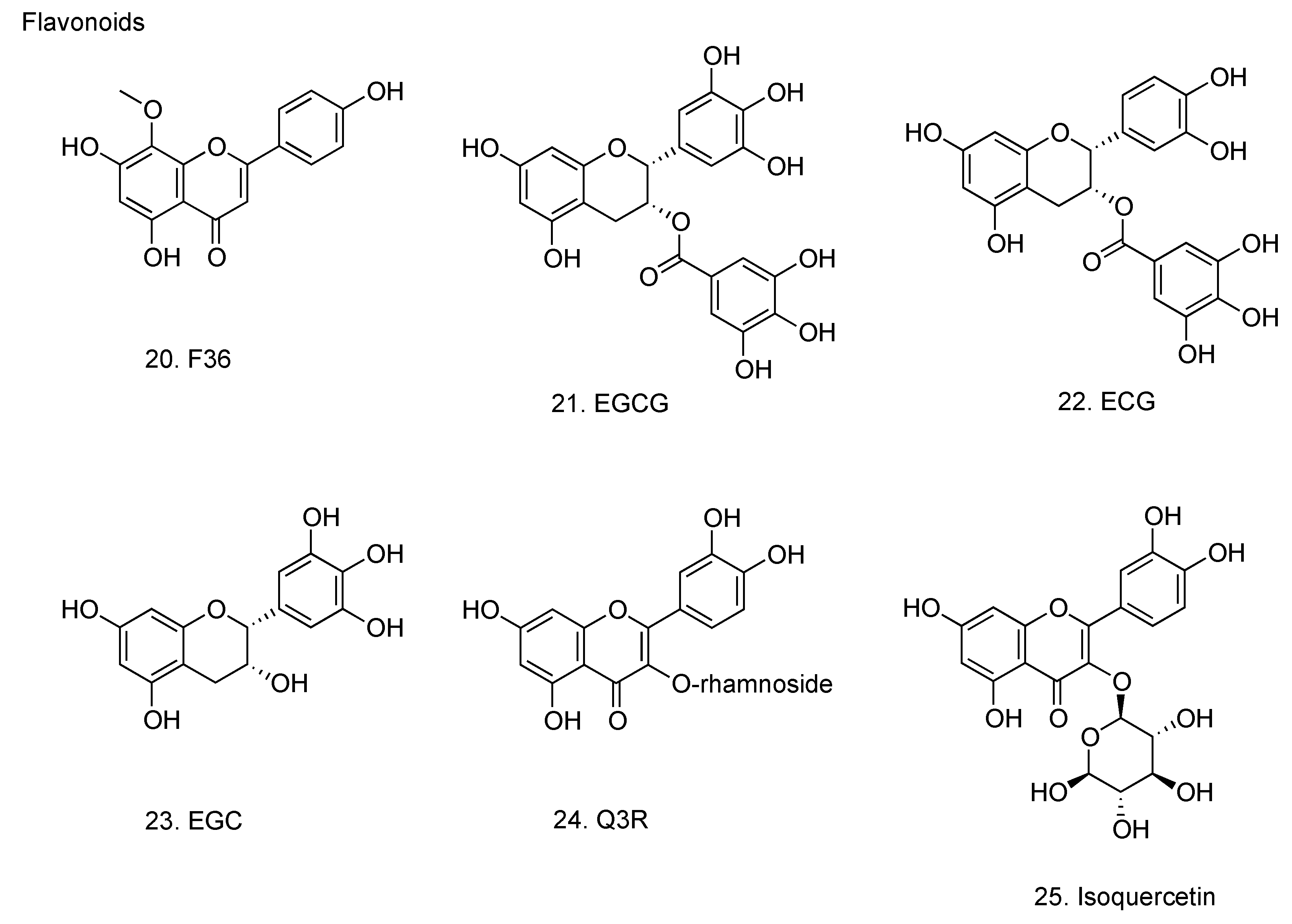
2.4.1. 5,7,4’-Trihydroxy-8-methoxyflavone
2.4.2. Catechins
2.4.3. Quercetin 3-rhamnoside
2.4.4. Isoquercetin
2.4.5. Oligonol
3. Conclusions
Acknowledgements
References
- Peiris, J.S.; Tu, W.W.; Yen, H.L. A novel H1N1 virus causes the first pandemic of the 21st century. Eur. J. Immunol. 2009, 39, 2946–2954. [Google Scholar] [CrossRef] [PubMed]
- Chang, L.Y.; Shih, S.R.; Shao, P.L.; Huang, D.T.; Huang, L.M. Novel swine-origin influenza virus A (H1N1): The first pandemic of the 21st century. J. Formos. Med. Assoc. 2009, 108, 526–532. [Google Scholar] [CrossRef]
- Rothberg, M.B.; Haessler, S.D. Complications of seasonal and pandemic influenza. Crit. Care Med. 2010, 38, e91–e97. [Google Scholar] [CrossRef] [PubMed]
- Girard, M.P.; Tam, J.S.; Assossou, O.M.; Kieny, M.P. The 2009 A (H1N1) influenza virus pandemic: A review. Vaccine 2010, 28, 4895–4902. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Pandemic (H1N1) 2009 - update 111. Available online: http://www.who.int/csr/ don/2010_07_30/en/index.html (Accessed on 26 February 2011).
- World Health Organization. Influenza updates. Available online: http://www.who.int/csr/don/2010_09_10/ en/index.html (Accessed on 26 February 2011).
- Kamps, B.S.; Reyes-Terán, G. Influenza. In Influenza Report; Kamps, B.S., Hoffmann, C., Preiser, W., Eds.; Flying Publishers: Paris, France, 2006; pp. 17–47. [Google Scholar]
- Beigel, J.; Bray, M. Current and future antiviral therapy of severe seasonal and avian influenza. Antivir. Res. 2008, 78, 91–102. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). Update: Drug susceptibility of swine-origin influenza A (H1N1) viruses, April 2009. MMWR Morb. Mortal. Wkly. Rep. 2009, 58, 433–435. [Google Scholar]
- World Health Organization. Pandemic (H1N1) 2009 - update 60. Available online: http://www.who.int/csr/ don/2009_08_04/en/index.html (Accessed on 26 February 2011).
- World Health Organization. Update on oseltamivir-resistant pandemic A (H1N1) 2009 influenza virus: January 2010. Wkly. Epidemiol. Rec. 2010, 85, 37–40. [Google Scholar]
- Sugaya, N.; Ohashi, Y. Long-acting neuraminidase inhibitor laninamivir octanoate (CS-8958) versus oseltamivir as treatment for children with influenza virus infection. Antimicrob. Agents Chemother. 2010, 54, 2575–2582. [Google Scholar] [CrossRef] [PubMed]
- Bearman, G.M.; Shankaran, S.; Elam, K. Treatment of severe cases of pandemic (H1N1) 2009 influenza: Review of antivirals and adjuvant therapy. Recent Patents Antiinfect. Drug Discov. 2010, 5, 152–156. [Google Scholar]
- Nasu, T.; Ogawa, D.; Wada, J.; Makino, H. Peramivir for severe influenza infection in a patient with diabetic nephropathy. Am. J. Respir. Crit. Care Med. 2010, 182, 1209–1210. [Google Scholar] [CrossRef] [PubMed]
- Renaud, C.; Pergam, S.A.; Polyak, C.; Jain, R.; Kuypers, J.; Englund, J.A.; Corey, L.; Boeckh, M.J. Early emergence of an H275Y mutation in a hematopoietic cell transplant recipient treated with intravenous peramivir. Transpl. Infect. Dis. 2010, 12, 513–517. [Google Scholar] [CrossRef] [PubMed]
- Cunha, B.A.; Syed, U.; Mickail, N. Fulminant fatal swine influenza (H1N1): Myocarditis, myocardial infarction, or severe influenza pneumonia? Heart Lung 2010, 39, 453–458. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, J.T.; Hoopes, J.D.; Le, M.H.; Smee, D.F.; Patick, A.K.; Faix, D.J.; Blair, P.J.; de Jong, M.D.; Prichard, M.N.; Went, G.T. Triple combination of amantadine, ribavirin, and oseltamivir is highly active and synergistic against drug resistant influenza virus strains in vitro. PLoS One 2010, 5, e9332. [Google Scholar] [CrossRef] [PubMed]
- Tumpey, T.M.; García-Sastre, A.; Taubenberger, J.K.; Palese, P.; Swayne, D.E.; Pantin-Jackwood, M.J.; Schultz-Cherry, S.; Solórzano, A.; Van Rooijen, N.; Katz, J.M.; Basler, C.F. Pathogenicity of influenza viruses with genes from the 1918 pandemic virus: Functional roles of alveolar macrophages and neutrophils in limiting virus replication and mortality in mice. J. Virol. 2005, 79, 14933–14944. [Google Scholar] [CrossRef] [PubMed]
- Uchide, N.; Toyoda, H. Current status of monocyte differentiation-inducing (MDI) factors derived from human fetal membrane chorion cells undergoing apoptosis after influenza virus infection. Gene Regul. Syst. Biol. 2007, 1, 295–302. [Google Scholar] [CrossRef]
- Akaike, T.; Suga, M.; Maeda, H. Free radicals in viral pathogenesis: Molecular mechanisms involving superoxide and NO. Proc. Soc. Exp. Biol. Med. 1998, 217, 64–73. [Google Scholar] [CrossRef] [PubMed]
- Uchide, N.; Toyoda, H. Potential of selected antioxidants for influenza chemotherapy. Anti-Infect. Agents Med. Chem. 2008, 7, 73–83. [Google Scholar] [CrossRef]
- Uchide, N.; Toyoda, H. Future target molecules for influenza treatment. Mini-Rev. Med. Chem. 2008, 8, 491–495. [Google Scholar] [CrossRef] [PubMed]
- Oda, T.; Akaike, T.; Hamamoto, T.; Suzuki, F.; Hirano, T.; Maeda, H. Oxygen radicals in influenza-induced pathogenesis and treatment with pyran polymer-conjugated SOD. Science 1989, 244, 974–976. [Google Scholar] [CrossRef] [PubMed]
- Sidwell, R.W.; Huffman, J.H.; Bailey, K.W.; Wong, M.H.; Nimrod, A.; Panet, A. Inhibitory effects of recombinant manganese superoxide dismutase on influenza virus infections in mice. Antimicrob. Agents Chemother. 1996, 40, 2626–2631. [Google Scholar] [PubMed]
- Serkedjieva, J.; Roeva, I.; Angelova, M.; Dolashka, P.; Voelter, W.G. Combined protective effect of a fungal Cu/Zn-containing superoxide dismutase and rimantadine hydrochloride in experimental murine influenza a virus infection. Acta Virol. 2003, 47, 53–56. [Google Scholar] [PubMed]
- Akaike, T.; Ando, M.; Oda, T.; Doi, T.; Ijiri, S.; Araki, S.; Maeda, H. Dependence on O2- generation by xanthine oxidase of pathogenesis of influenza virus infection in mice. J. Clin. Invest. 1990, 85, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Suliman, H.B.; Ryan, L.K.; Bishop, L.; Folz, R.J. Prevention of influenza-induced lung injury in mice overexpressing extracellular superoxide dismutase. Am. J. Physiol. Lung Cell Mol. Physiol. 2001, 280, L69–L78. [Google Scholar] [CrossRef] [PubMed]
- Snelgrove, R.J.; Edwards, L.; Rae, A.J.; Hussell, T. An absence of reactive oxygen species improves the resolution of lung influenza infection. Eur. J. Immunol. 2006, 36, 1364–1373. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Niculescu, R.; Wang, D.; Patel, S.; Davenpeck, K.L.; Zalewski, A. Increased NAD(P)H oxidase and reactive oxygen species in coronary arteries after balloon injury. Arterioscler. Thromb. Vasc. Biol. 2001, 21, 739–745. [Google Scholar] [CrossRef] [PubMed]
- Uchide, N.; Ohyama, K. Antiviral function of pyrrolidine dithiocarbamate against influenza virus: The inhibition of viral gene replication and transcription. J. Antimicrob. Chemother. 2003, 52, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Uchide, N.; Ohyama, K.; Bessho, T.; Yuan, B.; Yamakawa, T. Effect of antioxidants on apoptosis induced by influenza virus infection: Inhibition of viral gene replication and transcription with pyrrolidine dithiocarbamate. Antivir. Res. 2002, 56, 207–217. [Google Scholar] [CrossRef]
- Knobil, K.; Choi, A.M.; Weigand, G.W.; Jacoby, D.B. Role of oxidants in influenza virus-induced gene expression. Am. J. Physiol. 1998, 274, L134–L142. [Google Scholar] [CrossRef] [PubMed]
- Lowy, R.J.; Dimitrov, D.S. Characterization of influenza virus-induced death of J774.1 macrophages. Exp. Cell Res. 1997, 234, 249–258. [Google Scholar] [CrossRef] [PubMed]
- McKinney, L.C.; Galliger, S.J.; Lowy, R.J. Active and inactive influenza virus induction of tumor necrosis factor-α and nitric oxide in J774.1 murine macrophages: Modulation by interferon-γ and failure to induce apoptosis. Virus Res. 2003, 97, 117–126. [Google Scholar] [CrossRef] [PubMed]
- Chen, K.; Long, Y.M.; Wang, H.; Lan, L.; Lin, Z.H. Activation of nuclear factor-κB and effects of pyrrolidine dithiocarbamate on TNBS-induced rat colitis. World J. Gastroenterol. 2005, 11, 1508–1514. [Google Scholar] [CrossRef] [PubMed]
- Cuzzocrea, S.; Chatterjee, P.K.; Mazzon, E.; Dugo, L.; Serraino, I.; Britti, D.; Mazzullo, G.; Caputi, A.P.; Thiemermann, C. Pyrrolidine dithiocarbamate attenuates the development of acute and chronic inflammation. Br. J. Pharmacol. 2002, 135, 496–510. [Google Scholar] [CrossRef] [PubMed]
- Mallick, I.H.; Yang, W.X.; Winslet, M.C.; Seifalian, A.M. Pyrrolidine dithiocarbamate reduces ischemia-reperfusion injury of the small intestine. World J. Gastroenterol. 2005, 11, 7308–7313. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.H.; Fang, I.M.; Lin, C.P.; Yang, C.M.; Chen, M.S. Effects of the NF-κB inhibitor pyrrolidine dithiocarbamate on experimentally induced autoimmune anterior uveitis. Invest. Ophthalmol. Vis. Sci. 2005, 46, 1339–1347. [Google Scholar] [CrossRef] [PubMed]
- Kim, C.H.; Kim, J.H.; Hsu, C.Y.; Ahn, Y.S. Zinc is required in pyrrolidine dithiocarbamate inhibition of NF-κB activation. FEBS Lett. 1999, 449, 28–32. [Google Scholar] [CrossRef]
- Oxford, J.S.; Perrin, D.D. Inhibition of the particle-associated RNA-dependent RNA polymerase activity of influenza viruses by chelating agents. J. Gen. Virol. 1974, 23, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Si, X.; McManus, B.M.; Zhang, J.; Yuan, J.; Cheung, C.; Esfandiarei, M.; Suarez, A.; Morgan, A.; Luo, H. Pyrrolidine dithiocarbamate reduces coxsackievirus B3 replication through inhibition of the ubiquitin-proteasome pathway. J. Virol. 2005, 79, 8014–8023. [Google Scholar] [CrossRef] [PubMed]
- Krenn, B.M.; Holzer, B.; Gaudernak, E.; Triendl, A.; van Kuppeveld, F.J.; Seipelt, J. Inhibition of polyprotein processing and RNA replication of human rhinovirus by pyrrolidine dithiocarbamate involves metal ions. J. Virol. 2005, 79, 13892–13899. [Google Scholar] [CrossRef] [PubMed]
- Uchide, N.; Ohyama, K.; Bessho, T.; Toyoda, H. Effects of mitogen-activated protein kinase inhibitors on tumor necrosis factor-α gene expression and apoptosis induction in cultured human fetal membrane chorion cells infected with influenza virus. Intervirology 2007, 50, 99–107. [Google Scholar] [CrossRef] [PubMed]
- Chiou, W.F.; Chen, C.C.; Wei, B.L. 8-Prenylkaempferol suppresses influenza A virus-induced RANTES production in A549 cells via blocking PI3K-mediated transcriptional activation of NF-κB and IRF3. Evid. Based Complement. Altern. Med. 2009. In Press. [Google Scholar] [CrossRef] [PubMed]
- Kelly, G.S. Clinical applications of N-acetylcysteine. Altern. Med. Rev. 1998, 3, 114–127. [Google Scholar] [PubMed]
- Saito, T.; Tanaka, M.; Yamaguchi, I. Effect of brefeldin A on influenza A virus-induced apoptosis in vitro. J. Vet. Med. Sci. 1996, 58, 1137–1139. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.; Zimmer, S.G.; Lu, Z.; Holland, R.E., Jr.; Dong, Q.; Chambers, T.M. The involvement of a stress-activated pathway in equine influenza virus-mediated apoptosis. Virology 2001, 287, 202–213. [Google Scholar] [CrossRef] [PubMed]
- Geiler, J.; Michaelis, M.; Naczk, P.; Leutz, A.; Langer, K.; Doerr, H.W.; Cinatl, J., Jr. N-acetyl-l-cysteine (NAC) inhibits virus replication and expression of pro-inflammatory molecules in A549 cells infected with highly pathogenic H5N1 influenza A virus. Biochem. Pharmacol. 2009, 79, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Ungheri, D.; Pisani, C.; Sanson, G.; Bertani, A.; Schioppacassi, G.; Delgado, R.; Sironi, M.; Ghezzi, P. Protective effect of N-acetylcysteine in a model of influenza infection in mice. Int. J. Immunopathol. Pharmacol. 2000, 13, 123–128. [Google Scholar] [PubMed]
- Ghezzi, P.; Ungheri, D. Synergistic combination of N-acetylcysteine and ribavirin to protect from lethal influenza viral infection in a mouse model. Int. J. Immunopathol. Pharmacol. 2004, 17, 99–102. [Google Scholar] [CrossRef] [PubMed]
- Garozzo, A.; Tempera, G.; Ungheri, D.; Timpanaro, R.; Castro, A. N-acetylcysteine synergizes with oseltamivir in protecting mice from lethal influenza infection. Int. J. Immunopathol. Pharmacol. 2007, 20, 349–354. [Google Scholar] [CrossRef] [PubMed]
- De Flora, S.; Grassi, C.; Carati, L. Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur. Respir. J. 1997, 10, 1535–1541. [Google Scholar] [CrossRef] [PubMed]
- Lai, K.Y.; Ng, W.Y.; Osburga Chan, P.K.; Wong, K.F.; Cheng, F. High-dose N-acetylcysteine therapy for novel H1N1 influenza pneumonia. Ann. Intern. Med. 2010, 152, 687–688. [Google Scholar] [CrossRef] [PubMed]
- Cai, J.; Chen, Y.; Seth, S.; Furukawa, S.; Compans, R.W.; Jones, D.P. Inhibition of influenza infection by glutathione. Free Radic. Biol. Med. 2003, 34, 928–936. [Google Scholar] [CrossRef]
- Nencioni, L.; Iuvara, A.; Aquilano, K.; Ciriolo, M.R.; Cozzolino, F.; Rotilio, G.; Garaci, E.; Palamara, A.T. Influenza A virus replication is dependent on an antioxidant pathway that involves GSH and Bcl-2. FASEB J. 2003, 17, 758–760. [Google Scholar] [CrossRef] [PubMed]
- Floriano-Sanchez, E.; Villanueva, C.; Medina-Campos, O.N.; Rocha, D.; Sanchez-Gonzalez, D.J.; Cardenas-Rodriguez, N.; Pedraza-Chaverri, J. Nordihydroguaiaretic acid is a potent in vitro scavenger of peroxynitrite, singlet oxygen, hydroxyl radical, superoxide anion and hypochlorous acid and prevents in vivo ozone-induced tyrosine nitration in lungs. Free Radic. Res. 2006, 40, 523–533. [Google Scholar] [CrossRef] [PubMed]
- Lü, J.M.; Nurko, J.; Weakley, S.M.; Jiang, J.; Kougias, P.; Lin, P.H.; Yao, Q.; Chen, C. Molecular mechanisms and clinical applications of nordihydroguaiaretic acid (NDGA) and its derivatives: An update. Med. Sci. Monit. 2010, 16, RA93–RA100. [Google Scholar] [PubMed]
- Uchide, N.; Ohyama, K.; Bessho, T.; Toyoda, H. Inhibition of influenza virus-induced apoptosis in chorion cells of human fetal membranes by nordihydroguaiaretic acid. Intervirology 2005, 48, 336–340. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Andrea, B.J. Scalable synthetic process for making terameprocol. WO/2010/014936, 4 February 2010. [Google Scholar]
- Eads, D.; Hansen, R.; Oyegunwa, A.; Cecil, C.; Culver, C.; Scholle, F.; Petty, I.; Laster, S. Terameprocol, a methylated derivative of nordihydroguaiaretic acid, inhibits production of prostaglandins and several key inflammatory cytokines and chemokines. J. Inflamm. (Lond.) 2009, 6, 2. [Google Scholar] [CrossRef] [PubMed]
- Craigo, J.; Callahan, M.; Huang, R.C.; DeLucia, A.L. Inhibition of human papillomavirus type 16 gene expression by nordihydroguaiaretic acid plant lignan derivatives. Antivir. Res. 2000, 47, 19–28. [Google Scholar] [CrossRef]
- Tagaya, M.; Henomatsu, N.; Yoshimori, T.; Yamamoto, A.; Tashiro, Y.; Mizushima, S. Inhibition of vesicle-mediated protein transport by nordihydroguaiaretic acid. J. Biochem. 1996, 119, 863–869. [Google Scholar] [CrossRef] [PubMed]
- Nozoe, T. Uber die farbstoffe im holzteile des “Hinoki” baumes. I. hinokitin und hinokitiol. Bull. Chem. Soc. Jpn. 1936, 11, 295–298. [Google Scholar] [CrossRef]
- Miyamoto, D.; Kusagaya, Y.; Endo, N.; Sometani, A.; Takeo, S.; Suzuki, T.; Arima, Y.; Nakajima, K.; Suzuki, Y. Thujaplicin-copper chelates inhibit replication of human influenza viruses. Antivir. Res. 1998, 39, 89–100. [Google Scholar] [CrossRef]
- Arima, Y.; Hatanaka, A.; Tsukihara, S.; Fujimoto, K.; Fukuda, K.; Sakurai, H. Scavenging activities of α-, β- and γ-thujaplicins against active oxygen species. Chem. Pharm. Bull. 1997, 45, 1881–1886. [Google Scholar] [CrossRef]
- Martinez, J.; Moreno, J.J. Effect of resveratrol, a natural polyphenolic compound, on reactive oxygen species and prostaglandin production. Biochem. Pharmacol. 2000, 59, 865–870. [Google Scholar] [CrossRef]
- Palamara, A.T.; Nencioni, L.; Aquilano, K.; De Chiara, G.; Hernandez, L.; Cozzolino, F.; Ciriolo, M.R.; Garaci, E. Inhibition of influenza A virus replication by resveratrol. J. Infect. Dis. 2005, 191, 1719–1729. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.L.; Loke, S.H.; Hsu, C.C.; Chiou, W.F. (+)-Vitisin A inhibits influenza A virus-induced RANTES production in A549 alveolar epithelial cells through interference with Akt and STAT1 phosphorylation. Planta Med. 2008, 74, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.L.; Tsai, W.J.; Shen, C.C.; Chen, C.C. Resveratrol derivatives from the roots of Vitis thunbergii. J. Nat. Prod. 2005, 68, 217–220. [Google Scholar] [CrossRef] [PubMed]
- García-Alonso, M.; Rimbach, G.; Rivas-Gonzalo, J.C.; De Pascual-Teresa, S. Antioxidant and cellular activities of anthocyanins and their corresponding vitisins A - studies in platelets, monocytes, and human endothelial cells. J. Agric. Food Chem. 2004, 52, 3378–3384. [Google Scholar] [CrossRef] [PubMed]
- Huang, K.S.; Lin, M.; Cheng, G.F. Anti-inflammatory tetramers of resveratrol from the roots of Vitis amurensis and the conformations of the seven-membered ring in some oligostilbenes. Phytochemistry 2001, 58, 357–362. [Google Scholar] [CrossRef]
- Huang, Y.L.; Loke, S.H.; Hsu, C.C.; Chiou, W.F. (+)-Vitisin A inhibits influenza A virus-induced RANTES production in A549 alveolar epithelial cells through interference with Akt and STAT1 phosphorylation. Planta Med. 2008, 74, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Germouty, J.; Jirou-Najou, J.L. Clinical efficacy of ambroxol in the treatment of bronchial stasis. Clinical trial in 120 patients at two different doses. Respiration 1987, 51, 37–41. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Yao, D.F.; Ohuchi, M.; Ide, M.; Yano, M.; Okumura, Y.; Kido, H. Ambroxol suppresses influenza-virus proliferation in the mouse airway by increasing antiviral factor levels. Eur. Respir. J. 2002, 19, 952–958. [Google Scholar] [CrossRef] [PubMed]
- Gillissen, A.; Bartling, A.; Schoen, S.; Schultze-Werninghaus, G. Antioxidant function of ambroxol in mononuclear and polymorphonuclear cells in vitro. Lung 1997, 175, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Gillissen, A.; Schärling, B.; Jaworska, M.; Bartling, A.; Rasche, K.; Schultze-Werninghaus, G. Oxidant scavenger function of ambroxol in vitro: A comparison with N-acetylcysteine. Res. Exp. Med. (Berl.) 1997, 196, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Nobata, K.; Fujimura, M.; Ishiura, Y.; Myou, S.; Nakao, S. Ambroxol for the prevention of acute upper respiratory disease. Clin. Exp. Med. 2006, 6, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, Y.; Hashizume, H.; Yamazaki, M. Superoxide radical scavenging activity of phenolic compounds. Int. J. Biochem. 1993, 25, 491–494. [Google Scholar] [PubMed]
- Jariwalla, R.J.; Roomi, M.W.; Gangapurkar, B.; Kalinovsky, T.; Niedzwiecki, A.; Rath, M. Suppression of influenza A virus nuclear antigen production and neuraminidase activity by a nutrient mixture containing ascorbic acid, green tea extract and amino acids. Biofactors 2007, 31, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Furuya, A.; Uozaki, M.; Yamasaki, H.; Arakawa, T.; Arita, M.; Koyama, A.H. Antiviral effects of ascorbic and dehydroascorbic acids in vitro. Int. J. Mol. Med. 2008, 22, 541–545. [Google Scholar] [PubMed]
- Kimbarowski, J.A.; Mokrow, N.J. Colored precipitation reaction of the urine according to Kimbarowski (FARK) as an index of the effect of ascorbic acid during treatment of viral influenza. Dtsch. Gesundheitsw. 1967, 22, 2413–2418. [Google Scholar] [PubMed]
- Banerjee, D.; Kaul, D. Combined inhalational and oral supplementation of ascorbic acid may prevent influenza pandemic emergency: A hypothesis. Nutrition 2010, 26, 128–132. [Google Scholar] [CrossRef] [PubMed]
- Robak, J.; Gryglewski, R.J. Bioactivity of flavonoids. Pol. J. Pharmacol. 1996, 48, 555–564. [Google Scholar] [PubMed]
- Nagai, T.; Miyaichi, Y.; Tomimori, T.; Yamada, H. Inhibition of mouse liver sialidase by plant flavonoids. Biochem. Biophys. Res. Commun. 1989, 163, 25–31. [Google Scholar] [CrossRef]
- Nagai, T.; Miyaichi, Y.; Tomimori, T.; Suzuki, Y.; Yamada, H. Inhibition of influenza virus sialidase and anti-influenza virus activity by plant flavonoids. Chem. Pharm. Bull. (Tokyo) 1990, 38, 1329–1332. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Miyaichi, Y.; Tomimori, T.; Suzuki, Y.; Yamada, H. In vivo anti-influenza virus activity of plant flavonoids possessing inhibitory activity for influenza virus sialidase. Antivir. Res. 1992, 19, 207–217. [Google Scholar] [CrossRef]
- Nagai, T.; Suzuki, Y.; Tomimori, T.; Yamada, H. Antiviral activity of plant flavonoid, 5,7,4’-trihydroxy-8-methoxyflavone, from the roots of Scutellaria baicalensis against influenza A (H3N2) and B viruses. Biol. Pharm. Bull. 1995, 18, 295–299. [Google Scholar] [CrossRef] [PubMed]
- Nagai, T.; Moriguchi, R.; Suzuki, Y.; Tomimori, T.; Yamada, H. Mode of action of the anti-influenza virus activity of plant flavonoid, 5,7,4’-trihydroxy-8-methoxyflavone, from the roots of Scutellaria baicalensis. Antivir. Res. 1995, 26, 11–25. [Google Scholar] [CrossRef]
- Song, J.M.; Lee, K.H.; Seong, B.L. Antiviral effect of catechins in green tea on influenza virus. Antivir. Res. 2005, 68, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Kashima, M. Effects of catechins on superoxide and hydroxyl radical. Chem. Pharm. Bull. (Tokyo) 1999, 47, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Yamada, H.; Takuma, N.; Daimon, T.; Hara, Y. Gargling with tea catechin extracts for the prevention of influenza infection in elderly nursing home residents: A prospective clinical study. J. Altern. Complement. Med. 2006, 12, 669–672. [Google Scholar] [CrossRef] [PubMed]
- Mori, S.; Miyake, S.; Kobe, T.; Nakaya, T.; Fuller, S.D.; Kato, N.; Kaihatsu, K. Enhanced anti-influenza A virus activity of (-)-epigallocatechin-3-O-gallate fatty acid monoester derivatives: Effect of alkyl chain length. Bioorg. Med. Chem. Lett. 2008, 18, 4249–4952. [Google Scholar] [CrossRef] [PubMed]
- Kuzuhara, T.; Iwai, Y.; Takahashi, H.; Hatakeyama, D.; Echigo, N. Green tea catechins inhibit the endonuclease activity of influenza A virus RNA polymerase. PLoS. Curr. 2009, 1, RRN1052. [Google Scholar] [CrossRef]
- Choi, H.J.; Song, J.H.; Park, K.S.; Kwon, D.H. Inhibitory effects of quercetin 3-rhamnoside on influenza A virus replication. Eur. J. Pharm. Sci. 2009, 37, 329–333. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.; Narayanan, S.; Chang, K.O. Inhibition of influenza virus replication by plant-derived isoquercetin. Antivir. Res. 2010, 88, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Sun, B.; Nishioka, H.; Hirose, A.; Aruoma, O.I. Evaluation of the safety and toxicity of the oligomerized polyphenol Oligonol. Food Chem. Toxicol. 2007, 45, 378–387. [Google Scholar] [CrossRef] [PubMed]
- Gangehei, L.; Ali, M.; Zhang, W.; Chen, Z.; Wakame, K.; Haidari, M. Oligonol a low molecular weight polyphenol of lychee fruit extract inhibits proliferation of influenza virus by blocking reactive oxygen species-dependent ERK phosphorylation. Phytomedicine 2010, 17, 1047–1056. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 8, 9 and 10 are available from the authors. |
| Name | Oligonol | Lychee fruit polyphenol | |
|---|---|---|---|
| Monomer | (+)-Catechin | 0.1%> | 0.1%> |
| (-)-Epicatechin | 7.5% | 6.4% | |
| (-)-ECG | 2.1% | n.d. | |
| Dimer | Procyanidin A1 | 4.2% | 4.0% |
| Procyanidin A2 | 5.1% | 3.3% | |
| Procyanidin B1 | 1.4% | 0.8% | |
| Procyanidin B2 | 2.9% | 1.7% | |
| Trimer | (-)-Epicatechin-(4β→8,2β→o-7)-epicatechin-(4β→8)-epicatechin | 4.0% | 3.6% |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Uchide, N.; Toyoda, H. Antioxidant Therapy as a Potential Approach to Severe Influenza-Associated Complications. Molecules 2011, 16, 2032-2052. https://doi.org/10.3390/molecules16032032
Uchide N, Toyoda H. Antioxidant Therapy as a Potential Approach to Severe Influenza-Associated Complications. Molecules. 2011; 16(3):2032-2052. https://doi.org/10.3390/molecules16032032
Chicago/Turabian StyleUchide, Noboru, and Hiroo Toyoda. 2011. "Antioxidant Therapy as a Potential Approach to Severe Influenza-Associated Complications" Molecules 16, no. 3: 2032-2052. https://doi.org/10.3390/molecules16032032
APA StyleUchide, N., & Toyoda, H. (2011). Antioxidant Therapy as a Potential Approach to Severe Influenza-Associated Complications. Molecules, 16(3), 2032-2052. https://doi.org/10.3390/molecules16032032




