Hepatoprotective Potential of Clitoria ternatea Leaf Extract Against Paracetamol Induced Damage in Mice
Abstract
:1. Introduction
2. Results and Discussion
2.1. Radial Scavenging (DPPH) Assay and IC50 Determination
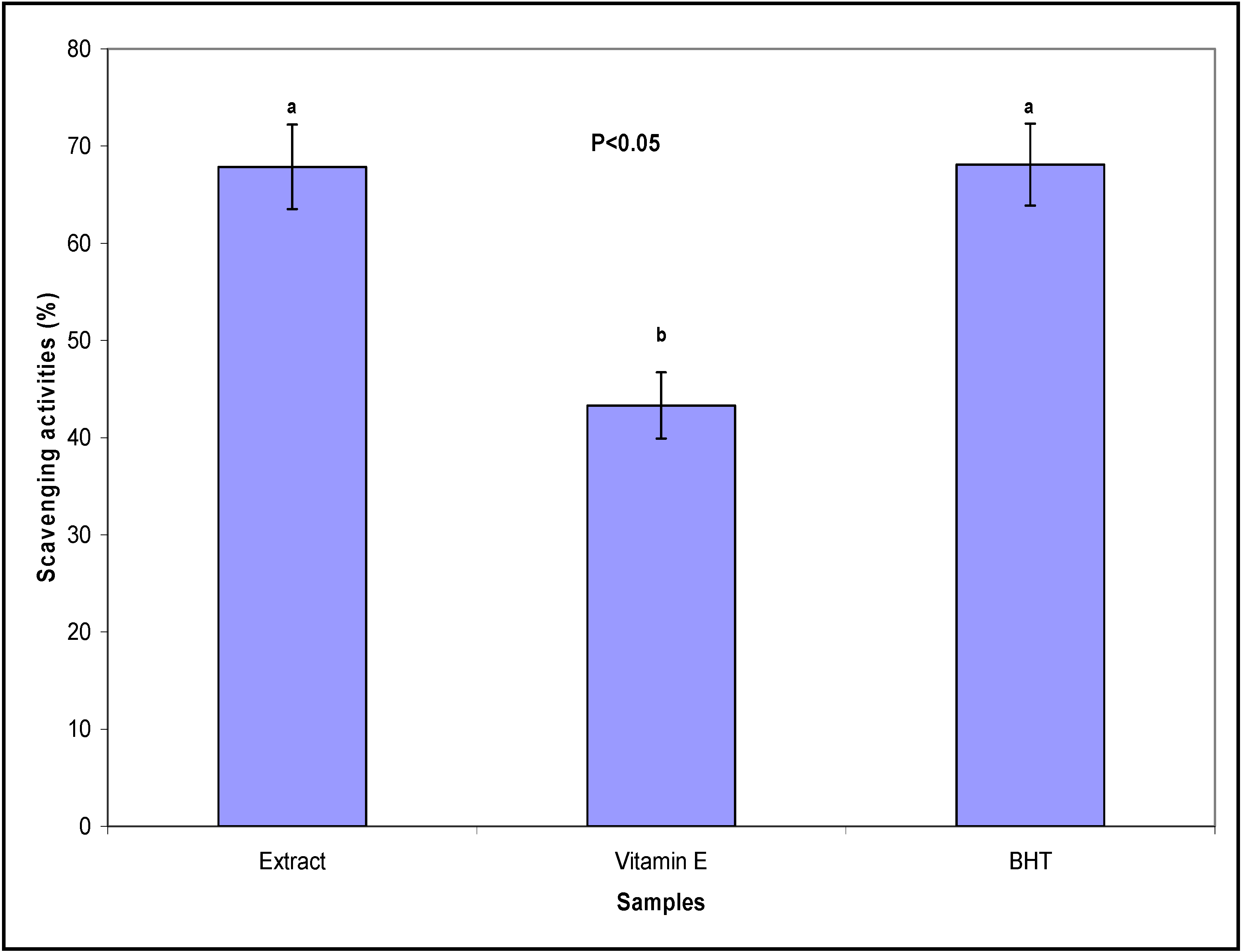
2.2. Total Phenolic and Flavonoid Content
2.3. Biochemical Parameters
| Parameters | Control | Paracetamol Treated | Extract Treated |
|---|---|---|---|
| AST (IU/L) | 38.83 ± 5.32 | 108.65 ± 12.21 ** | 42.1 ± 6.25 * |
| ALT (IU/L) | 32.58 ± 6.12 | 98.23 ± 10.12 ** | 39.23 ± 5.32 * |
| Billirubin (mg/L) | 1.5 ± 0.5 | 8.7 ± 3.9 ** | 2.3 ± 1.3 * |
2.4. Histopathology Analysis
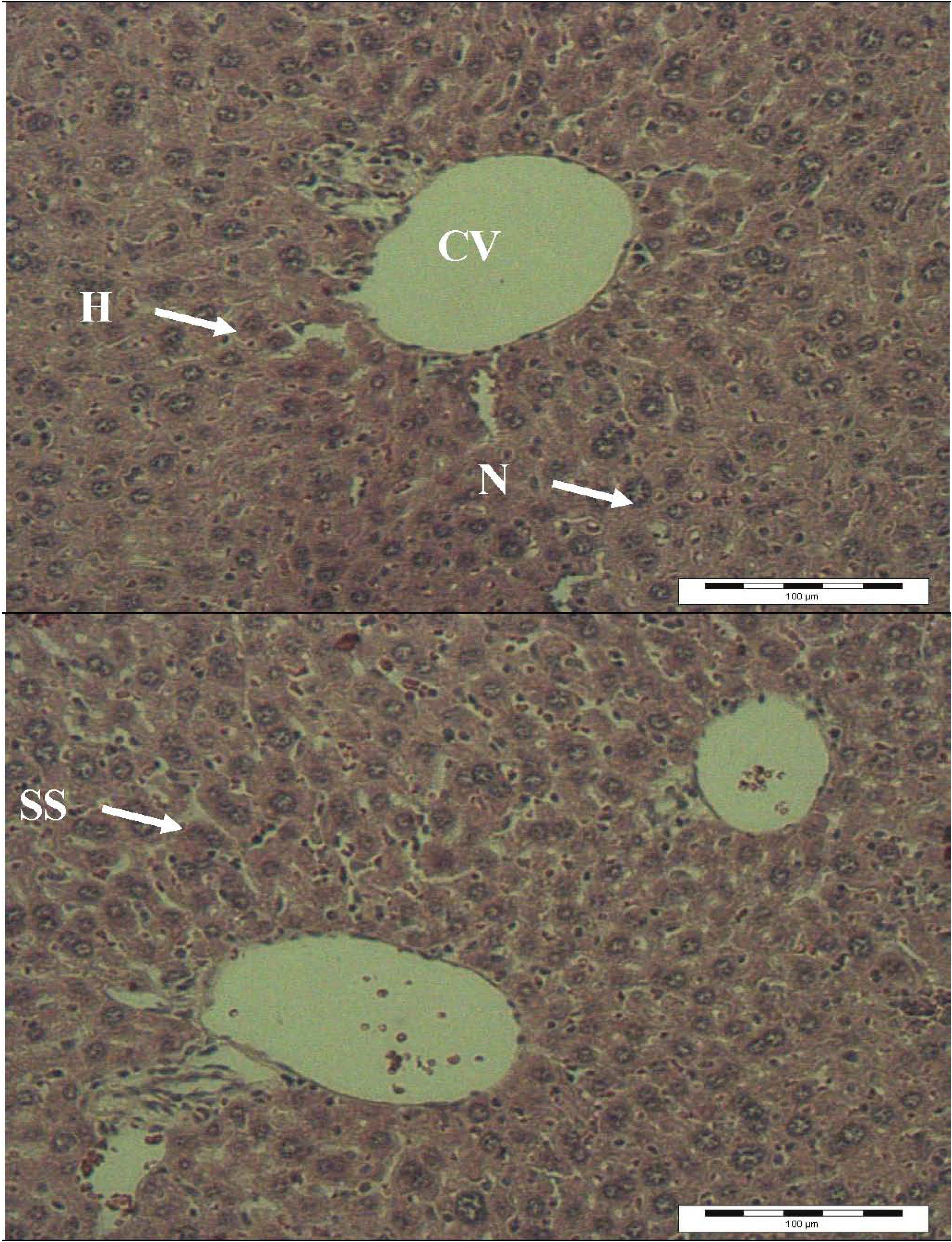
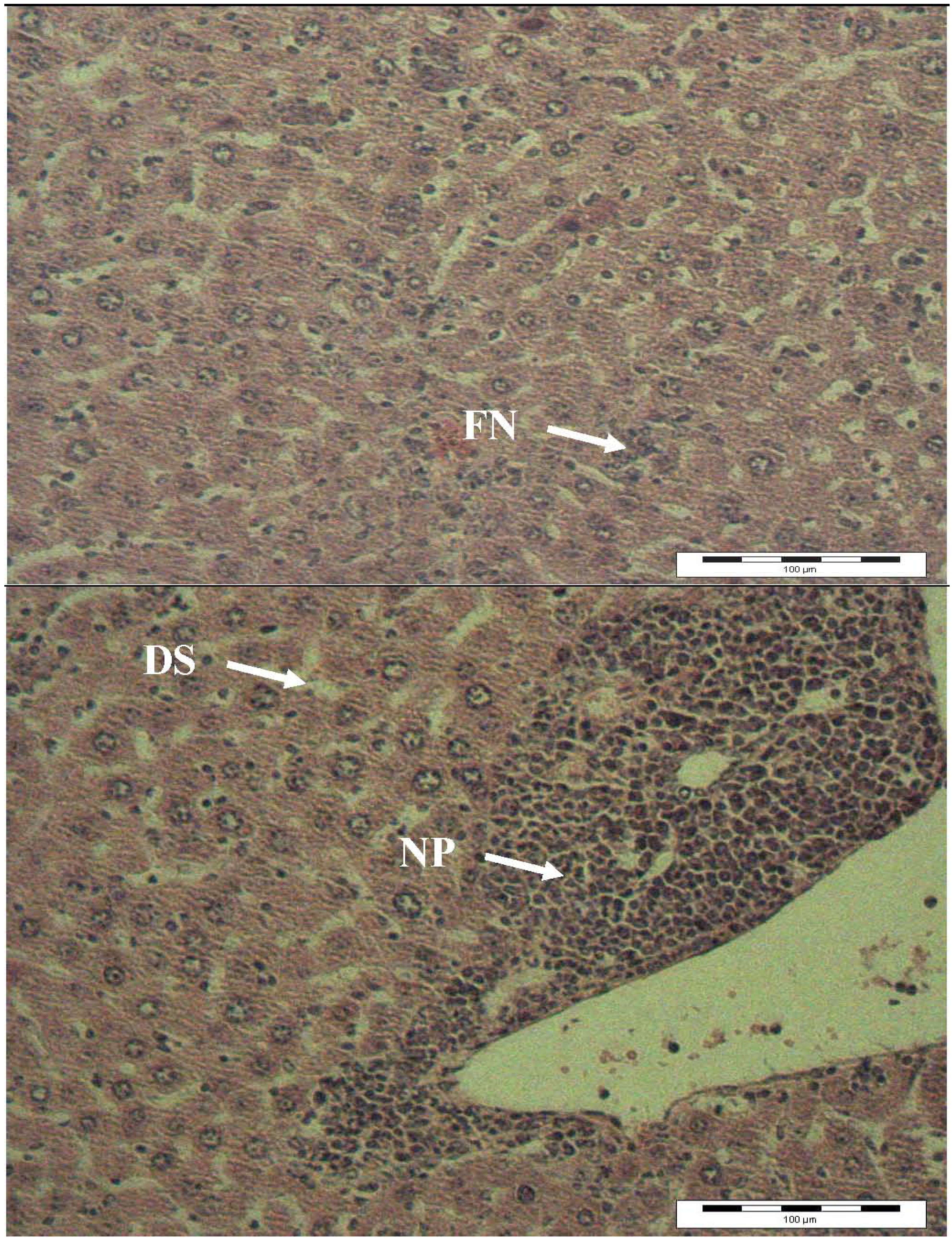
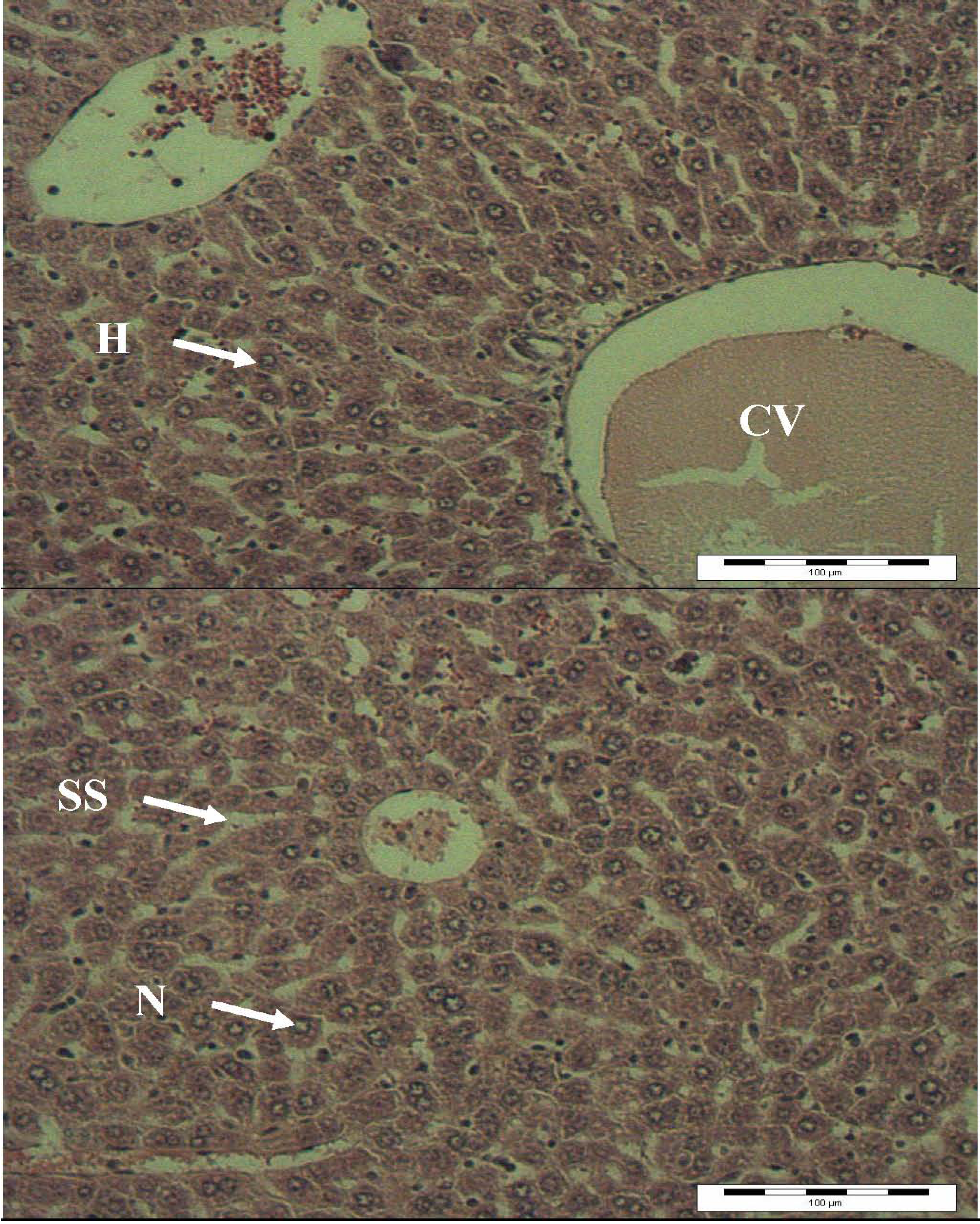
3. Experimental
3.1. Sample Collection
3.2. Extraction Procedure
3.3. Antioxidant Activity Assays
3.4. IC50 Determination
3.5. Determinations of Total Phenolic Contents
3.6. Determination of Total Flavonoid Content
3.7. Hepatoprotective Activity of C. ternatea Leaf Extract
3.7.1. Animals
3.7.2. Paracetamol Dose Regimen
3.7.3. Grouping of Mice and Treatments
3.7.4. Sacrifice and Organ Harvesting
3.7.5. Biochemical Parameters
3.7.6. Statistical Analysis
4. Conclusions
Acknowledgments
Conflict of Interest
References and Notes
- Mukherjee, P.K.; Kumar, V.; Kumar, N.S.; Heinrich, M. The Ayurvedic medicine Clitoria ternatea from traditional use to scientific assessment. J. Ethnopharmacol. 2008, 120, 291–301. [Google Scholar] [CrossRef]
- Patil, A.P.; Patil, V.R. Clitoria ternatea Linn.: An overview. Int. J. Pharm. Res. 2011, 3, 20–23. [Google Scholar]
- Anonymous, Indian Medicinal Plants; Orient Longman: Madras, India, 1995; Volume 2, pp. 129–132.
- Kirtikar, K.R.; Basu, B.D. Indian Medicinal Plants; Lalit Mohan Basu Publications: Allahabad, India, 1935; p. 802. [Google Scholar]
- Mukherjee, P.K.; Kumar, V.; Houghton, P.J. Screening of Indian medicinal plants for acetyl cholinesterase inhibitory activity. Phytother. Res. 2007, 21, 1142–1145. [Google Scholar] [CrossRef]
- Huang, B.; Ban, X.; He, J.; Tong, J.; Tian, J.; Wang, Y. Hepatoprotective and antioxidant activity of ethanolic extracts of edible lotus (Nelumbo nucifera Gaertn.) leaves. Food Chem. 2010, 120, 873–878. [Google Scholar] [CrossRef]
- Nayak, S.S.; Jain, R.; Sahoo, A.K. Hepatoprotective activity of Glycosmis pentaphylla against paracetamol-induced hepatotoxicity in Swiss albino mice. Pharm. Biol. 2011, 49, 111–117. [Google Scholar] [CrossRef]
- Bhaskar, V.H.; Balakrishnan, N. Protective effects of Pergularia daemia roots against paracetamol and carbon tetrachloride-induced hepatotoxicity in rats. Pharm. Biol. 2010, 48, 1265–1272. [Google Scholar] [CrossRef]
- Fakurazi, S.; Hairuszah, I.; Nanthini, U. Moringa oleifera Lam prevents acetaminophen induced liver injury through restoration of glutathione level. Food Chem. Toxicol. 2008, 46, 2611–2615. [Google Scholar] [CrossRef]
- Sabir, S.M.; Rocha, J.B. Water-extractable phytochemicals from Phyllanthus niruri exhibit distinct in vitro antioxidant and in vivo hepatoprotective activity against paracetamol-induced liver damage in mice. Food Chem. 2008, 111, 845–851. [Google Scholar] [CrossRef]
- Bhathal, P.S.; Rose, N.R.; Mackay, I.R.; Whittingham, S. Strain differences in mice in carbon tetrachloride-induced liver injury. Br. J. Exp. Pathol. 1983, 64, 524–533. [Google Scholar]
- Yousef, M.I.; Omar, S.A.; El-Guendi, M.I.; Abdelmegid, L.A. Potential protective effects of quercetin and curcumin on paracetamol-induced histological changes, oxidative stress, impaired liver and kidney functions and haematotoxicity in rat. Food Chem. Toxicol. 2010, 48, 3246–3261. [Google Scholar] [CrossRef]
- Choi, J.H.; Choi, C.Y.; Lee, K.J.; Hwang, Y.P.; Chung, Y.C.; Jeong, H.G. Hepatoprotective effects of an anthocyanin fraction from purple-fleshed sweet potato against acetaminophen-induced liver damage in mice. J. Med. Food. 2009, 12, 320–326. [Google Scholar] [CrossRef]
- Sharma, N.; Shukla, S. Hepatoprotective potential of aqueous extract of Butea monosperma against CCl4 induced damageinrats. Exp. Toxicol. Pathol. 2011, 63, 671–676. [Google Scholar] [CrossRef]
- Ajiboye, T.O.; Salau, A.K.; Yakubu, M.T.; Oladiji, A.T.; Akanji, M.A.; Okogun, J.I. Acetaminophen perturbed redox homeostasis in Wistar rat liver: Protective role of aqueous Pterocarpus osun leaf extract. Drug Chem. Toxicol. 2010, 33, 77–87. [Google Scholar] [CrossRef]
- Adeneye, A.A. Protective activity of the stem bark aqueous extract of Musanga cecropioides in carbon tetrachloride- and acetaminophen-induced acute hepatotoxicity in rats. Afr. J. Tradit. Complement. Altern. Med. 2009, 6, 131–138. [Google Scholar]
- Raghavendran, H.B.; Sathivel, A.; Devaki, T. Defensive nature of Sargassum polycystum (Brown alga) against acetaminophen-induced toxic hepatitis in rats: Role of drug metabolizing microsomal enzyme system, tumor necrosis factor-alpha and fate of liver cell structural integrity. World J. Gastroenterol. 2006, 12, 3829–3834. [Google Scholar]
- Iwalokun, B.A.; Efedede, B.U.; Alabi-Sofunde, J.A.; Oduala, T.; Magbagbeola, O.A.; Akinwande, A.I. Hepatoprotective and antioxidant activities of Vernonia amygdalina on acetaminophen-induced hepatic damage in mice. J. Med. Food. 2006, 9, 524–530. [Google Scholar] [CrossRef]
- Huang, B.; Ban, X.; He, J.; Tong, J.; Tian, J.; Wang, Y.W. Hepatoprotective and antioxidant activity of ethanolic extracts of edible lotus (Nelumbo nucifera Gaertn.) leaves. Food Chem. 2010, 120, 873–878. [Google Scholar] [CrossRef]
- Sasidharan, S.; Aravindran, S.; Latha, L.Y.; Vijenthi, R.; Saravanan, D.; Amutha, S. In Vitro antioxidant activity and hepatoprotective effects of Lentinula edodes against paracetamol-induced hepatotoxicity. Molecules 2010, 15, 4478–4489. [Google Scholar] [CrossRef]
- Sasidharan, S.; Darah, I.; Jain, N.M.K.M. Free radical scavenging activity and total phenolic compounds of Gracilaria changii. Int. J. Nat. Eng. Sci. 2007, 1, 115–117. [Google Scholar]
- Aderogba, M.A.; Okoh, E.K.; Adelanwa, T.A.; Obuotor, E.M. Antioxidant properties of the Nigerian Piliostigma species. J. Biol. Sci. 2004, 4, 501–503. [Google Scholar] [CrossRef]
- Sabir, S.M.; Rocha, J.B.T. Antioxidant and hepatoprotective activity of aqueous extract of Solanum fastigiatum (false “Jurubeba”) against paracetamol-induced liver damage in mice. J. Ethnopharmacol. 2008, 120, 226–232. [Google Scholar] [CrossRef]
- Djeridane, A.; Yousfi, M.; Nadjemi, B.; Boutassouna, D.; Stocker, P.; Vidal, N. Antioxidant activity of some Algerian medicinal plants extracts containing phenolic compounds. Food Chem. 2006, 97, 654–660. [Google Scholar] [CrossRef]
- Zimmerman, M. Ethical guidelines for investigation of experimental pain in conscious animal. Pain 1983, 16, 109–110. [Google Scholar] [CrossRef]
- Rao, P.G.; Rao, G.; Ramnarayan, K.; Srinivasan, K.K. Effect of hepatogard on paracetamolinduced liver injury in male albino rats. Indian Drugs 1993, 30, 41–46. [Google Scholar]
- da Rocha, R.P.; de Miranda Paquola, A.C.; do Valle Marques, M.; Menck, C.F.M.; Galhardo, R.S. Characterization of the SOS regulon of Caulobacter crescentus. J. Bacteriol. 2008, 190, 1209–1218. [Google Scholar] [CrossRef]
- Reitman, S.; Frankel, S. A colorimetric method for the determination of serum glutamic oxalacetic and glutamic piruvic transaminases. Am. J. Clin. Pathol. 1957, 28, 56–63. [Google Scholar]
- Malloy, H.T.; Evelyn, K.A. The determination of bilirubin with the photochemical colorimeter. J. Biol. Chem. 1937, 119, 481–490. [Google Scholar]
- Sample Availability: Samples of the compounds are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Nithianantham, K.; Shyamala, M.; Chen, Y.; Latha, L.Y.; Jothy, S.L.; Sasidharan, S. Hepatoprotective Potential of Clitoria ternatea Leaf Extract Against Paracetamol Induced Damage in Mice. Molecules 2011, 16, 10134-10145. https://doi.org/10.3390/molecules161210134
Nithianantham K, Shyamala M, Chen Y, Latha LY, Jothy SL, Sasidharan S. Hepatoprotective Potential of Clitoria ternatea Leaf Extract Against Paracetamol Induced Damage in Mice. Molecules. 2011; 16(12):10134-10145. https://doi.org/10.3390/molecules161210134
Chicago/Turabian StyleNithianantham, Kuppan, Murugesan Shyamala, Yeng Chen, Lachimanan Yoga Latha, Subramanion L. Jothy, and Sreenivasan Sasidharan. 2011. "Hepatoprotective Potential of Clitoria ternatea Leaf Extract Against Paracetamol Induced Damage in Mice" Molecules 16, no. 12: 10134-10145. https://doi.org/10.3390/molecules161210134
APA StyleNithianantham, K., Shyamala, M., Chen, Y., Latha, L. Y., Jothy, S. L., & Sasidharan, S. (2011). Hepatoprotective Potential of Clitoria ternatea Leaf Extract Against Paracetamol Induced Damage in Mice. Molecules, 16(12), 10134-10145. https://doi.org/10.3390/molecules161210134




