Monosaccharide-NAIM Derivatives for D-, L-Configurational Analysis
Abstract
:1. Introduction
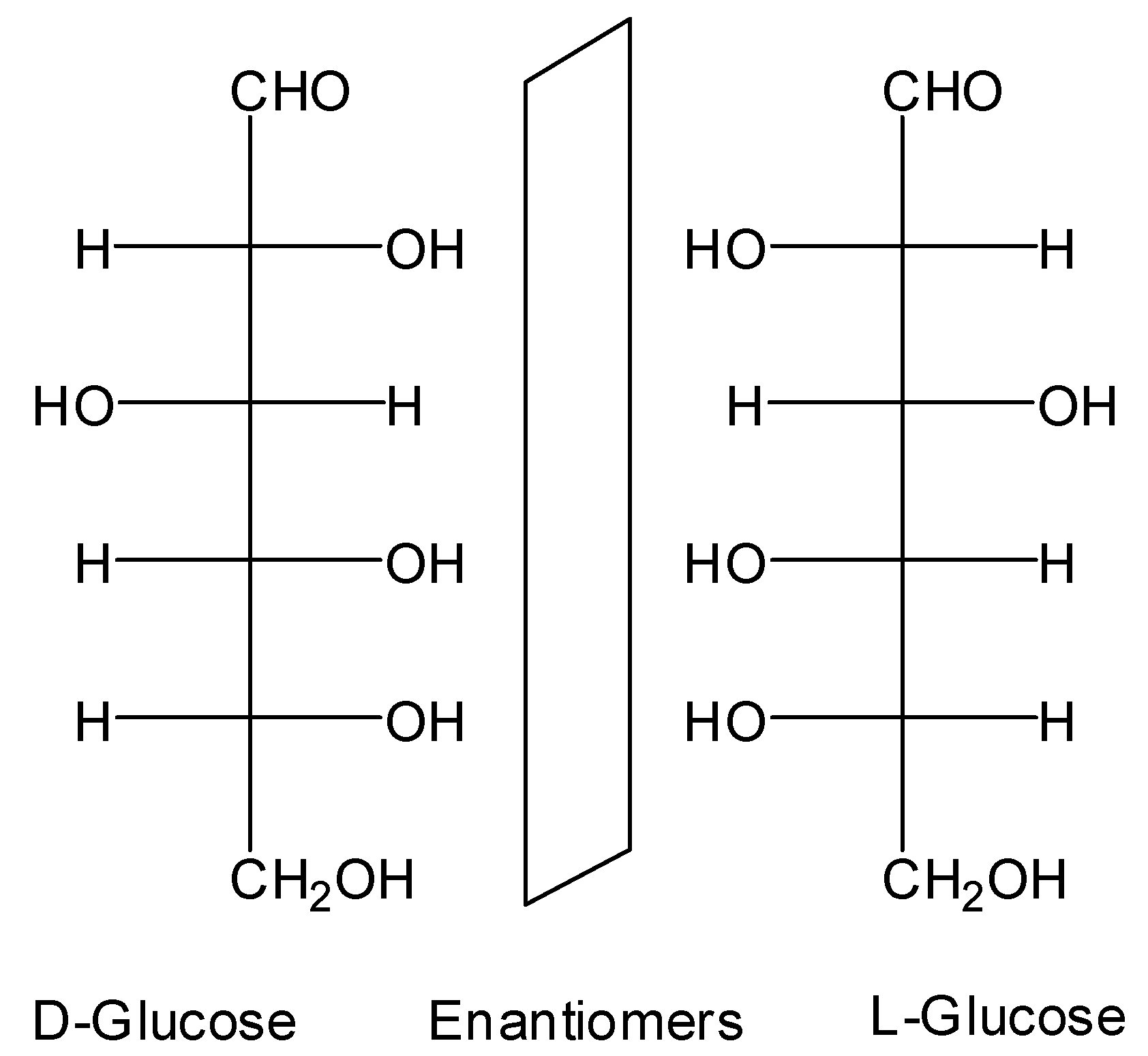
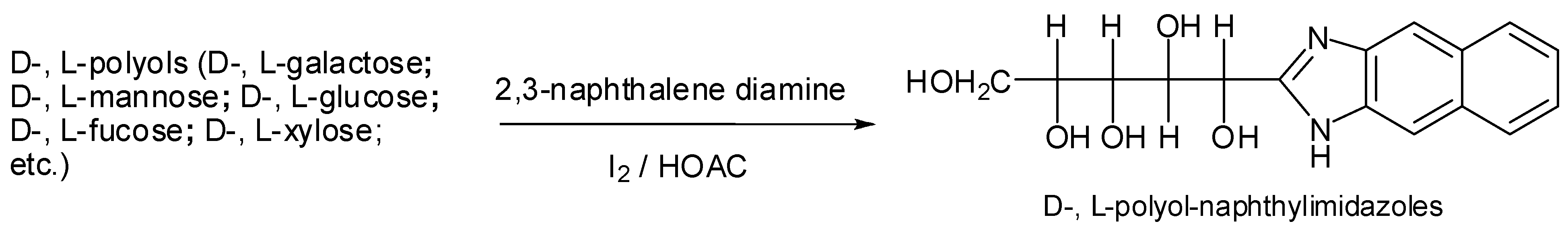
2. Results and Discussion
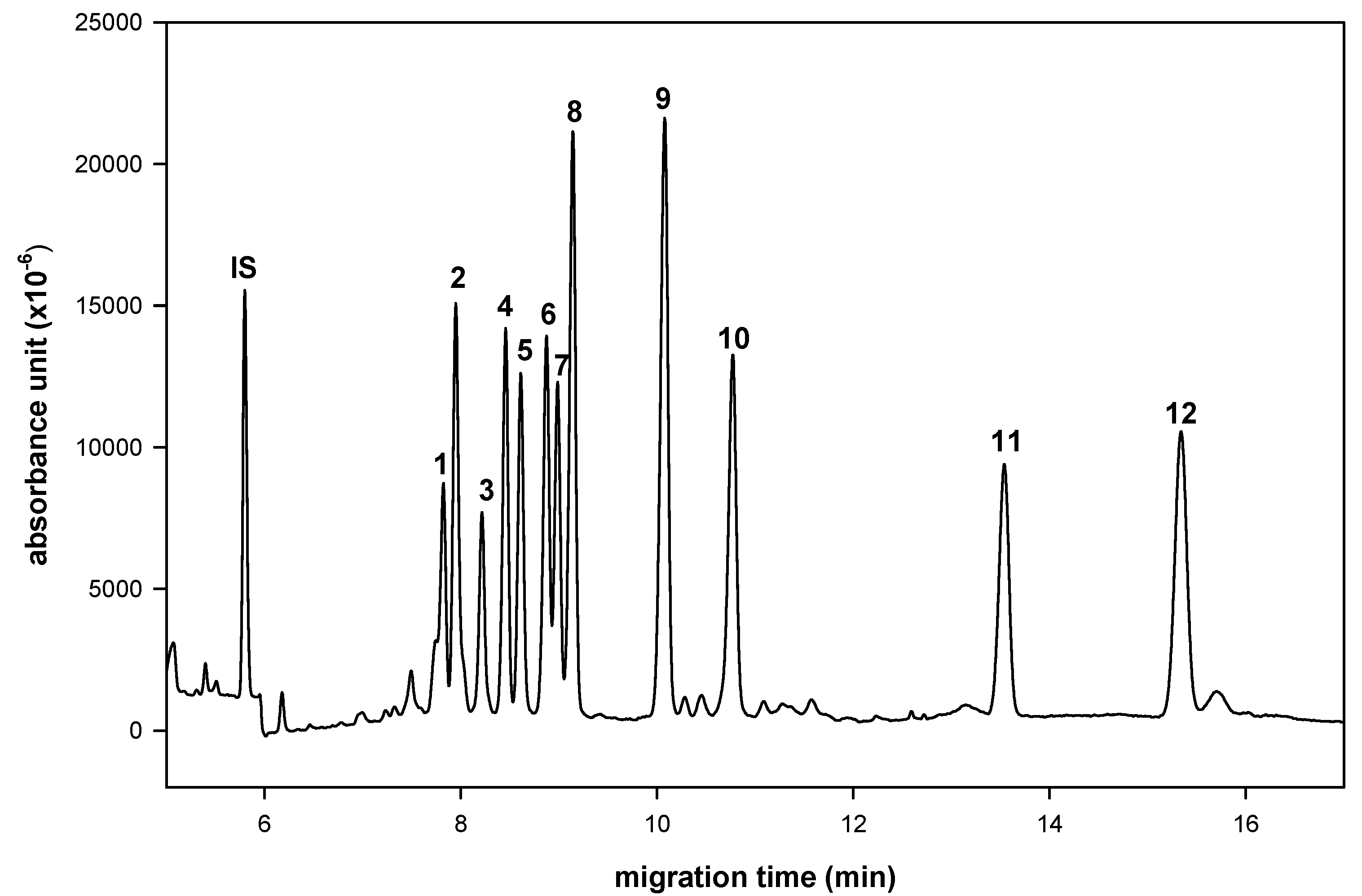
| Peak | aldo-NAIM | Migration Time (min) |
|---|---|---|
| 1 | Rha-NAIM | 7.7 |
| 2 | GalNAc-NAIM | 7.9 |
| 3 | SA-NAQ | 8.2 |
| 4 | Xyl-NAIM | 8.4 |
| 5 | Rib-NAIM | 8.5 |
| 6 | Glc-NAIM | 8.8 |
| 7 | Man-NAIM | 8.9 |
| 8 | Ara-NAIM | 9.1 |
| 9 | Fuc-NAIM | 10.1 |
| 10 | Gal-NAIM | 10.8 |
| 11 | GlcA-NAIM | 13.5 |
| 12 | GalA-NAIM | 15.3 |
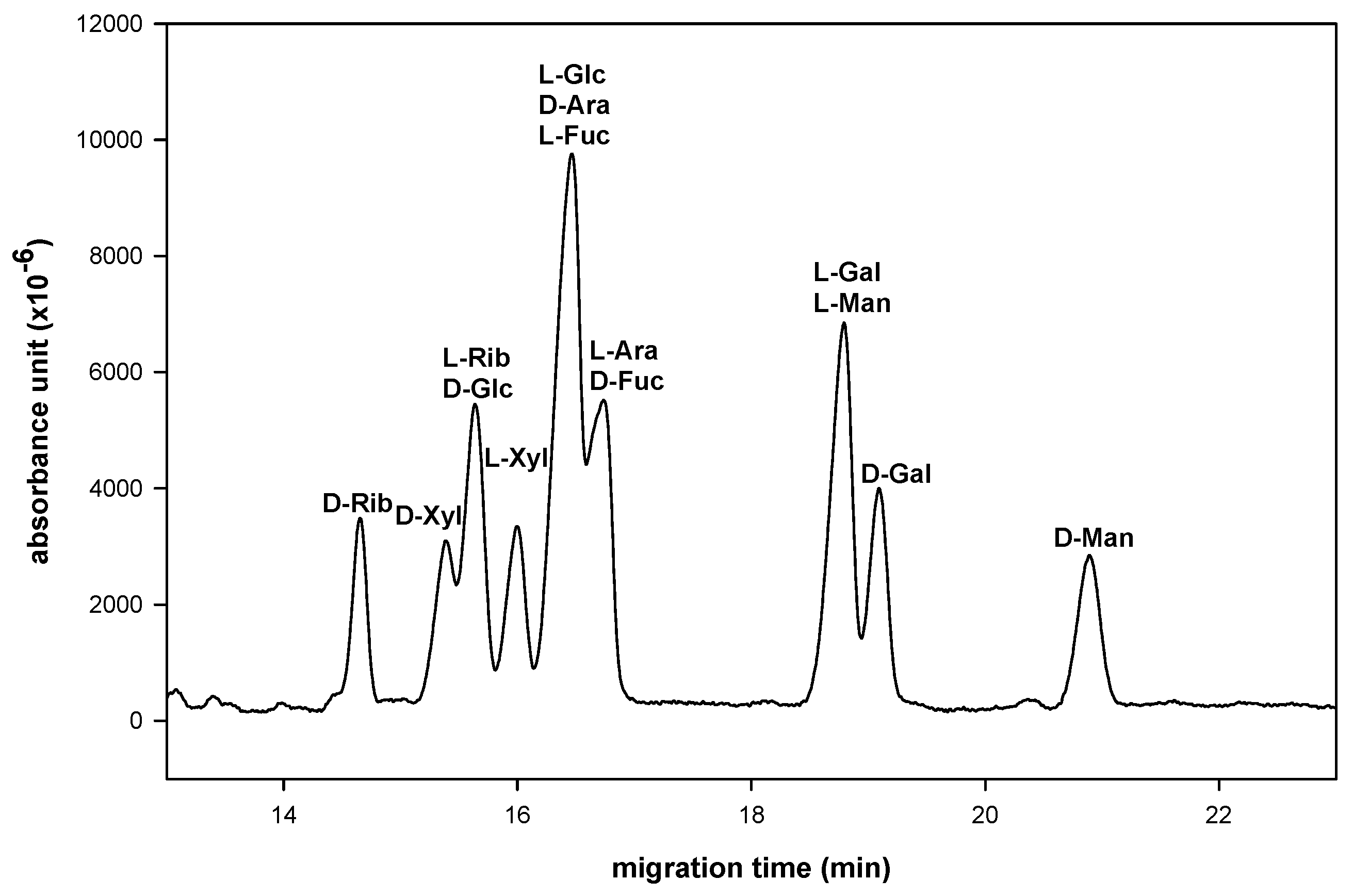
| D-enantiomer | Migration time (min) | L-enantiomer | Migration time (min) |
|---|---|---|---|
| D-Manno-NAIM | 20.4 | L-Manno-NAIM | 18.7 |
| D-Galacto-NAIM | 18.5 | L-Galacto-NAIM | 18.9 |
| D-Gluco-NAIM | 16.0 | L-Gluco-NAIM | 16.6 |
| D-Fuco-NAIM | 17.2 | L-Fuco-NAIM | 17.0 |
| D-Xylo-NAIM | 16.0 | L-Xylo-NAIM | 16.6 |
| D-Ribo-NAIM | 15.4 | L-Ribo-NAIM | 16.3 |
| D-Arabino-NAIM | 16.9 | L-Arabino-NAIM | 17.2 |


3. Experimental
3.1. General
3.2. Chemicals and reagents
3.3. Preparation of aldo-NAIMs
3.4. CE system
3.5. Optimization of the separation of 12 aldo-NAIM derivatives


3.6. Validation of experimental data
4. Conclusions
Acknowledgements
References
- Guttman, A. Analysis of monosaccharide composition by capillary electrophoresis. J. Chromatogr. A 1997, 763, 271–277. [Google Scholar] [CrossRef]
- Volpi, N.; Maccari, F.; Linhardt, R.J. Capillary electrophoresis of complex natural polysaccharides. Electrophoresis 2008, 29, 3095–3106. [Google Scholar] [CrossRef]
- Bertozzi, C.R.; Kiessling, L.L. Chemical glycobiology. Science 2001, 291, 2357–2364. [Google Scholar] [CrossRef]
- Price, N.P.J.; Bowman, M.J.; Gall, S.L.; Berhow, M.A.; Kendra, D.F.; Lerouge, P. Functionalized C-glycoside ketohydrazones: carbohydrate derivatives that retain the ring integrity of the terminal reducing sugar. Anal. Chem. 2010, 82, 2893–2899. [Google Scholar]
- Colombeau, L.; Traoré, T.; Compain, P.; Martin, O.R. Metal-free one-pot oxidative amidation of aldoses with functionalized amines. J. Org. Chem. 2008, 73, 8647–8650. [Google Scholar] [CrossRef]
- Lin, C.; Hung, W.T.; Kuo, C.Y.; Liao, K.S.; Liu, Y.C.; Yang, W.B. I2-catalyzed oxidative condensation of aldoses with diamines: synthesis of aldo-naphthimidazoles for carbohydrate analysis. Molecules 2010, 15, 1340–1353. [Google Scholar] [CrossRef]
- Lin, C.; Lai, P.T.; Liao, K.S.; Hung, W.T.; Yang, W.B.; Fang, J.M. Using molecular iodine in direct oxidative condensation of aldoses with diamines: An improved synthesis of aldo-benzimidazoles and aldo-naphthimidazoles for carbohydrate analysis. J. Org. Chem. 2008, 73, 3848–3853. [Google Scholar] [CrossRef]
- Ward, T.J.; Baker, B.A. Chiral separations. Anal.Chem. 2008, 80, 4363–4372. [Google Scholar] [CrossRef]
- Preinerstorfer, B.; Lämmerhofer, M.; Lindner, W. Advances in enantioselective separations using electromigration capillary techniques. Electrophoresis 2009, 30, 100–132. [Google Scholar] [CrossRef]
- Underberg, W.J.; Waterval, J.C. Derivatization trends in capillary electrophoresis: a update. Electrophoresis 2002, 23, 3922–3933. [Google Scholar] [CrossRef]
- Gübitz, G.; Schmid, M.G. Chiral separation by capillary electromigration techniques. J. Chromatogr. A 2008, 1204, 140–156. [Google Scholar] [CrossRef]
- Chankvetadze, B. Enantiomer migration order in chiral capillary electrophoresis. Electrophoresis 2002, 23, 4022–4035. [Google Scholar] [CrossRef]
- Honda, S.; Suzuki, S.; Taga, A. Analysis of carbohydrates as 1-phenyl-3-methyl-5-pyrazolone derivatives by capaillary/microchip electrophoresis and capillary electrochromatography. J. Pharm. Biomed. Anal. 2003, 30, 1689–1714. [Google Scholar] [CrossRef]
- Kodama, S.; Aizawa, S.; Taga, A.; Yamashita, T.; Kemmei, T.; Yamamoto, A.; Hayakawa, K. Simultaneous chiral resolution of monosaccharides as 8-aminonaphthalene-1,3,6-trisulfonate derivatives by ligand-exange CE using borate as a central ion of the chiral selector. Electrophoresis 2007, 28, 3930–3933. [Google Scholar] [CrossRef]
- Stefansson, M.; Novotny, M. Electrophoretic resolution of monosaccharide enantiomers in borate-oligosaccharide complexation media. J. Am. Chem. Soc. 1993, 115, 11573–11580. [Google Scholar] [CrossRef]
- Kodama, S.; Aizawa, S.; Taga, A.; Yamashita, T.; Yamamoto, A. Chiral resolution of monosaccharides as 1-phenyl-3-methyl-5-pyrazolone derivatives by ligand-exchange CE using borate anion as a central ion of the chiral selector. Electrophoresis 2006, 27, 4730–4734. [Google Scholar] [CrossRef]
- Stalcup, A.M.; Agyei, N.M. Heparin: a chiral mobile-phase additive for capillary zone electrophoresis. Anal.Chem. 1994, 66, 3054–3059. [Google Scholar] [CrossRef]
- Kuhn, R.; Stoecklin, F.; Erni, F. Chiral separations by host-guest complexation with cyclodextrin and crown ether in capillary zone electrophoresis. Chromatographia 1992, 33, 32–36. [Google Scholar] [CrossRef]
- Clark, C.R.; Barksdale, J.M. Synthesis and liquid chromatographic evaluation of some chiral derivatizing agents for resolution of amine enantiomers. Anal. Chem. 1984, 56, 958–962. [Google Scholar] [CrossRef]
- Sun, X.X.; Sun, L.Z.; Aboul-Enein, Y. Chiral derivatization reagents for drug enantioseparation by high-performance liquid chromatography based upon pre-column derivatization and formation of diastereomers. Biomed.Chromatogr. 2001, 15, 116–132. [Google Scholar] [CrossRef]
- Lamari, F.N.; Kuhn, R.; Karamanos, N.K. Derivatization of carbohydrates for chromatographic, electrophoretic and mass spectrometric structure analysis. J. Chromatogr. 2003, 793, 15–36. [Google Scholar] [CrossRef]
- Tietze, L.F. Domino reactions in organic synthesis. Chem. Rev. 1996, 96, 115–136. [Google Scholar] [CrossRef]
- Chmberland, S.; Gruschow, S.; Sherman, D.H.; Willams, R.M. Synthesis of potential early-stage intermediates in the biosynthesis of FR900482 and mitomycin C. Org. Lett. 2009, 11, 791–794. [Google Scholar] [CrossRef]
- Sample Availability: Samples of aldo-NAIM are available from the authors.
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lin, C.; Kuo, C.-Y.; Liao, K.-S.; Yang, W.-B. Monosaccharide-NAIM Derivatives for D-, L-Configurational Analysis. Molecules 2011, 16, 652-664. https://doi.org/10.3390/molecules16010652
Lin C, Kuo C-Y, Liao K-S, Yang W-B. Monosaccharide-NAIM Derivatives for D-, L-Configurational Analysis. Molecules. 2011; 16(1):652-664. https://doi.org/10.3390/molecules16010652
Chicago/Turabian StyleLin, Chunchi, Chien-Yuan Kuo, Kuo-Shiang Liao, and Wen-Bin Yang. 2011. "Monosaccharide-NAIM Derivatives for D-, L-Configurational Analysis" Molecules 16, no. 1: 652-664. https://doi.org/10.3390/molecules16010652
APA StyleLin, C., Kuo, C.-Y., Liao, K.-S., & Yang, W.-B. (2011). Monosaccharide-NAIM Derivatives for D-, L-Configurational Analysis. Molecules, 16(1), 652-664. https://doi.org/10.3390/molecules16010652




