Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Phenolic, flavonoid and antioxidant activities
| Solvent | Phenolic Content1 | Flavonoid Content2 |
|---|---|---|
| Ethanol | 6.3 ± 0.01b | 2.9 ± 0.02c |
| Water | 5.7 ± 0.04c | 3.8 ± 0.09b |
| Methanol | 6.5 ± 0.02a | 5.8 ± 0.12a |
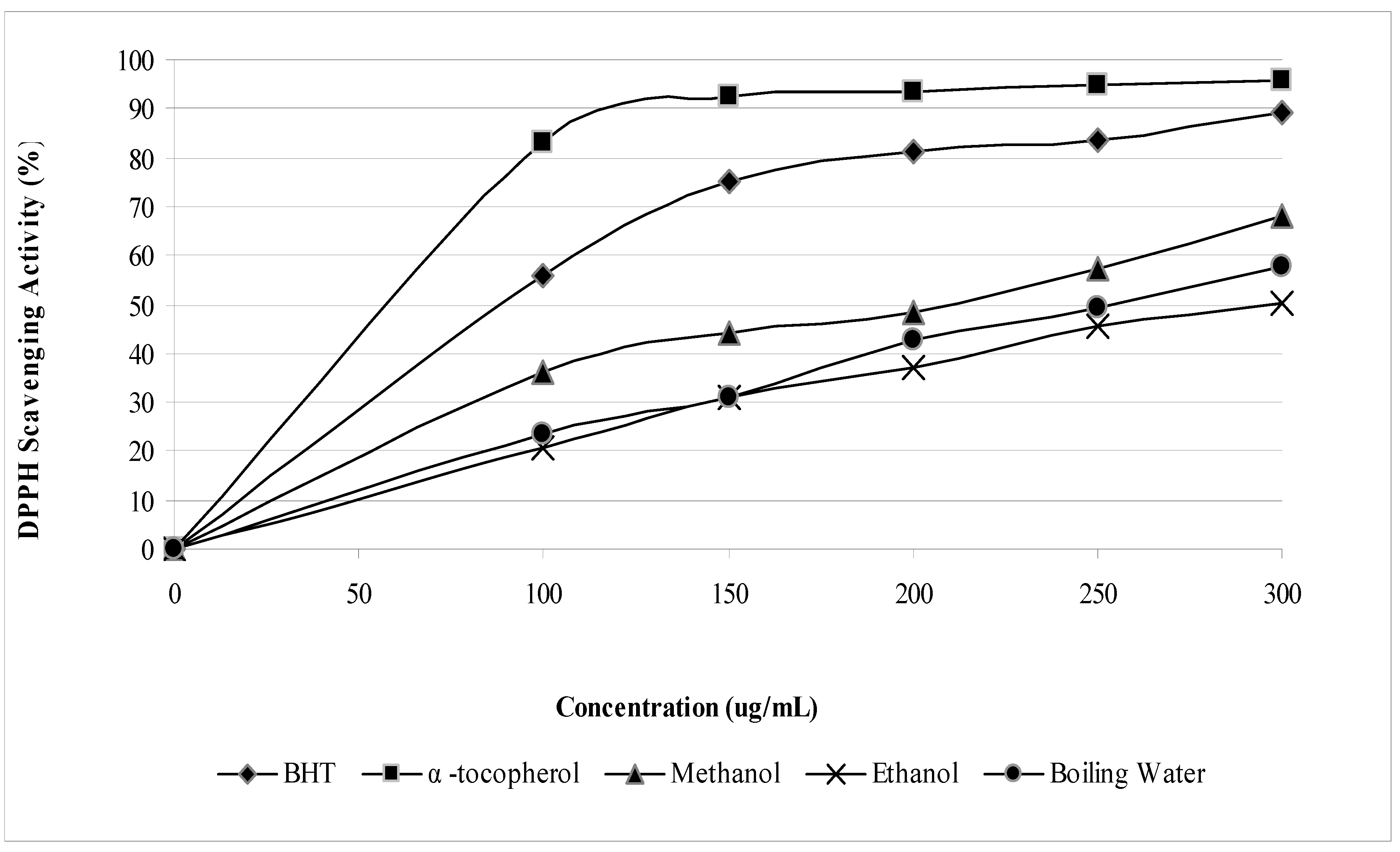
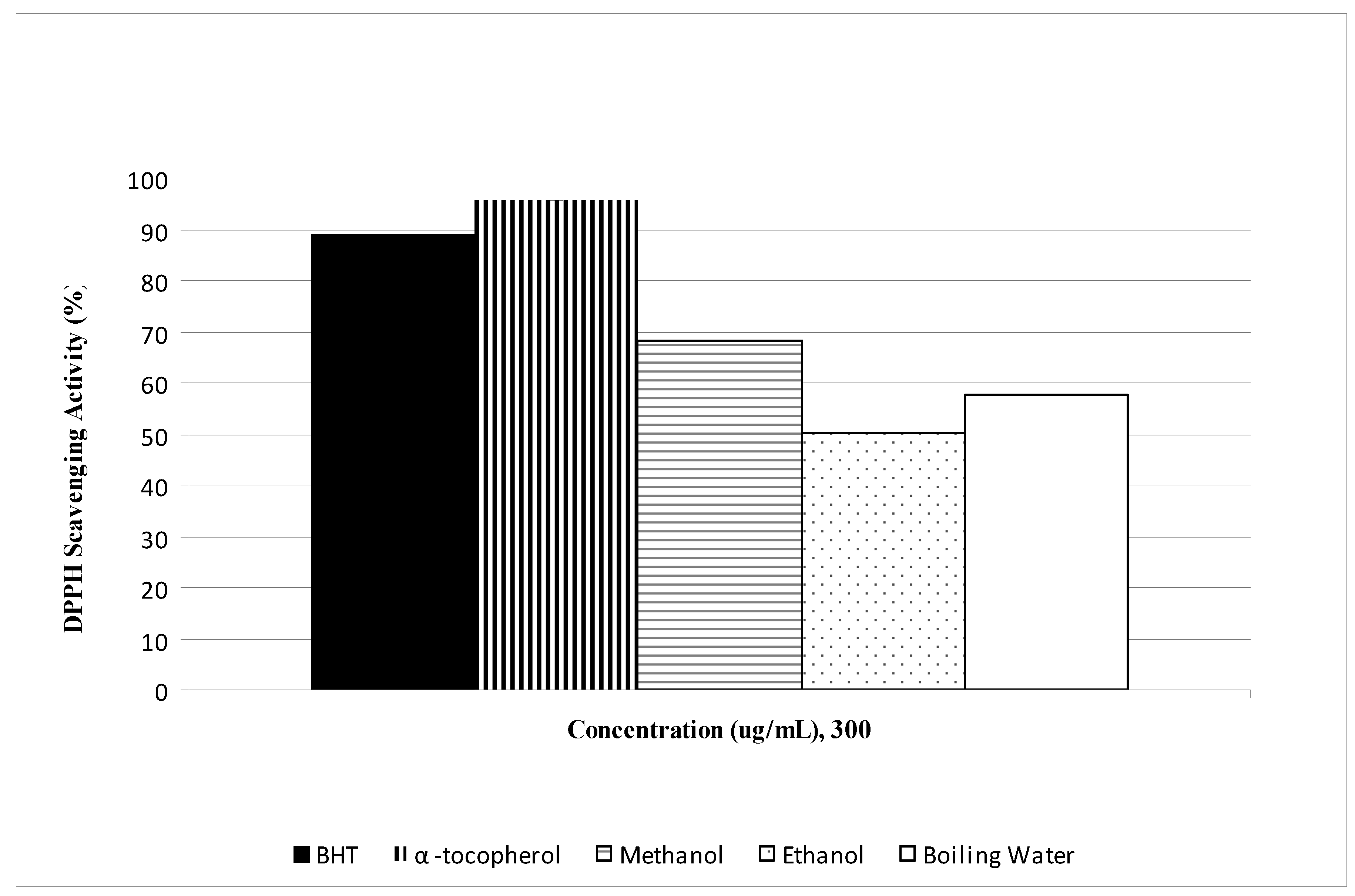
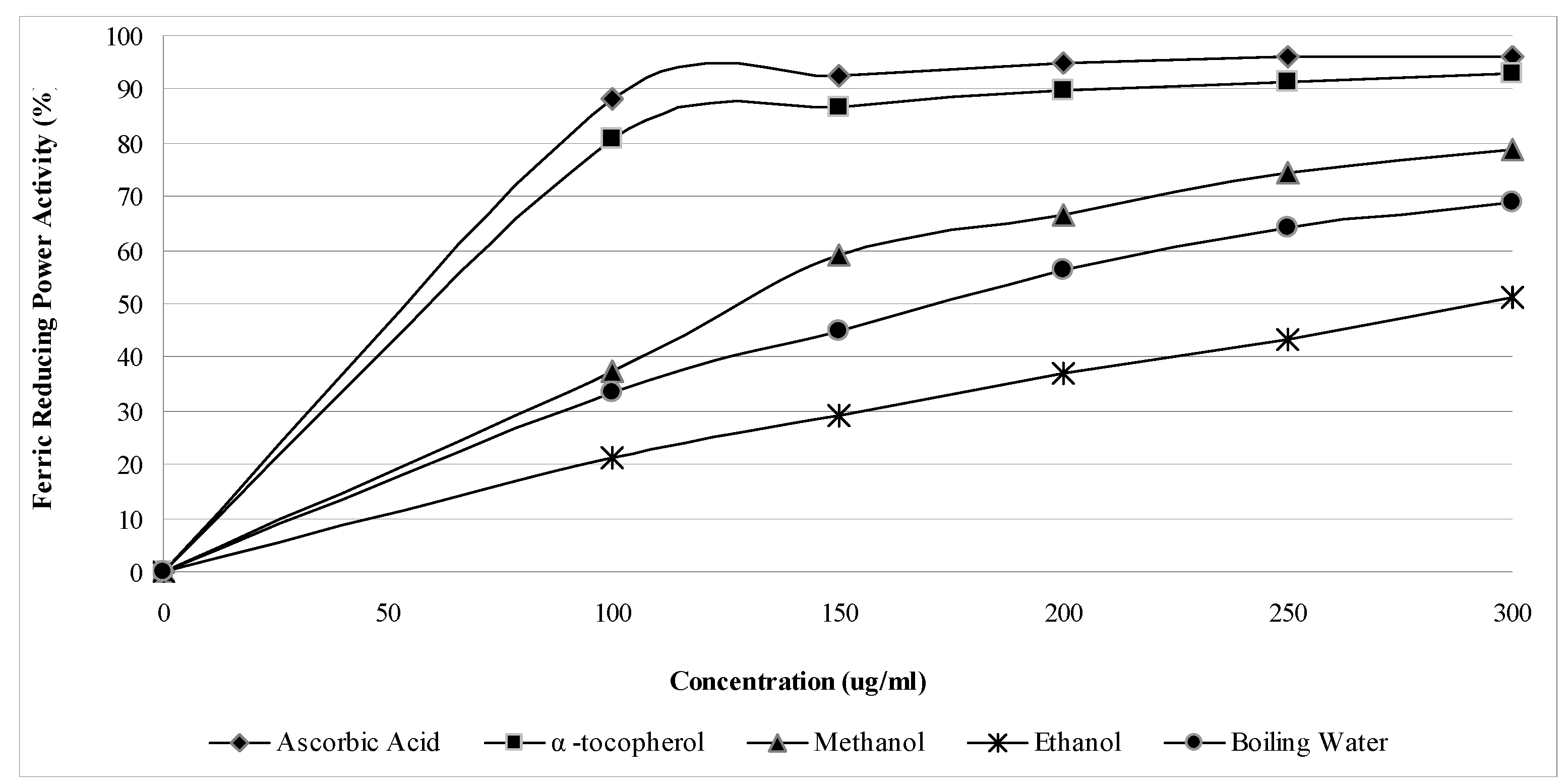
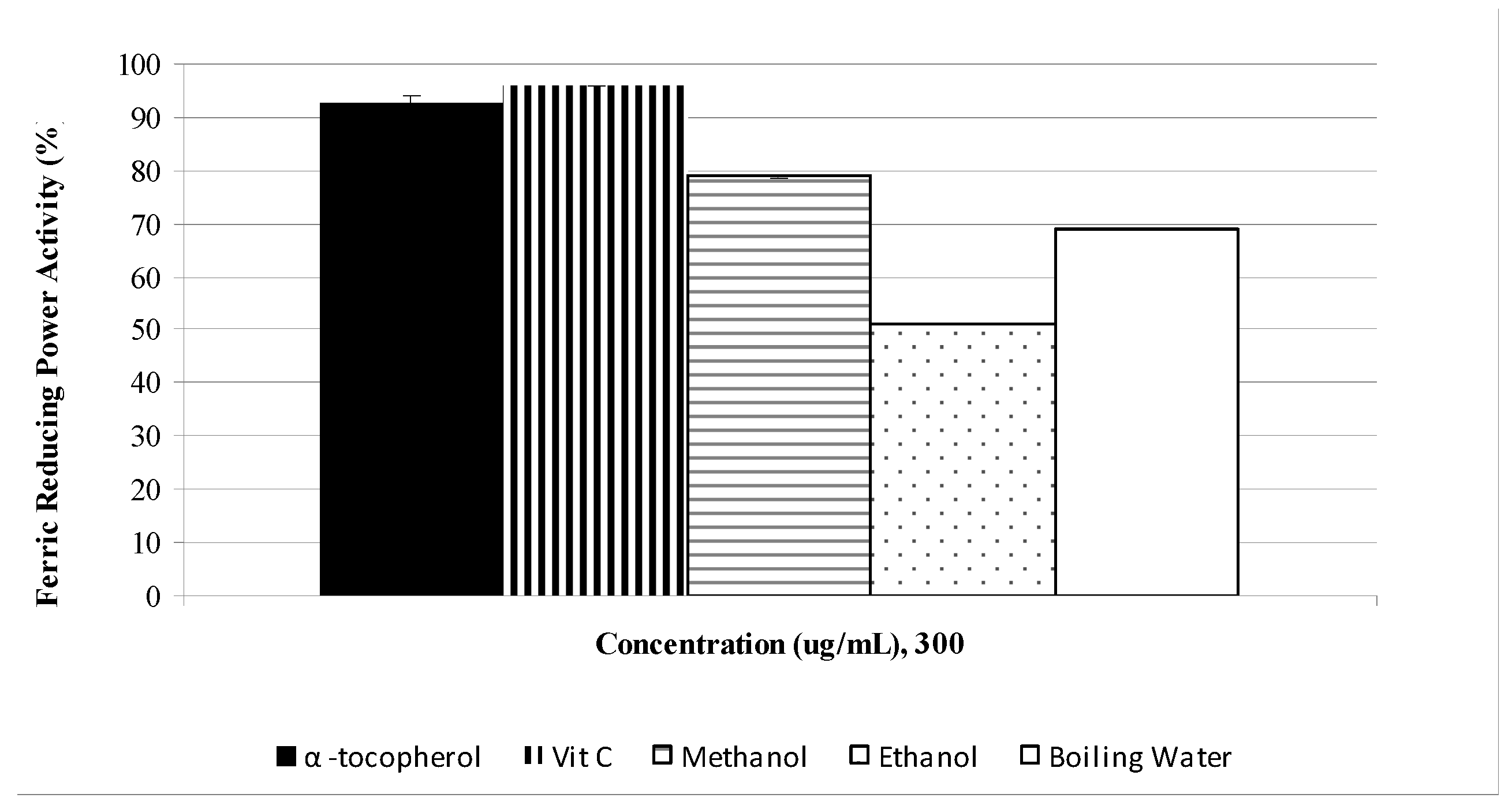
| Phenolic contents (mg/g dry sample) | ||||
|---|---|---|---|---|
| Gallic acid | Salicylic acid | Caffeic acid | Vanillic acid | Syringic acid |
| 1.82 ± 0.02 | ND | ND | ND | ND |
| Flavonoid contents (mg/g dry sample) | ||||||
|---|---|---|---|---|---|---|
| Apigenin | Kaempferol | Myricetin | Naringin | Quercetin | Pyrogallol | Rutin |
| ND | ND | ND | ND | ND | 1.4 ± 0.05 | ND |
2.2. Determination of phenolic and flavonoid compounds by HPLC
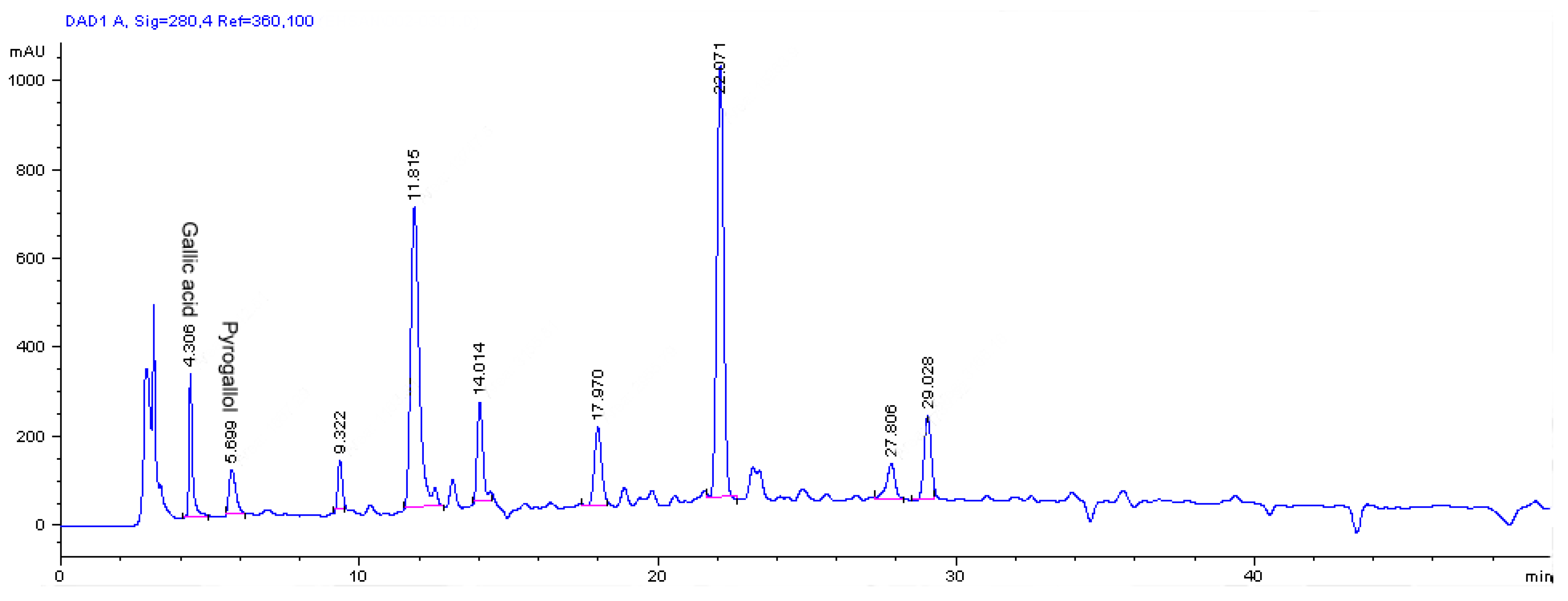
3. Experimental
3.1. Plant material
3.2. Preparation of saffron extracts
3.3. Total phenolic content
3.4. Total flavonoid content
3.5. Antioxidant activity (DPPH free radical scavenging activity)
3.6. Ferric reducing antioxidant power (FRAP)
3.7. Determination of phenolic and flavonoid compounds by HPLC
3.8. Statistical analysis
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds are available from the authors.
References and Notes
- Devasagayam, P.A.; Tilak, J.C.; Boloor, K.K.; Sane, K.S.; Ghaskadbi, S.S.; Lele, R.D. Free radicals and antioxidants in human health: current status and future prospects. J. Assoc. Phys. Ind. 2004, 52, 794–804. [Google Scholar]
- Serafini, M.; Bellocco, R.; Wolk, A.; Ekstrom, A.M. Total antioxidant potential of fruit and vegetables and risk of gastric cancer. Gastroenterology 2002, 123, 985–991. [Google Scholar] [CrossRef]
- Madsen, H.L.; Nielsen, B.R.; Bertelsen, G.; Skibsted, L.H. Screening of antioxidative activity of spices: Comparisons between assays based on ESR spin trapping and electrochemical measurement of oxygen consumption. Food Chem. 1996, 57, 331–337. [Google Scholar] [CrossRef]
- Shobana, S.; Naidu, K.A. Antioxidant activity of selected Indian spices. Prostagland., Leuk. Essent. Fatty Acids 2000, 62, 107–110. [Google Scholar]
- Reddy, A.C.; Lokesh, B.R. Studies on spice principles as antioxidants in the inhibition of lipid peroxidation of rat liver microsomes. Mol. Cell. Biochem. 1992, 111, 117–124. [Google Scholar]
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. J. Agric. Food Chem. 2001, 49, 5165–5170. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.C. Lipid peroxidation: a radical chain reaction. In Free Radical in Biology and Medicine, 2nd ed; Clarendon Press: Oxford, UK, 1989; pp. 189–267. [Google Scholar]
- Argolo, A.C.C.; Sant-Ana, A.E.G.; Pletsch, M.; Coelho, L.C.B.B. Antioxidant activity of leaf extracts from Bauhinia monandra. Bioresource Technol. 2004, 95, 229–233. [Google Scholar] [CrossRef]
- Tepe, B.; Sokmen, M.; Akpulat, H.A.; Sokmen, A. In vitro antioxidant activities of the methanol extracts of four Helichrysum species from Turkey. Food Chem. 2005, 90, 685–689. [Google Scholar] [CrossRef]
- Velioglu, Y.S.; Mazza, G.; Gao, L.; oomah, B.D. Antioxidant activity and total phenolics in selected fruits vegetables and grain products. J. Agric. Food Chem. 1998, 46, 4113–4117. [Google Scholar] [CrossRef]
- Robards, K.; Prenzler, P.D.; Tucker, G.; Swatsitang, P.; Glower, W. Phenolic compounds and their role in oxidative processes in fruits. Food Chem. 1999, 66, 401–436. [Google Scholar] [CrossRef]
- Caballero-Ortega, H.; Pereda-Miranda, R.; Abdullaev, F.I. HPLC quantification of major active components from 11 different saffron (Crocus sativus L.) sources. Food Chem. 2007, 100, 1126–1131. [Google Scholar] [CrossRef]
- Basker, D.; Negbi, M. Uses of saffron. Econ. Bot. 1983, 3, 228–235. [Google Scholar] [CrossRef]
- Locock, R.A. Alternatives saffron. Can. Pharm. J. 1995, 127, 45–46. [Google Scholar]
- Robinson, A. Notes on the saffron plant (Crocus sativus L.). Pharm. Hist. (Lond) 1995, 25, 2–3. [Google Scholar]
- Fernandez, J.A. Anticancer properties of saffron, Crocus sativus Linn. Adv. Phytomed. 2006, 313–330. [Google Scholar] [CrossRef]
- Wang, Y.C.; Chuang, Y.; Hsu, H. The flavonoid, carotenoid and pectin content in peels of citrus cultivated in Taiwan. Food Chem. 2008, 106, 277–284. [Google Scholar] [CrossRef]
- Chung, K.T.; Wong, T.Y.; Huang, Y.W.; Lin, Y. Tannins and human health: A review. Crit. Rev.Food Sci. Nutr. 1998, 38, 421–464. [Google Scholar] [CrossRef]
- Cassidy, A.; Hanley, B.; Lamuela-Raventos, R.M. Isoflavones, lignans and stilbenes: origins, metabolism and potential importance to human health. J. Sci. Food Agric. 2000, 80, 1044–1062. [Google Scholar] [CrossRef]
- Gao, D.; Kakuma, M.; Oka, S.; Sugino, K.; Sakurai, H. Reaction of β-alkannin (shikonin) with reactive oxygen species: detection of β-alkannin free radicals. Bioorg. Med. Chem. 2000, 8, 2561–2569. [Google Scholar] [CrossRef]
- Tapiero, H.; Tew, K.D.; Nguyen, B.G.; Mathé, G. Polyphenols: Do they play a role in the prevention of human pathologies? Biomed. Pharmacother. 2002, 56, 200–207. [Google Scholar] [CrossRef]
- Larson, R.A. The antioxidants of higher plants. Photochemistry 1998, 27, 969–978. [Google Scholar] [CrossRef]
- Frankel, E. Nutritional benefits of flavonids. In Proceedings of International Conference on Food Factors: Chemistry and Cancer Prevention, Hamamatsu, Japan, 10-15 December 1995.
- Buettner, G.R. The pecking order of free radicals and antioxidants: lipid peroxidation, α-tocopherol, and ascorbate. Arch. Biochem. Biophys. 1993, 300, 535–543. [Google Scholar] [CrossRef]
- Bravo, L. Polyphenols: Chemistry, dietary sources, metabolism, and nutritional significance. Nutr. Rev. 1998, 56, 317–333. [Google Scholar] [CrossRef]
- Assimopoulou, A.N.; Sinakos, Z.; Papageorgiou, V.P. Radical scavenging activity of Crocus sativus L. extract and its bioactive constituents. Phytother. Res. 2005, 19, 997–1000. [Google Scholar] [CrossRef]
- Chen, Y.; Zhang, H.; Tian, X.; Zhao, C.; Cai, L.; Liu, Y.; Jia, L.; Yin, H.X.; Chen, C. Antioxidant potential of crocins and ethanol extracts of Gardenia jasminoides ELLIS and Crocus sativus L.: A relationship investigation between antioxidant activity and crocin contents. Food Chem. 2008, 109, 484–492. [Google Scholar] [CrossRef]
- VonGadow, A.; Joubert, E.; Hansmann, C.F. Comparison of the antioxidant activity of aspalathin with that of other plant phenols of rooibos tea (Asalathus linearis), R-tocopherol, BHT, and BHA. J. Agric. Food Chem. 1997, 45, 632–638. [Google Scholar]
- Chatterjee, S.; Poduval, T.B.; Tilak, J.C.; Devasagayam, T.P.A. A modified, economic, sensitive method for measuring total antioxidant capacities of human plasma and natural compounds using Indian saffron (Crocus sativus). Clin. Chim. Acta 2005, 352, 155–163. [Google Scholar] [CrossRef]
- Yen, G.C.; Duh, P.D.; Tsai, C.L. Relationship between antioxidant activity and maturity of peanut hulls. J. Agric. Food Chem. 1993, 41, 67–70. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Mohan, P.S.; Becker, K. Studies on the antioxidant activity of Indian Laburnum (Cassia fistula L.): A preliminary assessment of crude extracts from stem bark, leaves, flowers and fruit pulp. Food Chem. 2002, 79, 61–67. [Google Scholar] [CrossRef]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef]
- Shan, B.; Cai, Y.Z.; Sun, M.; Corke, H. Antioxidant capacity of 26 spice extracts and characterization of their phenolic constituents. J. Agric. Food Chem. 2005, 53, 7749–7759. [Google Scholar] [CrossRef]
- Murugaian, P.; Srikumar, R.; Thangaraj, R. Isolation and estimation of phytochemical content in aqueous and ethanolic extract of Piper betel. J. Nat. Sci. Technol. - Life Sc. Bioinform. 2009, 1, 29–31. [Google Scholar]
- Proestos, C.; Chrianopoulos, N.; Nychas, G.-J.E.; Komaitis, M. RP-HPLC analysis of the phenolic compounds of plant extracts: Investigation of their antioxidant capacity and antimicrobial activity. J. Agric. Food Chem. 2005, 53, 1190–1195. [Google Scholar] [CrossRef]
- Sroka, Z.; Cisowski, W. Hydrogen peroxide scavenging, antioxidant and anti-radical activity of some phenolic acids. Food Chem. Toxicol. 2003, 41, 753–758. [Google Scholar] [CrossRef]
- Ramma, A.L.; Bahorun, T.; Crozier, A.; Zbarsky, V.; Datla, K.P.; Dexter, D.T.; Aruoma, O.I. Characterization of the antioxidant functions of flavonoids and proanthocyanidins in Mauritian black teas. Food Res. Int. 2005, 38, 357–367. [Google Scholar] [CrossRef]
- Middleton, E.; Kandaswami, J.R.C.; Theoharides, T.C. The effect of plant flavonoids on mammalian cells: implications for inflammation, heart disease and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar]
- Behbahani, M.; Ali, A.; Muse, R.; Mohd, N. Anti-oxidant and anti-inflammatory activities of leaves of Barringtonia racemosa. J. Med. Plant Res. 2007, 95–102. [Google Scholar]
- Charoensiri, R.; Kongkachuichai, R.; Suknicom, S.; Sungpuag, P. Beta-carotene, lycopene, and alpha-tocopherol contents of selected Thai fruits. Food Chem. 2009, 113, 202–207. [Google Scholar] [CrossRef]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A. Antioxidant activities, total phenolics and flavonoids content in two varieties of Malaysia young ginger (Zingiber officinale Roscoe). Molecules 2010, 15, 4324–4333. [Google Scholar] [CrossRef]
- Crozier, A.; Lean, M.E.J.; Mc Donald, M.S.; Black, C. Quantitative analysis of the flavonoid content of commercial tomatoes, onions, lettuce and celery. J. Agric. Food Chem. 1997, 45, 590–595. [Google Scholar] [CrossRef]
- Gulcin, I.; Sat, I.G.; Beydemir, S.; Elmastar, M.; Kufrevioglu, O.I. Comparisan of antioxidant activity of clove (Eugenia caryophylata Thunb) buds and lavender (Lavandula stoechs L.). Food Chem. 2004, 87, 393–400. [Google Scholar] [CrossRef]
- Halicia, M.; Odabasoglua, F.; Suleymanb, H.; Cakirc, A.; Asland, A.; Bayir, Y. Effects of water extract of Usnea longissima on antioxidant enzyme activity and mucosal damage caused by indomethacin in rats. Phytomedecine 2005, 12, 656–662. [Google Scholar] [CrossRef]
- Zhishen, J.; Mengcheng, T.; Jinming, W. The determination of flavonoid contents in mulberry and their scavenging effects on superoxide radicals. Food Chem. 1999, 64, 555–559. [Google Scholar] [CrossRef]
- Yen, G.C.; Duh, P.D. Scavenging effect methanolic extracts of peanut hulls on free-radical and active oxygen species. J. Agric. Food Chem. 1994, 42, 629–632. [Google Scholar] [CrossRef]
- Yen, G.C.; Chen, H.Y. Antioxidant activity of various tea extracts in relation to their antimutagenicity. J. Agric. Food Chem. 1995, 43, 27–32. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H.Z.E. Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity. Molecules 2010, 15, 6244-6256. https://doi.org/10.3390/molecules15096244
Karimi E, Oskoueian E, Hendra R, Jaafar HZE. Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity. Molecules. 2010; 15(9):6244-6256. https://doi.org/10.3390/molecules15096244
Chicago/Turabian StyleKarimi, Ehsan, Ehsan Oskoueian, Rudi Hendra, and Hawa Z.E. Jaafar. 2010. "Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity" Molecules 15, no. 9: 6244-6256. https://doi.org/10.3390/molecules15096244
APA StyleKarimi, E., Oskoueian, E., Hendra, R., & Jaafar, H. Z. E. (2010). Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity. Molecules, 15(9), 6244-6256. https://doi.org/10.3390/molecules15096244




