Synthesis and Odor Evaluation of Five New Sulfur-Containing Ester Flavor Compounds from 4-Ethyloctanoic Acid
Abstract
:1. Introduction
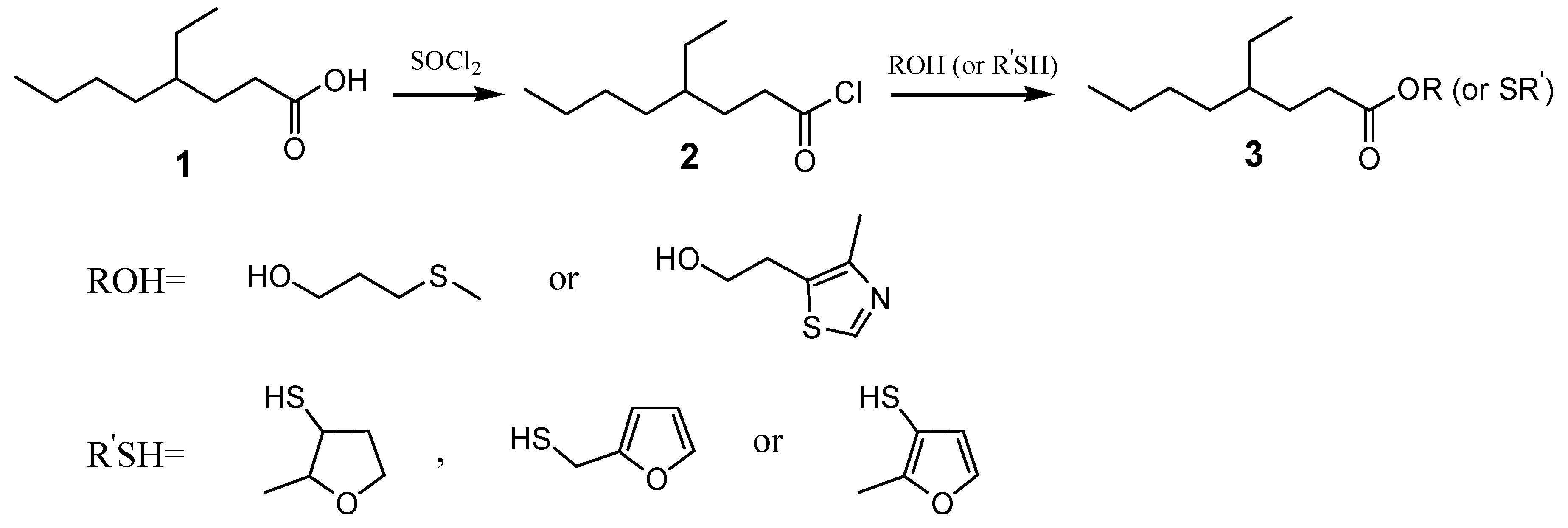
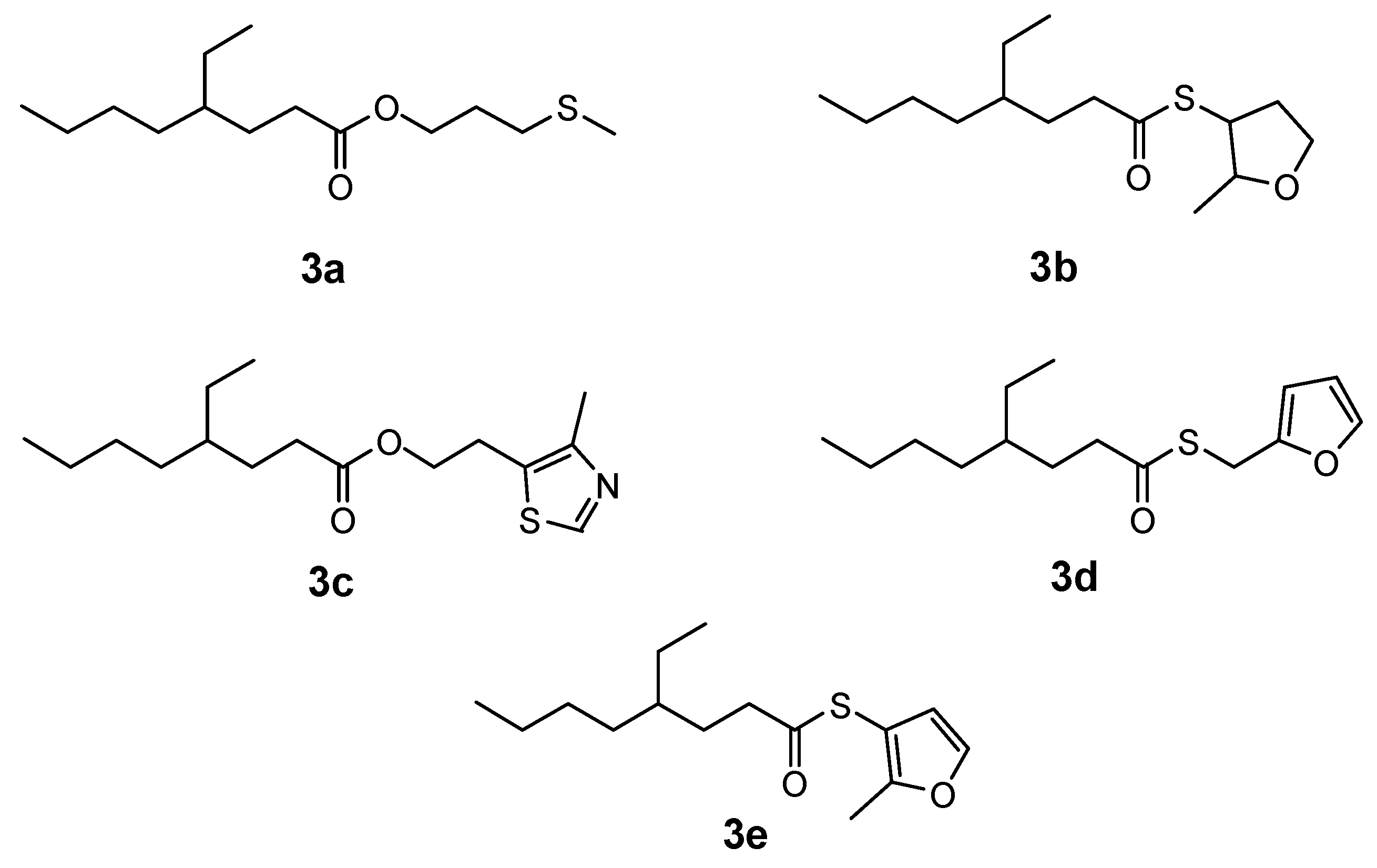
2. Results and Discussion
| Compound | Yield (%) | ||
|---|---|---|---|
| n (alcohol or mercaptan) : n (4-ethyloctanoyl chloride) | |||
| 1.2:1 | 1.6:1 | 2.0: 1 | |
| 3a | 85% | 90% | 94% |
| 3b | 67% | 78% | 94% |
| 3c | 93% | 89% | 87% |
| 3d | 93% | 94% | 95% |
| 3e | 79% | 84% | 86% |
| Compound | Odor characteristics |
|---|---|
| 3a | meaty, sauce, savory odor with a slight potato, radish and sulfury nuance |
| 3b | meaty, onion, metallic, sulfury odor with a slight hot nuance |
| 3c | a slight meaty, roasted and, burnt odor with sesame oil and fatty nuance |
| 3d | meaty, roasted meat, onion, sulfury odor with a slight garlic nuance |
| 3e | meaty, brothy, sulfury roasted odor with a slight sweet note |
3. Experimental
3.1. General
3.2. Synthesis of 4-ethyloctanoyl chloride (2)
3.3. General Synthesis of Compounds 3a-c
3.4. General Synthesis of Compound 3d-e
3.5. Odor evaluation
4. Conclusions
Acknowledgements
- Sample Availability: Samples of the compounds 3a-3e are available from the authors.
References and Notes
- Habib, F.; Nasser, I.; Mohammad, A. Pronounced catalytic effect of a micellar solution of sodium dodecyl sulfate (SDS) on the efficient C-S bond formation via an odorless thia-Michael addition reaction through the in situ generation of S-alkylisothiouronium salts. Adv. Synth. Catal. 2009, 351, 755–766. [Google Scholar]
- Habib, F.; Nasser, I.; Mohammad, A. A facile generation of C-S bonds via one-pot, odourless and efficient thia-Michaeladdition reactions using alkyl, aryl or allyl halides, thiourea and electron-deficientalkenes in wet polyethylene glycol (PEG 200) under mild reaction conditions. Tetrahedron 2009, 65, 5293–5301. [Google Scholar] [CrossRef]
- Beletskaya, I.P.; Tarasenko, E.A.; Khokhlov, A.R. Poly(N-vinylimidazole) as efficient and recyclable catalyst for the addition of thiols to Michael acceptors in aqueous medium. Russ. J. Org. Chem. 2007, 43, 1733–1736. [Google Scholar] [CrossRef]
- Tachibana, S.; Kasemura, K.; Ohtani, T. Synthesis and physiological activity of S-furfuryl thiocarboxylates. Nippon Nogei Kagaku Kaishi 2002, 76, 962–969. [Google Scholar] [CrossRef]
- Shimizu, H.; Sagara, R.; Aizawa, S. Preparation of amino group-containing esters by using solid superstrong acid catalysts. JP 06321878, 22 November 1994. [Google Scholar]
- Boustany, K.S. Addition of methanethiol to some α,β-unsaturated aldehydes and study of the reduction and acetalization of the addition products. J. Chem. U.A.R. 1966, 9, 317–322. [Google Scholar]
- Liu, Y.P.; Sun, B.G.; Zheng, F.P. Synthesis of 3-(methylthio)propyl acetate. Shipin gongye keji 2005, 26, 159–160. [Google Scholar]
- Tadashi, S. Comparison of the activity of different aneurine esters of organic acids. Bull. Chem. Soc. Jpn. 1944, 19, 185–205. [Google Scholar]
- Ayers, J.A.; Anderson, S.R. A preparative scale reduction of alkyl disulfides with tributyl phophine and water. Synth. Commun. 1999, 29, 351–358. [Google Scholar] [CrossRef]
- Winter, M.; Gautschi, F.; Flament, I. Flavoring agent. US Pat. 3,931,245, 6 January 1976. [Google Scholar]
- Evers, W.J.; Heinsohn, H.H.; Mayers, B.J. Novel flavoring compositions and products comprising 2,5-dimethyl-3-thioisovaleryl furan. US Pat. 3,958,029, 18 May 1976. [Google Scholar]
- Evers, W.J.; Heinsohn, H.H.; Mayers, B.J. Novel flavoring compositions and products containing 2-methyl-3-thio-(2-methylbutyryl)-furan. US Pat. 3,961,093, 1 June 1976. [Google Scholar]
- Evers, W.J.; Heinsohn, H.H.; Mayers, B.J. 3-Furyl thioesters to impart meaty aroma and taste. US 3,917,869, 4 November 1975. [Google Scholar]
- Evers, W.J.; Mayers, B.J.; Heinson, H.H. Acylthiofuran derivatives and their use as flavors. DE 2437890, 20 February 1975. [Google Scholar]
- Huang, X.J.; Liu, C.C. Synthesis of perfume furfuryl thiopropionate. Riyong huaxue gongye 2007, 37, 417–418. [Google Scholar]
- Ouweland, V.D.; Maria, G.A.; Peer, H.G. Compounds for giving aroma to foods. AT 340748, 27 December 1977. [Google Scholar]
- Boelens, H.; Haring, H. G.; Rijke, D.D. Threshold values of and human preferences for 4-ethyl octanoic and 3-methyl butanoic acid. Perfum. Flavor. 1983, 8, 71–73. [Google Scholar]
- Mosciano, G. Organoleptic characteristics of flavor materials. Perfum. Flavor. 2007, 32, 16–18. [Google Scholar]
- Mosciano, G. Organoleptic characteristics of flavor materials. Perfum. Flavor. 2006, 31, 48–51. [Google Scholar]
- Mosciano, G.; Fasano, M.; Cassidy, J. Organoleptic characteristics of flavor materials. Perfum. Flavor. 1993, 18, 51–53. [Google Scholar]
- Sun, B.G. Sulfur-Containing Flavour Chemistry(in Chinese); Science Press: Beijing, China, 2007; pp. 41–70. [Google Scholar]
- Mosciano, G. Organoleptic characteristics of flavor materials. Perfum. Flavor. 2001, 26, 68–72. [Google Scholar]
- Wang, G.Y.; Liu, X.J.; Li, L.B. Synthesis of perfume furfuryl butyrate and its characterization. Jingxi Huagong 2004, 21, 589–590. [Google Scholar]
- Zheng, F.P.; Sun, B.G.; He, J. Syntheses of ethylthiol furoate and furfurylthiol furoate. Jingxi Huagong 1997, 14, 17–19. [Google Scholar]
- Wu, R.K. Aromatics—Method for Evaluation of Odor; GB/T 14454.2-1993; Standards press of China: Beijing, China, 1993. [Google Scholar]
- Xie, J.C.; Sun, B.G.; Wang, S.B. Aromatic constituents from Chinese traditional smoke-cured bacon of Mini-pig. Food Sci. Tech. Int. 2008, 14, 329–340. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liu, Y.; Chen, H.; Yin, D.; Sun, B. Synthesis and Odor Evaluation of Five New Sulfur-Containing Ester Flavor Compounds from 4-Ethyloctanoic Acid. Molecules 2010, 15, 5104-5111. https://doi.org/10.3390/molecules15085104
Liu Y, Chen H, Yin D, Sun B. Synthesis and Odor Evaluation of Five New Sulfur-Containing Ester Flavor Compounds from 4-Ethyloctanoic Acid. Molecules. 2010; 15(8):5104-5111. https://doi.org/10.3390/molecules15085104
Chicago/Turabian StyleLiu, Yuping, Haitao Chen, Decai Yin, and Baoguo Sun. 2010. "Synthesis and Odor Evaluation of Five New Sulfur-Containing Ester Flavor Compounds from 4-Ethyloctanoic Acid" Molecules 15, no. 8: 5104-5111. https://doi.org/10.3390/molecules15085104
APA StyleLiu, Y., Chen, H., Yin, D., & Sun, B. (2010). Synthesis and Odor Evaluation of Five New Sulfur-Containing Ester Flavor Compounds from 4-Ethyloctanoic Acid. Molecules, 15(8), 5104-5111. https://doi.org/10.3390/molecules15085104




