Identification of Two Polysaccharides from Prunella vulgaris L. and Evaluation on Their Anti-Lung Adenocarcinoma Activity
Abstract
:1. Introduction
2. Results and Discussion
2.1. Isolation of polysaccharide from PV
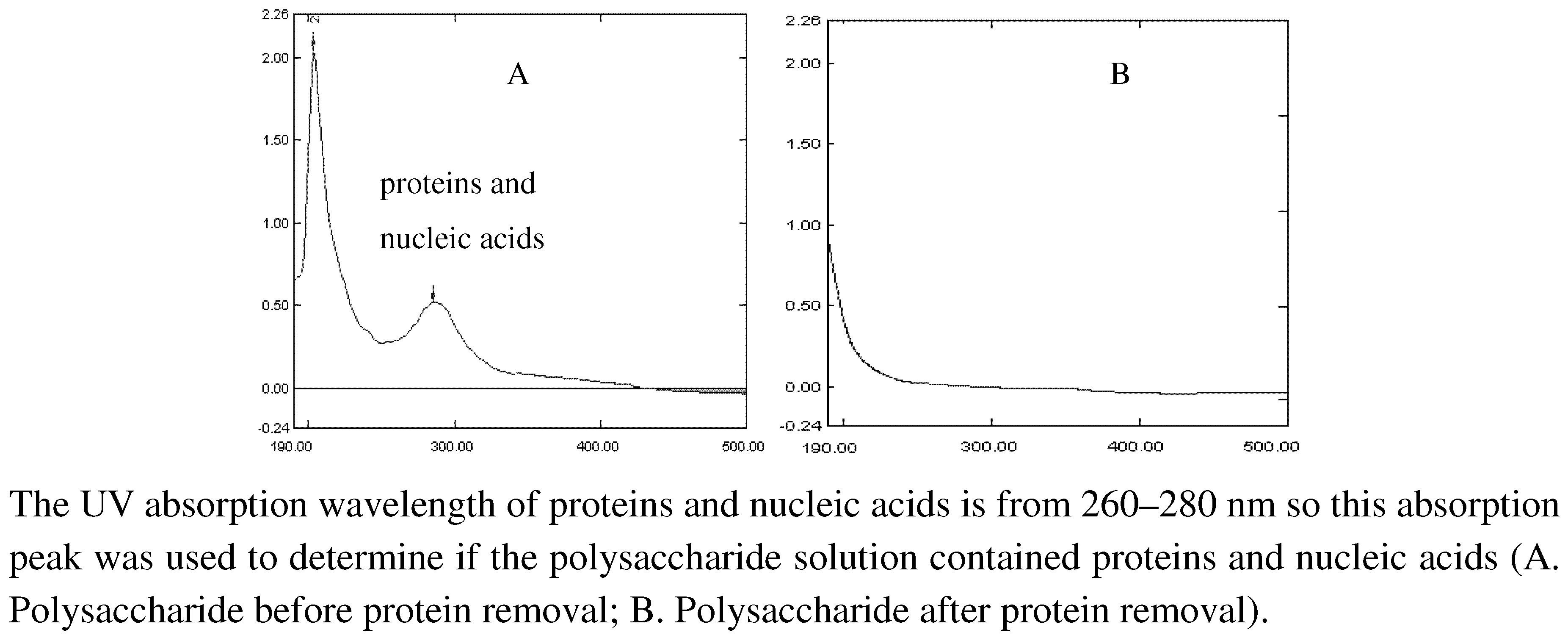
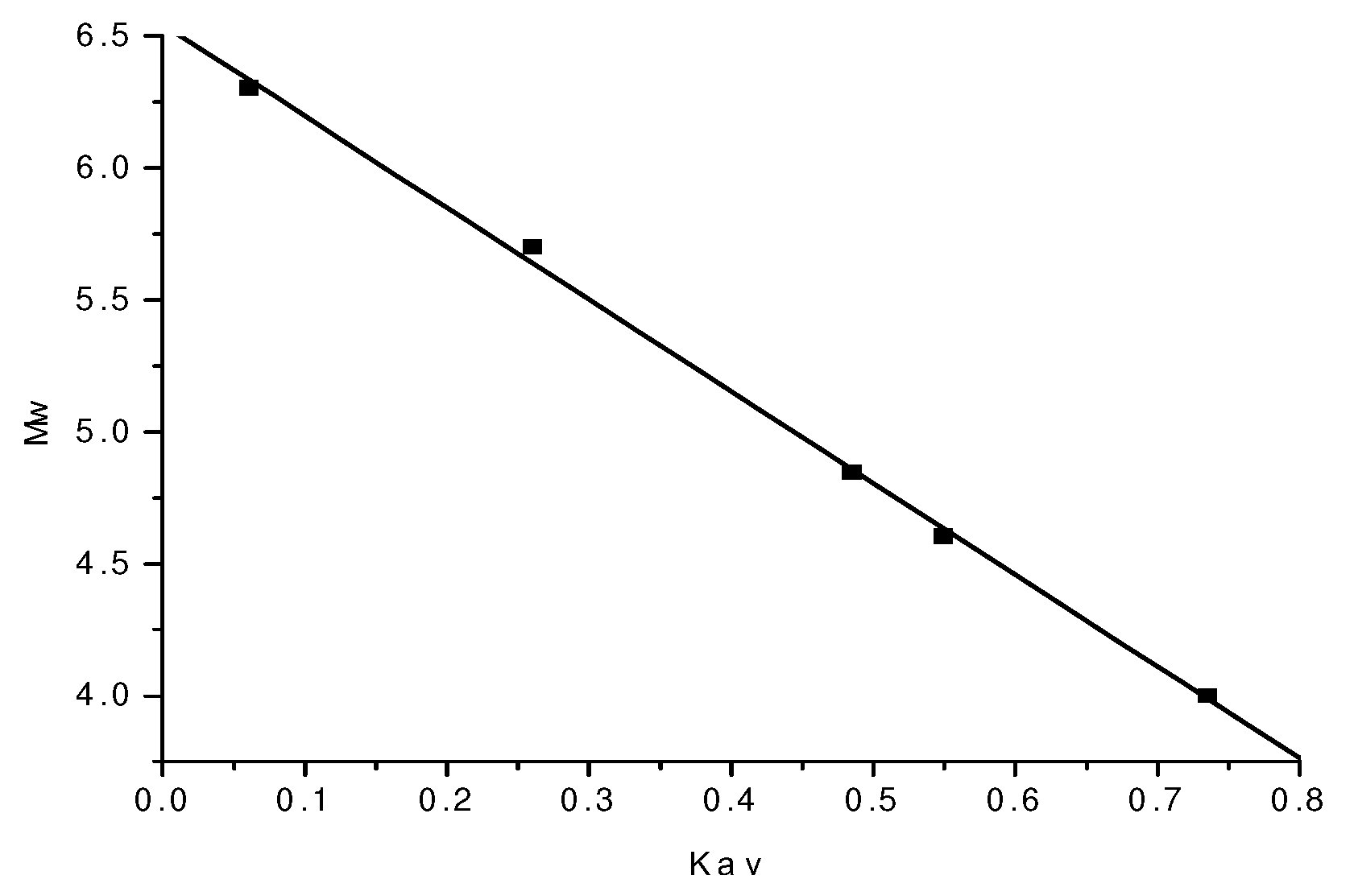
2.2. Molecular weight determination of polysaccharide
| Dextran | Mw | LgMw | Ve ( min) | Kav=(Ve-Vo)/(Vt-Vo) |
|---|---|---|---|---|
| T-10 | 10,000 | 4 | 12.643 | 0.7353 |
| T-40 | 40,000 | 4.602 | 11.344 | 0.5494 |
| T-70 | 70,000 | 4.845 | 10.895 | 0.4852 |
| T-500 | 500,000 | 5.699 | 9.325 | 0.2606 |
| T-2000 | 2,000,000 | 6.301 | 7.930 | 0.061 |
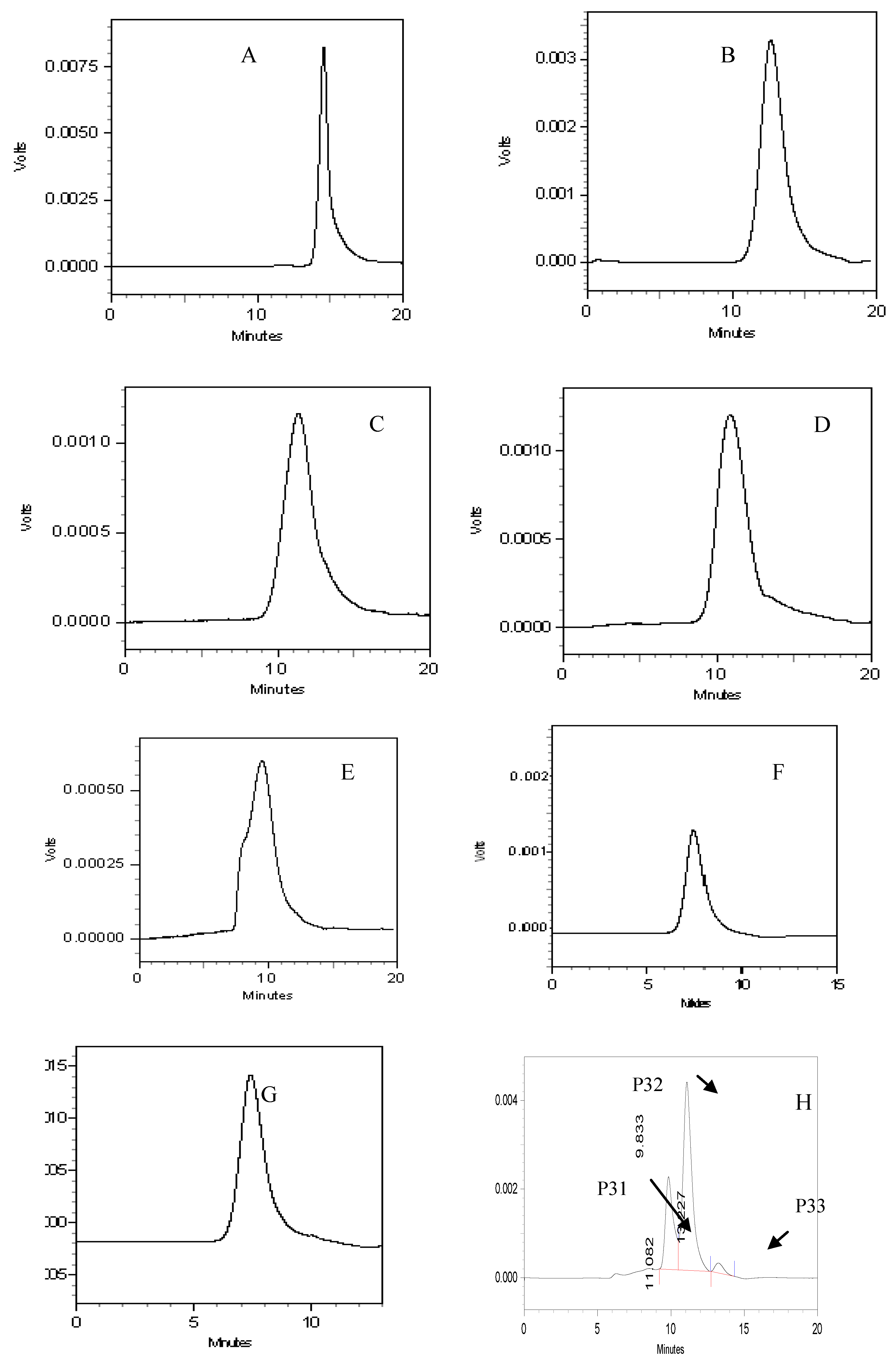
| Components | Retention time(min) | Molecular weight |
|---|---|---|
| P31 | 9.833 | 242,641 |
| P32 | 11.082 | 58,060 |
| P33 | 13.227 | 4,980 |
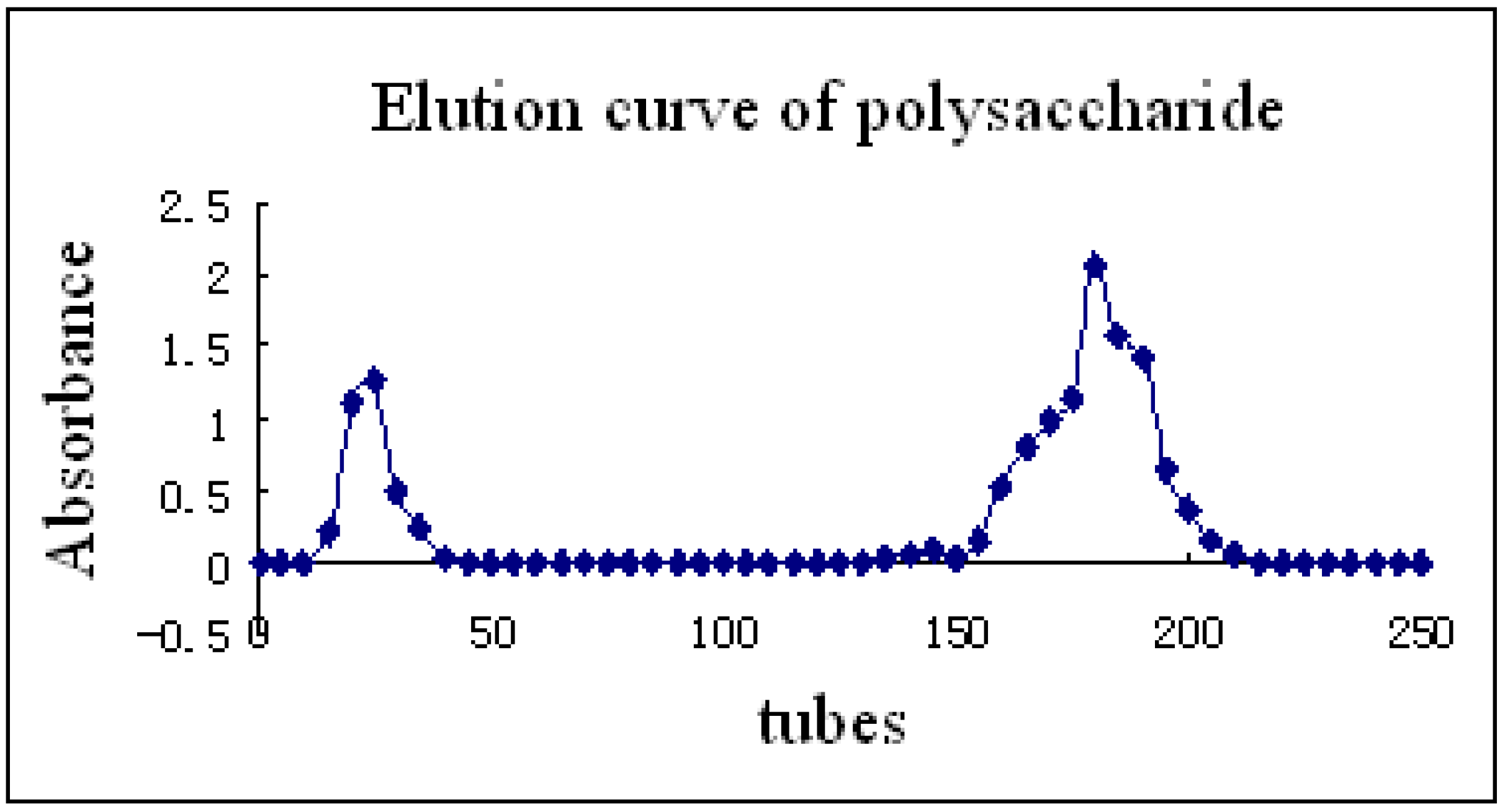
2.3. Monosaccharide composition of polysaccharide and its molar ratio
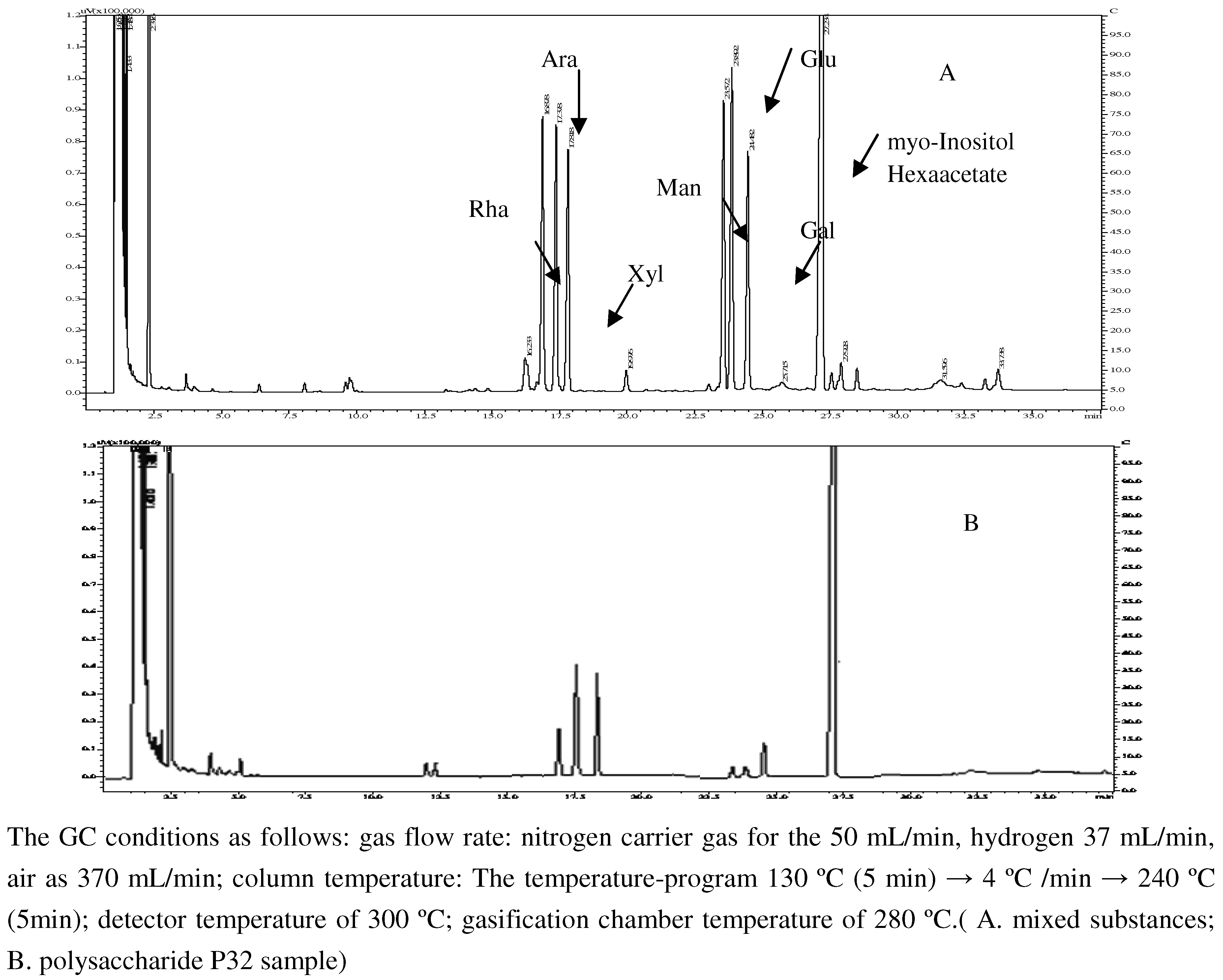
2.4. In vivo anti-lung adenocarcinoma activity of PV polysaccharide
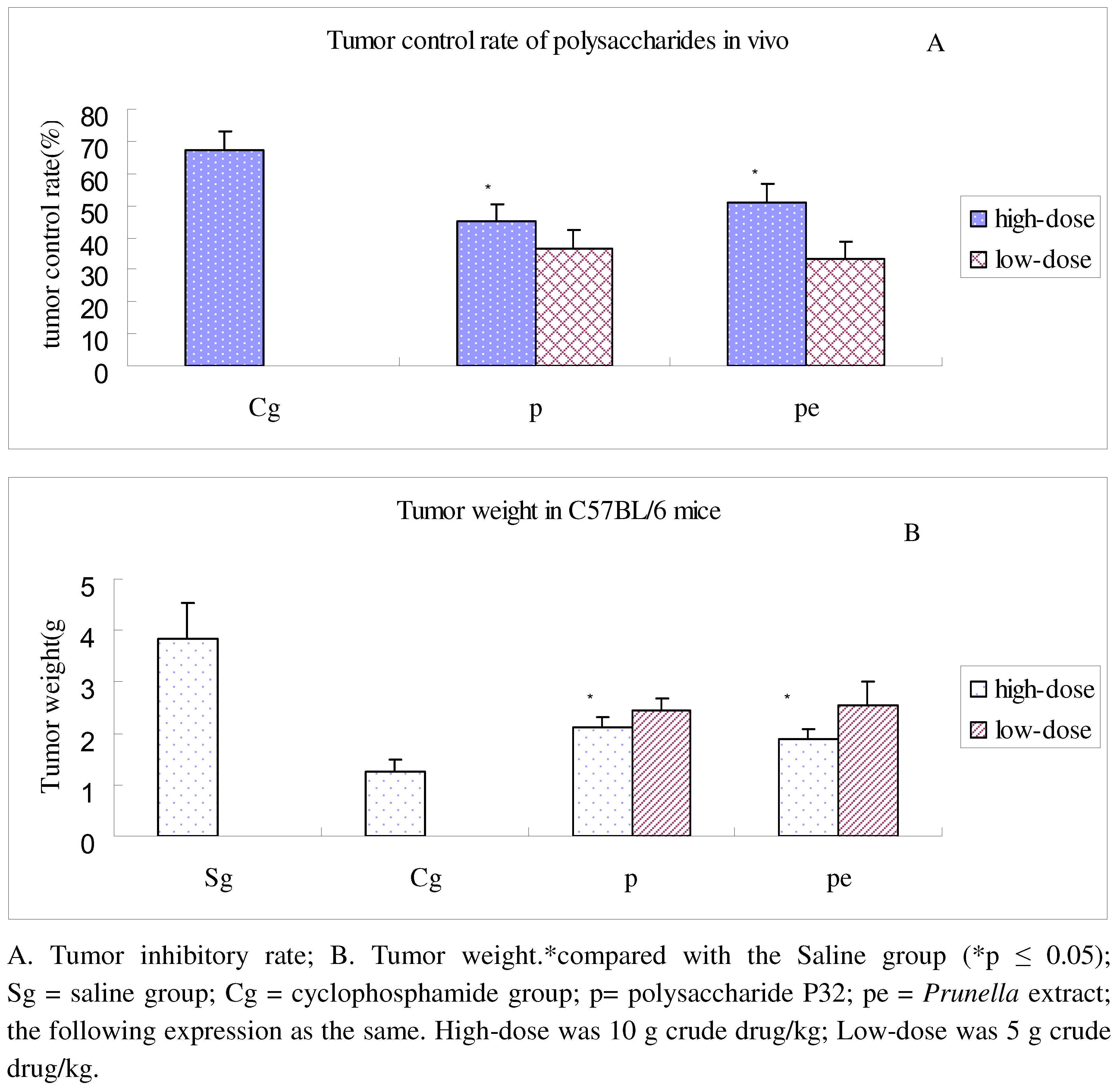
| Group | Thymus (mg) | Weight (g) | Thymus index (mg/g) |
|---|---|---|---|
| Sg | 35.71 ± 7.87 | 21.43 ± 1.72 | 1.67 ± 0.28 |
| Cg | 30.00 ± 8.16 | 20.86 ± 2.54 | 1.43 ± 0.30 |
| 10 g crude drug/kg of p | 46.25 ± 7.44 | 19.88 ± 2.36 | 2.33 ± 0.22** |
| 5 g crude drug/kg of p | 38.57 ± 3.78 | 20.43 ± 1.72 | 1.89 ± 0.16 |
| 10 g crude drug/kg of pe | 41.25 ± 6.40 | 21.63 ± 2.83 | 1.92 ± 0.26* |
| 5 g crude drug/kg of pe | 34.29 ± 7.87 | 19.43 ± 3.15 | 1.76 ± 0.25 |
| Group | Spleen(mg) | Weight (g) | Spleen index (mg/g) |
|---|---|---|---|
| Sg | 155.71 ± 16.18 | 21.43 ± 1.72 | 7.26 ± 0.31 |
| Cg | 105.71 ± 16.18 | 20.86 ± 2.54 | 5.06 ± 0.25 |
| 10 g crude drug/kg of p | 192.50 ± 26.05 | 19.88 ± 2.36 | 9.71 ± 0.90** |
| 5 g crude drug/kg of p | 150.00 ± 24.49 | 20.43 ± 1.72 | 7.32 ± 0.80 |
| 10 g crude drug/kg of pe | 168.75 ± 34.82 | 21.63 ± 2.83 | 7.79 ± 1.19 |
| 5 g crude drug/kg of pe | 155.71 ± 28.78 | 19.43 ± 3.15 | 8.00 ± 0.52 |
3. Experimental
3.1. General
3.2. Plant material
3.3. Extraction of crude polysaccharides
3.4. Deproteination of crude polysaccharides
3.5. Molecular weight determination
3.6. Gel column purification of protein-free polysaccharide
3.7. Preparation of myo-inositol hexaacetate as the internal standard
3.8. Hydrolysis and derivatization of monosaccharide and polysaccharide
3.9. Anti-lung adenocarcinoma activity of polysaccharide in C57BL/6 mice
3.10. Statistical analysis
4. Conclusions
Acknowledgements
- Sample Availability: Not Available.
References and Notes
- Tutin, T.G.; Burges, N.A.; Chater, A.O.; Edmondson, J.R.; Heywood, V.H.; Moore, D.M.; Valentine, D.H.; Walters, S.M.; Webb, D.A. Flora Europaea; Cambridge University Press,: Cambridge, UK, 1993; Volume 1-5. [Google Scholar]
- Jitka, P.; Milan, K.; Jaromír, S.; Zdenek, S.; Jaroslav, V.; Jitka, U. Biological Activities of Prunella vulgaris Extract. Phytother. Res. 2003, 17, 1082–1087. [Google Scholar] [CrossRef]
- Feng, L.; Chen, Y.; Jia, X.B.; Tan, X.B.; Gao, C.L.; Liu, G.M.; Shao, Z.Z. Evaluation of hierarchical cluster on fingerprint characteristic of Prunella species. China J. Trad. Chin. Med. Pharm. 2009, 9, 1216–1219. [Google Scholar]
- Han, E.H.; Choi, J.H.; Hwang, Y.P.; Park, H.J.; Choi, C.Y.; Chung, Y.C.; Seo, J.K.; Jeong, H.G. Immunostimulatory activity of aqueous extract isolated from Prunella vulgaris. Food Chem. Toxicol. 2009, 47, 62–69. [Google Scholar] [CrossRef]
- Harput, U.S.; Saracoglu, I.; Ogihara, Y. Effects of Two Prunella Species on Lymphocyte Pliferation and Nitric Oxide Production. Phytother. Res. 2006, 20, 157–159. [Google Scholar] [CrossRef]
- Fang, X.; Yu, M.M.; Yuen, W.H.; Zee, S.Y.; Chang, R.C. Immune modulatory effects of Prunella vulgaris L. on monocytes/macrophages. Int. J. Mol. Med. 2005, 16, 1109–1116. [Google Scholar]
- Fang, X.; Chang, R.C.; Yuen, W.H.; Zee, S.Y. Immune modulatory effects of Prunella vulgaris L. Int. J. Mol. Med. 2005, 15, 491–496. [Google Scholar]
- Tabba, H.D.; Chang, R.S.; Smith, K.M. Isolation, purification, and partial characterization of prunellin, an anti-HIV component from aqueous extracts of Prunella vulgaris. Antivir. Res. 1989, 11, 263–273. [Google Scholar] [CrossRef]
- Zhang, D.H. Isolation, Purification and Antioxidant Activity of the Polysaccharide of Prunella vugaris(Labiatae). Acta Botaniea Yunnaniea 2006, 28, 410–414. [Google Scholar]
- Chen, Y.; Xie, M.Y.; Wang, Y.X.; Nie, S.P.; Li, C. Analysis of the monosaccharide composition of purified polysaccharides in Ganoderma atrum by capillary gas chromatography. Phytochem. Anal. 2009, 20, 503–510. [Google Scholar]
- Zhang, Y.; Yu, Y.; Zhang, H. Analysis of monosaccharides in Radix Rehmanniae by GC. Zhongguo Zhong Yao Za Zhi 2009, 34, 419–422. [Google Scholar]
- Xu, D.J.; Xia, Q.; Wang, J.J.; Wang, P.P. Molecular Weight and Monosaccharide Composition of Astragalus Polysaccharides. Molecules 2008, 13, 2408–2415. [Google Scholar] [CrossRef]
- Liu, F.; Zhu, X.H.; Liu, M.; Zhou, J.; Che, X.P.; Han, R.F. Study on deproteinization and decoloration in extraction of polysaccharides from Rabdosia rubescens. Zhong Yao Cai 2008, 31, 751–753. [Google Scholar]
- Jiang, L.H.; Feng, Y.; Shen, L.; Xu, D.S. Research on method in avoiding the interference of poloxamer in determination of polysaccharides in thermosensitive gel. Zhong Yao Cai 2007, 30, 1443–1446. [Google Scholar]
- Gao, Z.; Zhang, D.; Guo, C. Paclitaxel Efficacy is Increased by Parthenolide via Nuclear Factor-KappaB Pathways in In Vitro and In Vivo Human Non-Small Cell Lung Cancer Models. Curr. Cancer Drug Targets 2010, in press. [Google Scholar]
- Choi, J.H.; Han, E.H.; Hwang, Y.P.; Choi, J.M.; Choi, C.Y.; Chung, Y.C.; Seo, J.K.; Jeong, H.G. Suppression of PMA-induced tumor cell invasion and metastasis by aqueous extract isolated from Prunella vulgaris via the inhibition of NF-κB-dependent MMP-9 expression. Food Chem. Toxicol. 2010, 48, 564–571. [Google Scholar] [CrossRef]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Feng, L.; Jia, X.-B.; Shi, F.; Chen, Y. Identification of Two Polysaccharides from Prunella vulgaris L. and Evaluation on Their Anti-Lung Adenocarcinoma Activity. Molecules 2010, 15, 5093-5103. https://doi.org/10.3390/molecules15085093
Feng L, Jia X-B, Shi F, Chen Y. Identification of Two Polysaccharides from Prunella vulgaris L. and Evaluation on Their Anti-Lung Adenocarcinoma Activity. Molecules. 2010; 15(8):5093-5103. https://doi.org/10.3390/molecules15085093
Chicago/Turabian StyleFeng, Liang, Xiao-Bin Jia, Feng Shi, and Yan Chen. 2010. "Identification of Two Polysaccharides from Prunella vulgaris L. and Evaluation on Their Anti-Lung Adenocarcinoma Activity" Molecules 15, no. 8: 5093-5103. https://doi.org/10.3390/molecules15085093
APA StyleFeng, L., Jia, X.-B., Shi, F., & Chen, Y. (2010). Identification of Two Polysaccharides from Prunella vulgaris L. and Evaluation on Their Anti-Lung Adenocarcinoma Activity. Molecules, 15(8), 5093-5103. https://doi.org/10.3390/molecules15085093




