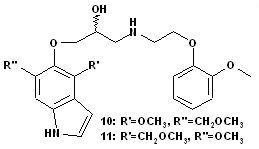
Synthesis and Adrenolytic Activity of New Propanolamines
Abstract
:1. Introduction
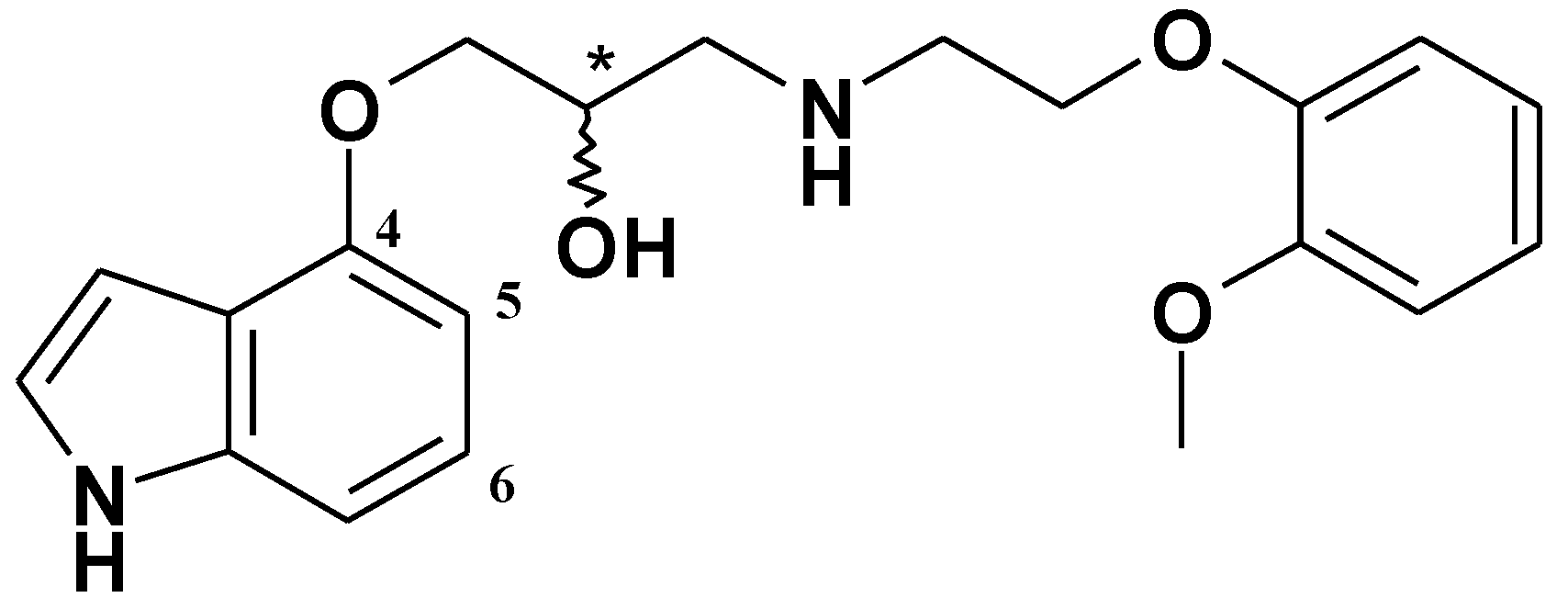
2. Results and Discussion
2.1. Chemistry

2.2. Pharmacology
2.2.1. Radioligand receptor binding assay for α- and β-adrenergic receptors
| Compound | [3H]Prazosin | [3H]Clonidine | [3H]CGP12177 |
|---|---|---|---|
| 10 | 5.9 ± 0.3 | 306.7 ± 11.7 | 6.9 ± 0,8 μM |
| 11 | 21.4 ± 1.3 | 166.5 ± 9.8 | 4.2 ± 0,6 μM |
| (R,S)-9 | 89.8 ± 9.5 | 1.4 ± 0.4 μM | 3.0 ± 0.6 |
| Carvedilol | 2.2 ± 0.2a | 3.4 ± 0.9 μM | 0.81 ± 0.06a |
2.2.2. Effect on normal electrocardiogram (ECG) in vivo in rats
| Compound Parameters | Time of observation (min) | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 1 | 5 | 15 | |||||||||
| 10 | P–Q (ms) | 40.4 ± 0.4 | 45.6 ± 1.6b | 46.8 ± 1.6c | 46.4 ± 1.6c | |||||||
| QRS (ms) | 27.6 ± 0.8 | 31.2 ± 0.5c | 31.2 ± 1.0c | 32.4 ± 0.8d | ||||||||
| Q–T (ms) | 94.4 ± 2.8 | 92.0 ± 4.2 | 100.0 ± 2.8 | 104.0 ± 1.4a | ||||||||
| Beats/min | 329.4 ± 12.2 | 334.0 ± 13.0 | 309.6 ± 13.7 | 297.4 ± 12.4 | ||||||||
| 11 | P–Q (ms) | 42.8 ± 1.2 | 47.2 ± 1.0c | 46.8 ± 0.5b | 48.0 ± 1.1c | |||||||
| QRS (ms) | 27.2 ± 1.4 | 30.4 ± 0.4b | 31.2 ± 0.8c | 32.4 ± 0.8d | ||||||||
| Q–T (ms) | 85.2 ± 0.8 | 87.2 ± 1.2 | 91.2 ± 3.3 | 97.2 ± 2.2d | ||||||||
| Beats/min | 337.8 ± 6.7 | 332.4 ± 10.6 | 320.6 ± 7.8 | 310.2 ± 9.4a | ||||||||
| (R,S)-9 | P–Q (ms) | 45.7 ± 2.0 | 47.2 ± 1.6 | 48.1 -± 2.4 | 46.2 ± 1.7 | |||||||
| QRS (ms) | 26.4 ± 1.3 | 28.4 ± 1.2 | 24.9 ± 1.3 | 26.9 ± 1.1 | ||||||||
| Q–T (ms) | 72.6 ± 1.7 | 69.2 ± 1.1 | 69.0 ± 1.6 | 72.8 ± 1.3 | ||||||||
| Beats/min | 304.9 ± 8.9 | 298.7 ± 7.2 | 294.5 ± 7.7 | 287.3 ± 7.9 | ||||||||
| Carvedilol | P–Q (ms) | 50.0 ± 3.2 | 50.0 ± 3.2 | 55.0 ± 5.5 | 54.6 ± 3.0 | |||||||
| QRS (ms) | 21.2 ± 0.8 | 22 ± 0.6 | 22.8 ± 1.0 | 23.2 ± 0.8 | ||||||||
| Q–T (ms) | 72.0 ± 3.1 | 74.4 ± 2.8 | 68.0 ± 3.7 | 74.0 ± 2.4 | ||||||||
| Beats/min | 356.7 ± 21.0 | 345.2 ± 17.7 | 340.3 ± 14.8 | 320.4 ± 10.2 | ||||||||
2.2.3. Effect on adrenaline-induced arrhythmia in rats
| Compound | ED50 iv (mg∙kg-1) | ED50 po (mg∙kg-1) |
|---|---|---|
| 10 | 0.35 (0.18–0.72) | 1.71 (1.52 ± 1.93) |
| 11 | 0.16 (0.10–0.23) | 0.86 (0.76 ± 0.96) |
| (R,S)-9 | 0.34 (0.23–0.51) | 0.44 (0.18 ± 1.10) |
| Carvedilol | 0.25 (0.12–0.53) | − |
| Propranolol | 1.05 (0.64–1.73)a | 19.5 (14.5 ± 26.1) |
2.2.4. Influence on blood pressure in rats
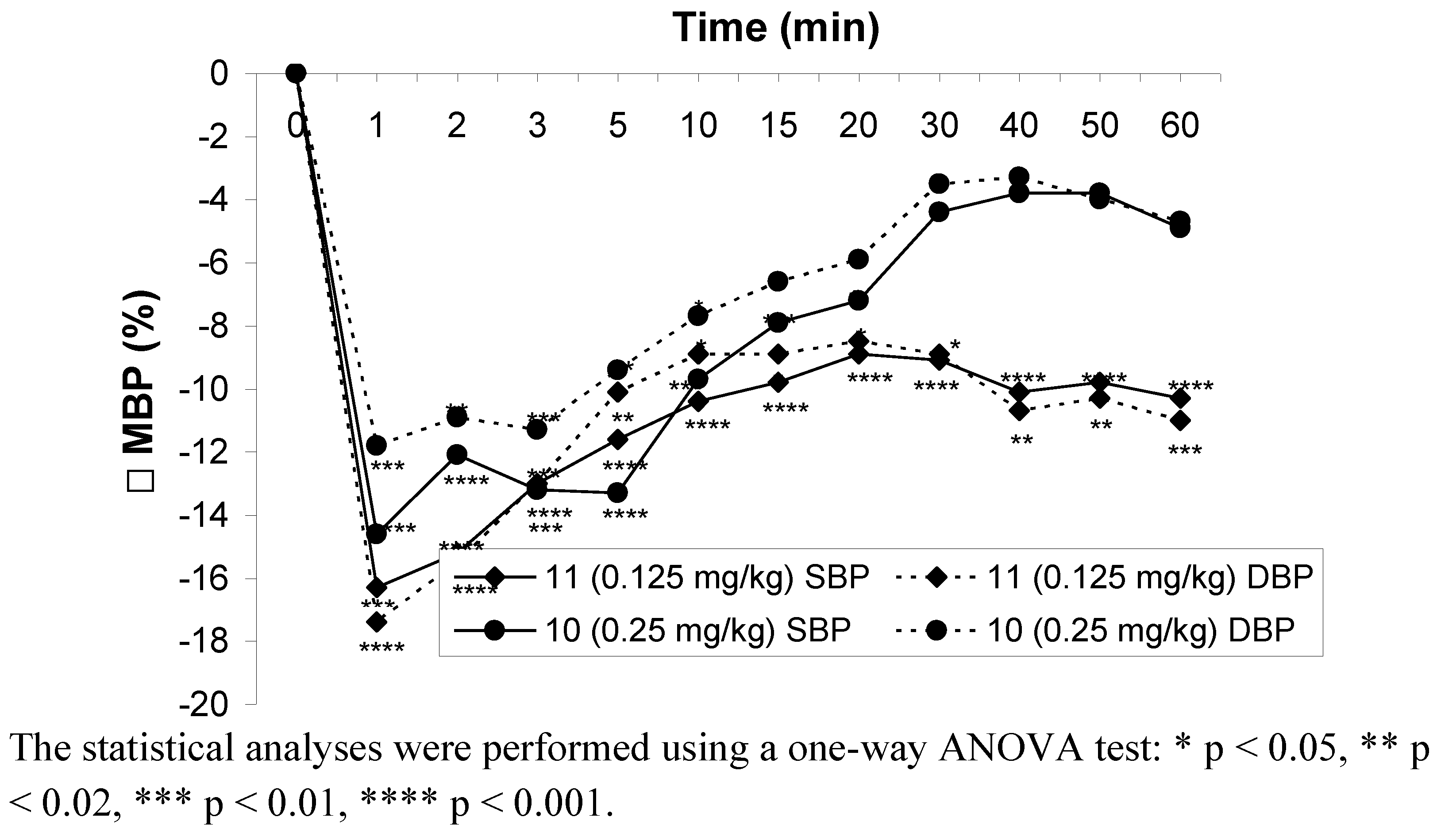
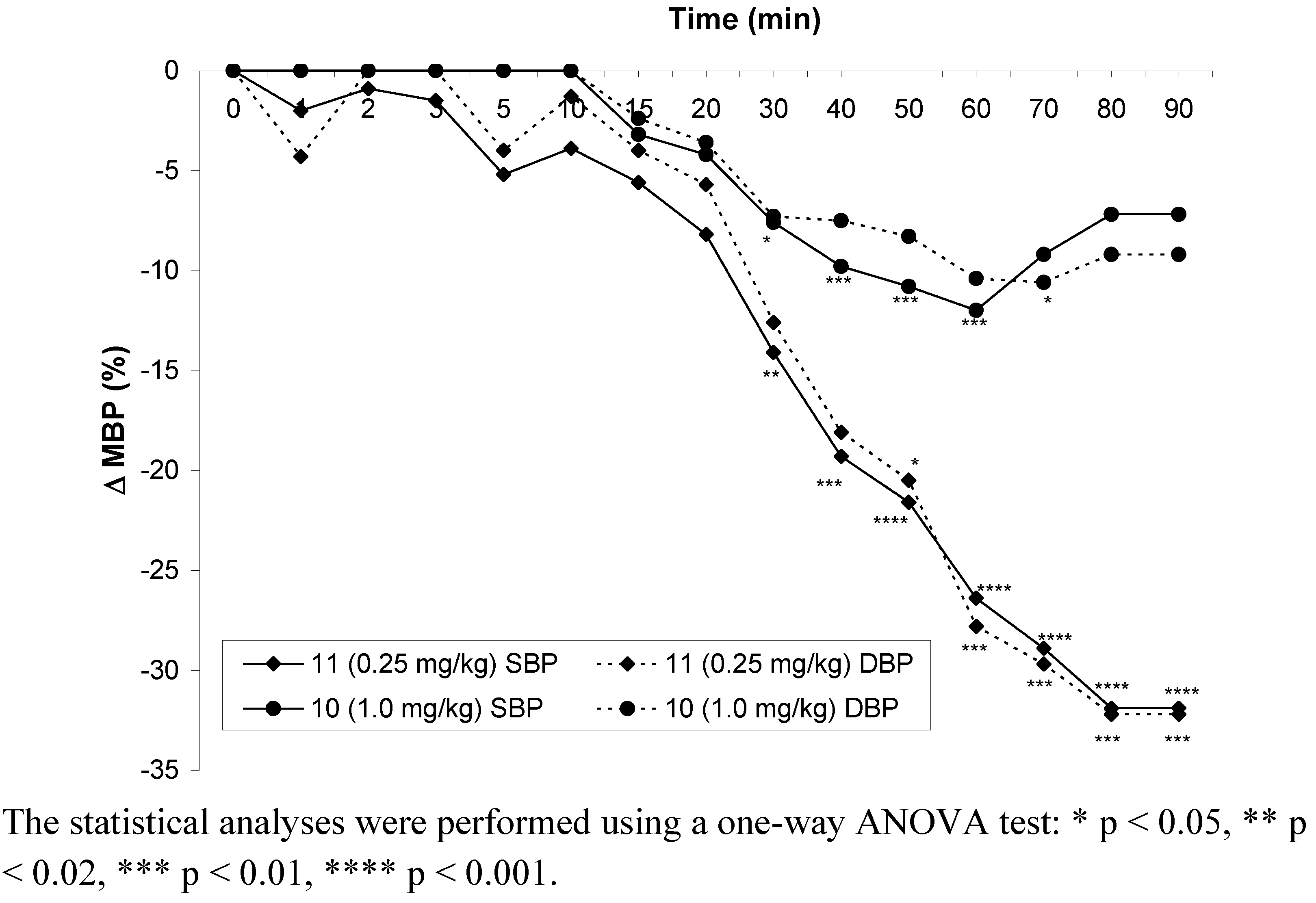
2.2.5. Influence on isolated rabbit ileum
| Compounds\Concentration | 0 | 10-9 [M] | 0 | 10-8 [M] | 0 | 10-7 [M] | 0 | 10-6 [M] | 0 | 10-5 [M] | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Amplitude [g] | 10 | 1.43 ± 0.06 | 1.56 ± 0.06 | 1.53 ± 0.06 | 1.31 ± 0.06b | 1.55 ± 0.10 | 1.29 ± 0.05a | 1.63 ± 0.10 | 1.10 ± 0.07c | 1.72 ± 0.11 | 1.11 ± 0.09c |
| 11 | 2.54 ± 0.28 | 2.59 ± 0.22 | 1.33 ± 0.09 | 1.68 ± 0.13a | 2.06 ± 0.21 | 166 ± 0.13 | 2.15 ± 0.12 | 2.02 ± 0.11 | 2.62 ± 0.19 | 1.82 ± 0.13b | |
| Frequency [min -1] | 10 | 12.74 ± 0.20 | 12.14 ± 0.22 | 11.77 ± 0.14 | 12.15 ± 0.18 | 12.05 ± 0.19 | 12.15 ± 0.15 | 12.24 ± 0.25 | 11.60 ± 0.22 | 11.61 ± 0.27 | 10.70 ± 0.25a |
| 11 | 6.90 ± 0.12 | 8.23 ± 0.95 | 11.48 ± 1.29 | 9.97 ± 1.13 | 10.44 ± 1.19 | 10.95 ± 1.19 | 12.08 ± 1.02 | 13.59 ± 0.67 | 11.40 ± 1.17 | 8.88 ± 0.85 | |
3. Experimental
3.1. Chemistry
3.1.1. General
3.1.2. Materials
3.1.3. 2,3-Dimethoxy-5-nitrobenzaldehyde (1)
3.1.4. 2-(Benzyloxy)-3-methoxy-5-nitrobenzaldehyde (2)
3.1.5. 2-(Benzyloxy)-1-methoxy-3-(methoxymethyl)-5-nitrobenzene (3)
3.1.6. (3-(Benzyloxy)-2-methoxy-4-(methoxymethyl)-6-nitrophenyl)acetonitrile (4) and (3-(benzyloxy)-4-methoxy-2-(methoxymethyl)-6-nitrophenyl)acetonitrile (5)
3.1.7. 4-Methoxy-6-(methoxymethyl)-1H-indol-5-ol (6) and 6-methoxy-4-(methoxymethyl)-1H-indol-5-ol (7)
3.1.8. 4-Methoxy-6-(methoxymethyl)-5-(oxiran-2-ylmethoxy)-1H-indole (8) and 6-methoxy-4-(methoxymethyl)-5-(oxiran-2-ylmethoxy)-1H-indole (9)
3.1.9. (2R,S)-1-(4-Methoxy-6-(methoxymethyl)-1H-indol-5-yloxy)-3-(2-(2-methoxyphenoxy)ethyl amino)propan-2-ol (10) and (2R,S)-1-(6-methoxy-4-(methoxymethyl)-1H-indol-5-yloxy)-3-(2-(2-methoxyphenoxy)ethylamino)propan-2-ol (11)
3.2. Pharmacology
3.2.1. Animals
3.2.2. Reference compound
3.2.3. Statistical analysis
3.2.4. Adrenoceptor radioligand binding assay
3.2.5. Effect on normal electrocardiogram (ECG)
3.2.6. Prophylactic antiarrhythmic activity in a model of adrenaline-induced arrhythmia according to Szekeres and Papp [27]
3.2.7. Influence on blood pressure in rats
3.2.8. Influence on isolated rabbit ileum
4. Conclusions
Acknowledgements
References
- Groszek, G.; Bednarski, M.; Dybała, M.; Filipek, B. Synthesis and adrenolytic activity of 1-(1H-indol-4-yloxy)-3-{[2-(2-methoxyphenoxy)ethyl]amino}propan-2-ol and its enantiomers. Part 1. Eur. J. Med. Chem. 2009, 44, 809–817. [Google Scholar] [CrossRef]
- Dulin, B.; Abraham, W.T. Pharmacology of Carvedilol. Am. J. Cardiol. 2004, 93, 3–6. [Google Scholar]
- Grassi, G.; Quarti-Trevano, F.; Seravalle, G.; Dell’Oro, R. Cardiovascular risk and adrenergic overdrive in the metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2007, 17, 473–481. [Google Scholar] [CrossRef]
- Shannon, R.; Chaudhry, M. Effect of α1-adrenergic receptors in cardiac pathophysiology. Am. Heart. J. 2006, 152, 842–850. [Google Scholar] [CrossRef]
- Kopecky, S.L. Effect of Beta Blockers, Particularly Carvedilol, on Reducing the Risk of Events after Acute Myocardial Infarction. Am. J. Cardiol. 2006, 98, 1115–1119. [Google Scholar] [CrossRef]
- Groszek, G.; Nowak-Król, A.; Wdowik, T.; Świerczyński, D.; Bednarski, M.; Otto, M.; Walczak, M.; Filipek, B. Synthesis and adrenolytic activity of 1-(1H-indol-4-yloxy)-3-(2-(2-methoxyphenoxy)ethylamino)propan-2-ol analogs and its enantiomers. Part 2. Eur. J. Med. Chem. 2009, 44, 5103–5111. [Google Scholar]
- Shibata, I.; Yoshida, T.; Baba, A.; Matsuda, H. Reduction of Aldehyde with Tributyltin Hydride-HMPA Combined System. Chem. Lett. 1989, 619–622. [Google Scholar]
- Goliński, J.; Mąkosza, M. "Vicarious” Nucleophilic Substitution of Hydrogen in Aromatic Nitro Compounds. Tetrahedron Lett. 1978, 37, 3495–3498. [Google Scholar]
- Pönicke, K.; Heinroth-Hoffmann, I.; Brodde, O. Differential effects of bucindolol and carvedilol on noradenaline-induced hypertrophic response in ventricular cardiomyocytes of adult rats. J. Pharmacol. Exp. Ther. 2002, 301, 71–76. [Google Scholar] [CrossRef]
- Filipek, B.; Sapa, J.; Malawska, B.; Kulig, K.; Antkiewicz-Michaluk, L. Search for new antiarrhythmic and hypotensive compounds. Synthesis, antiarrhythmic, antihypertensive, and alpha-adrenoceptor blocking activity of novel 1-[(2-hydroxy-3-amino)]propylpyrrolidin-2-one derivatives. Arch. Pharm. 1997, 330, 225–231. [Google Scholar] [CrossRef]
- Siemieniuk, A.; Szałkowska-Pągowska, H.; Lochyński, S.; Piątkowski, K.; Filipek, B.; Krupińska, J.; Czarnecki, R.; Librowski, T.; Białas, S. Propranolol analogs containing natural monoterpene structures: synthesis and pharmacological properties. Pol. J. Pharmacol. Pharm. 1992, 44, 557–593. [Google Scholar]
- Hool, L.C.; Oleksa, L.M.; Harvey, R.D. Role of G proteins in alpha1-adrenergic inhibition of the beta-adrenergically activated chloride current in cardiac myocytes. Mol. Pharmacol. 1997, 51, 853–860. [Google Scholar]
- Boyett, M.R.; Harrison, S.M.; Janvier, N.C.; McMorn, S.O.; Owen, J.M.; Shui, Z. A list of vertebrate cardiac ionic currents nomenclature, properties, function and cloned equivalents. Cardiovasc. Res. 1996, 32, 455–481. [Google Scholar]
- Carmeliet, E. Cardiac ionic currents and acute ischemia: From channels to arrhythmias. Physiol. Rev. 1999, 79, 917–1017. [Google Scholar]
- Hariman, R.J.; Gomes, J.A.C.; El-Sherif, N. Catecholamine-dependent atrioventricular nodal reentrant tachycardia. Circulation 1983, 67, 681–686. [Google Scholar] [CrossRef]
- Maze, M.; Smith, C.M. Identification of receptor mechanism mediating epinephrine-induced arrhythmias during halothane anesthesia in the dog. Anesthesiology 1983, 59, 322–326. [Google Scholar] [CrossRef]
- Billman, G.E. The effect of adrenergic receptor antagonists on cocaine-induced ventricular fibrillation: Alpha but not beta adrenergic receptor antagonists prevent malignant arrhythmias independent of heart rate. J. Pharmacol. Exp. Ther. 1994, 269, 409–416. [Google Scholar]
- Hieble, J.P. Adrenoceptor subclassification: An approach to improved cardiovascular therapeutics. Pharm. Acta Helv. 2000, 74, 163–171. [Google Scholar] [CrossRef]
- Jain, K.S.; Bariwal, J.B.; Kathiravan, M.K.; Phoujdar, M.S.; Sahne, R.S.; Chauhan, B.S.; Shah, A.K.; Yadav, M.R. Recent advances in selective alpha1-adrenoreceptor antagonists as antihypertensive agents. Bioorg. Med. Chem. 2008, 16, 4759–4800. [Google Scholar] [CrossRef]
- Grochowski, E.; Eckstein, Z. Synthesis of Some 2,2,2-Trichloro-1,1-bis(N-phenoxyacetamido)ethane Derivatives. Bull. Acad. Pol. Sc. 1963, XI(8), 443–446. [Google Scholar]
- Augstein, J.; Austin, W.C.; Boscott, R.J.; Green, S.M.; Worthing, C.R. Some Cardiovascular Effects of a Series of Aryloxyalkylamines. I. J. Med. Chem. 1965, 8, 356–367. [Google Scholar] [CrossRef]
- Fukuyama, Y.; Iwatsuki, Ch.; Kodama, M.; Ochi, M.; Katoaka, K.; Shibata, K. Antimicrobial Indolequinones from the Mid-Indestinal Glan of the Muricid Gastropod Drupella fragum. Tetrahedron 1998, 54, 10007–10016. [Google Scholar] [CrossRef]
- Mąkosza, M.; Danikiewicz, W.; Wojciechowski, K. Simple and General Synthesis of Hydroxy- and Methoxyindoles via Vicarious Nucleophilic Substitution of Hydrogen. Liebigs Ann. Chem. 1988, 203–208. [Google Scholar]
- Mąkosza, M.; Ziobrowski, T.; Serebriakov, M.; Kwast, A. Vicarious Nucleophilic Substitution of Hydrogen in Nitrophenyl Toluenesulfonates. Tetrahedron 1997, 53, 4739–4750. [Google Scholar] [CrossRef]
- Cheng, Y.C.; Prusoff, W.H. Relationship between the inhibition constant (K1) and the concentration of inhibitor which causes 50 per cent inhibition (I50) of an enzymatic reaction. Biochem. Pharmacol. 1973, 22, 3099–3108. [Google Scholar] [CrossRef]
- Hariman, R.J.; Gomes, J.A.C.; El-Sherif, N. Catecholamine-dependent atrioventricular nodal reentrant tachycardia. Circulation 1983, 67, 681–686. [Google Scholar] [CrossRef]
- Szekeres, L.; Papp, G. Handbook of Experimental Pharmacology; Schmier, J., Eichler, O., Eds.; Springer-Verlag: Berlin, Heidelberg, Germany, 1975. [Google Scholar]
- Walker, M.J.; Curtis, M.J.; Hearse, D.J.; Campbell, R.W.; Janse, M.J.; Yellon, D.M.; Cobbe, S.M.; Coker, S.J.; Harness, J.B.; Harron, D.W.; Higgins, A.J.; Julian, D.G.; Lab, M.J.; Manning, A.S.; Northover, B.J.; Parratt, J.R.; Riemersma, R.A.; Riva, E.; Russel, D.C.; Sheridan, D.J.; Winslow, E.; Woodward, B. The Lambeth Conventions: Guidelines for the study of arrhythmias in ischaemia infarction, and reperfusion. Cardiovasc. Res. 1988, 22, 447–455. [Google Scholar] [CrossRef]
- Magnus, R. Versuche am überlebenden Dünnalarm von Säugetieren. Pflügers Arch. 1904, 102, 123–151. [Google Scholar] [CrossRef]
- Orisadipe, A.; Amos, S.; Adesomoju, A.; Binda, L.; Emeje, M.; Okogun, J.; Wamebe, C.; Gamaniel, K. Spasmolytic activity of methyl angolensate: A triterpenoid isolated from Entandrophragma angolense. Biol. Pharm. Bull. 2001, 24, 364–367. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds 10 and 11 are available from the authors.
© 2010 by the authors;
Share and Cite
Groszek, G.; Bajek, A.; Bis, A.; Nowak-Król, A.; Bednarski, M.; Siwek, A.; Filipek, B. Synthesis and Adrenolytic Activity of New Propanolamines. Molecules 2010, 15, 3887-3904. https://doi.org/10.3390/molecules15063887
Groszek G, Bajek A, Bis A, Nowak-Król A, Bednarski M, Siwek A, Filipek B. Synthesis and Adrenolytic Activity of New Propanolamines. Molecules. 2010; 15(6):3887-3904. https://doi.org/10.3390/molecules15063887
Chicago/Turabian StyleGroszek, Grażyna, Agata Bajek, Agnieszka Bis, Agnieszka Nowak-Król, Marek Bednarski, Agata Siwek, and Barbara Filipek. 2010. "Synthesis and Adrenolytic Activity of New Propanolamines" Molecules 15, no. 6: 3887-3904. https://doi.org/10.3390/molecules15063887
APA StyleGroszek, G., Bajek, A., Bis, A., Nowak-Król, A., Bednarski, M., Siwek, A., & Filipek, B. (2010). Synthesis and Adrenolytic Activity of New Propanolamines. Molecules, 15(6), 3887-3904. https://doi.org/10.3390/molecules15063887