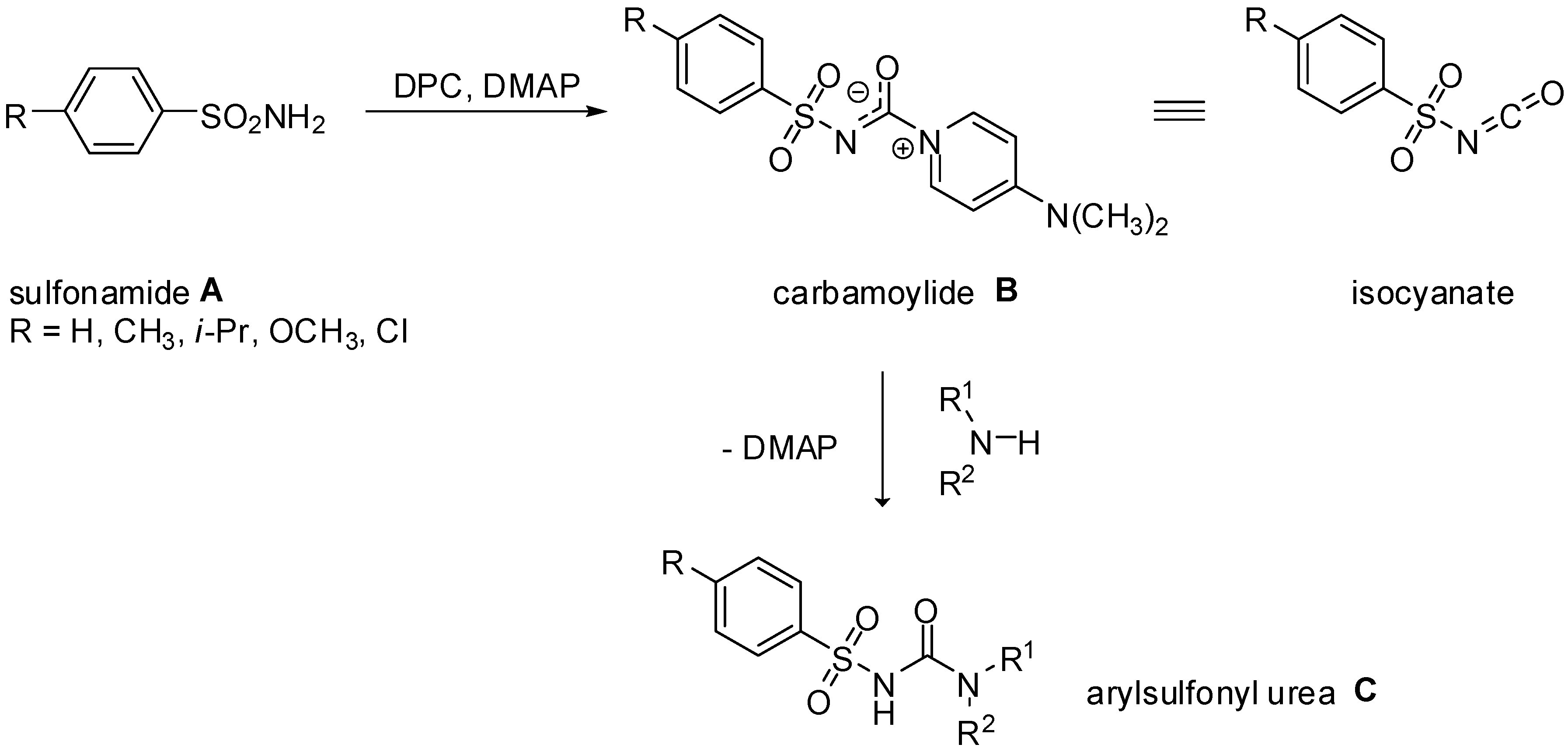
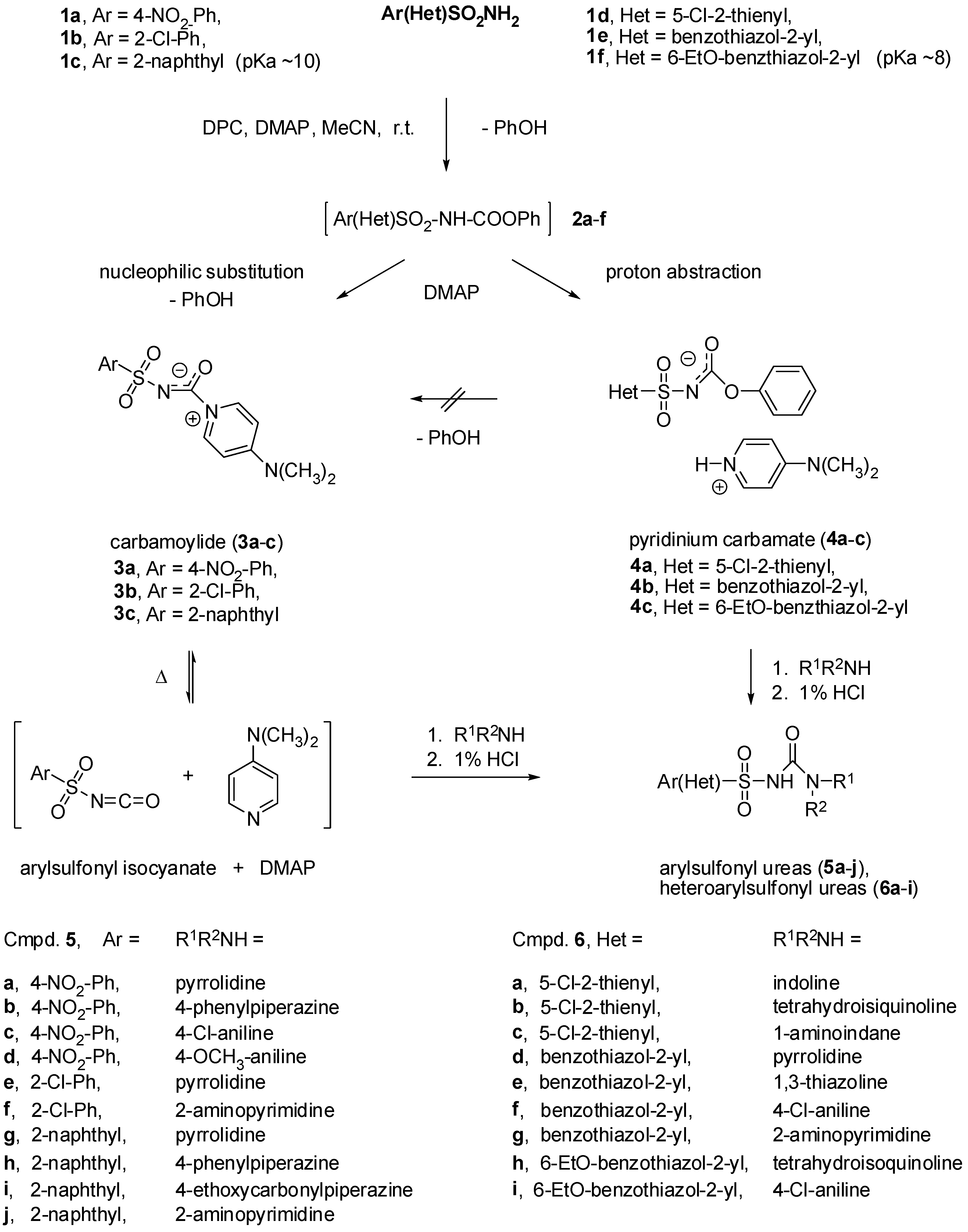
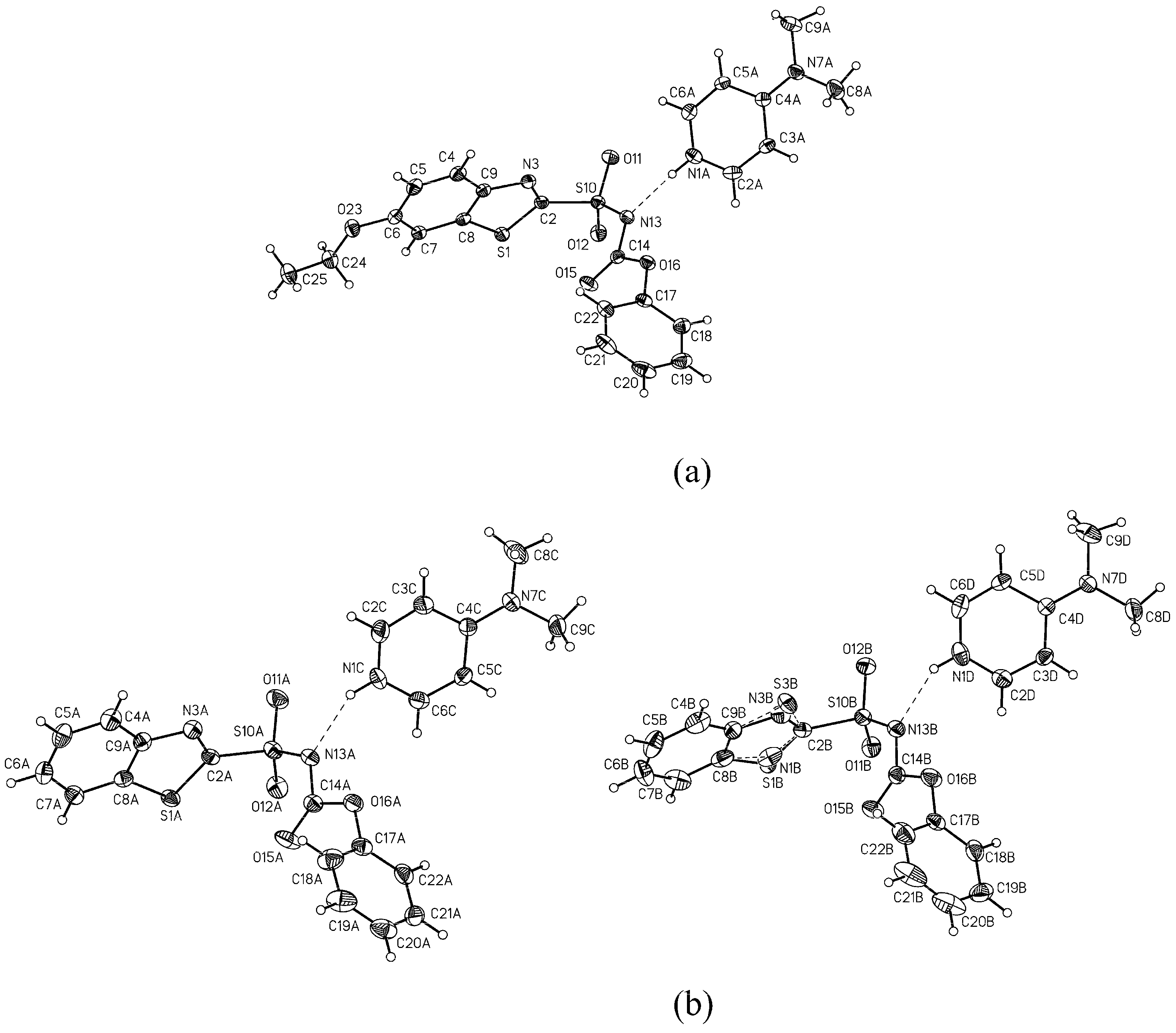
3.2. General procedure for the preparation of 4-dimethylaminopyridinium arylsulfonyl carbamoylides 3a-c and 4-dimethylaminopyridinium heteroarylsulfonyl carbamates 4a-c
A solution of appropriate arylsulfonamide 1a-c (33 mmol) in acetonitrile (40 mL) was treated with 4-dimethylaminopyridine (DMAP, 4.1 g, 66 mmol) and the reaction mixture was stirred at room temperature until sufonamide 1 had dissolved. Then diphenyl carbonate (DPC, 2.3 g, 37 mmol) was added and the reaction mixture was left overnight at room temperature. The solid that precipitated was separated by suction, washed with dry acetonitrile and dried to give pure carbamoylides 3a-d or carbamates 4a-d.
According to the above procedure the following compounds were obtained:
4-Dimethylaminopyridinium 4-nitrophenylsulfonyl carbamoylide (3a). Yield 56%; m.p. 175–177 °C (dec); IR (KBr) ν = 3099, 2937, 2683, 1691, 1645, 1578, 1520, 1349, 1294, 1257, 1215, 1159, 1072, 1078 cm-1; 1H-NMR (DMSO-d6) δ = 3.33 (s, 6H, CH3), 6.97 (d, 2H, CH, J = 7.2 Hz), 8.31 (d, 2H, CH, J = 8.7 Hz), 8.71 (d, 2H, CH, J = 8.7 Hz), 8.95 (d, 2H, CH, J = 7.2 Hz); Anal. Calcd. for C14H14 N4O5S (350.35) C, 47.99%; H, 4.03%; N, 15.99%; Found C, 47.72%; H, 3.87%; N, 15.83%.
4-Dimethylaminopyridinium 2-chlorophenylsulfonyl carbamoylide (3b). Yield: 54%; m.p. 112–115 °C; IR (KBr) ν = 3072, 2924, 1697, 1644, 1560, 1293, 1255, 1215, 1184, 1150, 1128, 1110, 1028 cm-1; 1H-NMR (DMSO-d6) δ = 3.16 (s, 6H, CH3), 6.96 (d, 2H, CH, J = 7.6 Hz), 7.60 (d, 1H, CH), 7.20-7.47 (m, 2H, CH), 7.95 (d, 1H, CH), 8.65 (d, 2H, CH, J = 7.6 Hz); Anal. Calcd. for C14H14ClN3O3S (339.80) C, 49.49%; H, 4.15%; N, 12.37%; Found C, 49.18%; H, 4.29%; N, 12.31%.
4-Dimethylaminopyridinium naphth-2-ylsulfonyl carbamoylide (3c). Yield: 53%; m.p. 153–156 °C (dec); IR (KBr) ν = 3087, 1707, 1648, 1572, 1289, 1253, 1217, 1150, 1090, 1065, 862, 834, 768 cm-1; 1H-NMR (DMSO-d6) δ = 3.22 (s, 6H, CH3), 6.95 (d, 2H, CH, Ј = 7.3 Hz), 7.60-7.69 (m, 2H, CH), 7.88-8.14 (m, 4H, CH), 8.43 (s, 1H, CH), 8.74 (d, 2H, CH, Ј = 7.3 Hz); Anal. Calcd. for C18H17N3O3S (355.41) C, 60.83%; H, 4.82%; N, 11.82%; Found C, 60.58%; H, 4.77%; N, 11.59%.
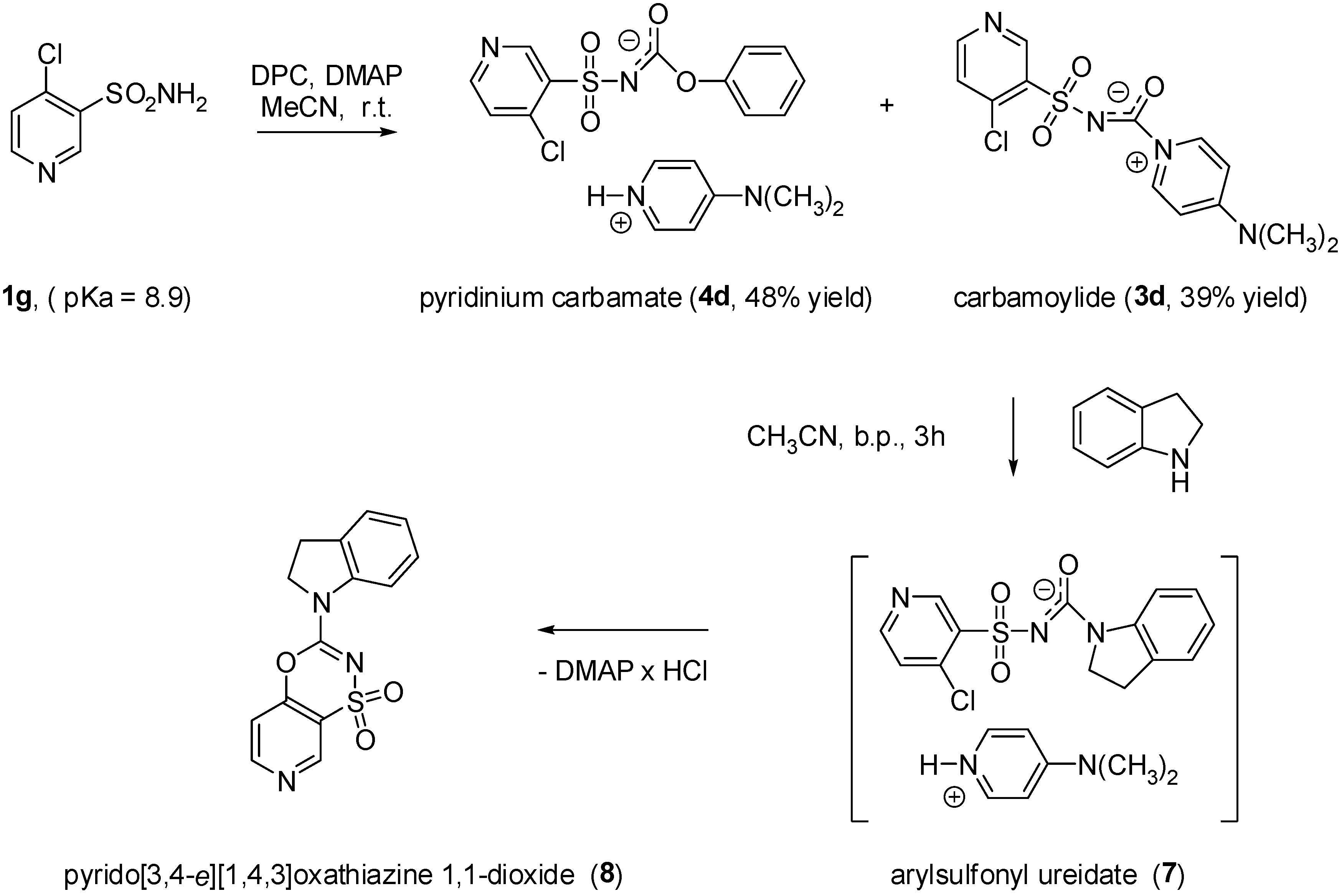
4-Dimethylaminopyridinium (4-chloropyridin-3-ylsulfonyl carbamoylide (3d). Yield: 39%; m.p. 151–153 °C (dec); IR (KBr) ν = 3097, 2934, 1702, 1645, 1572, 1559, 1449, 1395, 1296, 1260, 1089, 842, 824, 766, 596 cm-1; 1H-NMR (DMSO-d6) δ = 3.25 (s, 6H, CH3), 7.00 (d, 2H, CH, J = 8.1 Hz), 7.64 (d, 1H, CH, J = 5.2 Hz), 8.64 (d, 1H, CH, J = 5.2 Hz), 8.73 (d, 2H, CH, J = 8.1 Hz), 9.07 (s, 1H, CH); Anal. Calcd. for C13H13ClN4O3S (340.79) C, 45.82%; H, 3.85%; N, 16.44%; Found C, 45.61%; H, 3.90%; N, 16.16%.
4-(Dimethylamino)pyridinium (5-chlorothiophen-2-ylsulfonyl)(phenoxycarbonyl)amide (4a). Yield: 54%; m.p. 144–146 °C (dec); IR (KBr) ν = 3077, 2924, 2611, 1964, 1645, 1562, 1409, 1295, 1262, 1210, 1186, 1173, 1024, 988, 924, 887, 807, 626 cm-1; 1H-NMR (DMSO-d6) δ = 3.18 (s, 6H, CH3), 6.96-7.38 (m, 8H, CH), 7.42 (d, 1H, CH, J = 3.9 Hz), 8.21 (d, 2H, CH, J = 7.6 Hz), 12.6 (bs, 1H, NH); Anal. Calcd. for C18H18ClN3O4S2 (439.94) C, 49.14%; H, 4.12%; N, 9.55%; Found C, 48.93%; H, 4.26%; N, 9.19%.
4-(Dimethylamino)pyridinium (benzothiazol-2-ylsulfonyl)(phenoxycarbonyl)amide (4b). Yield: 73%; m.p. 135–138 °C (dec); IR (KBr) ν = 3060, 2924, 1685, 1646, 1559, 1271 cm-1; 1H-NMR (DMSO-d6) δ = 3.14 (s, 6H, CH3), 6.92-6.96 (m, 4H, CH), 7.02-7.12 (t, 1H, J = 7.4 Hz), 7.25-732 (t, 2H, CH, J = 7.4 Hz), 7.45-7.62 (m, 2H, CH), 8.04-8.16 (m, 2H, CH), 8.29 (d, 2H, CH, J = 7.6 Hz); Anal. Calcd. for C21H20N4O4S2 (456.54) C, 55.25%; H, 4.42%; N, 12.27%; Found C, 55.21%; H, 4.62%; N, 11.92%. Upon treatment of the above pyridinium salt with aqueous 1% HCl at room temperature the free carbamate 4b’ was obtained; m.p. 206-208 °C; IR (KBr) ν = 3007, 2851, 2799, 1758, 1458, 1222, 1096, 922, 767, 634 cm-1; 1H-NMR (DMSO-d6) δ = 6.75 (d, 2H, CH, J = 7.8 Hz), 7.16 (t, 1H, CH, J = 7.8 Hz), 7.35 (t, 2H, CH, J = 7.8 Hz), 7.60-7.72 (m, 2H, CH), 8.16-8.30 (m, 2H, CH), 8.35 (s, 1H, NH); Anal. Calcd. for C14H10N2O4S2 (334.37) C, 50.29%; H, 3.01%; N, 8.38%; Found C, 50.22%; H, 3.32%; N, 8.61%.
4-(Dimethylamino)pyridinium (6-ethoxybenzothiazol-2-ylsulfonyl)(phenoxy-carbonyl)amide (4c). Yield: 29.7%; m.p. 251–255 °C; IR (KBr) ν = 3223, 3066, 2924, 1681, 1645, 1557, 1296 cm-1; 1H-NMR (DMSO-d6) δ = 1.35 (t, 3H, CH3, J = 8.1 Hz), 3.14 (s, 6H, CH3), 4.09 (q, 2H, CH2, J = 8.1 Hz), 6.92-6.96 (m, 4H, CH), 7.05-7.15 (dd, 1H, CH, Jortho = 6.75, Jmeta = 2.3 Hz), 7.22-7.30 (t, 2H, CH, J = 7.4 Hz), 7.65 (d, 1H, CH, Jmeta = 2.3 Hz), 7.93 (d, 1H, CH, Jortho = 6.75 Hz), 8.19 (d, 2H, CH, J = 7.7 Hz); Anal. Calcd. for C23H24N4O5S2 (500.59) C, 55.18%; H, 4.83%; N, 11.19%; Found C, 54.97%; H, 5.11%; N, 10.98%. Upon treatment of the above pyridinium salt with aqueous 1% HCl the free carbamate 4c’ was obtained; m.p. 206-208 °C; IR (KBr) ν = 2989, 2859, 2787, 2663, 1756, 1599, 1485, 1375, 1262, 1217, 1168, 1080, 1042, 842, 759, 688, 627 cm-1; 1H-NMR (DMSO-d6) δ = 1.37 (t, 3H, CH3, J = 8.2 Hz), 4.12 (q, 2H, CH2, J = 8.2 Hz), 6.75 (m, 1H, CH), 7.00-7.45 (m, 5H, CH), 7.80-7.95 (m, 1H, CH), 8.00-8.15 (m, 1H, CH), 8.75 (bs, 1H, NH); Anal. Calcd. for C16H14N2O5S2 (378.42) C, 50.78%; H, 3.73%; N, 7.40%; Found C, 50.53%; H, 3.89%; N, 7.06%.
4-(Dimethylamino)pyridinium (4-chloropyridin-3-ylsulfonyl)(phenoxycarbonyl)amide (4d). Yield: 48%; m.p. 191–193 °C; IR (KBr) ν = 3215, 3082, 2922, 1688, 1646, 1561, 1397, 1263, 1191, 1149, 1028, 916, 887, 806, 797 cm-1; 1H-NMR (DMSO-d6) δ = 3.16 (s, 6H, CH3), 6.88-6.93 (m, 2H, CH), 6.96 (d, 2H, CH, J = 7.7 Hz), 7.06 (t, 1H, CH, J = 7.1 Hz), 7.22-7.30 (m, 2H, CH), 7.56 (d, 1H, CH, J = 5.3 Hz), 8.20 (d, 2H, CH, J = 7.7 Hz), 8.53 (d, 1H, CH, J = 5.3 Hz), 8.96 (s, 1H, CH), 12.60 (bs, 1H, NH); Anal. Calcd. for C19H19ClN4O4S (434.90) C, 52.47%; H, 4.40%; N, 12.88%; Found C, 52.19%; H, 4.71%; N, 12.60%.
3.3. General procedure for preparation of arylsulfonyl ureas 5 and heteroarylsulfonyl ureas 6. Reaction of carbamoylides 3a-c and carbamates 4a-c with aliphatic and aromatic amines
A mixture of carbamoylide 3 or carbamate 4 (2.8 mmol) and appropriate aliphatic or aromatic amine (3 mmol) in acetonitrile (10 mL) was heated at reflux for 10 min (in case of aliphatic) or 1 h (in case of aromatic) amine. The reaction mixture was cooled to room temperature and the solvent was evaporated under reduced pressure to dryness. The oily residue was suspended in methanol and treated with 10% aqueous hydrochloric acid. The sulphonylureas 5 or 6 that precipitated were separated by suction, washed with methanol and water and re-crystallized from suitable solvent.
According to the above procedure the following sulphonylureas were obtained:
N-(4-Nitrophenylsulfonyl)pyrrolidine-1-carboxamide (5a). Yield: 17%; m.p. 187–193 °C (methanol); IR (KBr) ν = 3466, 3271, 3109, 2990, 2788, 1670, 1529, 1348, 1313, 1167, 1091, 1067 cm-1; 1H-NMR (DMSO-d6) δ = 1.60-1.90 (m, 4H, CH2CH2), 3.15-3.45 (m, 4H, N-CH2), 8.15 (d, 2H, CH, J = 5.2 Hz), 8.42 (d, 2H, CH, J = 5.2 Hz), 11.05 (bs, 1H, NH); Anal. Calcd. for C11H13N3O5S (299.30) C, 44.14%; H, 4.38%; N, 14.04%; Found C, 44.08%; H, 4.52; N, 13.96%.
N-4-Nitrophenylsulfonyl)-4-phenylopiperazine-1-carboxamide (5b). Yield: 31%; m.p. 105–107 °C (ethanol); IR (KBr) ν = 3419, 3105, 2882, 1669, 1600, 1529, 1494, 1350, 1235, 1166, 1091 cm-1; 1H-NMR (DMSO-d6) δ = 2.90-3.20 (m, 4H, CH2), 3.30-3.60 (m, 4H, CH2), 6.80 (t, 1H, CH, J = 7.4 Hz), 6.90 (d, 2H, CH, J = 7.4 Hz), 7.20 (t, 2H, CH, J = 7.4 Hz), 8.50 (d, 2H, CH, J = 9.7 Hz), 8.43 (d, 2H, CH, J = 9.7 Hz), 11.45 (bs, 1H, NH); Anal. Calcd. for C17H18N4O5S (390.41) C, 52.30%; H, 4.65%; N, 14.35%; Found C, 52.05%; H, 4.83%; N, 14.16%.
N-4-Chlorophenylcarbamoyl)-4-nitrophenylsulfonamide (5c). Yield: 42%; m.p. 165–170 °C (ethanol); IR (KBr) ν = 3438, 3351, 3112, 2890, 1715, 1607, 1542, 1351, 1158, 1086, 1039, 1013 cm-1; 1H-NMR (DMSO-d6) δ = 7.32 (d, 2H, CH, J = 8.8 Hz), 7.38 (d, 2H, CH, J = 8.8 Hz), 8.22 (d, 2H, CH, J = 8.8 Hz), 8.46 (d, 2H, CH, J = 8.8 Hz), 9.80 (bs, 1H, NH), 11.20 (bs, 1H, NH); Anal. Calcd. for C13H10ClN3O5S (355.75) C, 43.89%; H, 2.83%; N, 11.81%; Found C, 44.17%; H, 3.12%; N, 11.57%.
N-(4-Methoksyphenylcarbamoyl)-4-nitrophenylsulfonamide (5d). Yield: 46%; m.p. 148–152 °C (ethanol); IR (KBr) ν = 3482, 3316, 3117, 3071, 3013, 2959, 2836, 1698, 1530, 1514, 1436, 1348, 1311, 1252, 1163, 1089, 1023 cm-1; 1H-NMR (DMSO-d6) δ = 3.68 (s, 3H, OCH3), 6.84 (d, 2H, CH, J = 9 Hz), 7.24 (d, 2H, CH, J = 9 Hz), 8.20 (d, 2H, CH, J = 9 Hz), 8.45 (d, 2H, CH, J = 9 Hz); 8.93 (bs, 1H, NH), 11.2 (bs, 1H, NH); Anal. Calcd. for C14H13N3O6S (351.33) C, 47.86%; H, 3.73%; N, 11.96%; Found C, 47.74%; H, 3.95%; N, 11.71%.
N-(2-Chlorophenylsulfonyl)pyrrolidine-1-carboxamide (5e). Yield: 37%; m.p. 175–190 °C (methanol); IR (KBr) ν = 3400, 3271, 3102, 3068, 1694, 1575, 1487, 1436, 1255, 1236, 1185, 1168, 1132, 1115 cm-1; 1H-NMR (DMSO-d6) δ = 1.64-1-94 (m, 4H, CH2), 3.15-45 (m, 4H, N-CH2), 7.54-7.58 (m, 1H, CH), 7.60-7.68 (m, 2H, CH), 8.03 (d, 1H, CH, J = 7.7 Hz), 11.06 (s, 1H, NH); Anal. Calcd. for C11H13ClN2O3S (288.75) C, 45.75%; H, 4.54%; N, 9.70%; Found C, 45.61%; H, 4.82%; N, 9.47%.
2-Chloro-N-(pyrimidin-2-ylcarbamoyl)benzenesulfonamide (5f). Yield: 68%; m.p. 178–182 °C; IR (KBr) ν = 3148, 3067, 2970, 2920, 1715, 1582, 1470, 1449, 1402, 1353, 1282, 1155, 1130, 1075, 1006, 914, 866, 812 cm-1; 1H-NMR (DMSO-d6) δ = 7.42-7.50 (m, 1H, CH), 7.65-7.80 (m, 3H, CH), 7.97-8.03 (d, 1H, CH, J = 7.6 Hz), 8.70-8.77 (m, 2H, CH), 10.69 (s, 1H, NH), 12.63 (bs, 1H, NH); Anal. Calcd. for C11H9 ClN4O3S (312.73); C, 42.25%; H, 2.90%; N, 17.92%; Found C, 41.89%; H, 3.27%; N, 17.65%.
N-(Naphthalene-2-ylsulfonyl)pyrrolidine-1-carboxamide(5g). Yield: 48%; m.p. 203–208 °C (ethanol); IR (KBr) ν = 3266, 2975, 2870, 1692, 1441, 1328, 1157, 1057, 861, 748, 658 cm-1; 1H-NMR (DMSO-d6) δ = 1.60-1.90 (m, 4H, CH2), 3.15-3.45 (m, 4H, N-CH2), 7.62-7.75 (m, 2H, CH), 7.91-8.21 (m, 4H, CH), 8.57 (s, 1H, CH), 10.77 (s, 1H, NH); Anal. Calcd. for C15H16N2O3S (304.36) C, 59.19%; H, 5.30%; N, 9.20%; Found C, 58.98%; H, 5.43%; N, 9.01%.
N-(Naphthalene-2-ylsulfonyl)-4-phenylpiperazine-1-carboxamide (5h). Yield: 98%; m.p. 119–123 °C (ethanol); IR (KBr) ν = 3277, 3059, 3016, 2959, 2936, 1676, 1484, 1338, 1252, 1162, 1125, 1070, 1021, 876, 745 cm-1; 1H-NMR (DMSO-d6) δ = 3.28-3.36 (m, 4H, CH2), 3.66-3.74 (m, 4H, CH2), 7.11-7.17 (m, 1H, CH), 7.30-7.40 (m, 4H, CH), 7.64-7.76 (m, 2H, CH), 7.92-8.22 (m, 4H, CH), 8.59 (s, 1H, CH), 11.35 (bs, 1H, NH); Anal. Calcd. for C21H21N3O3S (395.47) C, 63.78%; H, 5.35%; N, 10.63%; Found C, 63.56%; H, 5.50%; N, 10.33%.
Ethyl 4-(naphthalen-2-ylsulfonylcarbamoyl)piperazine-1-carboxylate (5i). Yield: 48%; m.p. 143–147 °C; IR (KBr) ν = 3196, 3060, 2978, 2930, 2868, 1709, 1677, 1469, 1441, 1257, 1230, 1163, 1072 cm-1; 1H-NMR (DMSO-d6) δ = 1.16 (t, 3H, CH3, Ј = 7 Hz), 3.20-3.42 (m, 8H, CH2), 4.02 (q, 2H, CH2, Ј = 7 Hz), 7.67-7.73 (m, 2H, CH), 7.88-7.93 (m, 1H, CH), 8.02-8.21 (m, 3H, CH), 8.55 (s, 1H, CH), 11.20 (bs, 1H, NH); Anal. Calcd. for C18H21N3O5S (391.44) C, 55.23%; H, 5.41%; N, 10.73%; Found C, 54.94%; H, 5.72%; N, 10.66%.
N-(Pyrimidin-2-ylcarbamoyl)naphthalene-2-sulfonamide (5j). Yield: 68%; m.p. 177–182 °C; IR (KBr) ν = 3148, 3067, 2971, 2920, 1716, 1583, 1471, 1449, 1354, 1283, 1156, 1075 cm-1; 1H-NMR (DMSO-d6) δ = 7.20-7.24 (m, 1H, CH), 7.66-7.78 (m, 2H, CH), 7.98-8.26 (m, 4H, CH), 8.70-8.72 (m, 3H, CH), 10.70 (s, 1H, NH), 12.65 (bs, 1H, NH); Anal. Calcd. for C15H12N4O3S (328.35) C, 54.87%; H, 3.68%; N, 17.06%; Found C, 55.02%; H, 3.84%; N, 16.81%.
N-(5-Chlorothiophen-2-ylsulfonyl)indoline-1-carboxamide (6a). Yield: 73%; m.p. 182–184 °C; IR (KBr) ν = 3635, 3450, 2920, 1659, 1486, 1464, 1400, 1348, 1165, 1137, 1091, 998, 808, 871, 679, 620, 604 cm-1; 1H-NMR (DMSO-d6) δ = 3.11 (t, 2H, CH2, J = 8.4 Hz), 4.03 (t, 2H, CH2, J = 8.4 Hz), 6.92-7.01 (m, 1H, CH), 7.08-7.23 (m, 2H, CH), 7.27 (d, 1H, CH, J = 4.2 Hz), 7.69 (d, 1H, CH, J = 4.2 Hz), 10.9 (bs, 1H, NH); Anal. Calcd. for C13H11ClN2O3S2 (342.82) C, 45.55%; H, 3.23%; N, 8.17%; Found C, 45.38%; H, 3.59%; N, 7.93%.
N-(5-Chlorothiophen-2-ylsulfonyl)-3,4-dihydroisoquinoline-2(1H)-carboxamide (6b). Yield: 61%; m.p. 169–172 °C; IR (KBr) ν = 3107, 3020, 2896, 2805, 2751, 1646, 1481, 1410, 1344, 1235, 1167, 1001, 757, 616, 572 cm-1; 1H-NMR (DMSO-d6) δ = 2.81-2.82 (m, 2H, CH2), 3.59-3.61 (m, 2H, CH2), 4.53 (s, 2H, CH2), 7.16-7.20 (m, 4H, CH), 7.25 (d, 1H, CH, J = 3.9 Hz), 7.64 (d, 1H, CH, J = 3.9 Hz), 11.44 (bs, 1H, NH); Anal. Calcd. for C14H13ClN2O3S2 (356.85) C, 47.12%; H, 3.67%; N, 7.85%; Found C, 46.84%; H, 3.73%; N, 7.57%.
5-Chloro-N-(2,3-dihydro-1H-inden-1-ylcarbamoyl)thiophene-2-sulfonamide (6c). Yield: 49%, m.p. 144–148 °C; IR (KBr) ν = 3353, 3094, 1687, 1650, 1541, 1466, 1409, 1367, 1168, 995, 752, 679 cm-1; 1H-NMR (DMSO-d6) δ = 1.71-1.86 (m, 1H, CH), 2.30-2.48 (m, 1H, CH), 2.68-2.96 (m, 2H, CH), 5.00-5.18 (m, 1H, CH, J = 8.0 Hz), 6.99 (d, 1H, NH, J =8.0 Hz), 7.10-7.24 (m, 4H, CH), 7.29 (d, 1H, CH, J = 4.1 Hz), 7.65 (d, 1H, CH, J = 4.1 Hz), 10.90 (bs, 1H, NH); Anal. Calcd. for C14H13ClN2O3S2 (356.85) C, 47.12%; H, 3.67%; N, 7.85%; Found C, 46.97%; H, 3.95%; N, 7.82%.
N-(Benzothiazol-2-ylsulfonyl)pyrrolidine-1-carboxamide (6d). Yield: 80%; m.p. 220–222 °C; IR (KBr) ν = 3277, 2877, 1690, 1454, 1168, 1056, 860, 765, 625 cm-1; 1H-NMR (DMSO-d6) δ = 1.60-1.95 (m, 4H, CH2), 3.25-3.65 (m, 4H, CH2), 7.61-7.73 (m, 2H, CH), 8.16-8.32 (m, 2H, CH), 11.80 (bs, 1H, NH); Anal. Calcd. for C12H13N3O3S2 (311.38); C, 46.29%; H, 4.21%; N, 13.49%; Found C, 45.97%; H, 4.53%; N, 13.42%.
N-(Benzothiazol-2-ylsulfonyl)thiazolidine-1-carboxamide (6e). Yield: 79%; m.p. 240–241 °C; IR (KBr) ν = 3068, 2887, 2791, 1681, 1455, 1181, 1385, 1092, 859, 626 cm-1; 1H-NMR (DMSO-d6) δ = 2.98-3.04 (m, 2H, CH2), 3.59-3.63 (m, 2H, CH2), 4.42 (s, 2H, CH2), 7.61-7.74 (m, 2H, CH), 8.16-8.22 (m, 1H, CH), 8.26-8.33 (m, 1H, CH), 11.20 (bs, 1H, NH); Anal. Calcd. for C11H11N3O3S3 (329.42) C, 40.11%; H, 3.37%; N, 12.76%; Found C, 39.87%; H, 3.42%; N, 12.40%.
N-(4-Chlorophenylcarbamoyl)benzothiazole-2-sulfonamide (6f). Yield: 81%, m.p. 182–184 °C; IR (KBr) ν = 3281, 1709, 1597, 1532, 1367, 1166, 1028, 923, 673 cm-1; 1H-NMR (DMSO-d6) δ = 7.28 (d, 1H, CH, J = 7.6 Hz,), 7.38 (d, 2H, CH, J = 7.6 Hz), 7.62-7.72 (m, 2H, CH), 8.16-8.23 (m, 1H, CH), 8.25-8.34 (m, 1H, CH), 9.39 (bs, 1H, NH), 12.6 (bs, 1H, NH); Anal. Calcd. for C14H10ClN3O3S2 (367.83) C, 45.71%; H, 2.74%; N, 11.42%; Found C, 45.34%; H, 3.11%; N, 11.10%.
N-(Pyrimidin-2-ylcarbamoyl)benzothiazole-2-sulfonamide (6g). Yield: 70%; m.p. 312–314 °C; IR (KBr) ν = 3067, 2972, 2919, 1646, 1583, 1449, 1374, 1161, 1028, 917, 761 cm-1; 1H-NMR (DMSO-d6) δ = 7.27-7.32 (m, 1H, CH), 7.56-7.69 (m, 2H, CH), 8.10-8.16 (m, 1H, CH), 8.19-8.27 (m, 1H, CH), 8.73-8.76 (m, 2H, CH), 11.43 (bs, 2H, NH); Anal. Calcd. for C12H9N5O3S2 (335.36) C, 42.98%; H, 2.70%; N, 20.88%; Found C, 42.71%; H, 3.02%; N, 20.57%.
N-(6-Ethoxybenzothiazol-2-ylsulfonyl)-3,4-dihydroisoquinoline-2(1H)-carboxamide (6h). Yield: 87%; m.p. 203–206 °C; IR (KBr) ν = 3145, 3088, 2956, 2876, 1648, 1555, 1432, 1386, 1145, 1128, 1034, cm-1; 1H-NMR (DMSO-d6) δ = 1.37 (t, 3H, CH3, J = 7.8 Hz), 2.80 (t, 2H, CH2, J = 6.8 Hz), 3.60 (t, 2H, CH2, J = 6.8 Hz), 4.12 (q, 2H, CH2, J = 7.8 Hz), 4.51 (s, 2H, CH2), 7.05-7.2 (m, 4H, CH), 7.23 (dd, 1H, CH, Jortho = 6.8 Hz, Jmeta = 2.0 Hz), 7.80 (d, 1H, CH, Jortho = 6.8 Hz), 8.05 (d, 1H, CH, Jmeta = 2.0 Hz), 11.30 (bs, 1H, NH); Anal. Calcd. for C19H19N3O4S2 (417.50) C, 54.66%; H, 4.59%; N, 10.06%; Found 54.56%; H, 4.85%; N, 10.03%.
N-(4-Chlorophenylcarbamoyl)-6-ethoxybenzothiazol-2-ylsulfonamide (6i). Yield: 75%; m.p. 238–240 °C; IR (KBr) ν = 3326, 3194, 3088, 2967, 2902, 2887, 1642, 1558, 1442, 1390, 1148, 1148, 1142, 1044 cm-1; 1H-NMR (DMSO-d6) δ = 1.37 (t, 3H, CH3, J = 7.7 Hz), 4.16 (q, 2H, CH2, J = 7.7 Hz), 7.26 (dd, 1H, CH, Jortho = 6.9 Hz, Jmeta = 2.1 Hz), 7.30 (d, 2H, CH, J = 7.0 Hz), 7.38 (d, 2H, CH, J = 7.0 Hz), 7.80 (d, 1H, CH, J = 2.1 Hz), 8.06 (d, 1H, CH, J = 6.9 Hz), 9.30 (s, 1H, NH), 11.80 (bs, 1H, NH); Anal. Calcd. for C16H14ClN3O4S2 (411.88) C, 46.66%; H, 3.43%; N, 10.20%; Found C, 46.73%; H, 3.68%; N, 9.83%.