Elevated Carbon Dioxide Increases Contents of Flavonoids and Phenolic Compounds, and Antioxidant Activities in Malaysian Young Ginger (Zingiber officinale Roscoe.) Varieties
Abstract
:1. Introduction
2. Results and Discussion
2.1. HPLC Analysis of Flavonoids
| Flavonoid compounds | (a) Halia Bentong | |||
|---|---|---|---|---|
| 400 | 800 | |||
| Leaves | Rhizomes | Leaves | Rhizomes | |
| Quercetin | 0.972 ± 0.013c | 0.895 ± 0.039c | 1.22 ± 0.07b | 1.138 ± 0.023b |
| Rutin | 0.171 ± 0.0028de | 0.452 ± 0.004a | 0.141 ± 0.031e | 0.388 ± 0.026b |
| Epicatechin | 0.122 ± 0.018a | 0.083 ± 0.007bc | 0.073 ± 0.008c | 0.048 ± 0.018d |
| Catechin | 0.409 ± 0.027d | 0.491 ± 0.019cd | 0.673 ± 0.044ab | 0.637 ± 0.034b |
| Kaempferol | 0.042 ± 0.002e | 0.053 ± 0.003de | 0.118 ± 0.014c | 0.148 ± 0.023b |
| Naringenin | 0.089 ± 0.0052c | 0.047 ± 0.003d | 0.127 ± 0.022b | 0.083 ± 0.004c |
| Fisetin | 0.986 ± 0.012e | 0.633 ± 0.033f | 2.05 ± 0.27c | 2.82 ± 0.19a |
| Morin | 0.514 ± 0.027e | 0.463 ± 0.014e | 0.49 ± 0.052e | 0.875 ± 0.036a |
| Flavonoid compounds | (b) Halia Bara | |||
| 400 | 800 | |||
| Leaves | Rhizomes | Leaves | Rhizomes | |
| Quercetin | 1.19 ± 0.122ab | 0.986 ± 0.032c | 1.33 ± 0.134a | 1.27 ± 0.01a |
| Rutin | 0.174 ± 0.007d | 0.334 ± 0.009c | 0.151 ± 0.025de | 0.404 ± 0.016b |
| Epicatechin | 0.12 ± 0.004a | 0.103 ± 0.0035ab | 0.096 ± 0.022bc | 0.037 ± 0.009d |
| Catechin | 0.668 ± 0.079ab | 0.533 ± 0.034c | 0.733 ± 0.014a | 0.682 ± 0.05ab |
| Kaempferol | 0.051 ± 0.002de | 0.068 ± 0.005d | 0.163 ± 0.011ab | 0.181 ± 0.009a |
| Naringenin | 0.061 ± 0.004d | 0.028 ± 0.003e | 0.155 ± 0.027a | 0.121 ± 0.011b |
| Fisetin | 1.53 ± 0.121d | 1.32 ± 0.12d | 2.38 ± 0.395b | 3.11 ± 0.185a |
| Morin | 0.765 ± 0.024b | 0.606 ± 0.006d | 0.661 ± 0.029c | 0.515 ± 0.025e |
| Flavonoid compounds | Halia Bentong | Halia Bara | ||
|---|---|---|---|---|
| Leaves | Rhizomes | Leaves | Rhizomes | |
| Quercetin | +25.5 | +27.2 | +9.2 | +28.8 |
| Rutin | -17.5 | -14.2 | -13.2 | +21.0 |
| Epicatechin | -40.2 | -42.2 | -20.0 | -64.1 |
| Catechin | +64.5 | +29.7 | +9.7 | +28.0 |
| Kaempferol | +181.0 | +179.2 | +219.6 | +166.2 |
| Naringenin | +42.7 | +76.6 | +154.1 | +332.1 |
| Fisetin | +107.9 | +345.5 | +55.6 | +135.6 |
| Morin | -4.7 | +89.0 | -13.6 | -15.0 |
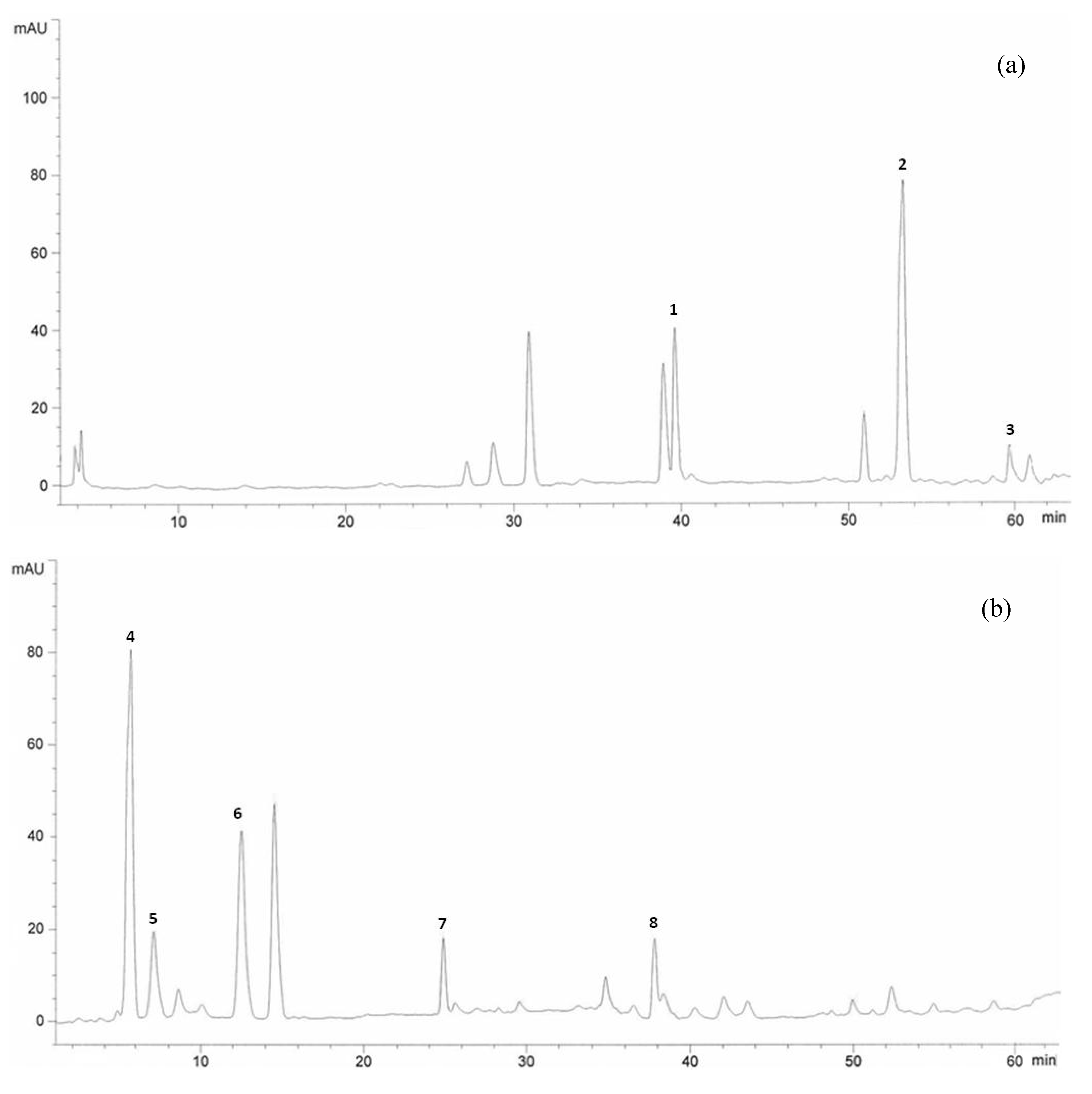
2.2. HPLC Analysis of Phenolic Compounds
| Phenolic compounds | (a) Halia Bentong | |||
|---|---|---|---|---|
| 400 | 800 | |||
| Leaves | Rhizomes | Leaves | Rhizomes | |
| Gallic acid | 0.173 ± 0.0091d | 0.141 ± 0.031d | 0.576 ± 0.049b | 0.489 ± 0.043c |
| Vanillic acid | nd | nd | 0.229 ± 0.058b | 0.335 ± 0.028a |
| Ferulic acid | 0.081 ± 0.022f | 0.116 ± 0.016ef | 0.117 ± 0.026de | 0.21 ± 0.022b |
| Tannic acid | 0.388 ± 0.072a | nd | nd | nd |
| Cinnamic acid | nd | nd | 0.134 ± 0.027a | 0.0336 ± 0.255b |
| Salicylic acid | nd | nd | 0.22 ±0.021b | 0.037 ± 0.0125c |
| Phenolic compounds | (b) Halia Bara | |||
| 400 | 800 | |||
| Leaves | Rhizomes | Leaves | Rhizomes | |
| Gallic acid | 0.191±0.008d | 0.152+0.0081d | 0.645±0.066a | 0.537±0.034bc |
| Vanillic acid | 0.082±0.016c | nd | 0.24±0.052b | 0.357±0.038a |
| Ferulic acid | 0.071±0.017f | 0.148+0.017cd | 0.162±0.014c | 0.285±0.038a |
| Tannic acid | 0.224±0.041b | nd | nd | nd |
| Cinnamic acid | nd | nd | 0.125±0.027a | 0.0457±0.01b |
| Salicylic acid | nd | nd | 0.269±0.027a | 0.0417±0.044c |
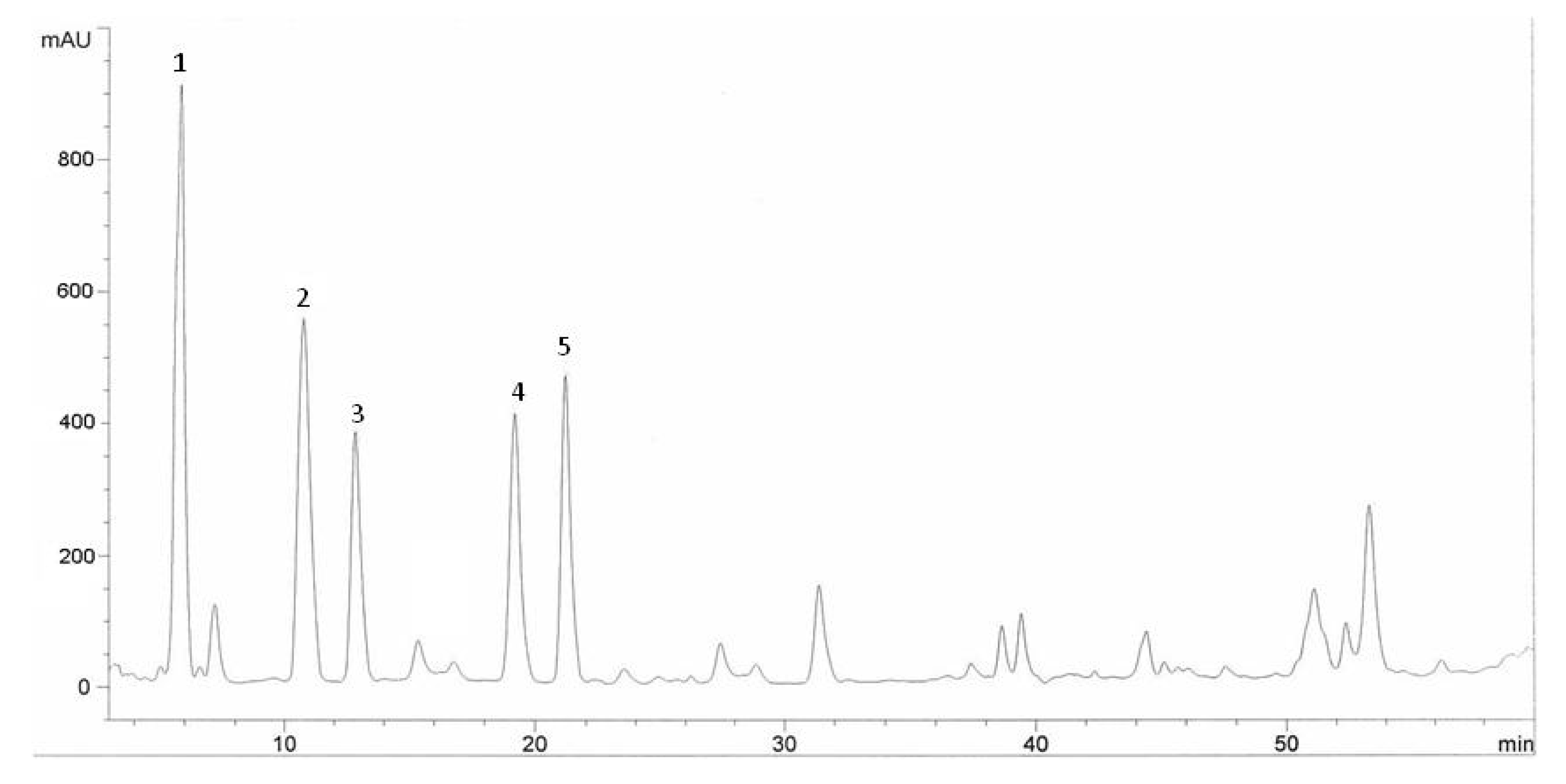
| Phenolic compounds | Halia Bentong | Halia Bara | ||
|---|---|---|---|---|
| Leaves | Rhizomes | Leaves | Rhizomes | |
| Gallic acid | +232.4 | +246.8 | +252.4 | +262.8 |
| Vanillic acid | +100 | +100 | +192.6 | +100 |
| Ferulic acid | +44.4 | +81 | +128.2 | +92.5 |
| Tannic acid | -100 | 0 | -100 | 0 |
| Cinnamic acid | +100 | +100 | +100 | +100 |
| Salicylic acid | +100 | +100 | +100 | +100 |
2.3. Radical Scavenging Activity (DPPH)
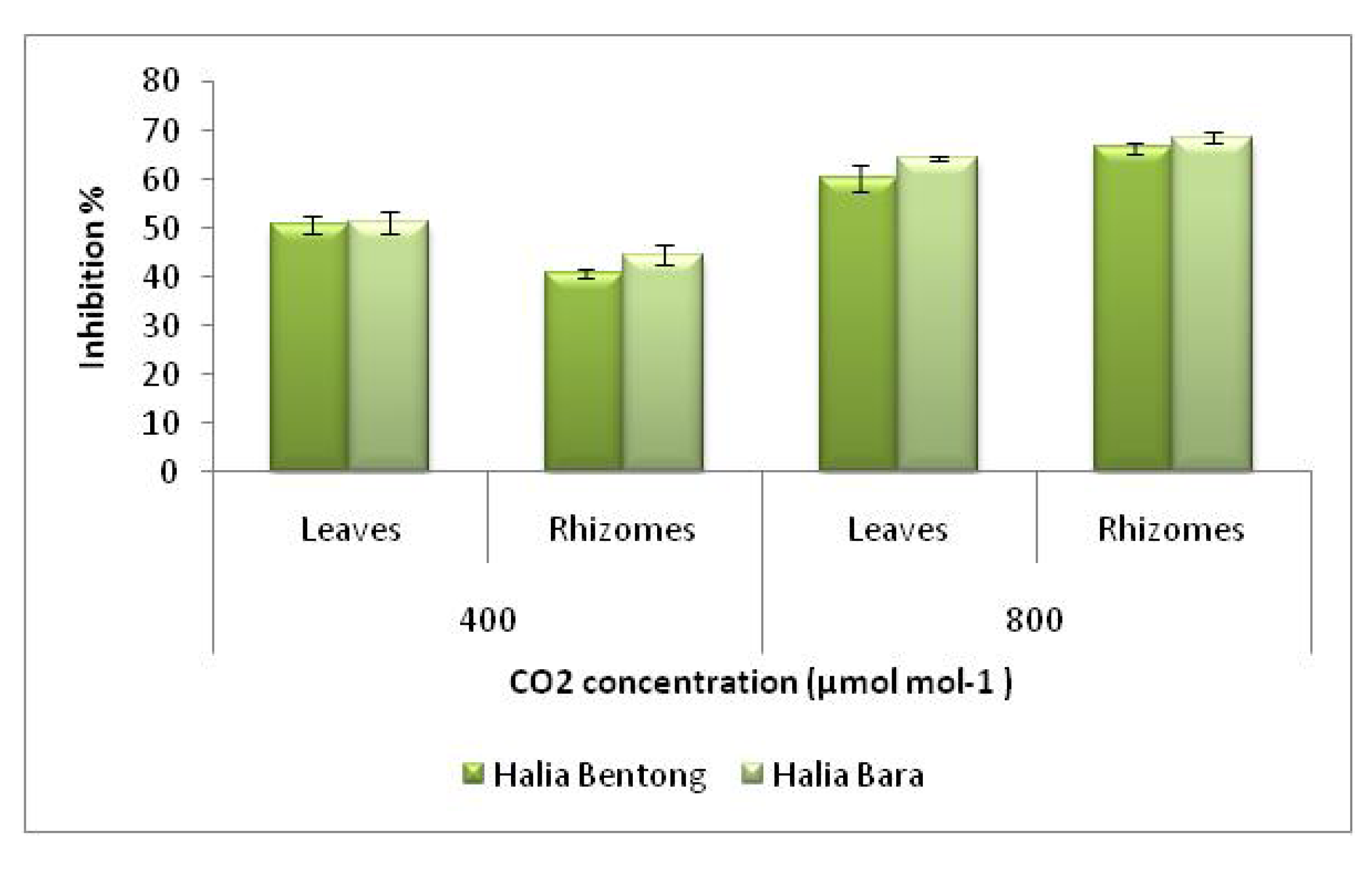
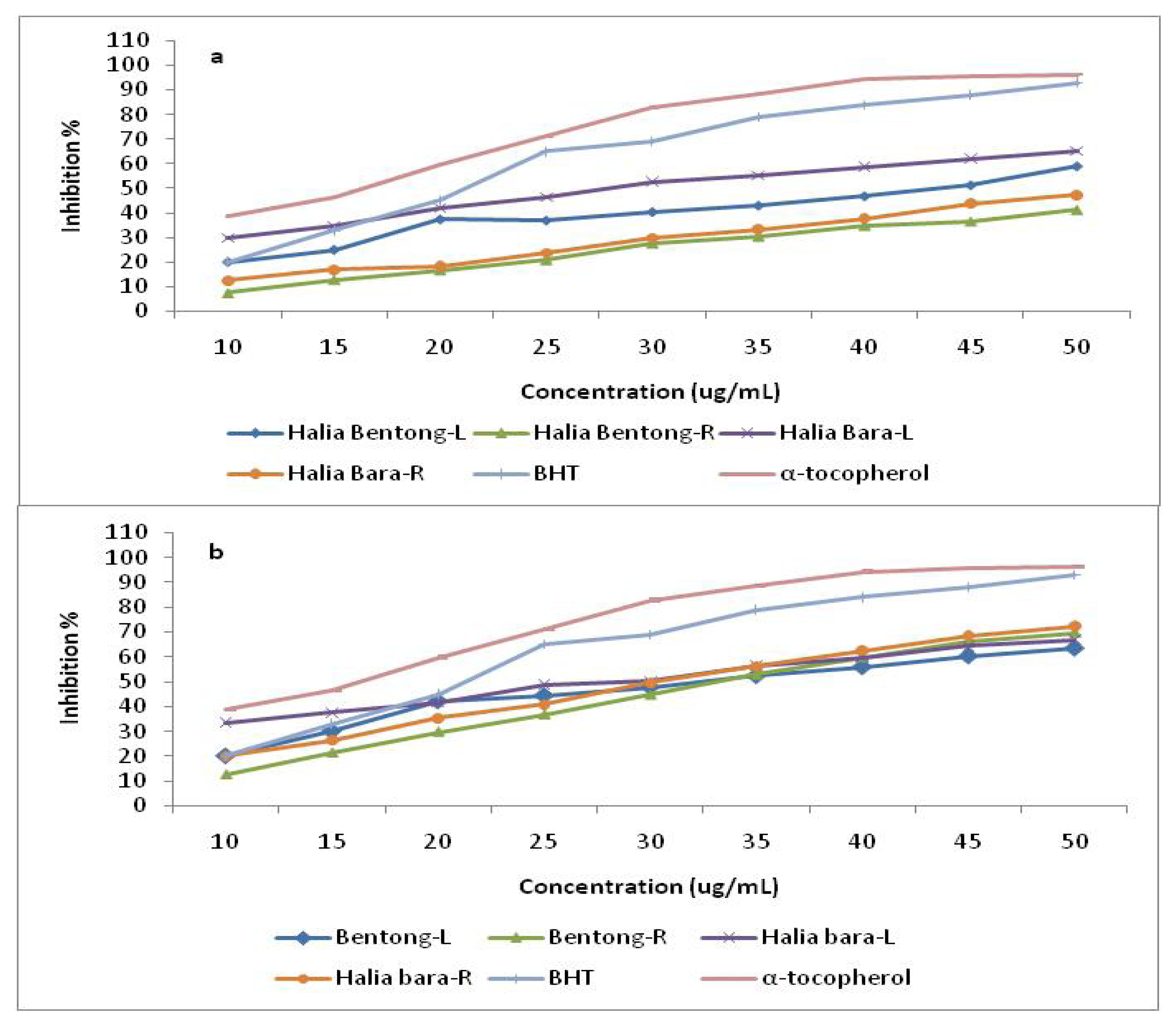
3. Experimental
3.1. Plant Materials
3.2. Growth Chamber Microclimate
3.3. High Performance Liquid Chromatography (HPLC)
3.3.1. Flavonoid Extract Preparation
3.3.2. Analysis of Flavonoids Composition by HPLC
3.3.3. Preparation of Phenolics Extracts
3.3.4. Analysis of Phenolics Acids Composition by HPLC
3.4. Determination of Antioxidant Activities
Radical Scavenging Assay (DPPH)
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
References and Notes
- Bazzaz, F.A. The response of natural ecosystems to the rising global CO2 levels. Annu. Rev. Ecol. Syst. 1990, 21, 167–196. [Google Scholar]
- Lavola, A.; Julkunen, T.R. The effect of elevated carbon dioxide and fertilization on primary and secondary metabolites in Befula pendula (Roth). Oecologia 1994, 99, 315–321. [Google Scholar] [CrossRef]
- Mark, S.J.; Jackson, S.B. Growth responses of Quercus petraea, Fraxinus excelsior and Pinus sylvestris to elevated carbon dioxide, ozone and water supply. New Phytol. 2000, 146, 437–451. [Google Scholar] [CrossRef]
- Tisserat, B.; Herman, C.; Silman, R.; Bothast, R.J. Using ultrahigh carbon dioxide levels enhances plantlet growth in vitro. Horttechnology 1997, 7, 282–289. [Google Scholar]
- Brevoort, P. The blooming United State botanical market: a new overview. Herbalgram 1998, 44, 33–46. [Google Scholar]
- Jaafar, H.Z.E. Carbon dioxide enrichment technology for improved productivity under controlled environment system in the tropics. Acta Hort. 2006, 742, 353–363. [Google Scholar]
- Ibrahim, M.H.; Jaafar, H.Z.E.; Harun, M.H.; Yusop, R. Changes in the growth and photosynthetic patterns of oil palm (Elaeis guineensis Jacq.) seedlings exposed to short term CO2 enrichment in a closed top chamber. Acta Physiol. Plant. 2010, 32, 305–313. [Google Scholar] [CrossRef]
- Mattson, W.J.; Julkunen-Tiitto, R.; Herms, D.A. CO2 enrichment and carbon partitioning to phenolics: do plant responses accord better with the protein competition or the growth-differentiation balance models? Oikos 2005, 111, 337–347. [Google Scholar] [CrossRef]
- Wang, Y.S.H.; Bunce, A.J.; Maas, L.J. Elevated carbon dioxide increases contents of antioxidant compounds in field-grown strawberries. J. Agr. Food Chem. 2003, 51, 4315–4320. [Google Scholar] [CrossRef]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A.; Wahab, P.E.M.; Halim, M.R.A. Effect of Different Light Intensities on Total Phenolics and Flavonoids Synthesis and Anti-oxidant Activities in Young Ginger Varieties (Zingiber officinale Roscoe). Int. J. Mol. Sci. 2010, 11, 3885–3897. [Google Scholar]
- Booker, F.L. Influence of carbon dioxide enrichment, ozone and nitrogen fertilization on cotton (Gossypium hirsutum L.) leaf and root composition. Plant Cell Environ. 2000, 23, 573–583. [Google Scholar] [CrossRef]
- Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A. Identification and Concentration of Some Flavonoid Components in Malaysian Young Ginger (Zingiber officinale Roscoe) Varieties by a High Performance Liquid Chromatography Method. Molecules 2010, 15, 6231–6243. [Google Scholar] [CrossRef]
- Yao, L.H. Flavonoids in food and their health benefits. Plant Food. Hum. Nutr. 2004, 59, 113–122. [Google Scholar]
- Ho, C.T.; Lee, C.Y.; Hungan, M.T. Phenolic compounds in food and their effects on health (analysis, occurrence, and chemistry). Amer. Chem. Soc. 1992, 2–19. [Google Scholar]
- Byers, T.; Guerrero, N. Epidemilogic evidence for vitamin C and vitamin E in cancer prevention. Amer. J. Clin. Nutr. 1995, 62, 1385–1392. [Google Scholar]
- Namiki, M. Antioxidant/antimutagens in food, critical reviews of food science and nutrition. Food Sci. Nutr. 1990, 29, 273–300. [Google Scholar]
- Heijnen, C.G.; Haenen, G.R.; Vanacker, F.A.; Vijgh, W.J.; Bast, A. Flavonoids as peroxynitrite scavengers:the role of the hydroxyl groups. Toxicol. Vitro 2001, 15, 3–6. [Google Scholar] [CrossRef]
- Chun, O.K.; Kim, D.O.; Lee, C.Y. Superoxide radical scavenging activity of the major polyphenols in fresh plums. J. Agr. Food Chem. 2003, 51, 8067–8072. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research science. Phytochemistry 2000, 55, 481–504. [Google Scholar] [CrossRef]
- Chen, G. Effect of low fat and/or high fruit and vegetable diets on plasma level of 8-isoprostane-F2alpha in nutrition and breast health study. Nutr. Cancer 2004, 50, 155–160. [Google Scholar] [CrossRef]
- Sung-Jin, P.; Hoon, M.; Young-Youn, K.; Jun-Young, P.; Jun-Woo, P.; Myung-Jin, K.; Soon-Min, H. Anticancer effects of genistein, green tea catechins, and cordycepin on oral squamous cell carcinoma. J. Korean Oral Maxillofac. Surg. 2008, 34, 1–10. [Google Scholar]
- Tepe, B.; Sokmen, M.; Akpulat, H.A.; Sokmen, A. Screening of the antioxidant potentials of six Salvia species from Turkey. Food Chem. 2006, 95, 200–204. [Google Scholar] [CrossRef]
- Chan, E.W.C.; Lim, Y.Y.; Wong, L.F.; Lianto, F.S.; Wong, S.K.; Lim, K.K.; Joe, C.E.; Lim, T.Y. Antioxidant and tyrosinase inhibition properties of leaves and rhizomes of ginger species. Food Chem. 2008, 109, 477–483. [Google Scholar] [CrossRef]
- Kikuzaki, H.; Nakatani, N. Antioxidant effect of some ginger constituents. J. Food Sci. 1993, 578, 1407–1410. [Google Scholar] [CrossRef]
- Chen, C.H.; Kuo, M.; Wu, C.H; Ho, C.H. Pungent compounds of ginger (Zingiber officinale .L) Rosc.) extracted by liquid carbon dioxide. J. Agr. Food Chem. 1986, 34, 477–480. [Google Scholar] [CrossRef]
- Ramanthan, L.; Das, N.P. Effect of natural copper chelating compounds on the pro-oxidant activity of ascorbic acid in steam-cooked ground fish. Int. J. Food Sci. Technol. 1993, 28, 279–288. [Google Scholar]
- Kaufman, P.B.; Cseke, L.J.; Warber, S.; Duke, J.A.; Brielmann, H.L. Natural Products from Plant; CRC Press: Boca Raton, FL, USA, 1999. [Google Scholar]
- Wink, M. Introduction: Biochemistry, Role and Biotechnology of Secondary Products; CRC Press: Boca Raton, FL, USA, 1999; pp. 1–16. [Google Scholar]
- Liu, L.; King, J.S.; Giardina, P.C.H. Effects of elevated concentrations of atmospheric CO2 and tropospheric O3 on leaf litter production and chemistry in trembling aspen and paper birch communities. Tree Physiol. 2005, 25, 1511–1522. [Google Scholar] [CrossRef]
- Malikov, V.M.; Yuledashev, M.P. Phenolic compounds of plants of the Scutellaria L. genus: distribution, structure, and properties. Chem. Nat. Compd. 2002, 28, 358–406. [Google Scholar]
- Lindroth, R.L.; Kinney, K.K.; Platz, C.L. Responses of deciduous trees to elevated atmospheric CO2: Productivity, phytochemistry, and insect performance. Ecology 1993, 74, 763–777. [Google Scholar] [CrossRef]
- Stutte, G.W.; Eraso, I. Carbon dioxide enrichment enhances growth and flavonoid content of two Scutellaria species. J. Amer. Soc. Hort. Sci. 2008, 133, 631–638. [Google Scholar]
- Caldwell, C.R.; Britz, S.J.; Mirecki, R.M. Effect of temperature, elevated carbon dioxide, and drought during seed development on the isoflavone content of dwarf soybean (Glycine max (L.) Merrill] grown in controlled environments. J. Agr. Food Chem. 2005, 53, 1125–1129. [Google Scholar] [CrossRef]
- Khoo, H.M.; Suhaila, M. Flavonoid (myricetin, quercetin, kaempferol, luteolin, and apigenin) content of edible tropical plants. J. Agr. Food Chem. 2001, 49, 3106–3112. [Google Scholar] [CrossRef]
- Geraets, L.; Haegens, A.; Brauers, K.; Haydock, J.A.; Vernooy, J.H.; Wouters, E.F.; Bast, A.; Hageman, G.J. Inhibition of LPS-induced pulmonary inflammation by specific flavonoids. Biochem. Biophys. Res. Commun. 2009, 382, 598–603. [Google Scholar] [CrossRef]
- Tolonen, M.; Taipale, M.; Viander, B.; Pihlava, J.M.; Korhonen, H.; Ryhänen, E.L. Plant-derived biomolecules in fermented cabbage. J. Agr. Food. Chem. 2002, 50, 6798–6803. [Google Scholar]
- Kim, D.O.; Padilla-Zakour, O.I.; Griffiths, P.D. Flavonoids and antioxidant capacity of various cabbage genotypes at juvenile stage. J. Food Sci. 2004, 69, 685–689. [Google Scholar]
- Park, H.H.; Lee, S.; Son, H.Y.; Park, S.B.; Kim, M.S.; Choi, E.J.; Singh, T.S.; Ha, J.H.; Lee, M.G.; Kim, J.E.; Hyun, M.C.; Kwon, T.K.; Kim, Y.H.; Kim, S.H. Flavonoids inhibit histamine release and expression of proinflammatory cytokines in mast cells. Arch. Pharm. Res. 2008, 31, 1303–1311. [Google Scholar] [CrossRef]
- Lim, D.O.Y.; Park, J.H. Induction of P53 contributes to apoptosis of HCT-116 human colon cancer cells induced by the dietary compound fisetin. Amer. J. Phys. Gastrointest. Liver Physiol. 2009, 296, 1060–1068. [Google Scholar] [CrossRef]
- Brown, J.; Prey, J.; Harrison, P.R. Enhanced sensitivity of human oral tumours to the flavonol, morin, during cancer progression: involvement of the Akt and stress kinase pathways. Carcinogenesis 2003, 24, 171–177. [Google Scholar] [CrossRef]
- Kawabata, K.; Tanaka, T.; Honjo, S.; Kakumoto, M.; Hara, A.; Makita, H.; Tatematsu, N.; Ushida, J; Tsuda, H.; Mori, H. Chemopreventive effect of dietary flavonoid morin on chemically induced rat tongue carcinogenesis. Int. J. Cancer 1999, 83, 381–386. [Google Scholar] [CrossRef]
- Song, Y.M.; Kang, J.W.; Zhou, J.; Wang, Z.H.; Lua, X.Q.; Wang, L.F.; Gao, J.Z. Study on the fluorescence spectra and electrochemical behavior of ZnL2 and morin with DNA. Spectrochim. Acta PT A: Mol. Bio. 2000, 56, 2491–2497. [Google Scholar] [CrossRef]
- Wijeratne, S.S.K.; Abou-Zaid, M.M.; Shahidi, F. Antioxidant polyphenols in almond and its coproducts. J. Agr. Food Chem. 2006, 54, 312–318. [Google Scholar] [CrossRef]
- Aggarwal, B.B.; Shishodia, S. Molecular targets of dietary agents for prevention and therapy of cancer. Biochem. Pharmacol. 2006, 71, 1397–1421. [Google Scholar] [CrossRef]
- Nagasubramaniam, A.; Pathmanabhan, G.; Mallika, V. Studies on improving production potential of baby corn with foliar spray of plant growth regulators. Annu. Rev. Plant Physiol. Plant Mol. Biol. 2007, 21, 154–157. [Google Scholar]
- Jeyakumar, P.; Velu, G.; Rajendran, C.; Amutha, R.; Savery, M.A.J.R.; Chidambaram, S. Varied responses of blackgram (Vigna munga) to certain foliar applied chemicals and plant growth regulators. Legume Res. Int. J. 2008, 31, 110–113. [Google Scholar]
- Crozier, A; Jensen, E.; Lean, M.E.J; Mc Donald, M.S. Quantitative analysis of flavonoids by reversed-phase high performance liquid chromatography. J. Chromatogr. 1997, 761, 315–321. [Google Scholar] [CrossRef]
- Wang, T.C.; Chuang, Y.C.; Ku, Y.H. Quantification of bioactive compounds in citrus fruits cultivated in Taiwan. Food Chem. 2007, 102, 1163–1171. [Google Scholar] [CrossRef]
- Standard Operating Protocol, HPLC Analysis of Phenolic acids (SOP), SOP No.: CB0103; Botanical Center for Age-Related Diseases: West Lafayette, IN, USA, 2001; p. 9.
- Mensor, L.L.; Menezes, F.S.; Leitao, G.G.; Reis, A.S.; Santos, T.S.; Coube, C.S. Screening of Brazilian plant extracts for antioxidant activity by the use of DPPH free radical method. Phytother. Res. 2001, 15, 127–130. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ghasemzadeh, A.; Jaafar, H.Z.E.; Rahmat, A. Elevated Carbon Dioxide Increases Contents of Flavonoids and Phenolic Compounds, and Antioxidant Activities in Malaysian Young Ginger (Zingiber officinale Roscoe.) Varieties. Molecules 2010, 15, 7907-7922. https://doi.org/10.3390/molecules15117907
Ghasemzadeh A, Jaafar HZE, Rahmat A. Elevated Carbon Dioxide Increases Contents of Flavonoids and Phenolic Compounds, and Antioxidant Activities in Malaysian Young Ginger (Zingiber officinale Roscoe.) Varieties. Molecules. 2010; 15(11):7907-7922. https://doi.org/10.3390/molecules15117907
Chicago/Turabian StyleGhasemzadeh, Ali, Hawa Z.E. Jaafar, and Asmah Rahmat. 2010. "Elevated Carbon Dioxide Increases Contents of Flavonoids and Phenolic Compounds, and Antioxidant Activities in Malaysian Young Ginger (Zingiber officinale Roscoe.) Varieties" Molecules 15, no. 11: 7907-7922. https://doi.org/10.3390/molecules15117907
APA StyleGhasemzadeh, A., Jaafar, H. Z. E., & Rahmat, A. (2010). Elevated Carbon Dioxide Increases Contents of Flavonoids and Phenolic Compounds, and Antioxidant Activities in Malaysian Young Ginger (Zingiber officinale Roscoe.) Varieties. Molecules, 15(11), 7907-7922. https://doi.org/10.3390/molecules15117907






