The Roles of Phytochemicals in Bronchial Asthma
Abstract
:Introduction
Oxidative Stress in Asthma
Antioxidant System in the Lung – Balancing Intracellular Oxidation
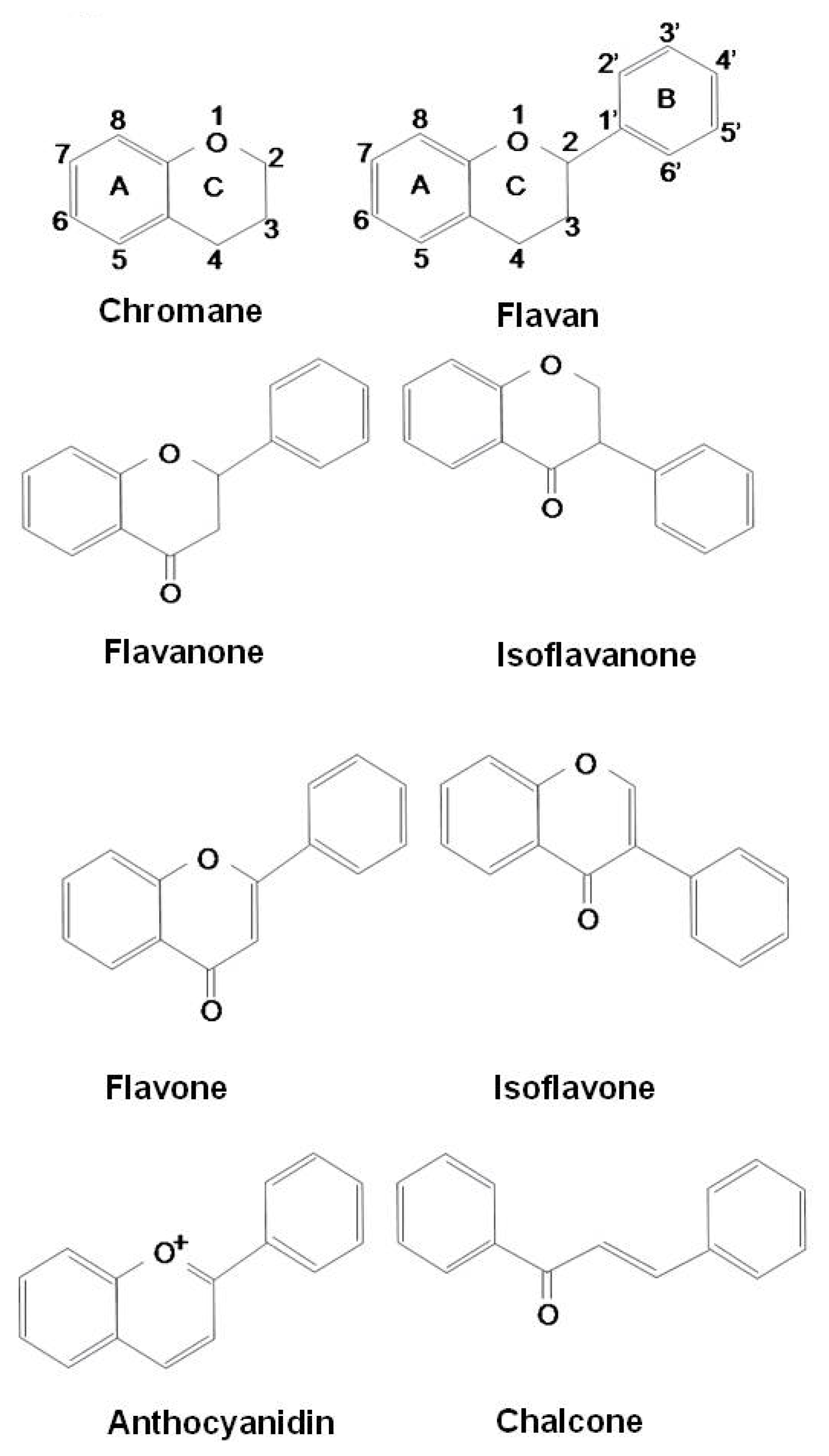
Antioxidant Molecules in Plants and the Their Role in Bronchial Asthma
| Bioactive compounds | Activity and Potential mechanisms of effect | |
|---|---|---|
| Polyphenols | ||
| Flavonoids | Flavans | Donation of hydrogen atom to radicals |
| Flavanones | Chelation of redox-active metals | |
| Isoflavanones | Inhibition of lipid peroxidation | |
| Flavones | Regulation of the enzyme activities | |
| Isoflavones | Inhibition of mast cell/basophil activation | |
| Inhibition of eosinophlic degranulation | ||
| Anthocyanidins | Switching allergic immune response to Th1 profile | |
| Chalcones | Regulation of various transcription factors and mediators involving angiogenesis: HIF-1, VEGF, MMPs, EGFR and inhibit NF-κB, PI3K/Akt, and ERK1/2 signaling pathways | |
| Flavonolignans | ||
| Curcumin | Prevention of lipid peroxydation | |
| Radical scavenger/neutralizer | ||
| Decrease the levels of iNOS | ||
| Regulation of cytokines such as IL-2, IL-5, and GM-CSF through maintaining HDAC2 activity | ||
| Inhibition of mast cell activation | ||
| Inhibition of neutrophil function | ||
| Resveratrol | Inducing and stabilizing antioxidant enzymes | |
| Inhibition of prostaglandin production | ||
| Decrease the phosphorylation of ERK1/2, cyclooxygenase-2 activity, and activity of various transcription factors including NF-κB, STAT3, HIF-1α, and β-catenin | ||
| Inhibit protein kinases (src, PI3K, JNK and Akt) | ||
| Inhibit production of inflammatory mediators (IFN-γ, TNF, COX-2, iNOS, CRP and various interleukins) | ||
| Sirtuin1 activation | ||
| Modulate innate immune response | ||
| Inhibition of angiogenetic pathway that is mediated through expression of MMPs, VEGF, cathepsin D, ICAM-1 and E-selectin | ||
| Antioxidant vitamins | ||
| Carotenoids | Lycopene | Quenches singlet oxygen without degradation |
| Lutein | Regulation of various transcription factors (AP-1, NF-κB) | |
| β-cryptoxanthin | Supression production of inflammatory cytokines | |
| α-carotene | Reduce induction of IGF-1 | |
| β-carotene | ||
| Vitamin C | Ascorbic acid | Donation of hydrogen atom to radicals |
| Dehydro-ascorbic acid | Inhibiting the JNK/AP-1 signaling pathways | |
| prostaglandin inhibition | ||
| Vitamin E | Tocopherols | Hydroperoxide scavenger |
| (α, β, γ, and δ) | Modulation of the functional activity of T-lymphocytes and enhance the phagocytic activity of peripheral granulocytes | |
| Tocotrienols | ||
| (α, β, γ, and δ) | ||
| Inhibit monocyte response to LPS and LPS-induced degrdation of IκB and JNK activation | ||
| Regulation of endothelial cell signals | ||
| Membrane stabilization | ||
| Inhibition of IgE production | ||
| Organosulfur compounds | ||
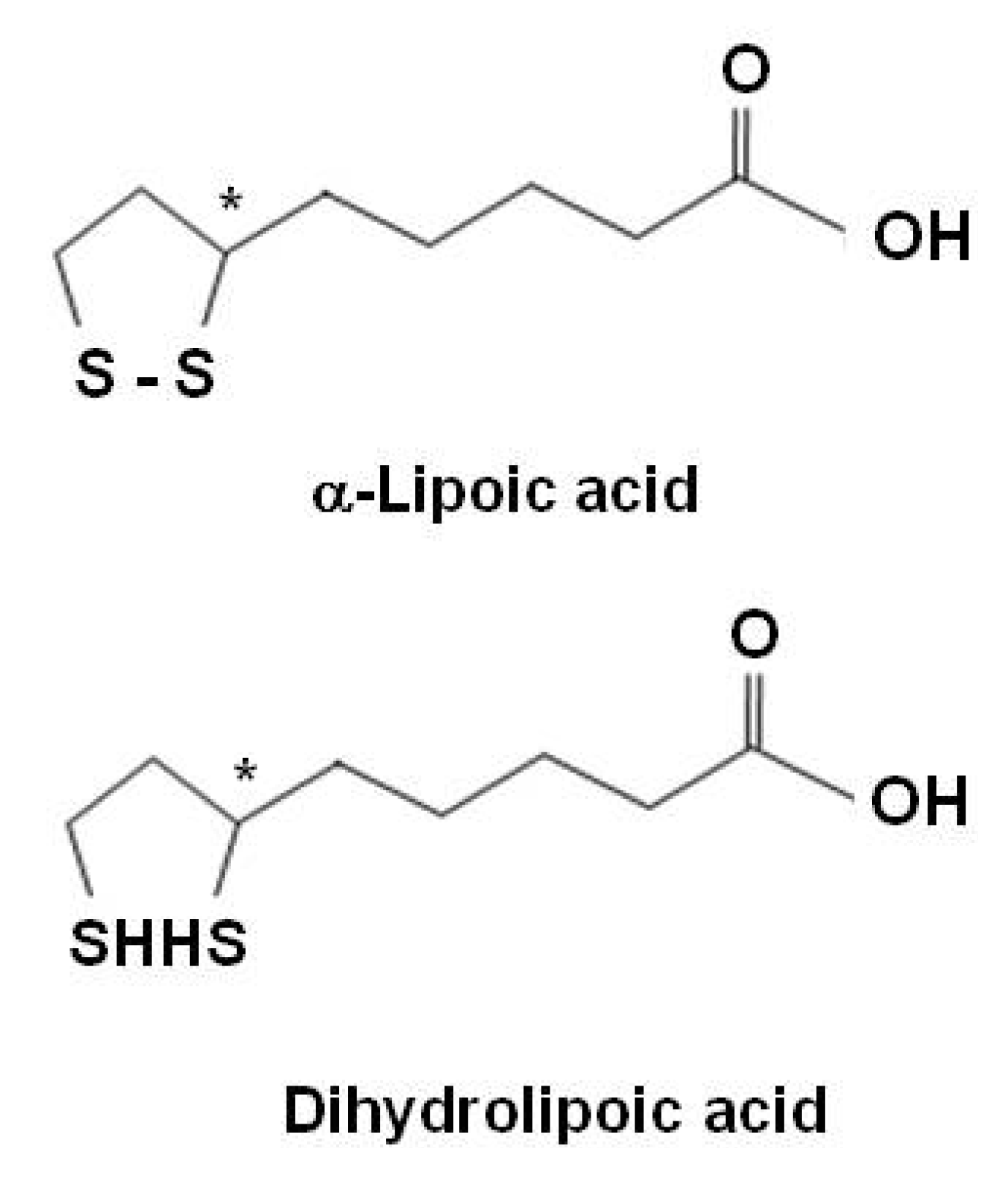
| α-lipoic acid | Quenches reactive oxygen species | |
| Regenerates/recycles endogenous and exogenous antioxidants | ||
| Chelates redox metals | ||
| Modulate the activity of transcription factors | ||
| Volatile compounds | ||
| Phytoncides | Insecticide | |
| Antibacterial/antifungal activity | ||
| Radical scavenging activity | ||
| Enhance the activity of NK cells | ||
| Restoring antioxidants | ||
| Modulate the activity of transcription factor, NF-κB | ||
| Attenuate allergic inflammation | ||
Flavonoids
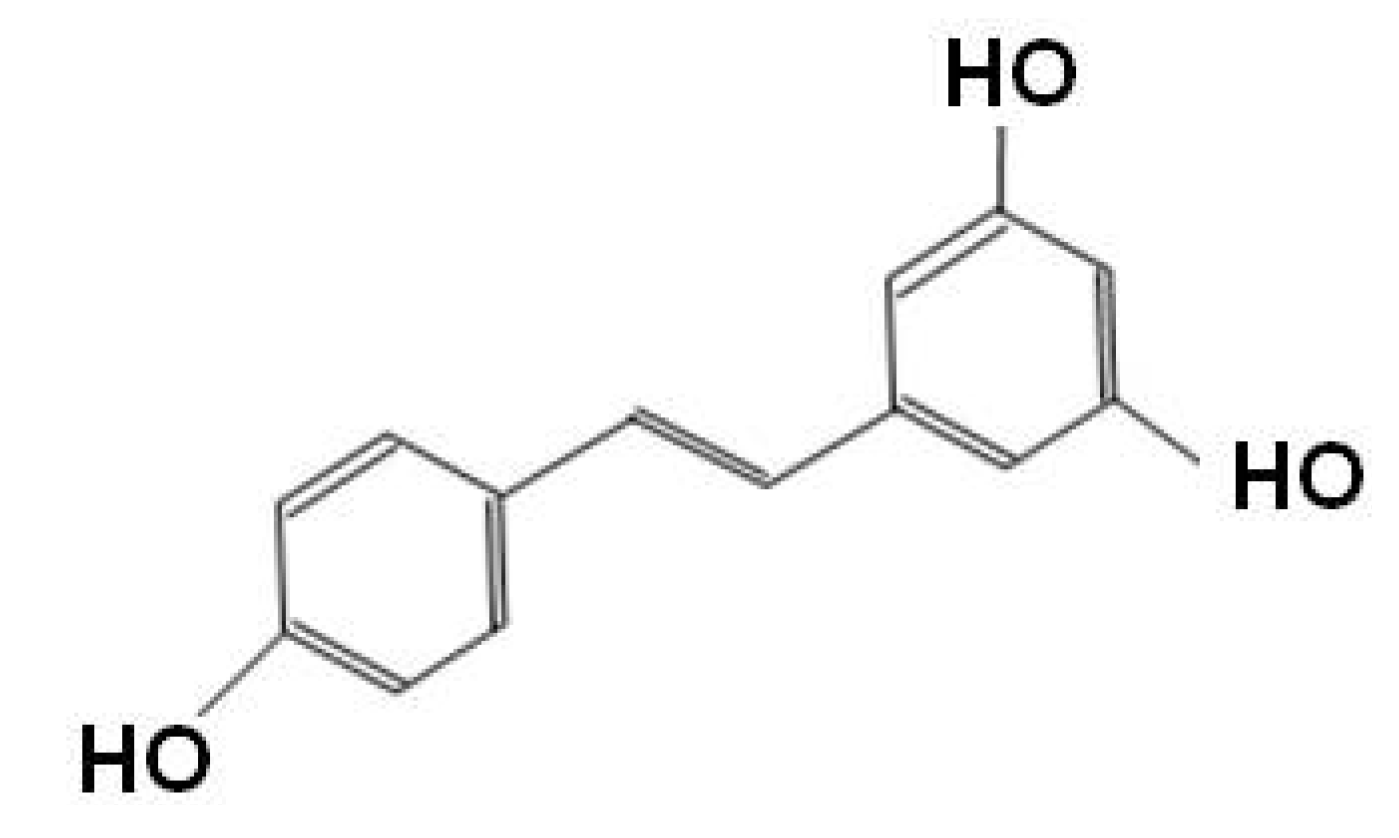
Curcumin (diferuloylmethane)
Resveratrol
Antioxidant Vitamins
Carotenoids
Vitamin C
Vitamin E
Alpha-lipoic acid
Selenium
Antioxidative Diet
Volatile Compounds
Phytoncides
Conclusions and Closing Remarks
Acknowledgments
References
- Manson, M.M.; Farmer, P.B.; Gescher, A.; Steward, W.P. Innovative agents in cancer prevention. Recent Results Cancer Res. 2005, 166, 257–275. [Google Scholar] [CrossRef]
- Park, H.S.; Kim, S.R.; Lee, Y.C. Impact of oxidative stress on lung diseases. Respirology 2009, 14, 27–38. [Google Scholar] [CrossRef]
- Nohl, H.; Kozlov, A.V.; Gille, L.; Staniek, K. Cell respiration and formation of reactive oxygen species: facts and artefacts. Biochem. Soc. Trans. 2003, 31, 1308–1311. [Google Scholar]
- Liu, P.L.; Chen, Y.L.; Chen, Y.H.; Lin, S.J.; Kou, Y.R. Wood smoke extract induces oxidative stress-mediated caspase-independent apoptosis in human lung endothelial cells: Role of AIF and EndoG. Am. J. Physiol. Lung Cell. Mol. Physiol. 2005, 289, L739–L749. [Google Scholar] [CrossRef]
- Vayssier-Taussat, M.; Camilli, T.; Aron, Y.; Meplan, C.; Hainaut, P.; Polla, B.B.; Weksler, B. Effects of tobacco smoke and benzo[a]pyrene on human endothelial cell and monocyte stress responses. Am. J. Physiol. Heart Circ. Physiol. 2001, 280, H1293–H1300. [Google Scholar]
- Wilson, M.; Lightbody, J.H.; Donaldson, K.; Sales, J.; Stone, V. Interactions between ultrafine particles and transition metals in vivo and in vitro. Toxicol. Appl. Pharmacol. 2002, 84, 172–179. [Google Scholar]
- Henricks, P.A.; Nijkamp, F.P. Reactive oxygen species as mediators in asthma. Pulm. Pharmacol. Ther. 2001, 14, 409–420. [Google Scholar] [CrossRef]
- Halliwell, B.; Gutteridge, J.M.; Cross, C.E. Free radicals, antioxidants, and human disease: Where are we now? J. Lab. Clin. Med. 1992, 119, 598–620. [Google Scholar]
- MacNee, W. Oxidative stress and lung inflammation in airway disease. Eur. J. Pharmacol. 2001, 429, 195–207. [Google Scholar] [CrossRef]
- Repine, J.E.; Bast, A.; Lankhorst, I. Oxidative stress in chronic obstructive pulmonary disease. Oxidative Stress Study Group. Am. J. Respir. Crit. Care Med. 1997, 156, 341–357. [Google Scholar]
- Dworski, R. Oxidant stress in asthma. Thorax 2000, 55, S51–S53. [Google Scholar] [CrossRef]
- Kay, A.B. Asthma and inflammation. J. Allergy Clin. Immunol. 1991, 87, 893–910. [Google Scholar] [CrossRef]
- Bousquet, J.; Jeffery, P.K.; Busse, W.W.; Johnson, M.; Vignola, A.M. Asthma. From bronchoconstriction to airways inflammation and remodeling. Am. J. Respir. Crit. Care Med. 2000, 161, 1720–1745. [Google Scholar]
- Barnes, P.J. Reactive oxygen species in asthma. Eur. Respir. Rev. 2000, 10, 240–243. [Google Scholar]
- Schauer, U.; Leinhaas, C.; Jäger, R.; Rieger, C.H. Enhanced superoxide generation by eosinophils from asthmatic children. Int. Arch. Allergy Appl. Immunol. 1991, 96, 317–321. [Google Scholar] [CrossRef]
- Teramoto, S.; Shu, C.Y.; Ouchi, Y.; Fukuchi, Y. Increased spontaneous production and generation of superoxide anion by blood neutrophils in patients with asthma. J. Asthma 1996, 33, 149–155. [Google Scholar] [CrossRef]
- Rochelle, L.G.; Fischer, B.M.; Adler, K.B. Concurrent production of reactive oxygen and nitrogen species by airway epithelial cells in vitro. Free Radic. Biol. Med. 1998, 24, 863–868. [Google Scholar] [CrossRef]
- Bruijnzeel, P.L.; Koenderman, L.; Kok, P.T.; Hameling, M.L.; Verhagen, J. Platelet activating factor (PAF-acether) induced leukotriene C4 formation and luminal dependent chemiluminescence of human eosinophils. Pharm. Res. Comm. 1986, 18, 61–69. [Google Scholar]
- Chihara, J.; Hayashi, N.; Kakazu, T.; Yamamoto, T.; Kurachi, D.; Nakajima, S. RANTES augments radical oxygen products from eosinophils. Int. Arch. Allergy Immunol. 1994, 104, 52–53. [Google Scholar] [CrossRef]
- Tenscher, K.; Metzner, B.; Schopf, E.; Norgauer, J.; Czech, W. Recombinant human eotaxin induces oxygen radical production, upregulation in human eosinophils via a pertussis toxin-sensitive heterotrimeric guanine nucleotide-binding protein. Blood 1996, 88, 3195–3199. [Google Scholar]
- Nagata, M.; Sedgwick, J.B.; Vrtis, R.; Busse, W.W. Endothelial cells upregulate eosinophil superoxide generation via VCAM-1 expression. Clin. Exp. Allergy 1998, 29, 550–561. [Google Scholar]
- Rankin, J.A.; Harris, P.; Ackerman, S.J. The effect of eosinophil granule major basic protein on lung-macrophage superoxide anion generation. J. Allergy Clin. Immunol. 1992, 89, 746–752. [Google Scholar] [CrossRef]
- Health effects of outdoor air pollution. Committee of the environmental and occupational health assembly of the American Thoracic Society. Am. J. Respir. Crit. Care Med. 1996, 153, 3–50.
- Rahman, I.; MacNee, W. Reactive oxygen species. In Asthma and COPD; Barnes, P.J., Drazen, J., Rennard, S., Thomson, N., Eds.; Academic Press: London, UK, 2002; pp. 243–254. [Google Scholar]
- Barnes, P.J.; Chung, K.F.; Page, C.P. Inflammatory mediators of asthma: An update. Pharmacol. Rev. 1998, 50, 515–596. [Google Scholar]
- Barnes, P.J. Cytokines as mediators of chronic asthma. Am. J. Respir. Crit. Care Med. 1994, 150, 42–49. [Google Scholar]
- Hulsmann, A.R.; Raatgeep, H.R.; den Hollander, J.C.; Stijnen, T.; Saxena, P.R.; Kerrebijn, K.F.; de Jongste, J.C. Oxidative epithelial damage produces hyperresponsiveness of human peripheral airways. Am. J. Respir. Crit. Care Med. 1994, 149, 519–525. [Google Scholar]
- Cortijo, J.; Marti-Cabrera, M.; de la Asuncion, J.G.; Pallardó, F.V.; Esterase, A.; Bruseghini, L.; Viña, J.; Morcillo, E.J. Contraction of human airways by oxidative stress protection by N-acetylcysteine. Free Radic. Biol. Med. 1999, 27, 392–400. [Google Scholar] [CrossRef]
- Sadeghi-Hashjin, G.; Folkerts, G.; Henricks, P.A.; Verheyen, A.K.; van der Linde, H.J.; van Ark, I.; Coene, A.; Nijkamp, F.P. Peroxynitrite induces airway hyperresponsiveness in guinea pigs in vitro and in vivo. Am. J. Respir. Crit. Care Med. 1996, 153, 1697–1701. [Google Scholar]
- Lee, K.S.; Kim, S.R.; Park, S.J.; Park, H.S.; Min, K.H.; Lee, M.H.; Jin, S.M.; Jin, G.Y.; Yoo, W.H.; Lee, Y.C. Hydrogen peroxide induces vascular permeability via regulation of vascular endothelial growth factor. Am. J. Respir. Cell Mol. Biol. 2006, 35, 190–197. [Google Scholar] [CrossRef]
- Adam, L.; Bouvier, M.; Jones, T.L. Nitric oxide modulates beta(2)-adrenergic receptor palmitoylation and signaling. J. Biol. Chem. 1999, 274, 26337–26343. [Google Scholar]
- Owen, S.; Pearson, D.; Suarez-Mendez, V.; O’Driscoll, R.; Woodcock, A. Evidence of free-radical activity in asthma. N. Engl. J. Med. 1991, 325, 586–587. [Google Scholar] [CrossRef]
- Cho, Y.J.; Seo, M.S.; Kim, J.K.; Lim, Y.; Chae, G.; Ha, K.S.; Lee, K.H. Silica-induced generation of reactive oxygen species in Rat2 fibroblasts: Role in activation of mitogen-activated protein kinase. Biochem. Biophys. Res. Commun. 1999, 262, 708–712. [Google Scholar] [CrossRef]
- Ding, M.; Shi, X.; Dong, Z.; Chen, F.; Lu, Y.; Castranova, V.; Vallyathan, V. Freshly fractured crystalline silica induces activator protein-1 activation through ERKs and p38 MAPK. J. Biol. Chem. 1999, 274, 30611–30616. [Google Scholar]
- Lopez-Ilasaca, M.; Crespo, P.; Pellici, P.G.; Gutkind, J.S.; Wetzker, R. Linkage of G protein-coupled receptors to the MAPK signaling pathway through PI 3-kinase gamma. Science 1997, 275, 394–397. [Google Scholar]
- Lee, Y.C.; Lee, K.S.; Park, S.J.; Park, H.S.; Lim, J.S.; Park, K.H.; Im, M.J.; Choi, I.W.; Lee, H.K.; Kim, U.H. Blockade of airway hyperresponsiveness and inflammation in a murine model of asthma by a prodrug of cysteine, L-2-oxothiazolidine-4-carboxylic acid. FASEB J. 2004, 18, 1917–1919. [Google Scholar]
- Lee, K.S.; Park, H.S.; Park, S.J.; Kim, S.R.; Min, K.H.; Jin, S.M.; Li, L.; Lee, Y.C. An antioxidant modulates expression of receptor activator of NF-κB in asthma. Exp. Mol. Med. 2006, 38, 217–229. [Google Scholar]
- Schenk, H.; Klein, M.; Erdbrugger, W.; Dröge, W.; Schulze-Osthoff, K. Distinct effects of thioredoxin and antioxidants on the activation of transcription factors NF-κB and AP-1. Proc. Natl. Acad. Sci. USA 1994, 91, 1672–1676. [Google Scholar]
- Harper, R.; Wu, K.; Chang, M.M.; Yoneda, K.; Pan, R.; Reddy, S.P.; Wu, R. Activation of nuclear factor-kappa b transcriptional activity in airway epithelial cells by thioredoxin but not by N-acetyl-cysteine and glutathione. Am. J. Respir. Cell Mol. Biol. 2001, 25, 178–185. [Google Scholar]
- Shaulian, E.; Karin, M. AP-1 as a regulator of cell life and death. Nat. Cell Biol. 2002, 4, E131–E136. [Google Scholar] [CrossRef]
- Tikoo, K.; Lau, S.S.; Monks, T.J. Histone H2 phosphorylation is coupled to poly(ADP-ribosylation) during reactive oxygen species-induced cell death in renal proximal tubular epithelial cells. Mol. Pharmacol. 2001, 60, 394–402. [Google Scholar]
- Zor, U.; Ferber, E.; Gergely, P.; Szücs, K; Dombrádi, V.; Goldman, R. Reactive oxygen species mediate phorbol ester-regulated tyrosine phosphorylation and phospholipase A2 activation: potentiation by vanadate. Biochem. J. 1993, 295, 879–888. [Google Scholar]
- Goldman, R.; Ferber, E.; Zort, U. Reactive oxygen species are involved in the activation of cellular phospholipase A2. FEBS Lett. 1992, 309, 190–192. [Google Scholar] [CrossRef]
- Liu, T.; Castro, S.; Brasier, A.R.; Jamaluddin, M.; Garofalo, R.P.; Casola, A. Reactive oxygen species mediate virus-induced STAT activation: Role of tyrosine phosphatase. J. Biol. Chem. 2004, 279, 2461–2469. [Google Scholar]
- Hsu, H.Y.; Wen, M.H. Lipopolysaccharide-mediated reactive oxygen species and signal transduction in the regulation of interleukin-1 gene expression. J. Biol. Chem. 2002, 277, 22131–22139. [Google Scholar]
- Seifried, H.E.; Anderson, D.E.; Fisher, E.I.; Milner, J.A. A review of the interaction among dietary antioxidants and reactive oxygen species. J. Nutr. Biochem. 2007, 18, 567–579. [Google Scholar] [CrossRef]
- Chaudiere, J.; Ferrari-Iliou, R. Intracellular antioxidants from chemical to biochemical mechanisms. Food Chem. Toxicol. 1999, 37, 949–962. [Google Scholar] [CrossRef]
- Heffner, J.A.; Repine, J.E. State of the art: Pulmonary strategies of antioxidant defense. Am. Rev. Respir. Dis. 1989, 140, 531–554. [Google Scholar] [CrossRef]
- Toth, K.M.; Clifford, D.P.; Berger, E.M.; White, C.W.; Repine, J.E. Intact human erythrocyte prevent hydrogen peroxide mediated damage to isolated perfused rat lungs and cultured bovine pulmonary artery endothelial cells. J. Clin. Invest. 1984, 74, 292–295. [Google Scholar] [CrossRef]
- Van Asbeck, B.S.; Hoidal, J.; Vercelloti, G.M.; Schwartz, B.A.; Moldow, C.F.; Jacob, H.S. Protection against lethal hyperoxia by tracheal insufflation of erythrocytes: role of red cell glutathione. Science 1985, 227, 756–759. [Google Scholar]
- Wood, L.G; Gibson, P.G.; Garg, M.L. Biomarkers of lipid peroxidation, airway inflammation and asthma. Eur. Respir. J. 2003, 21, 177–186. [Google Scholar] [CrossRef]
- Devereux, G.; Seaton, A. Diet as a risk factor for atopy and asthma. J. Allergy Clin. Immunol. 2005, 115, 1109–1117. [Google Scholar] [CrossRef]
- Atkinson, C.J.; Nestby, R.; Fod, Y.Y.; Dodds, P.A. Enhancing beneficial antioxidants in fruits: A plant physiological perspective. BioFactors 2005, 23, 229–234. [Google Scholar] [CrossRef]
- Klein, B.P.; Kurilich, A.C. Processing effects on dietary antioxidants from plant foods. HortScience 2000, 35, 580–584. [Google Scholar]
- Prior, R.L.; Cao, G. Antioxidant phytochemicals in fruits and vegetables: Diet and health implications. HortScience 2000, 35, 588–592. [Google Scholar]
- Balsano, C.; Alisi, A. Antioxidant effects of natural bioactive compounds. Curr. Pharmaceu. Des. 2009, 15, 3063–3073. [Google Scholar] [CrossRef]
- Middleton, E.J.; Kandaswami, C.; Theoharides, T.C. The effects of plant flavonoids on mammalian cells: Implications for inflammation, heart disease, and cancer. Pharmacol. Rev. 2000, 52, 673–751. [Google Scholar]
- Williams, C.A.; Grayer, R.J. Anthocyanins and other flavonoids. Nat. Prod. Rep. 2004, 21, 539–573. [Google Scholar] [CrossRef]
- Hollman, P.C.; Katan, M.B. Health effects and bioavailability of dietary flavonols. Free Rad. Res. 1999, 31, S75–S80. [Google Scholar] [CrossRef]
- Harborne, J.B.; Williams, C.A. Advances in flavonoid research since 1992. Phytochemistry 2000, 55, 481–504. [Google Scholar]
- Schroeter, H.; Boyd, C.; Spencer, J.P.; Williams, R.J.; Cadenas, E.; Rice-Evans, C. MAPK signaling in neurodegeneration: Influences of flavonoids and of nitric oxide. Neurobiol. Aging 2002, 23, 861–880. [Google Scholar] [CrossRef]
- Rice-Evans, C. Flavonoid antioxidants. Curr. Med. Chem. 2001, 8, 797–807. [Google Scholar] [CrossRef]
- Polovka, M.; Brezova, V.; Stasko, A. Antioxidant properties of tea investigated by EPR spectroscopy. Biophys. Chem. 2003, 106, 39–56. [Google Scholar] [CrossRef]
- Shaheen, S.O.; Sterne, J.A.; Thompson, R.L.; Songhurst, C.E.; Margetts, B.M.; Burney, P.G. Dietary antioxidants and asthma in adults: population-based case-control study. Am. J. Respir. Crit. Care Med. 2001, 164, 1823–1828. [Google Scholar]
- Knekt, P.; Kumpulainen, J.; Jarvinen, R.; Rissanen, H.; Hellövaara, M.; Reunanen, A.; Hhakulinen, T.; Aromaa, A. Flavonoid intake and risk of chronic diseases. Am. J. Clin. Nutr. 2002, 76, 560–568. [Google Scholar]
- Middleton, E. The role of hydrogen peroxide in basophil histamine release and the effect of selected flavonoids. J. Allergy Clin. Immunol. 1986, 78, 321–328. [Google Scholar] [CrossRef]
- Sloan, R.; Boran-Ragotzy, R.; Ackerman, S.J.; Drzewiecki, G.; Middleton, E. The effect of plant flavonoids on eosinophil degranulation. J. Allergy Clin. Immunol. 1991, 282. abstract. [Google Scholar]
- Li, R.R.; Pang, L.L.; Du, Q.; Shi, Y.; Dai, W.J.; Yin, K.S. Apigenin inhibits allergen-induced airway inflammation and switches immune response in a murine model of asthma. Immunopharmacol. Immunotoxicology 2010, in press.. [Google Scholar]
- Park, H.S.; Kim, S.Y.; Kim, S.R.; Lee, Y.C. Targeting abnomrla airway vasculatiry as a therapeutical strategy in asthma. Respirology 2010, 15, 459–471. [Google Scholar] [CrossRef]
- Oak, M.H.; El Bedoui, J.; Schini-Kerth, V.B. Antiangiogenic properties of naturalpolyphenols from red wine and green tea. J. Nutr. Biochem. 2005, 16, 1–8. [Google Scholar] [CrossRef]
- Cao, Y.; Cao, R. Angiogenesis inhibited by drinking tea. Nature 1999, 398, 381. [Google Scholar]
- Fang, J.; Zhou, Q.; Liu, L.Z.; Xia, C.; Hu, X.; Shi, X.; Jiang, B.H. Angiogenin inhibits tumor angiogenesis through decreasing HIF-1alpha and VEGF expression. Carcinogenesis 2007, 28, 858–864. [Google Scholar]
- Liu, L.Z.; Fang, J.; Zhou, Q.; Hu, X.; Shi, X.; Jiang, B.H. Apigenin inhibits expressionof vascular endothelial growth factor and angiogenesis in human lung cancercells: Implication of chemoprevention of lung cancer. Mol. Pharmacol. 2005, 68, 635–643. [Google Scholar]
- Fang, J.; Xia, C.; Cao, Z.; Zheng, J.Z.; Reed, E.; Jiang, B.H. Apigenin inhibits VEGF andHIF-1 expression via PI3K/AKT/p70S6K1 and HDM2/p53 pathways. FASEB J. 2005, 19, 342–353. [Google Scholar] [CrossRef]
- Mojžišová, G.; Mojžiš, J.; Pilátová, M.; Vohárová, V.; Varinská, L.; Perjesi, P.; Sarišský, M.; Mirossay, L. Antiproliferative and antiangiogenic effects of selected chalcones. Acta Pharmacol. Sin. 2006, 27, 338. [Google Scholar]
- Mojžiš, J.; Varinská, L.; Perjesi, P.G.; Mojžišová, G. Antiangiogenic effect of newlysynthesized chalcones. Eur. J. Cancer 2007, 5, 87. [Google Scholar]
- Mojzis, J.; Varinska, L.; Mojzisova, G.; Kostova, I.; Mirossay, L. Antiangiogenic effects of flavonoids and chalcones. Pharmacol. Res. 2008, 57, 259–265. [Google Scholar] [CrossRef]
- Oak, M.H.; El Bedoui, J.; Schini-Kerth, V.B. Antiangiogenic properties of natural polyphenols from red wine and green tea. J. Nutr. Biochem. 2005, 16, 1–8. [Google Scholar] [CrossRef]
- Kurup, V.P.; Barrios, C.S. Immunomodulatory effects of curcumin in allergy. Mol. Nutr. Food Res. 2008, 52, 1031–1039. [Google Scholar]
- Biswas, S.; Rahman, I. Modulation of steroid activity in chronic inflammation: A novel anti-inflammatory role for curcumin. Mol. Nutr. Food Res. 2008, 52, 987–994. [Google Scholar] [CrossRef]
- Mortellini, R.; Foresti, R.; Bassi, R.; Green, C.J. Curcumin, an antioxidant and anti-inflammatory agent, induces heme oxygenase-1 and protects endothelial cells against oxidative stress. Free Radic. Biol. Med. 2000, 28, 1301–1312. [Google Scholar]
- Brouet, I.; Ohshima, H.; Curcumin. An anti-tumour promoter and anti-inflammatory agent, inhibits induction of nitric oxide synthetase in activated macrophages. Biochem. Biophys. Res. Commun. 1995, 206, 533–540. [Google Scholar] [CrossRef]
- Barnes, P.J. Histone deacetylase-2 and airway disease. Ther. Adv. Respir. Dis. 2009, 3, 235–243. [Google Scholar] [CrossRef]
- Meja, K.K.; Rajendrasozhan, S.; Adenuga, D.; Biswas, S.K.; Sundar, I.K.; Spooner, G.; Marwick, J.A.; Chakravarty, P.; Fletcher, D.; Whittaker, P.; et al. Curcumin restores corticosteroid function in monocytes exposed to oxidants by maintaining HDAC2. Am. J. Respir. Cell Mol. Biol. 2008, 39, 312–323. [Google Scholar] [CrossRef]
- Bremner, P.; Heinrich, M. Natural products as targeted modulators of the nuclear factor-kappaB pathway. J. Pharm. Pharmacol. 2002, 54, 453–472. [Google Scholar]
- de la Lastra, C.A.; Villegas, I. Resveratrol as an antioxidant and prooxidant agent: Mechanisms and clinical implications. Biochem. Soc. Trans. 2007, 35, 1156–1160. [Google Scholar] [CrossRef]
- Das, S.; Das, D.K. Resveratrol: A therapeutic promise for cardiovascular diseases. Recent Pat. Cardiovasc. Drug Discov. 2007, 2, 133–138. [Google Scholar] [CrossRef]
- Shankar, S.; Singh, G.; Srivastava, R.K. Chemoprevention by resveratrol: molecular mechanisms and therapeutic potential. Front. Biosci. 2007, 12, 4839–4854. [Google Scholar] [CrossRef]
- Pirola, L.; Fröjdö, S. Resveratrol: one molecule, many targets. IUBMB Life 2008, 60, 323–332. [Google Scholar]
- Leonard, S.; Xia, C.; Jiang, B.H.; Stinefelt, B.; Klandorf, H.; Harris, G.K.; Shi, X. Resveratrol scavenges reactive oxygen species and effects radical-induced cellular responses. Biochem. Biophys. Res. Commun. 2003, 309, 1017–1026. [Google Scholar] [CrossRef]
- Das, D.K.; Maulik, N. Resveratrol in cardioprotection: A therapeutic promise of alternative medicine. Mol. Interven. 2006, 6, 36–47. [Google Scholar] [CrossRef]
- Youn, J.; Lee, J.S.; Na, H.K.; Kundu, J.K.; Surh, Y.J. Resveratrol and piceatannol inhibit iNOS expression and NF-kappaB activation in dextran sulfate sodium-induced mouse colitis. Nutr. Cancer 2009, 61, 847–854. [Google Scholar] [CrossRef]
- Li, T.; Wang, W.; Chen, H.; Li, T.; Ye, L. Evaluation of anti-leukemia effect of resveratrol by modulating STAT3 signaling. Int. Immunopharmacol. 2010, 10, 18–25. [Google Scholar]
- Tang, X.; Zhang, Q.; Nishitani, J.; Brown, J.; Shi, S; Le, A.D. Overexpression of human papillomavirus type 16 oncoproteins enhances hypoxia-inducible factor 1 alpha protein accumulation and vascular endothelial growth factor expression in human cervical carcinoma cells. Clin. Cancer Res. 2007, 13, 2568–2576. [Google Scholar] [CrossRef]
- Wu, H.; Liang, X.; Fang, Y.; Qin, X.; Zhang, Y.; Liu, J. Resveratrol inhibits hypoxia-induced metastasis potential enhancement by restricting hypoxia-induced factor-1 alpha expression in colon carcinoma cells. Biomed. Pharmacother. 2008, 62, 613–621. [Google Scholar] [CrossRef]
- Park, S.Y.; Jeong, K.J.; Lee, J.; Yoon, D.S.; Choi, W.S.; Kim, Y.K.; Han, J.W.; Kim, Y.M.; Kim, B.K.; Lee, H.Y. Hypoxia enhances LPA-induced HIF-1alpha and VEGF expression: Their inhibition by resveratrol. Cancer Lett. 2007, 258, 63–69. [Google Scholar] [CrossRef]
- Bourguignon, L.Y.; Xia, W.; Wong, G. Hyaluronan-mediated CD44 Interaction with p300 and SIRT1 Regulates β-Catenin Signaling and NFkappaB-specific Transcription Activity Leading to MDR1 and Bcl-xL Gene Expression and Chemoresistance in Breast Tumor Cells. J. Biol. Chem. 2009, 284, 2657–2671. [Google Scholar]
- Rachon, D.; Rimoldi, G.; Wuttke, W. In vitro effects of genistein and resveratrol on the production of interferon-gamma (IFNgamma) and interleukin-10 (IL-10) by stimulated murine splenocytes. Phytomedicine 2006, 13, 419–424. [Google Scholar] [CrossRef]
- Tao, H.Y.; Wu, C.F.; Zhou, Y.; Gong, W.H.; Zhang, X.; Iribarren, P.; Zhao, Y.Q.; Le, Y.Y.; Wang, J.M. The grape component resveratrol interferes with the function of chemoattractant receptors on phagocytic leukocytes. Cell Mol. Immunol. 2004, 1, 50–56. [Google Scholar]
- Wirleitner, B.; Schroecksnadel, K.; Winkler, C.; Schennach, H.; Fuchs, D. Resveratrol suppresses interferon-gamma-induced biochemical pathways in human peripheral blood mononuclear cells in vitro. Immunol. Lett. 2005, 100, 159–163. [Google Scholar] [CrossRef]
- Gao, X.; Deeb, D.; Media, J.; Divine, G.; Jiang, H.; Chapman, R.A.; Gautam, S.C. Immunomodulatory activity of resveratrol: discrepant in vitro and in vivo immunological effects. Biochem. Pharmacol. 2003, 66, 2427–2435. [Google Scholar] [CrossRef]
- Gao, X.; Xu, Y.X.; Janakiraman, N.; Chapman, R.A.; Gautam, S.C. Immunomodulatory activity of resveratrol: Suppression of lymphocyte proliferation, development of cell-mediated cytotoxicity, and cytokine production. Biochem. Pharmacol. 2001, 62, 1299–1308. [Google Scholar] [CrossRef]
- Harikumar, K.B.; Aggarwal, B.B. Resveratrol: a multitargeted agent for age-associated chronic diseases. Cell Cycle 2008, 7, 1020–1035. [Google Scholar]
- Kolthur-Seetharam, U.; Dantzer, F.; McBurney, M.W.; de Murcia, G.; Sassone-Corsi, P. Control of AIF-mediated cell death by the functional interplay of SIRT1 and PARP-1 in response to DNA damage. Cell Cycle 2006, 5, 873–877. [Google Scholar] [CrossRef]
- Alcaín, F.J.; Villalba, J.M. Sirtuin activators. Expert Opin. Ther. Pat. 2009, 19, 403–414. [Google Scholar] [CrossRef]
- Kim, G,Y.; Cho, H.; Ahn, S.C.; Oh, Y.H.; Lee, C.M.; Park, Y.M. Resveratrol inhibits phenotypic and functional maturation of murine bone marrow-derived dendritic cells. Int. Immunopharmacol. 2004, 4, 245–253. [Google Scholar] [CrossRef]
- Rao, A.V.; Rao, L.G. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar]
- Gallicchio, L.; Boyd, K.; Matanoski, G.; Tao, X.G.; Chen, L.; Lam, T.K.; Shiels, M.; Hammond, E.; Robinson, K.A.; Caulfield, L.E.; et al. Carotenoids and the risk of developing lung cancer: A systematic review. Am. J. Clin. Nutr. 2008, 88, 372–383. [Google Scholar]
- Young, C.Y.; Yuan, H.Q.; He, M.L.; Zhang, J.Y. Carotenoids and prostate cancer risk. Mini Rev. Med. Chem. 2008, 8, 529–537. [Google Scholar] [CrossRef]
- Melendez, G.V; Okani, E.T.; Kiertsman, B.; Roncada, M.J. Vitamin A status in children with pneumonia. Eur. J. Clin. Nutr. 1995, 49, 379–384. [Google Scholar]
- Kukukbay, H.; Yakinci, C.; Kukukbay, F.Z.; Tutgut, M. Serum vitamin A and beta-carotene levels in children with recurrent acute respiratory infections and diarrhoea in Malarya. J. Trop. Pediatr. 1997, 43, 337–340. [Google Scholar]
- Mayne, S.T. Beta-carotene, carotenoids, and disease prevention in humans. FASEB J. 1996, 10, 690–701. [Google Scholar]
- Mortensen, A.; Skibsted, L.H.; Truscott, T.G. The interaction of dietary carotenoids with radical species. Arch. Biochem. Biophys. 2001, 385, 13–19. [Google Scholar]
- Schock, B.C.; Young, I.S.; Brown, V.; Fitch, P.S.; Shields, M.D.; Ennis, M. Antioxidants and oxidative stress in BAL fluid of atopic asthmatic children. Pediatr. Res. 2003, 53, 375–381. [Google Scholar] [CrossRef]
- Barasi, ME. Water soluble vitamins. In Human Nutrition; Arnold Publishers: London, UK, 1997; pp. 171–175. [Google Scholar]
- Rice-Evans, C.A.; Sampson, J.; Bramley, P.M.; Holloway, D.E. Why do we expect carotenoids to be antioxidants in vivo? Free Radic. Res. 1997, 26, 381–398. [Google Scholar] [CrossRef]
- Patrick, L. Beta-carotene: the controversy continues. Altern. Med. Rev. 2000, 5, 530–545. [Google Scholar]
- Wood, L.G.; Garg, M.L.; Blake, R.J.; Garcia-Caraballo, S.; Gibson, P.G. Airway and circulating levels of carotenoids in asthma and healthy controls. J. Am. Coll. Nutr. 2005, 24, 448–455. [Google Scholar]
- Zachman, R.D. Role of vitamin A in lung development. J. Nutr. 1994, 125, 1634S–1638S. [Google Scholar]
- Chandra, R.K.; Vyas, D. Vitamin A, immunocompetence and infection. Fed. Nutr. Bull. 1989, 11, 12–19. [Google Scholar]
- Bloem, M.W.; Wedel, M.; Egger, R.J. Mild vitamin A deficiency and risk of respiratory tract diseases and diarrhea in preschool and school children in northeastern Thailand. Am. J. Epidemiol. 1990, 131, 332–339. [Google Scholar]
- Villamor, E.; Fawzi, W.W. Vitamin A supplementation: Implications for morbidity and mortality in children. J. Infect. Dis. 2000, 182, S122–S133. [Google Scholar] [CrossRef]
- Niles, R.M. Signaling pathways in retinoid chemoprevention and treatment of cancer. Mutat. Res. 2004, 555, 81–96. [Google Scholar]
- Sharoni, Y.; Danilenko, M.; Dubi, N.; Ben-Dor, A.; Levy, J. Carotenoids and transcription. Arch. Biochem. Biophys. 2004, 430, 89–96. [Google Scholar]
- Karas, M.; Amir, H.; Fishman, D.; Danilenko, M.; Segal, S.; Nahum, A.; Koifmann, A.; Giat, Y.; Levy, J.; Sharoni, Y. Lycopene interferes with cell cycle progression and insulin-like growth factor I signaling in mammary cancer cells. Nutr. Cancer 2000, 36, 101–111. [Google Scholar] [CrossRef]
- Riccioni, G.; Barbara, M.; Bucciarelli, T.; de Ilio, C.; D’Orazio, N. Antioxidant vitamin supplementation in asthma. Ann. Clin. Lab. Sci. 2007, 37, 96–101. [Google Scholar]
- Retsky, K.L.; Chen, K.; Zeind, J.; Frei, B. Inhibition of copperinduced LDL oxidation by Vitamin C is associated with decreased copperbinding to LDL and 2-oxo-histidine formation. Free Radic. Biol. Med. 1999, 26, 90–98. [Google Scholar] [CrossRef]
- Cameron, E.; Pauling, L. Supplemental ascorbate in supportive treatment of cancer—prolongation of survival times in terminal human cancer. Part 1. Proc. Natl. Acad. Sci. USA 1976, 73, 3685–3689. [Google Scholar] [CrossRef]
- You, W.C.; Zhang, L.; Gail, M.H.; Chang, Y.S.; Liu, W.D.; Ma, J.L.; Li, J.Y.; Jin, M.L.; Hu, Y.R.; Yang, C.S.; et al. Gastric dysplasia and gastric cancer: Helicobacter pylori, serum vitamin C, and other risk factors. J. Natl. Cancer Inst. 2000, 92, 1607–1612. [Google Scholar] [CrossRef]
- Knekt, P.; Jarvinen, R.; Seppanen, R.; Rissanen, A.; Aromaa, A.; Heinonen, O.P.; Albanes, D.; Heinonen, M.; Pukkala, E.; Teppo, L. Dietary antioxidants and the risk of lung-cancer. Am. J. Epidemiol. 1991, 134, 471–479. [Google Scholar]
- Divisi, D.; Di Tommaso, S.; Salvemini, S.; Garramone, M.; Crisci, R. Diet and cancer. Acta Biomed. 2006, 77, 118–123. [Google Scholar]
- Romieu, I.; Trenga, C. Diet and obstructive lung diseases. Epidemiol. Rev. 2001, 23, 268–287. [Google Scholar] [CrossRef]
- Allen, S.; Britton, J.R.; Leonardi-Bee, J.A. Association between antioxidant vitamins and asthma outcome measures: systematic review and meta-analysis. Thorax 2009, 64, 610–619. [Google Scholar] [CrossRef]
- Catani, M.V.; Rossi, A.; Costanzo, A.; Sabatini, S.; Levrero, M.; Melino, G.; Avigliano, L. Induction of gene expression via activator protein-1 in the ascorbate protection against UV-induced damage. Biochem. J. 2001, 356, 77–85. [Google Scholar] [CrossRef]
- Kaur, B.; Rowe, B.H.; Arnold, E. Vitamin C supplementation for asthma. Cochrane Database Syst. Rev. 2009, 1, CD000993. [Google Scholar]
- Riccioni, G.; D’Orazio, N. The role of selenium, zinc and antioxidant vitamin supplementation in the treatment of bronchial asthma: Adjuvant therapy or not? Expert Opin. Investig. Drugs 2005, 14, 1145–1155. [Google Scholar] [CrossRef]
- Burton, G.W.; Ingold, K.U. Vitamin E as an in vitro and in vivo antioxidant. Ann. N.Y. Acad. Sci. 1989, 570, 7–22. [Google Scholar] [CrossRef]
- Atkinson, J.; Epand, R.F.; Epand, R.M. Tocopherols and tocotrienols in membranes: A critical review. Free Radic. Biol. Med. 2008, 44, 739–764. [Google Scholar]
- De Luca, H.F.; Zierold, C. Mechanisms and functions of vitamin D. Nutr. Rev. 1998, 56, 4–10. [Google Scholar] [CrossRef]
- Moriguchi, S.; Muraga, M. Vitamin E and immunity. Vitam. Horm. 2000, 59, 305–336. [Google Scholar] [CrossRef]
- Pryor, W.A. Vitamin E and heart disease: Basic science to clinical intervention trials. Free Radic. Biol. Med. 2000, 28, 141–164. [Google Scholar]
- Devarah, S.; Harris, A.; Jialal, I. Modulation of monocyte-macrophage function with alpha-tocopherol: implications for atherosclerosis. Nutr. Rev. 2002, 60, 8–14. [Google Scholar] [CrossRef]
- Yoshikawa, T.; Yoshida, N. Vitamin E and leukocyte-endothelial cell interactions. Antioxid. Redox Signal 2000, 2, 821–825. [Google Scholar] [CrossRef]
- Kojo, S.; Vitamin, C. basic metabolism and its function as an index of oxidative stress. Curr. Med. Chem. 2004, 11, 1041–1064. [Google Scholar] [CrossRef]
- Pletsityi, K.D.; Vasipa, S.B.; Daydova, T.V.; Fomina, V.G. Vitamin E: Immunocorrecting effect in bronchial asthma patients. Vopr. Med. Khim. 1995, 41, 33–36. [Google Scholar]
- Picado, C.; Deulofeu, R.; Lleonart, R.; Agusti, M.; Mullol, J.; Quintó, L.; Torra, M. Dietary. micronutrients/antioxidants and their relationship with bronchial asthma severity. Allergy 2001, 56, 43–49. [Google Scholar]
- Troisi, R.J.; Willett, W.C.; Weiss, S.T.; Trichopoulos, D.; Rosner, B.; Speizer, F.E. A prospective study of diet and adult-onset asthma. Am. J. Respir. Crit. Care Med. 1995, 151, 1401–1408. [Google Scholar]
- Trenga, C.; Koenig, J.Q,; Williams, P.V. Dietary antioxidants and ozone-induced bronchial hyperresponsiveness in adults with asthma. Arch. Environ. Health. 2001, 56, 242–249. [Google Scholar] [CrossRef]
- Pearson, P.J.; Lewis, S.A.; Britton, J.; Fogarty, A. Vitamin E supplements in asthma: A parallel group randomized placebo controlled trial. Thorax 2004, 59, 652–656. [Google Scholar] [CrossRef]
- Wiser, J.; Alexis, N.E.; Jiang, Q.; We, W.; Robinnette, C.; Roubey, R.; Peden, D.B. In vivo gamma tocopherol supplementation decreases systemic oxidative stress and cytokine responses of human monocytes in normal and asthmatic subjects. Free Radic. Biol. Med. 2008, 45, 40–49. [Google Scholar] [CrossRef]
- Berdnikovs, S.; Abdala-Valencia, H.; McCary, C.; Somand, M.; Cole, R.; Garcia, A.; Bryce, P.; Cook-Mills, J.M. Isoforms of vitamin E have opposing immunoregulatory functions during inflammation by regulating leukocyte recruitment. J. Immunol. 2009, 182, 4395–4405. [Google Scholar]
- Wolf, G. How an increased intake of α-tocopherol can suppress the bioavailability of γ-tocopherol. Nutr. Rev. 2006, 64, 295–299. [Google Scholar] [CrossRef]
- Packer, L.; Suzuki, Y.J. Vitamin-E and alpha-lipoate — role in antioxidant recycling and activation of the NF-kappa B transcription factor. Mol. Aspects Med. 1993, 14, 229–239. [Google Scholar] [CrossRef]
- Bustamante, J.; Lodge, J.K.; Marcocci, L.; Tritschler, H.J.; Packer, L.; Rihn, B.H. Alpha-lipoic acid in liver metabolism and disease. Free Radic. Biol. Med. 1998, 24, 1023–1039. [Google Scholar] [CrossRef]
- Cao, X.; Phillis, J.W. The free radical scavenger, alpha-lipoic acid, protects against cerebral ischemia-reperfusion injury in gerbils. Free Radic. Res. 1995, 23, 365–370. [Google Scholar] [CrossRef]
- Van Dam, P.S. Oxidative stress and diabetic neuropathy: Pathophysiological mechanisms and treatment perspectives. Diabetes Metab. Res. Rev. 2002, 18, 176–184. [Google Scholar]
- Ametov, A.S.; Barinov, A.; Dyck, P.J.; Hermann, R.; Kozlova, N.; Litchy, W.J.; Low, P.A.; Nehrdich, D.; Novosadova, M.; O'Brien, P.C.; et al. The sensory symptoms of diabetic polyneuropathy are improved with alpha-lipoid acid: The SYDNEY trial. Diabetes Care 2003, 26, 770–776. [Google Scholar] [CrossRef]
- Ramakrishnan, N.; Wolfe, W.W.; Catravas, G.N. Radioprotection of hematopoietic tissues in mice by lipoic acid. Radic. Res. 1992, 130, 360–365. [Google Scholar] [CrossRef]
- Cho, Y.S.; Lee, J.C.; Lee, T.H.; Lee, E.Y.; Lee, K.U.; Park, J.Y.; Moon, H.B. α-Lipoic acid inhibits airway inflammation and hyperresponsiveness in a mouse model of asthma. J. Allergy Clin. Immunol. 2004, 114, 429–435. [Google Scholar] [CrossRef]
- Lee, K.S.; Park, H.S.; Park, S.J.; Kim, S.R.; Min, K.H.; Jin, S.M.; Park, K.H.; Kim, U.H.; Kim, C.Y.; Lee, Y.C. A prodrug of cysteine, L-2-oxothiazolidine-4-carboxylic acid, regulates vascular permeability by reducing vascular endothelial growth factor expression in asthma. Mol. Pharmacol. 2005, 68, 1281–1290. [Google Scholar] [CrossRef]
- Schwarz, S. Essentiality and metabolic functions of selenium. Med. Clin. North Am. 1976, 60, 745–758. [Google Scholar]
- Saito, Y.; Sato, N.; Hirashima, M.; Takebe, G.; Nagasawa, S.; Takahashi, K. Domain structure of bi-functional selenoprotein P. Biochem. J. 2004, 381, 841–846. [Google Scholar] [CrossRef]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar]
- Gromer, S.; Eubel, J.K.; Lee, B.L.; Jacob, J. Human selenoproteins at a glance. Cell Mol. Life Sci. 2005, 62, 2414–2437. [Google Scholar] [CrossRef]
- Anonymous. Selenium-monograph. Altern. Med. Rev. 2003, 8, 63–71. [Google Scholar]
- Bainbridge, D.R. Use of (75Se)L-Selenomethionine as a label for lymphoid cells. Immunology 1976, 30, 135–144. [Google Scholar]
- Gromer, S.; Eubel, J.K.; Lee, B.L.; Jacob, J. Human selenoproteins at a glance. Cell Mol. Life Sci. 2005, 62, 2414–2437. [Google Scholar] [CrossRef]
- Pretorius, E.; Oberholzer, H.M.; Vieira, W.A.; Smit, E. Ultrastructure of platelets and fibrin networks of asthmatic mice exposed to selenium and Withania somnifera. Anat. Sci. Int. 2009, 84, 210–217. [Google Scholar] [CrossRef]
- Jahnova, E.; Horvathova, M.; Gazdik, F.; Weissova, S. Effects of selenium supplementation on expression of adhesion molecules in corticoid-dependent asthmatics. Bratisl. Lek. Listy 2002, 103, 12–16. [Google Scholar]
- Brown, K.M.; Arthur, J.R. Selenium, selenoproteins and human health: A review. Public Health Nutr. 2001, 4, 593–599. [Google Scholar]
- Horváthová, M.; Jahnová, E.; Gazdik, F. Effect of selenium supplementation in asthmatic subjects on the expression of endothelial cell adhesion molecules in culture. Biol. Trace Elem. Res. 1999, 69, 15–26. [Google Scholar]
- Hoffmann, P.R.; Berry, M.J. The influence of selenium on immune responses. Mol. Nutr. Food Res. 2008, 52, 1273–1280. [Google Scholar] [CrossRef]
- Allam, M.F.; Lucena, R.A. Selenium supplementation for asthma. Cochrane DatabaseSyst. Rev. 2009, 1, CD003538. [Google Scholar]
- Riccioni, G. Plasma lycopene and antioxidant vitamins in asthma: The PLAVA study. J. Asthma 2007, 44, 429–432. [Google Scholar] [CrossRef]
- Ray, A.L.; Semba, R.D.; Walston, J.; Ferrucci, L.; Cappola, A.R.; Ricks, M.O.; Xue, Q.L.; Fried, L.P. Low serum selenium and total carotenoids predict mortality among older women living in the community: the women’s health and aging studies. J. Nutr. 2006, 136, 172–176. [Google Scholar]
- Hisama, M.; Matsuda, S.; Tanaka, T.; Shibayama, H.; Nomura, M.; Iwaki, M. Suppression of mutagens-induced SOS response by phytoncide solution using salmonella typhimurium TA 1535/pSK1002 umu Test. J. Oleo. Sci. 2008, 57, 381–390. [Google Scholar] [CrossRef]
- Aoshima, H.; Hamamoto, K. Potentiation of GABAA receptors expressed in Xenopus oocytes by perfume and phytoncide. Biosci. Biotechnol. Biochem. 1999, 63, 743–748. [Google Scholar] [CrossRef]
- Hossain, S.J.; Aoshima, H.; Koda, H.; Kiso, Y. Potentiation of the ionotropioc GABA receptor response by whiskey fragrance. J. Agric. Food Chem. 2002, 50, 6828–6834. [Google Scholar] [CrossRef]
- Li, Q.; Morimoto, K.; Inagaki, H.; Katsumata, M.; Hirata, Y.; Hirata, K.; Shimizu, T.; Li, Y.J.; Wakayama, Y. A forest bathing trip increases human natural killer activity and expression of anti-cancer proteins in female subjects. J. Biol. Regul. Homeost. Agents 2008, 22, 45–55. [Google Scholar]
- Sadlon, A.E.; Lamson, D.W. Immune-modifying and antimicrobial effects of Eucalyptus oil and simple inhalation devices. Altern. Med. Rev. 2010, 15, 33–47. [Google Scholar]
- Nam, J.H.; Jung, H.J.; Choi, J.; Lee, K.T.; Park, H.J. The anti-gastropathic and anti-rheumatic effect of nigaichigoside F1 and 23-hydroxytormentic acid isolated from the unripe fruits of Rubus Coreanus in a rat model. Biol. Pharm. Bull. 2006, 29, 967–970. [Google Scholar] [CrossRef]
- Oikawa, S. Sequence-specific DNA damage by reactive oxygen species: Implications for carcinogenesis and aging. Environ. Health Prev. Med. 2005, 10, 65–71. [Google Scholar]
- Nose, K. Role of reactive oxygen species in the regulation of physiological functions. Bio. Pharm. Bull. 2000, 23, 897–903. [Google Scholar] [CrossRef]
- Li, Q.; Nakadai, A.; Matsushima, H.; Miyazaki, Y.; Krensky, A.M.; Kawada, T.; Morimoto, K. Phytoncides (wood essential oils) induce human natural killer cell activity. Immunopharmacol. Immunotoxicol. 2006, 28, 319–333. [Google Scholar]
- Komori, T.; Fujiwara, R.; Tanida, M.; Nomura, J.; Yokoyama, M.M. Effects of citrus fragrance on immune function and depressive states. Neuroimmunomodulation 1995, 2, 174–180. [Google Scholar] [CrossRef]
- Shibata, H.; Fujiwara, R.; Iwamoto, M.; Matsuoka, H.; Yokoyama, M.M. Immunological and behavioral effects of fragrance in mice. Int. J. Neurosci. 1991, 57, 151–159. [Google Scholar] [CrossRef]
- Sample Availability: Not Available.
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Park, H.S.; Kim, S.R.; Kim, J.O.; Lee, Y.C. The Roles of Phytochemicals in Bronchial Asthma. Molecules 2010, 15, 6810-6834. https://doi.org/10.3390/molecules15106810
Park HS, Kim SR, Kim JO, Lee YC. The Roles of Phytochemicals in Bronchial Asthma. Molecules. 2010; 15(10):6810-6834. https://doi.org/10.3390/molecules15106810
Chicago/Turabian StylePark, Hee Sun, So Ri Kim, Ju Ock Kim, and Yong Chul Lee. 2010. "The Roles of Phytochemicals in Bronchial Asthma" Molecules 15, no. 10: 6810-6834. https://doi.org/10.3390/molecules15106810
APA StylePark, H. S., Kim, S. R., Kim, J. O., & Lee, Y. C. (2010). The Roles of Phytochemicals in Bronchial Asthma. Molecules, 15(10), 6810-6834. https://doi.org/10.3390/molecules15106810




